TS PGECET 2023 Metallurgical Engineering Question Paper with Answer key PDF is available here for download. TS PGECET 2023 was conducted by JNTU Hyderabad on behalf of TSCHE on May 31, 2023. TS PGECET 2023 MT Question Paper consisted of 120 questions carrying 1 mark for each.
TS PGECET 2023 Metallurgical Engineering Question Paper
| TS PGECET 2023 MT Question Paper with Answer Key | Download PDF | Check Solution |

Question 1:
If \(A = \begin{pmatrix} 2019 & 2020 & 2021
2020 & 2021 & 2022
2021 & 2022 & 2023 \end{pmatrix}\), then \(|A| =\)
View Solution
To find the determinant of matrix \( A \), observe the pattern in the rows: each row increases by 1 from the previous row, suggesting possible linear dependence. Perform row operations to simplify:
% Option
(A) \( R_2 \to R_2 - R_1 \):
\[ \begin{pmatrix} 2020 - 2019 & 2021 - 2020 & 2022 - 2021 \end{pmatrix} = \begin{pmatrix} 1 & 1 & 1 \end{pmatrix} \]
Matrix becomes:
\[ \begin{pmatrix} 2019 & 2020 & 2021
1 & 1 & 1
2021 & 2022 & 2023 \end{pmatrix} \]
% Option
(B) \( R_3 \to R_3 - 2R_2 \):
\[ \begin{pmatrix} 2021 - 2 \cdot 1 & 2022 - 2 \cdot 1 & 2023 - 2 \cdot 1 \end{pmatrix} = \begin{pmatrix} 2019 & 2020 & 2021 \end{pmatrix} \]
Matrix becomes:
\[ \begin{pmatrix} 2019 & 2020 & 2021
1 & 1 & 1
2019 & 2020 & 2021 \end{pmatrix} \]
Since Rows 1 and 3 are identical, the matrix is singular, and thus \( |A| = 0 \). Alternatively, computing the determinant via cofactor expansion along the second row confirms this, as the resulting 2x2 determinants yield terms that cancel out. Quick Tip: To quickly assess a determinant, check for linearly dependent rows or columns (e.g., one row being a linear combination of others). If found, the determinant is zero. For numerical matrices, row reduction or cofactor expansion can confirm this efficiently.
Let \(A\) be a \(3 \times 3\) matrix with eigenvalues 0, 0, and 14. Which of the following is true?
View Solution
By the Cayley-Hamilton theorem, a matrix satisfies its characteristic equation. For a 3x3 matrix with eigenvalues \( 0, 0, 14 \), the characteristic polynomial is: \[ p(\lambda) = (\lambda - 0)(\lambda - 0)(\lambda - 14) = \lambda^2 (\lambda - 14) = \lambda^3 - 14\lambda^2 \]
Thus, the characteristic equation is \( \lambda^3 - 14\lambda^2 = 0 \), so: \[ A^3 - 14A^2 = 0 \]
Verify by checking eigenvalues:
For \( \lambda = 0 \): \( 0^3 - 14 \cdot 0^2 = 0 \)
For \( \lambda = 14 \): \( 14^3 - 14 \cdot 14^2 = 2744 - 14 \cdot 196 = 2744 - 2744 = 0 \)
This matches option (3). Other options fail (e.g., for option (4), \( A^3 - 14A = 0 \), test \( \lambda = 14 \): \( 14^3 - 14 \cdot 14 = 2744 - 196 = 2548 \neq 0 \)). Quick Tip: Use the Cayley-Hamilton theorem to form the characteristic polynomial from the eigenvalues. Substitute the matrix \( A \) to find the matrix equation. Verify by testing each eigenvalue in the resulting polynomial to ensure it equals zero.
