AP PGECET Question Paper 2025 for Pharmacy is available for download here with answer key and solution PDF. AP PGECET 2025 was conducted from June 6 to June 8 in two shifts.
AP PGECET Question Paper 2025 consists of 120 MCQ-based questions in total carrying 1 mark each to be attempted in the duration of 2 hours.
AP PGECET Pharmacy Question Paper with Solution PDF
Question 1:
The drug ‘Pectin’ belongs to the category of _______.
- (1) polyuronide
- (2) disaccharide
- (3) alkaloid
- (4) Monosaccharide
Question 2:
Source of pale catechu is _______.
- (1) Eucalyptus rostrata
- (2) Uncaria gambir
- (3) Terminalia chebula
- (4) Punica granatum
Question 3:
Trichomes with quadracellular head \& sessible stalk are seen in ___.
- (1) Digitalis
- (2) Belladonna
- (3) Hyoscyamus
- (4) Vasaka
Question 4:
Which of the following crude drug is obtained from mineral origin?
- (1) Digitalis
- (2) Sandalwood
- (3) Nux vomica
- (4) Kaolin
Question 5:
The natural oil of winter green majorly contains ____.
- (1) methyl salicylate
- (2) salicylic acid
- (3) phenol
- (4) \( \alpha \)-napthol
Question 6:
Mangifera indica belongs to the family:
- (1) Phyllanthaceae
- (2) Combretaceae
- (3) Meliaceae
- (4) Anacardiaceae
Question 7:
Steroidal saponins are biosynthesized via ____.
- (1) shikimic acid pathway
- (2) mevalonic acid pathway
- (3) aceto-acetate pathway
- (4) Calvin’s cycle
Question 8:
The swelling index of a crude drug is used to evaluate the ____.
- (1) presence of alkaloids
- (2) moisture content
- (3) mucilage content
- (4) presence of volatile oils
Question 9:
The biological source of cotton fiber is ______.
- (1) Cyamopsis tetragonolobus
- (2) Gossypium barbadense
- (3) Saraca indica
- (4) Arachis hypogaea
Question 10:
Number of isoprene units present in diterpenes are ____.
Question 11:
Which is the specific test for the identification of cardiac glycoside?
- (1) Baljet’s test
- (2) Legal test
- (3) Keller – kiliani test
- (4) 3,5–dinitro benzoic acid test
Question 12:
Match the following correctly.
\[ \begin{array}{ll} A) Goldbeater skin test & 1) Carbohydrates
B) Molisch test & 2) Terpenoids
C) Mayer’s test & 3) Tannins
D) Salkowski test & 4) Alkaloids
\end{array} \]
- (1) A–2; B–3; C–4; D–1
- (2) A–4; B–1; C–2; D–1
- (3) A–3; B–1; C–4; D–2
- (4) A–1; B–4; C–2; D–3
Question 13:
Glass wool is primarily used in pharmaceutical applications for ___.
- (1) drug formulation
- (2) insulation and filtration
- (3) textile production
- (4) capsule coating
Question 14:
An alkaloid containing one of the nitrogen atoms as quaternary nitrogen is:
- (1) Atropine
- (2) Ephedrine
- (3) Tubocurarine
- (4) Mescaline
Question 15:
Which of the following plant cell culture technique is generally regarded as a “closed system” technique?
- (1) Continuous cultures
- (2) Batch suspension cultures
- (3) Semi continuous cultures
- (4) Petri plate cultures
Question 16:
Rasburicase is a newer drug used in gout. It acts by _____.
- (1) decreasing urate synthesis
- (2) increasing urate oxidation
- (3) decreasing intestinal absorption of uric acid
- (4) increasing renal excretion of uric acid
Question 17:
1-[(5-methylpyrazin-2-yl-carboxamide) ethyl phenyl sulphonyl]-3-cyclohexyl urea is _____.
- (1) Glimepiride
- (2) Glipizide
- (3) Glyburide
- (4) Repaglinide
Question 18:
Select the INCORRECT statement with respect to the SAR of adrenergic agonists with specific reference to 3’,5’-dihydroxy ring substitution pattern.
