AP PGECET 2024 Nano Technology Question Paper is available for download here. Sri Venkateswara University, Tirupati on behalf of APSCHE conducted AP PGECET 2024 Nano Technology on May 31 in Shift 2 from 2.30 PM to 4.30 PM. AP PGECET Question Paper 2024 consists of 120 MCQ-based questions in total carrying 1 mark each to be attempted in the duration of 2 hours.
AP PGECET 2024 Nano Technology Question Paper with Answer Key PDF
| AP PGECET 2024 Nano Technology Question Paper with Answer Key | Check Solution |

Question 1:
When several forces act at a point and their vector sum is zero, the forces are said to be
Question 2:
The centroid of a composite plane figure is found by
Question 3: The theorem relates the moment of inertia about any axis to the moment of inertia about a parallel axis through the centroid?
Question 4: Polar moment of inertia is used primarily for calculating the stresses in
Question 5: The equation F=kx describes a force that is
Question 6: D'Alembert's Principle is used to
Question 7: Impulse can be defined as
Question 8: Which type of motion does a rigid body undergo about a fixed axis?
Question 9: Mohr's Circle is used to
Question 10: The bending moment diagram represents
Question 11: Which of the following is not a type of support in structural analysis?
Question 12: Shearing force in a beam tends to cause
Question 13: The concept of mass moment of inertia is significant in the analysis of
Question 14: Torsion in circular shafts is analyzed to determine
Question 15: Principal stresses occur where the shear stress is
Question 16: If three forces acting in equilibrium at a point are 3 N, 4 N, and 5 N, the angle between the 3 N and 4 N forces is closest to:
Question 17: The centroid of a rectangle of height h and width w is located at
Question 18: For a thin plate shaped like a quarter circle, the polar moment of inertia at the center of the circle is given by
Question 19: The moment of inertia of a rectangle with base b and height h about an axis through its centroid parallel to the base is
Question 20: A mass undergoes simple harmonic motion (SHM) with an amplitude of 10 cm. Its maximum acceleration is 5 m/s\(^2\). The angular frequency \(\omega\) of the mass is
Correct Answer: 2. 7 rad/s
View Solution
View Solution
Question 21: Why the inertia torque acts in the opposite direction to the accelerating couple?
Question 22: A beam is supported at two points with a uniform load across its length. The type of bending moment diagram this beam will have is
Question 23: A circular shaft subjected to torsion experiences a shear stress \(\tau\). If the radius of the shaft doubles, the shear stress will
Question 24: A particle moves with constant acceleration of 2 m/s\(^2\) starting from rest. Its velocity after 3 seconds is
Question 25: In a 2D stress system on an element, if the normal stresses on the x and y axes are \(\sigma_x\) and \(\sigma_y\), and the shear stress is \(\tau\) (\(\tau_{xy}\)), the normal stress on a plane inclined at 45\(^\circ\) to these axes is:
Question 26: What characterizes a turbulent flow compared to a laminar flow?
Question 27: Which type of flow has no rotation of fluid elements about their center of mass?
Question 28: What does the conservation of mass principle state for a fluid in motion?
Question 29: Euler's equation is used to describe the motion of fluids under the influence of what forces?
Question 30: Bernoulli's equation is applicable under which of the following conditions?
Question 31: What is the primary feature of Couette flow?
Question 32: The Buckingham \(\pi\) theorem is used for which purpose in fluid mechanics?
Question 33: Which type of heat transfer is primarily utilized in furnaces for metallurgical processes?
Question 34: Which equation would best describe the conservation of momentum for fluid flow in pipes?
Question 35: In which scenario is Bernoulli's equation modified to include a term for head loss?
Question 36: The critical Reynolds number for the transition from laminar to turbulent flow in a pipe is approximately
Question 37: A fluid flows through a pipe with a velocity that varies with time at a given point. What type of flow is this?
Question 38: If a fluid has a Reynolds number of 5000 in a pipe. The type of flow is
Question 39: A fluid traveling through a horizontal pipe with a decreasing cross-sectional area experiences what kind of pressure change, assuming inviscid flow?