If \(\vec{f} = 3xy\hat{i} - y^2\hat{j}\), then \(\int_C \vec{f} \cdot d\vec{r}\), where \(C\) is the curve \(y=2x^2\) in the \(xy\)-plane from \((0,0)\) to \((1,2)\), is
View Solution
To compute the line integral, parametrize the curve \(C: y = 2x^2\) using \(x = t\), so \(y = 2t^2\). The position vector is \(\vec{r}(t) = t\hat{i} + 2t^2\hat{j}\), \(0 \leq t \leq 1\). The differential is: \[ d\vec{r} = \frac{d\vec{r}}{dt} dt = (1 \hat{i} + 4t \hat{j}) dt \]
Substitute \(x = t\), \(y = 2t^2\) into \(\vec{f} = 3xy \hat{i} - y^2 \hat{j}\): \[ \vec{f}(t) = 3(t)(2t^2) \hat{i} - (2t^2)^2 \hat{j} = 6t^3 \hat{i} - 4t^4 \hat{j} \]
Compute the dot product: \[ \vec{f} \cdot d\vec{r} = (6t^3 \hat{i} - 4t^4 \hat{j}) \cdot (1 \hat{i} + 4t \hat{j}) dt = (6t^3 \cdot 1 - 4t^4 \cdot 4t) dt = (6t^3 - 16t^5) dt \]
Evaluate: \[ \int_C \vec{f} \cdot d\vec{r} = \int_0^1 (6t^3 - 16t^5) dt = \left[ \frac{6t^4}{4} - \frac{16t^6}{6} \right]_0^1 = \frac{6}{4} - \frac{16}{6} = \frac{3}{2} - \frac{8}{3} = \frac{9 - 16}{6} = -\frac{7}{6} \]
To confirm, note that \(\vec{f}\) is not conservative (curl \(\vec{f} = -5y \hat{k} \neq 0\)), so the integral depends on the path, validating the parametrization approach. Quick Tip: Choose a parametrization that simplifies the curve’s equation for line integrals. Compute \(\vec{f} \cdot d\vec{r}\) carefully. Check if the vector field is conservative by computing the curl; if zero, the integral depends only on endpoints, simplifying to the potential function’s difference.
The series \(x + 2x^2 + 3x^3 + 4x^4 + \dots\) is
View Solution
The series is \(\sum_{n=1}^\infty n x^n\). Apply the ratio test: \[ \lim_{n \to \infty} \left| \frac{(n+1) x^{n+1}}{n x^n} \right| = |x| \lim_{n \to \infty} \frac{n+1}{n} = |x| \cdot 1 = |x| \]
The series converges if \(|x| < 1\) and diverges if \(|x| > 1\). Test endpoints:
At \(x = 1\): \(\sum_{n=1}^\infty n = 1 + 2 + 3 + \dots\), which diverges (harmonic series).
At \(x = -1\): \(\sum_{n=1}^\infty n (-1)^n = -1 + 2 - 3 + 4 - \dots\), which diverges as terms \(|n|\) do not approach 0.
Thus, the series converges for \(|x| < 1\) (i.e., \(-1 < x < 1\)) and diverges for \(|x| \geq 1\). Option (2) aligns, interpreting ``convergent when \(x < 1\)'' as \(|x| < 1\) in the power series context. Alternatively, for \(|x| < 1\), the sum is: \[ \sum_{n=1}^\infty n x^n = x \cdot \frac{d}{dx} \left( \sum_{n=0}^\infty x^n \right) = x \cdot \frac{1}{(1-x)^2} = \frac{x}{(1-x)^2} \]
This confirms convergence for \(|x| < 1\). Quick Tip: For power series \(\sum n x^n\), use the ratio test to find the radius of convergence (\(|x| < 1\)). Test endpoints separately, as convergence depends on the resulting series (e.g., harmonic or alternating). Use geometric series derivatives to compute the sum and verify the convergence interval.
The Fourier series of an odd function of period \(2l\) is
View Solution
For a function \(f(x)\) with period \(2l\), the Fourier series is: \[ f(x) = \frac{a_0}{2} + \sum_{n=1}^{\infty} \left( a_n \cos\frac{n\pi x}{l} + b_n \sin\frac{n\pi x}{l} \right) \]
Since \(f(x)\) is odd (\(f(-x) = -f(x)\)), compute coefficients:
\(a_0 = \frac{1}{l} \int_{-l}^{l} f(x) dx = 0\), as \(f(x)\) is odd.