- (1) Increases the drug distribution
- (2) Increases resistance to metabolism by COMT
- (3) Provides selectivity for \(\beta_2\)-receptors
- (4) Orally active bronchodilator
Question 19:
The term “bioisosterism” in drug design aims to maintain or improve the molecule's biological activity by:
- (1) Replacement of a functional group with another that enhances lipophilicity
- (2) Replacement of a chemical group in a molecule with another that has similar physical and chemical properties
- (3) Structural modification to increase water solubility
- (4) Conversion of a drug into a salt form
Question 20:
Pharmacophore modelling involves ______.
- (1) Identifying excipients for formulation development with enhanced bioavailability
- (2) Monitoring plasma levels of drugs for better pharmacodynamics
- (3) Predicting half-life of drugs
- (4) Creating a 3D model of the necessary features to interact with a target
Question 21:
IUPAC name of aspirin is ______.
- (1) Salicylic anhydride
- (2) 2-ethyl salicylate
- (3) 2-acetoxybenzoic acid
- (4) 4-hydroxybenzoic acid
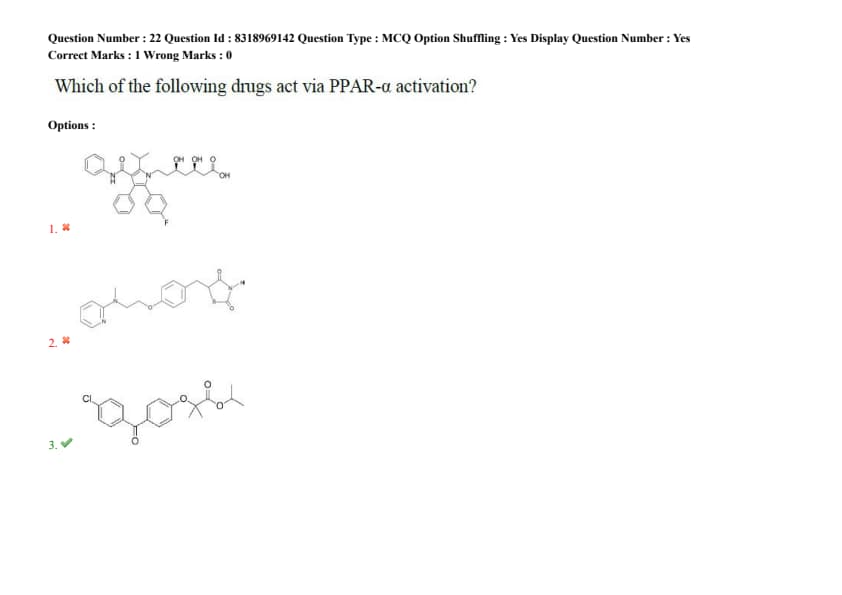
Question 22:
Which of the following drugs act via PPAR-\(\alpha\) activation?
Question 23:
What does “QSAR” stand for in drug design?
- (1) Qualitative Selective-Activity Response
- (2) Quick Structure-Activity Response
- (3) Quantitative Structure-Activity Relationship
- (4) Quantitative Selective-Activity Relationship
Question 24:
What is the primary function of dental abrasives?
- (1) Removing plaque and stains
- (2) Enhancing digestion
- (3) Neutralizing stomach acid
- (4) Regulating blood pressure
Question 25:
Which of the following statements is true regarding the following structure (nevirapine)?
![]()
- (1) It is a nucleoside reverse transcriptase inhibitor (NRTI).
- (2) It binds to an allosteric binding site next to the substrate binding site of reverse transcriptase
- (3) It is an achiral molecule
- (4) It is an example of a second-generation drug of its class
Question 26:
When 6-OH of morphine is replaced by 6-O-acetyl group, then the activity will \hspace{1cm}.
- (1) increase
- (2) decrease
- (3) no effect
- (4) Antagonistic
Question 27:
Which of the following drugs causes severe birth defects due to stereochemical differences in its enantiomers?
- (1) Tamoxifen
- (2) Methamphetamine
- (3) Thalidomide
- (4) Paclitaxel
Question 28:
The antihypertensive drug with a tetrazole nucleus that binds to AT1 receptor with high affinity is \hspace{1cm}.
- (1) diazoxide
- (2) valsartan
- (3) talupidine
- (4) fosinopril
Question 29:
The E/Z nomenclature is used to describe \hspace{1cm} isomerism.