Question 40: In plane Couette flow, what determines the shear stress between the plates?
Question 41: In a duct, if the flow enters at 1 kg/s and exits at 0.5 kg/s, what additional information is needed to use the continuity equation effectively?
Question 42: Which method is used to improve the efficiency of heat transfer in regenerators?
Question 43: For a fluid element in a steady, uniform flow, which of the following statements is true regarding the flow velocity at any point in the flow field?
Question 44: In heat transfer, the Log Mean Temperature Difference (LMTD) is used in the design of
Question 45: What parameter in boundary layer theory represents the thickness of the layer in which the velocity increases from zero at the surface to 99% of the free stream velocity?.
Question 46: The key factor in classifying furnaces used in metallurgical industries is
Question 47: A cubic crystal has a unit cell edge length of 0.4 nm. What is the volume of the unit cell?
Question 48: If a face-centered cubic (FCC) crystal has a lattice parameter of 0.5 nm, calculate the atomic radius.
Question 49: Calculate the Burgers vector magnitude for a body-centered cubic (BCC) crystal with a lattice constant of 0.3 nm, assuming the dislocation is along the shortest lattice vector.
Question 50: A material is deformed by slip along a plane with a critical resolved shear stress (CRSS) of 150 MPa. What is the applied stress needed if the orientation factor is 0.45?
Question 51: During cold working, a metal experiences a 10% reduction in cross-sectional area. If the original cross-sectional area was 1 cm\(^2\), what is the new cross-sectional area?
Question 52: A material undergoes recrystallization at 250\(^\circ\)C. If the material is heated to 300\(^\circ\)C, what process is most likely occurring?
Question 53: An aluminum alloy sample has a yield stress of 250 MPa and a modulus of elasticity of 70 GPa. What is the strain at yield point?
Question 54: In a tensile test, a ductile material exhibits necking after reaching its ultimate tensile strength. If the ultimate tensile strength is 500 MPa, what happens to the stress in the necked region?
Question 55: A steel bar exhibits a Hall-Petch relationship with a yield strength of 300 MPa when the average grain size is 10 micrometers. What trend in yield strength would you expect if the grain size is reduced to 5 micrometers?
Question 56: A cast iron beam shows a brittle fracture on the stress-strain diagram. What does this indicate about its elongation at break?
Question 57: The primary result of grain growth in a material is
Question 58: The typical characteristic of a stress-strain diagram for cast iron
Question 59: Which of the following is the effect of recrystallization on the properties of a material?
Question 60: The primary mechanism for deformation by twinning is
Question 61: The effect of grain growth on the mechanical properties of a metal is
Question 62: Cold Working is primarily characterized by
Question 63: The main mechanism of plastic deformation in metals is
Question 64: Which of the following crystal system has axes of equal length intersecting at 90-degree angles?
Question 65: The type of defect involves an atom from an impurity substituting for a lattice atom is
Question 66: How does dislocation movement by cross-slip differ from climb?
Question 67: Consider a process where the entropy change of the system is negative. What can be inferred if the process is spontaneous?
Question 68: Identify the equation typically used to describe the efficiency of a cyclic process.
Question 69: How does the Clausius-Clapeyron Equation help in meteorology?
Question 70: A system undergoes isothermal reversible expansion. What is true about the work done by the system?
Question 71: What is the significance of the Gibbs-Helmholtz Equation in chemical thermodynamics?
Question 72: If the enthalpy change for a process at constant pressure is negative, what type of process is it likely to be?
Question 73: How can the concept of maximum work be used to determine the efficiency of a thermodynamic cycle?
Question 74: A reaction's Gibbs free energy change (\(\Delta G\)) is found to be positive at 298 K but becomes negative at 350 K. What does this imply about the reaction?
Question 75: A 10 kg block slides down a frictionless incline from a height of 5 meters. Assuming the gravitational constant g=9.8 m/s\(^2\), calculate the kinetic energy of the block at the bottom of the incline.