\(a_n = \frac{1}{l} \int_{-l}^{l} f(x) \cos\frac{n\pi x}{l} dx = 0\), as \(f(x) \cos\frac{n\pi x}{l}\) is odd.
\(b_n = \frac{1}{l} \int_{-l}^{l} f(x) \sin\frac{n\pi x}{l} dx = \frac{2}{l} \int_0^l f(x) \sin\frac{n\pi x}{l} dx\), as \(f(x) \sin\frac{n\pi x}{l}\) is even.
Thus, the series is \(f(x) = \sum_{n=1}^{\infty} b_n \sin\frac{n\pi x}{l}\). For option (1), the coefficient \(b_n = \frac{1}{l} \int_0^{2l} f(x) \sin\frac{n\pi x}{l} dx\) covers one period, and since \(f(x) \sin\frac{n\pi x}{l}\) is even: \[ \int_0^{2l} f(x) \sin\frac{n\pi x}{l} dx = 2 \int_0^l f(x) \sin\frac{n\pi x}{l} dx \]
Thus, \(b_n\) matches the standard form. Options (2) and (3) include cosine terms, and option (4) uses incorrect sine arguments. Quick Tip: For odd functions with period \(2l\), the Fourier series contains only sine terms, as cosine terms vanish due to symmetry. Compute coefficients over \([-l, l]\) or \([0, 2l]\), using even/odd properties to simplify integrals to \([0, l]\).
If from a group of 10 dozens of matchboxes, selecting 5 matchboxes successively with replacement is a random experiment, then the number of elements in the sample space is
View Solution
A group of 10 dozens of matchboxes means \(10 \times 12 = 120\) matchboxes. Selecting 5 matchboxes with replacement means each of the 5 selections has 120 choices, as the matchboxes are returned after each selection. Thus, the sample space size is: \[ 120 \times 120 \times 120 \times 120 \times 120 = 120^5 \]
Other options misinterpret the number of matchboxes: \(10^5\) assumes 10 matchboxes, \(12^5\) assumes one dozen (12 matchboxes), and \(102^5\) is likely a typo. Hence, the correct answer is \(120^5\). Quick Tip: For sampling with replacement, the sample space size is \(n^k\), where \(n\) is the number of choices per trial and \(k\) is the number of trials. Ensure clarity in units (e.g., ``dozens'' means 12 items) to correctly determine \(n\).
Let \(X\) be a random variable that follows the Binomial distribution with expectation \(E(X) = 4\) and variance \(V(X) = 3\). Then the ratio between the probability of success \(p\) and its mode is
View Solution
For a binomial random variable \(X \sim Binomial(n, p)\), we have:
\(E(X) = np = 4\)
\(V(X) = np(1-p) = 3\)
Divide variance by expectation: \[ \frac{V(X)}{E(X)} = \frac{np(1-p)}{np} = 1-p = \frac{3}{4} \implies p = \frac{1}{4} \]
Solve for \(n\): \[ np = 4 \implies n \cdot \frac{1}{4} = 4 \implies n = 16 \]
The mode is \(\lfloor (n+1)p \rfloor = \lfloor (16+1) \cdot \frac{1}{4} \rfloor = \lfloor 4.25 \rfloor = 4\). Thus, the ratio \(p\) to mode is: \[ \frac{1}{4} : 4 = \frac{1}{4} \cdot \frac{1}{4} = \frac{1}{16} = 1:16 \]
Verify: \(V(X) = 16 \cdot \frac{1}{4} \cdot \frac{3}{4} = 3\), which holds. Other options (e.g., 1:4 implies mode = 1) are inconsistent. Quick Tip: For a binomial distribution, use \(E(X) = np\) and \(V(X) = np(1-p)\) to solve for \(n\) and \(p\). The mode is \(\lfloor (n+1)p \rfloor\), but check if \((n+1)p\) is an integer, as the mode may include \(\lfloor np \rfloor\).