- (1) optical
- (2) geometrical
- (3) chain
- (4) functional group
Question 30:
Which enzyme is targeted by beta-lactam antibiotics?
- (1) DNA gyrase
- (2) Transpeptidase
- (3) Reverse transcriptase
- (4) Topoisomerase
Question 31:
Lipinski’s rule of five is linked with \hspace{2cm}.
- (1) drug’s metabolic pathway
- (2) potency of drug-receptor binding
- (3) drug-likeness for oral bioavailability
- (4) stability under acidic conditions
Question 32:
Which of the following is not the category of cardiotonic agents?
- (1) Calcium sensitizers
- (2) Phosphodiesterase III inhibitors
- (3) Beta-adrenergic antagonists
- (4) Cardiac glycosides
Question 33:
SAR studies of sulfonamides suggest that antibacterial activity is not lost when the \hspace{2cm}.
- (1) sulfonamide group is acetylated
- (2) amine group is replaced with halogens
- (3) aromatic ring is replaced with a non-aromatic moiety
- (4) sulfonyl group is replaced by a carboxyl group
Question 34:
What is the role of ethanol in the preparation of barium sulphate reagent?
- (1) To provide an acidic medium
- (2) To oxidize barium
- (3) To precipitate barium
- (4) To prevent super-saturation
Question 35:
Identify the key step in the synthesis of beta-blockers.
- (1) Condensation of amines with aromatic rings
- (2) Hydrolysis of esters
- (3) Oxidation of alcohols
- (4) Reduction of ketones
Question 36:
As compared to unfractionated heparin, low molecular weight heparins ______:
- (1) are absorbed more uniformly when given subcutaneously
- (2) require more frequent laboratory monitoring
- (3) can be given to patients with heparin induced thrombocytopenia
- (4) predispose to a higher risk of osteopenia
Question 37:
Anxiolytic drugs like lorazepam primarily act on ______ receptors.
- (1) serotonin
- (2) NMDA
- (3) GABA-A
- (4) dopamine
Question 38:
Which of the following is used as anticaries agent?
- (1) Calcium carbonate
- (2) Sodium lauryl sulphate
- (3) Zinc chloride
- (4) Sodium fluoride
Question 39:
The locally acting antacid(s) known for its constipation effect is/are:
- (1) Sodium bicarbonate
- (2) Aluminium hydroxide
- (3) Magnesium hydroxide
- (4) Sucralose
Question 40:
Match the following limit test reagents correctly.
% Matching Table
\begin{tabular{ll
A) Arsenic & 1) Dithizone
B) Lead & 2) Thioglycolic acid
C) Iron & 3) Barium chloride
D) Barium & 4) Mercuric chloride
\end{tabular
- (1) A-4; B-1; C-2; D-3
- (2) A-3; B-2; C-4; D-1
- (3) A-2; B-1; C-4; D-3
- (4) A-3; B-4; C-2; D-1
Question 41:
In general, Kraft point phenomenon is generally not exhibited by which of the following surfactant?
- (1) Tweens
- (2) Sodium lauryl sulphate
- (3) Calcium oleate
- (4) Sodium stearate
Question 42:
The angle of repose is primarily used to determine \hspace{2cm}.
- (1) particle density
- (2) flowability of powders
- (3) surface charge
- (4) particle solubility
Question 43:
A first-order reaction has a half-life of 24 hours. What is its rate constant (k)?
- (1) 0.0289 hr\(^{-1}\)
- (2) 0.693 hr\(^{-1}\)
- (3) 0.5 hr\(^{-1}\)
- (4) 1.2 hr\(^{-1}\)
Question 44:
What is water number?
- (1) Maximum amount of water that can be added to 100 g of a base at a given temperature
- (2) Minimum amount of water that can be added to 100 g of a base at a given temperature
- (3) Minimum amount of water that can be added to 5 g of a base at a given temperature
- (4) Minimum amount of water that can be added to 40 g of a base at a given temperature
Question 45:
What is a molecular phenomenon where by adding a second solute results in an increase in aqueous solubility of poorly soluble solute?
- (1) Peptization
- (2) Hydrotrophy
- (3) Eutectic mixture formation
- (4) Levigation
Question 46:
If pKa of a weak electrolyte is 4.2, how much percentage of it will be ionized at pH of 5.2?