Question 76: One mole of an ideal gas expands isothermally and reversibly from 2 liters to 6 liters at a temperature of 300 K. Calculate the work done by the gas. (R=8.314 J/K/mol)
Question 77: An ideal gas is compressed adiabatically, where its initial volume of 3 liters is reduced to 1 liter. If the initial pressure was 1 atm and \(\gamma=5/3\), calculate the final pressure of the gas.
Question 78: Calculate the heat transferred when 50 g of water is heated from 20\(^\circ\)C to 80\(^\circ\)C. The specific heat capacity of water is 4.18 J/\(^\circ\)C\(^{-1}\)g\(^{-1}\)
Question 79: A Carnot engine operates between a hot reservoir at 500 K and a cold reservoir at 300 K. Calculate the efficiency of the engine.
Question 80: A closed system undergoes a process resulting in a work output of 150 J while the heat removed from the system is 100 J. Calculate the change in internal energy of the system.
% Corrected from user key based on standard convention.
View Solution
View Solution
Question 81: During an isothermal expansion, 2 moles of an ideal gas expand from 1 L to 3 L at a constant temperature of 300 K. Using R=8.314 J/mol.K calculate the work done by the gas.
Correct Answer: 2 = -477.8J
View Solution
View Solution
Question 82: Calculate the increase in entropy when two identical containers, each containing 1 mole of an ideal gas at 300 K and 1 atm, are allowed to mix freely.
% Modified based on calculation matching key
% Modified based on calculation matching key
Question 83: During an isothermal expansion of an ideal gas, how does the entropy of the system change?
Question 84: Which process involves an increase in enthalpy due to the system absorbing heat from the surroundings?
Question 85: What does the Third Law of Thermodynamics state about the entropy of a perfect crystal at absolute zero temperature?
Question 86: Consider the properties of ceramics. Which of the following is a reason for their brittleness?
Ionic bonds also contribute.
View Solution
View Solution
Question 87: Design a study to test the effectiveness of different nano-material coatings to improve the wear resistance of industrial cutting tools. What would be a key variable to control?
Question 88: Which factor most significantly influences the optical properties of a material such as refractive index and absorption of light?
Question 89: Evaluate the potential benefits and drawbacks of using ceramic matrix composites (CMCs) in aerospace engine components.
Question 90: Analyze the implications of using a hard magnetic material in the stator of an electric motor instead of a soft magnetic material.
Question 91: Design an experiment to test the effectiveness of nanostructured coatings to improve the corrosion resistance of metals. What would be the primary performance metric to measure?
Question 92: Considering the unique properties of nano materials, evaluate their use in targeted drug delivery systems.
Question 93: Identify a bottom-up approach in nanotechnology:
Question 94: Calculate the electrical conductivity of a copper wire if the number density of free electrons is 8.5x10\(^{28}\) electrons/m\(^3\) and the electron mobility is 0.0035 m\(^2\)/Vs.
Question 95: A paramagnetic substance, in the form of a cube with sides 1 cm, has a magnetic dipole moment of 20 x 10\(^{-6}\) J/T, when a magnetic intensity of 60 x 10\(^3\) A/m is applied. Its magnetic susceptibility is
Question 96: Estimate the surface area of a spherical nanoparticle with a diameter of 10 nm.
Question 97: What is the primary challenge in synthesizing nanoparticles using the bottom-up approach?
Question 98: Which of the following statements best describes the effect of increasing the temperature on the conductivity of an intrinsic semiconductor?
Question 99: What is the primary mechanical advantage of cross-linked polymers over linear polymers?
Question 100: Which factor is most critical when selecting a matrix material for a high-temperature composite application?
Question 101: Which characteristic differentiates thermosetting polymers from thermoplastic polymers?
Question 102: What distinguishes hard magnetic materials from soft magnetic materials in terms of their application?
Question 103: What is a significant environmental challenge associated with the bottom-up approach in nanotechnology?
Question 104: A block of mass 5 kg is placed on a frictionless inclined plane. The angle of inclination of the plane is 30 degrees. Calculate the force required to keep the block in equilibrium.