If the differential equation \((x^3D^3 + 3x^2D^2 + xD + 8)y = \cos(\log x)\), where \(D^n = \frac{d^n}{dx^n}\), is reduced to a differential equation with constant coefficients, then its auxiliary equation is
View Solution
The equation is a Cauchy-Euler type. Substitute \(x = e^z\), so \(\log x = z\), and transform derivatives: \(x \frac{dy}{dx} = \frac{dy}{dz} = D'y\), where \(D' = \frac{d}{dz}\). Higher derivatives give: \[ x^2 \frac{d^2 y}{dx^2} = D'(D'-1)y, \quad x^3 \frac{d^3 y}{dx^3} = D'(D'-1)(D'-2)y \]
Substitute into \((x^3 D^3 + 3x^2 D^2 + xD + 8)y = \cos(\log x)\): \[ [D'(D'-1)(D'-2) + 3D'(D'-1) + D' + 8]y = \cos z \]
Simplify: \[ D'(D'-1)(D'-2) = D'^3 - 3D'^2 + 2D', \quad 3D'(D'-1) = 3D'^2 - 3D' \] \[ (D'^3 - 3D'^2 + 2D') + (3D'^2 - 3D') + D' + 8 = D'^3 + 8 \]
The equation becomes \((D'^3 + 8)y = \cos z\). The auxiliary equation for the homogeneous part is: \[ m^3 + 8 = 0 \]
Other options do not match the simplified operator. Quick Tip: For Cauchy-Euler equations, use \(x = e^z\) to transform \(x^n \frac{d^n y}{dx^n}\) into \(D'(D'-1)\cdots(D'-n+1)y\). Simplify the resulting operator to find the constant-coefficient auxiliary equation.
If \(\mathcal{L}\{f(t)\} = F(s)\), then \(\mathcal{L}\left\{ \int_0^t \int_0^{u_3} \int_0^{u_2} \int_0^{u_1} f(u) \, du \, du_1 \, du_2 \, du_3 \right\} =\)
View Solution
For a single integral, \(\mathcal{L}\left\{ \int_0^t f(u) \, du \right\} = \frac{F(s)}{s}\). For a four-fold integral: \[ g(t) = \int_0^t \int_0^{u_3} \int_0^{u_2} \int_0^{u_1} f(u) \, du \, du_1 \, du_2 \, du_3 \]
Apply the Laplace transform iteratively:
First: \(\mathcal{L}\left\{ \int_0^t f(u) \, du \right\} = \frac{F(s)}{s}\)
Second: \(\mathcal{L}\left\{ \int_0^t g_1(u_1) \, du_1 \right\} = \frac{1}{s} \cdot \frac{F(s)}{s} = \frac{F(s)}{s^2}\)
Third: \(\frac{F(s)}{s^3}\)
Fourth: \(\frac{F(s)}{s^4}\)
Thus, \(\mathcal{L}\{g(t)\} = \frac{F(s)}{s^4}\). Options (3) and (4) incorrectly involve s-domain integrals. Quick Tip: The Laplace transform of an \(n\)-fold integral of \(f(t)\) is \(\frac{F(s)}{s^n}\). Verify by applying the integral property iteratively, matching the number of integrations to the power of \(s\).
Using the given below values of \(t\) and \(f(t)\), \(\int_1^7 f(t) \, dt\) is evaluated by the Simpson's \(\frac{1}{3}\)rd rule and its value is \(\frac{1369}{3}\). Then the value of \(a\) is
\(t\) 1 2 3 4 5 6 7
\(f(t)\) 81 75 80 83 \(a\) 70 60
View Solution
Simpson's 1/3 rule for \(\int_1^7 f(t) \, dt\) with \(n=6\) intervals (\(h = \frac{7-1}{6} = 1\)) is: \[ \frac{h}{3} \left[ f(1) + 4(f(2) + f(4) + f(6)) + 2(f(3) + f(5)) + f(7) \right] = \frac{1369}{3} \]
Given \(f(1) = 81\), \(f(2) = 75\), \(f(3) = 80\), \(f(4) = 83\), \(f(5) = a\), \(f(6) = 70\), \(f(7) = 60\): \[ \frac{1}{3} \left[ 81 + 4(75 + 83 + 70) + 2(80 + a) + 60 \right] = \frac{1369}{3} \]
Compute: \[ 4(75 + 83 + 70) = 4 \cdot 228 = 912, \quad 81 + 912 + 60 = 1053, \quad 2(80 + a) = 160 + 2a \] \[ \frac{1053 + 160 + 2a}{3} = \frac{1213 + 2a}{3} = \frac{1369}{3} \] \[ 1213 + 2a = 1369 \implies 2a = 156 \implies a = 78 \]
Option (4) is correct. Quick Tip: Simpson's 1/3 rule requires an even number of intervals. Use \(\frac{h}{3} [f(x_0) + 4 \sum f(x_{odd}) + 2 \sum f(x_{even}) + f(x_n)]\), ensuring odd and even terms (excluding endpoints) are correctly indexed.