- (1) 50%
- (2) \(>\)50%
- (3) \(<\)50%
- (4) Cannot be predicted
Question 47:
The distance between two tangents on opposite sides of the particle parallel to some fixed direction is called as _____ diameter.
- (1) Martin diameter
- (2) Feret diameter
- (3) Projected area diameter
- (4) Projected number diameter
Question 48:
The density that is determined by displacement of helium is known as _____ density.
- (1) True
- (2) Intrinsic
- (3) Granule
- (4) Bulk
Question 49:
Which sterilization method is suitable for heat-labile parenteral solutions?
- (1) Autoclaving
- (2) Dry heat
- (3) Filtration
- (4) UV radiation
Question 50:
The solubility of the drug will be high when it is in its _____ form.
- (1) stable
- (2) metastable
- (3) unstable
- (4) crystalline
Question 51:
In a suspension, the purpose of a flocculating agent is to _______.
- (1) increase viscosity
- (2) prevent caking
- (3) enhance solubility
- (4) reduce particle size
Question 52:
Which suppository base is water-soluble?
- (1) Cocoa butter
- (2) PEG
- (3) Gelatin
- (4) Witepsol
Question 53:
Vanishing cream is an example of _____.
- (1) w/o emulsion
- (2) o/w
- (3) w/o/w
- (4) Microemulsion
Question 54:
Bulking agent used for parenteral preparation is _____.
- (1) sodium metabisulphite
- (2) benzyl alcohol
- (3) carboxylic acid
- (4) Sorbitol
Question 55:
The disintegration time for sugar coated tablet is _____ minutes.
- (1) 30
- (2) 45
- (3) 60
- (4) 75
Question 56:
Which of the following tablet problems cannot be eliminated by pre-compression?
- (1) Mottling
- (2) Lamination
- (3) Double impression
- (4) Capping
Question 57:
Which of the following is not used for the enteric coating of the tablets?
- (1) Cellulose acetate phthalate
- (2) Hydroxy propyl methyl cellulose
- (3) Shellac
- (4) Eudragit L
Question 58:
Study of adverse effects of drugs and toxic substances in the body is ______.
- (1) Clinical Toxicology
- (2) Biopharmaceutics
- (3) Clinical Pharmacokinetics
- (4) Toxicokinetics
Question 59:
LAL test is used as an in-process control in parenteral preparation to examine presence of ______.
- (1) pyrogens
- (2) endotoxins
- (3) exotoxins
- (4) proteinaceous matter
Question 60:
If the plasma half-life of a drug is 1.386 h and the volume of distribution is 10 L, the total body clearance of the drug without any unit conversion will be _____.
- (1) 5
- (2) 20
- (3) 13.86
- (4) 6.93
Question 61:
The best explanation for the absorption of salicylic acid from intestine, despite being ionized is:
- (1) High surface area of the drug molecules
- (2) Dissolution of drug
- (3) Availability of 1% of the un-ionized form
- (4) Availability of receptors
Question 62:
The area under the serum concentration time curve represents the ______.
- (1) biological half-life of the drug
- (2) amount of drug absorbed
- (3) amount of drug excreted in the urine
- (4) amount of drug in the original dosage form
Question 63:
Fumaric acid is used in gelatin capsule shell is used as _____.
- (1) plasticizer
- (2) antioxidant
- (3) solubilizer
- (4) opacifier
Question 64:
Wurster’s process is also better known as _____.
- (1) rotary plate process
- (2) air suspension coating
- (3) coacervation process
- (4) pan coating
Question 65:
What is the ideal particle size for topical powders?
- (1) 50 to 100 µm
- (2) 150 to 250 µm
- (3) 250 to 500 µm
- (4) Above 1000 µm
Question 66:
A super disintegrant in tablet formulation is _____.
- (1) sodium starch glycolate
- (2) starch
- (3) PVP
- (4) Mg-aluminium silicate
Question 67:
The creatinine clearance in normal 70 kg individuals is typically in the range of ____ mL/min.
- (1) 100–120
- (2) 200–250
- (3) 50–100
- (4) 120–200
Question 68:
A co-solvent used in the preparation of parenteral products is ____.