Question 105: The velocity profile for a Bingham plastic fluid flowing (under laminar conditions) in a pipe.
Question 106: Which of the following properties is typically NOT enhanced by the addition of carbon in steel?
Question 107: Calculate the drift velocity of the free electrons with mobility of 3.5\(\times\)10\(^{-3}\) m\(^2\)/Vs in copper for an electric field strength of 0.5 V/m.
Question 108: If \(\lambda\) is an eigenvalue of a non-singular matrix A . Then the eigenvalue of (adjA) is
Question 109: Let A and B be two real symmetric matrices of order n. Then which of the following is true?
Question 110: If any function is even, in Fourier series it contains
Question 111: Probability that a leap year has 53 Sundays is
Question 112: If \( \vec{a}, \vec{b}, \vec{c} \) are unit vectors, then \( |\vec{a}-\vec{b}|^2 + |\vec{b}-\vec{c}|^2 + |\vec{c}-\vec{a}|^2 \) does not exceed
Question 113: Find the greatest value of the directional derivative of the function \( f = x^2 y z^3 \) at (2,1,-1)
Question 114: By eliminating a \& b from z = ax + by + (a/b) then, P.D.E formed is \hspace{2cm}
Question 115: The particular integral for the differential equation \( (D^2 - 2D + 1)y = x^2 e^{3x} \) is
Question 116: Laplace transform of \( g(t) = \begin{cases} \cos(t-\frac{\pi}{3}), & if t > \frac{\pi}{3}
0, & if t < \frac{\pi}{3} \end{cases} \) is
0, & if t < \frac{\pi}{3} \end{cases} \) is
Question 117: Let X be a continuous random variable denoting the temperature measured. The range of temperature is [0, 100] degree Celsius and let the probability density function of X be f(x) = 0.01 for \(0 \le X \le 100\). The mean is
Question 118: The Laplace transform of the function f(t) = t sin t is
Question 119: If the mean and variance of a binomial variate are 12 \& 4, then the distribution is \hspace{2cm}









































































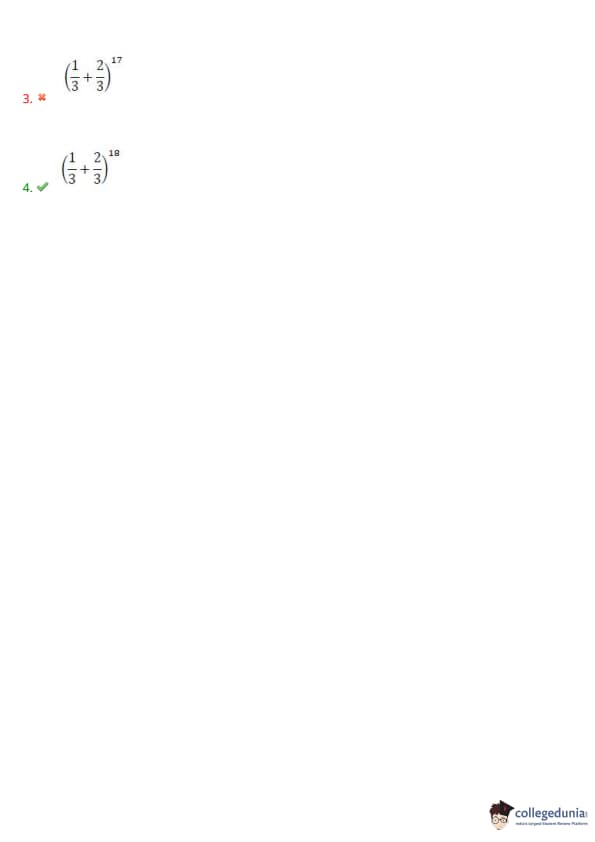







Comments