Which of the following materials are protected by passivation (i.e., formation of a thin adherent film on the surface) from corrosion?
View Solution
Passivation involves a thin, adherent oxide layer that prevents corrosion. Aluminium alloys form a stable \(Al_2O_3\) layer, and stainless steel forms a chromium oxide (\(Cr_2O_3\)) layer, both highly protective. Mild steel forms porous iron oxide (rust), which does not prevent further corrosion. Silver tarnishes slowly (e.g., forming silver sulfide), but its layer is less protective. Thus, only aluminium alloys and stainless steel (option 4) exhibit effective passivation. Quick Tip: Passivation occurs in metals like aluminium and stainless steel, forming stable, adherent oxide layers. Identify metals with self-healing oxide films for corrosion resistance.
Which one of the following is NOT an intensive property?
View Solution
Intensive properties are independent of the amount of substance, while extensive properties depend on it. Temperature, pressure, and refractive index are intensive, as they remain constant regardless of system size (e.g., temperature of water is the same for 1 mL or 1 L). Volume is extensive, as it scales with the amount of substance (e.g., doubling the material doubles the volume). Thus, volume (option 3) is not intensive. Quick Tip: Intensive properties (e.g., temperature, pressure) are independent of system size. Extensive properties (e.g., volume, mass) scale with quantity. Test by checking if doubling the substance changes the property.
In the Helmholtz free energy equation, \(TS\) basically represents
View Solution
The Helmholtz free energy is \(A = U - TS\), where \(U\) is internal energy, \(T\) is temperature, and \(S\) is entropy. The term \(TS\) represents the ``bound'' energy, unavailable for work due to the system's entropy. \(A\) is the free energy available for work at constant temperature and volume. Surface energy relates to interfaces, and activation energy pertains to reaction barriers, neither relevant here. Thus, \(TS\) is bound energy (option 1). Quick Tip: In Helmholtz free energy \(A = U - TS\), \(TS\) is the bound energy unavailable for work due to entropy. \(A\) represents usable energy at constant temperature and volume.
The effect of temperature and pressure on chemical equilibrium can be predicted by
View Solution
Le Chatelier's principle predicts how a chemical equilibrium shifts in response to temperature or pressure changes (e.g., higher pressure favors fewer gas moles, higher temperature favors endothermic reactions). The Van't Hoff equation quantifies temperature effects on the equilibrium constant but not pressure. The law of mass action defines the equilibrium constant, not its response to changes. The Clausius-Clapeyron equation applies to phase equilibria (e.g., vapor pressure vs. temperature), not chemical reactions. Thus, Le Chatelier's principle (option 2) is correct. Quick Tip: Use Le Chatelier's principle for qualitative predictions of equilibrium shifts due to temperature, pressure, or concentration. Use Van't Hoff for quantitative temperature effects and Clausius-Clapeyron for phase equilibria.