- (1) benzyl alcohol
- (2) methyl alcohol
- (3) dimethyl acetamide
- (4) phenol
Question 69:
If zeta potential of a suspension is high, then the system will be considered as _____.
- (1) deflocculated
- (2) flocculated
- (3) emulsion
- (4) sedimentation
Question 70:
Non-linear pharmacokinetics is most likely due to _____.
- (1) first-pass metabolism
- (2) saturation of metabolic enzymes
- (3) renal excretion
- (4) high plasma protein binding
Question 71:
Match the following:
\begin{tabular{ll
A) Schedule Y & 1) List of minimum equipment for efficient running of a pharmacy
B) Schedule G & 2) Life period of drugs
C) Schedule N & 3) Requirements and guidelines on clinical trials
& \quad\; for import and manufacture of new drugs
D) Schedule P & 4) List of drugs to be used under medical supervision
\end{tabular
- (1) A–2; B–3; C–4; D–1
- (2) A–4; B–1; C–2; D–3
- (3) A–3; B–4; C–1; D–2
- (4) A–2; B–4; C–2; D–3
Question 72:
The first schedule of the Drugs and Cosmetics Act, 1940 deals with _______.
- (1) standards to be complied with by imported drugs and by drugs manufactured for sale, stocked or exhibited for sale, sold or distributed
- (2) standard to be complied for cosmetics
- (3) authoritative books of Ayurvedic, Siddha and Unani Tibb system
- (4) standards to be complied for medical devices
Question 73:
The Drugs Controller General of India (DCGI) can issue an order to ban a drug on the recommendations of ______.
- (1) Indian Council of Medical Research
- (2) Pharmacy Council of India
- (3) Drugs Technical Advisory Board
- (4) National Pharmaceutical Pricing Authority
Question 74:
_____ is a psychotropic substance under the Narcotics and Psychotropic Substances Act.
- (1) Barbital
- (2) Ampicillin
- (3) Albendazole
- (4) Chloroquine
Question 75:
Who is the Ex-officio Member of Pharmacy Council of India?
- (1) Director, Central Drugs Research Laboratory
- (2) Director General of Health Services
- (3) President, Medical Council of India
- (4) Director, Central Research Institute
Question 76:
Which drug acts as a selective serotonin reuptake inhibitor (SSRI)?
- (1) Amitriptyline
- (2) Fluoxetine
- (3) Diazepam
- (4) Phenelzine
Question 77:
Atropine blocks the effects of acetylcholine at _____ receptors.
- (1) nicotinic
- (2) muscarinic
- (3) alpha-1
- (4) beta-2
Question 78:
Levothyroxine is a synthetic analogue of ______.
- (1) Triiodothyronine (T\textsubscript{3})
- (2) Thyroxine (T\textsubscript{4})
- (3) TSH
- (4) Calcitonin
Question 79:
Penicillins act by inhibiting ______.
- (1) cell membrane synthesis
- (2) DNA gyrase
- (3) cell wall synthesis
- (4) protein synthesis
Question 80:
Which of the following antidiabetic agent primarily enhances insulin sensitivity in target tissues?
- (1) Sulfonylureas
- (2) Biguanides
- (3) Thiazolidinediones
- (4) Alpha-glucosidase inhibitors
Question 81:
Which local anaesthetic is commonly used for spinal anaesthesia due to its long duration of action?
- (1) Lidocaine
- (2) Bupivacaine
- (3) Procaine
- (4) Mepivacaine
Question 82:
Development of drug resistance is not generally associated with overexpression of which of the following transporters?
- (1) BCRP
- (2) ABCB1
- (3) MDR1
- (4) Folate receptors
Question 83:
Which of the following cellular transport requires energy?
- (1) Active transport
- (2) Passive transport
- (3) Facilitated transport
- (4) Pinocytosis
Question 84:
Which of the following is not correct for the \( H_1 \) receptor antagonists?
- (1) Strongly block the increased capillary permeability caused by histamine
- (2) Suppress the flare and itching caused by intradermal injection
- (3) Always suppress the gastric secretion
- (4) Relieve the allergic reactions
Question 85:
Absorption of oral iron preparations can be facilitated by co-administering \hspace{1cm}.