Activation energy of a chemical reaction, homogeneous or heterogeneous, is graphically estimated from a plot between [where, \(k\) is the rate constant and \(T\) is the absolute temperature]
View Solution
The Arrhenius equation is \(k = A e^{-E_a / RT}\), where \(k\) is the rate constant, \(A\) is the pre-exponential factor, \(E_a\) is the activation energy, \(R\) is the gas constant, and \(T\) is the absolute temperature. Taking the natural logarithm: \[ \ln k = \ln A - \frac{E_a}{R} \cdot \frac{1}{T} \]
Plotting \(\ln k\) versus \(\frac{1}{T}\) gives a straight line with slope \(-\frac{E_a}{R}\), from which \(E_a\) is calculated. Other options (e.g., \(k\) vs. \(T\)) yield nonlinear plots, unsuitable for direct \(E_a\) estimation. Thus, option 4 is correct. Quick Tip: Use the Arrhenius equation \(k = A e^{-E_a / RT}\). Plot \(\ln k\) vs. \(1/T\) for a straight line, with slope \(-\frac{E_a}{R}\) giving the activation energy. Use Kelvin for temperatures.
Joule is a measure of
View Solution
A joule (J) is the SI unit of energy (\(1 \, J = 1 \, kg \cdot m^2 / s^2\)). Heat energy, the energy transferred due to temperature differences, is measured in joules. Specific heat is in \(J/(kg \cdot K)\), thermal capacity in \(J/K\), and entropy in \(J/K\). Thus, heat energy (option 2) is directly measured in joules. Quick Tip: A joule measures energy, including heat energy. Distinguish from derived units like \(J/(kg \cdot K)\) for specific heat or \(J/K\) for entropy and thermal capacity by checking the quantity’s definition.
\(\frac{\partial H}{\partial P}\) is the mathematical expression for
View Solution
Enthalpy is \(H = U + PV\). The specific heat at constant pressure is \(C_p = \left( \frac{\partial H}{\partial T} \right)_P\), the heat required to raise a unit mass by 1 K at constant pressure. The expression \(\frac{\partial H}{\partial P}\) likely contains a typo, intending \(\left( \frac{\partial H}{\partial T} \right)_P\). For \(\left( \frac{\partial H}{\partial P} \right)_T = V + P \left( \frac{\partial V}{\partial P} \right)_T\), which is zero for ideal gases but nonzero for non-ideal systems, not matching any option directly. Entropy change, Gibbs free energy (\(G = H - TS\)), and Helmholtz free energy (\(A = U - TS\)) are unrelated. Assuming the intended expression, option 1 is correct. Quick Tip: Specific heat at constant pressure is \(C_p = \left( \frac{\partial H}{\partial T} \right)_P\). For \(\frac{\partial H}{\partial P}\), use \(\left( \frac{\partial H}{\partial P} \right)_T = V + P \left( \frac{\partial V}{\partial P} \right)_T\). Verify partial derivative notation in thermodynamic questions.
When the rate of reaction is constant, the reaction is said to be
View Solution
In a zero-order reaction, the rate is constant (\(k\)), independent of reactant concentration \([A]\). For first-order (rate = \(k[A]\)), second-order (rate = \(k[A]^2\)), and third-order (rate = \(k[A]^3\)), the rate decreases as \([A]\) decreases. A constant rate is unique to zero-order reactions (option 1), as seen in processes like enzyme kinetics at high substrate concentrations. Quick Tip: A zero-order reaction has a constant rate, independent of concentration. Plot concentration vs. time (linear for zero-order) to confirm, unlike higher-order reactions where rate depends on concentration.
The migration of atoms in a pure material is known as
View Solution
Self-diffusion is the random movement of atoms in a pure material (e.g., copper atoms in pure copper), typically via vacancies. Substitutional diffusion involves impurity atoms replacing host atoms. Interstitial diffusion involves smaller atoms moving through lattice gaps. Diffusivity is a measure of diffusion rate (\(m^2/s\)), not the process. In a pure material, migration is self-diffusion (option 3). Quick Tip: Self-diffusion occurs in pure materials where identical atoms migrate, often via vacancies. Distinguish from substitutional (impurity atoms) and interstitial (small atoms in gaps) diffusion.
Diffusion is more rapid
View Solution
Diffusion is faster along grain boundaries due to their disordered structure, compared to slower lattice diffusion within grains. Fine-grained materials have smaller grains, increasing grain boundary area per unit volume, thus enhancing diffusion. Coarse grains have fewer boundaries, and diffusion within grains (coarse or fine) is slower. Therefore, diffusion is most rapid along fine grain boundaries (option 4). Quick Tip: Diffusion is faster along grain boundaries than within grains. Fine-grained materials have more boundary area, enhancing diffusion. Compare grain size effects by considering boundary density.