- (1) calcium carbonate
- (2) calcium phosphate
- (3) ascorbic acid
- (4) casein
Question 86:
The weight ratio of T4 and T3 in the combination preparations is \hspace{1cm}.
- (1) 1:3
- (2) 3:1
- (3) 1:4
- (4) 4:1
Question 87:
The antiplatelet action of aspirin involves \hspace{1cm} inhibition.
- (1) arachidonic acid pathway
- (2) ADP pathway
- (3) glycoprotein IIb/IIIa receptor
- (4) phosphodiesterase
Question 88:
The drug that is not metabolized using hydrolysis reaction is \hspace{1cm}.
- (1) aspirin
- (2) lidocaine
- (3) morphine
- (4) procaine
Question 89:
Which receptor type is primarily involved in the "fight or flight" response?
- (1) alpha-1 adrenergic
- (2) beta-2 adrenergic
- (3) Muscarinic
- (4) Nicotinic
Question 90:
Find the direct oral anticoagulant drug from the following.
- (1) Heparin
- (2) Dabigatran
- (3) Warfarin
- (4) Aspirin
Question 91:
Which of the following is true regarding the purpose of taking a medication history of patient?
- (1) To identify potential drug interactions and allergies
- (2) To know the cost of medications
- (3) Both a and b are correct
- (4) Both a and b are incorrect
Question 92:
Which of the following is acting as an antidiarrheal agent?
- (1) Bisacodyl
- (2) Docusate
- (3) Loperamide
- (4) Lactulose
Question 93:
For the emergency treatment of severe hypoglycaemia, which of the following is administered?
- (1) Pramlintide
- (2) Glucagon
- (3) Rosiglitazone
- (4) Metformin
Question 94:
Identify the drug used in androgen replacement therapy.
- (1) Cortisol
- (2) Estrogen
- (3) Progesterone
- (4) Testosterone enanthate
Question 95:
The primary action of diuretics in hypertension management is __________.
- (1) reducing urine output
- (2) reducing volume of blood
- (3) inhibiting calcium channels
- (4) increasing heart rate
Question 96:
When absorption energy is increased, then the shift is called ______.
- (1) Hypochromic shift
- (2) Hyperchromic shift
- (3) Bathochromic shift
- (4) Hypsochromic shift
Question 97:
What is the wavelength of fingerprint region?
- (1) 100–500 cm\(^{-1}\)
- (2) 500–1500 cm\(^{-1}\)
- (3) 1700–2000 cm\(^{-1}\)
- (4) 400–800 cm\(^{-1}\)
Question 98:
Beer Lambert’s law indicates _______.
- (1) absorbance is directly proportional to concentration
- (2) absorbance is indirectly proportional to concentration
- (3) absorbance is indirectly proportional to path length
- (4) absorbance is indirectly proportional to molar absorptivity
Question 99:
In HPLC analysis, what type of column is preferred?
- (1) A column with high HETP and high number of plates
- (2) A column with low HETP and low number of plates
- (3) A column with high HETP and low number of plates
- (4) A column with low HETP and high number of plates
Question 100:
Following are the desirable properties of the liquid phase used in GLC EXCEPT for one of the following. Identify the same.
- (1) It should be inert to the analytes
- (2) It should have high viscosity at operating temperature
- (3) It should have low vapour pressure at the operating temperature
- (4) It should have a high resolving power
Question 101:
Which of the following is a primary function of GLP?
- (1) To ensure the safety of consumers by testing the quality of food products.
- (2) To establish a framework for conducting non-clinical studies, ensuring the quality, integrity, and reproducibility of the data generated.
- (3) To certify the quality of pharmaceutical products after they have been manufactured.
- (4) To ensure the proper storage and disposal of hazardous waste generated in a laboratory.
Question 102:
The electrode potentials are calculated by ________.
- (1) Ilkovic equation
- (2) Stokes equation
- (3) Nernst equation
- (4) Ohm’s Law
Question 103:
ICH stands for ________.
- (1) International Committee on Harmonisation
- (2) International Conference on Harmonisation
- (3) International Council for Harmonisation
- (4) International Council of Harmony
Question 104:
Resolution of HPLC can not be modified by ________.
- (1) decreasing particle size
- (2) decreasing column diameter
- (3) changing flow rate
- (4) changing detector
Question 105:
The errors arising due to use of un-calibrated or improperly calibrated weights are known as:
- (1) Operational and personal errors
- (2) Instrumental and reagent errors
- (3) Errors of methods
- (4) Additive and proportional errors
Question 106:
For the \(pH\) range of 2.8 to 4.6, which indicator should be preferred?
- (1) Phenolphthalein
- (2) Methyl red
- (3) Thymol blue
- (4) Bromophenol blue
Question 107:
The phenomenon called "levelling effect" for weak acids will be observed in ________ solvents.
- (1) aprotic
- (2) protophilic
- (3) protogenic
- (4) amphiprotic
Question 108:
To obtain the best results in any of the quantitative TLC methods, the spots being used should have \( R_f \) values between _______.
- (1) 0.01 to 0.3
- (2) 0.3 to 0.7
- (3) 0.7 to 1.0
- (4) > 1.0
Question 109:
As per the Indian Pharmacopoeia, the test organism for the microbiological assay of amikacin is ______.
- (1) Saccharomyces cerevisiae
- (2) Micrococcus luteus
- (3) Klebsiella pneumoniae
- (4) Staphylococcus aureus
Question 110:
Subpart C of Good Laboratory Practice (GLP) regulations, specifically under 21 CFR Part-58, focuses on ______.
- (1) test and control articles
- (2) organisational and personnel
- (3) testing facilities operations
- (4) Facilities
Question 111:
The relationship between the atoms present in their ground-state and excited-state in flame photometry is given by ______ equation.
- (1) Arrhenius
- (2) Nernst
- (3) Boltzmann
- (4) de Broglie
Question 112:
Which of the following functional group(s) does not show two absorption peaks in IR spectroscopy:
- (1) Anhydride
- (2) Primary amine
- (3) Nitro
- (4) Secondary amine
Question 113:
If 10 mL of 0.1 M sulphuric acid solution requires 20 mL of NaOH solution, then the normality of NaOH solution is:
- (1) 0.1 N
- (2) 0.05 N
- (3) 0.2 N
- (4) 0.4 N
Question 114:
During titration of strong acid with a weak base, which one is correct with respect to conductance?
- (1) It increases till the end point
- (2) It decreases till the end point
- (3) End point cannot be determined using conductimetry
- (4) Conductance not altered
Question 115:
Which substance is commonly used titrant in iodometric titrations?
- (1) Potassium iodide
- (2) Sodium thiosulfate
- (3) Iodine solution
- (4) Potassium permanganate
Question 116:
Which of the following is correct about robustness and ruggedness in method validation?
- (1) Robustness evaluates inter-laboratory variations, while ruggedness evaluates small changes in method parameters
- (2) Ruggedness evaluates inter-laboratory variations, while robustness evaluates small changes in method parameters
- (3) Robustness focuses on analyst variations, while ruggedness evaluates intra-laboratory changes
- (4) Ruggedness focuses on temperature changes, while robustness focuses on pressure changes
Question 117:
Which of the following is a primary standard used in the calibration of spectrophotometers?
- (1) Sodium dichromate
- (2) Sodium chloride
- (3) Potassium dichromate
- (4) Potassium chloride
Question 118:
Find the correct sentence from the following about accuracy and precision in pharmaceutical analysis.
- (1) Accuracy refers to reproducibility, while precision refers to closeness to the true value
- (2) Accuracy refers to closeness to the true value, while precision refers to reproducibility
- (3) Both accuracy and precision refers to the same concept
- (4) Accuracy is related to random errors, while precision is related to systematic errors
Question 119:
Which guideline is commonly referred to for method validation in pharmaceutical analysis?
- (1) ICH Q3A
- (2) ICH Q2 (R1)
- (3) ICH Q1B
- (4) ICH Q4A
Question 120:
What will be change observed in liquid chromatography with increased mobile phase flow rate?
- (1) Decreased pressure
- (2) Increased resolution
- (3) Increased sensitivity
- (4) Reduced retention times




































































































Comments