Doping of silicon wafers for integrated electronic circuits involves
View Solution
Doping introduces impurities (e.g., phosphorus, boron) into silicon to modify its electrical properties. This is achieved via solid-state diffusion, where dopant atoms diffuse into the silicon lattice at high temperatures (e.g., 900–1100°C for phosphorus). Solid-state reduction involves chemical reduction, solid-state strengthening enhances mechanical properties, and segregation control manages phase separation, none of which are primary doping methods. Thus, solid-state diffusion (option 1) is correct. Quick Tip: Doping in semiconductors uses solid-state diffusion to incorporate impurities into the lattice. Distinguish from ion implantation, which may precede diffusion, followed by annealing to activate dopants.
The cathode in an electrochemical cell always carries
View Solution
In an electrochemical cell, the cathode is where reduction (gain of electrons) occurs. Electrons flow from the anode (oxidation) to the cathode via the external circuit, making the cathode positive relative to the anode as it attracts electrons. This holds for both galvanic and electrolytic cells (e.g., in a Cu-Zn cell, the copper cathode reduces \(Cu^{2+}\)). The charge is not zero, negative, or electrolyte-dependent. Thus, the cathode carries a positive charge (option 2). Quick Tip: The cathode is where reduction occurs, attracting electrons and carrying a positive charge relative to the anode. Remember: ``Reduction at cathode, oxidation at anode'' (REDOX).
The activation energy of a chemical reaction is
View Solution
Activation energy (\(E_a\)) is the minimum energy barrier for a chemical reaction, per the Arrhenius equation \(k = A e^{-E_a / RT}\). It is always positive, as it represents the energy required to reach the transition state (e.g., ~200 kJ/mol for methane combustion). \(E_a\) is intrinsic and does not change with temperature; temperature affects the rate constant \(k\). Negative \(E_a\) (option 3) is not physically realistic, and options 1 and 2 are incorrect. Thus, option 4 is correct. Quick Tip: Activation energy (\(E_a\)) is always positive, representing the reaction barrier. Temperature affects the rate via \(k = A e^{-E_a / RT}\), but \(E_a\) is constant. Use \(\ln k\) vs. \(1/T\) plots to estimate \(E_a\).
Nernst equation is given by the following relation
View Solution
The Nernst equation is typically \(E = E^0 - \frac{RT}{nF} \ln Q\), relating cell potential to standard potential and reaction conditions. The relation \(\Delta G = -nFE\) connects Gibbs free energy to cell potential, where \(n\) is the number of electrons, \(F\) is Faraday’s constant, and \(E\) is the cell potential. Option 1 is correct, as other options are dimensionally incorrect (2, 3) or have the wrong sign (4). Quick Tip: The Nernst equation is \(E = E^0 - \frac{RT}{nF} \ln Q\), but \(\Delta G = -nFE\) relates free energy to cell potential. Ensure consistent units and signs for electrochemical and thermodynamic quantities.
Partial molal quantities are important in the study of
View Solution
Partial molal quantities (e.g., partial molar volume, Gibbs free energy) describe a component’s contribution to a mixture’s thermodynamic properties. In non-ideal mixtures, complex interactions (e.g., deviations from Raoult’s law) require these quantities to account for properties like volume or free energy. Ideal gases and solutions have simpler mixing rules, and pure compounds lack mixing, making partial molal quantities less relevant. Thus, non-ideal mixtures (option 3) are correct. Quick Tip: Partial molal quantities are critical for non-ideal mixtures, where component interactions cause deviations from ideal behavior. Use them for properties like partial molar Gibbs free energy in complex systems.
Thermal diffusivity is minimum for
View Solution
Thermal diffusivity is a measure of how quickly a material can spread heat. Rubber, being a poor conductor of heat, has a very low thermal diffusivity compared to metals like aluminum, iron, and lead. Quick Tip: Low thermal diffusivity: Poor heat conductors (e.g., rubber).
The dimensionless number relevant in transient heat conduction is
View Solution
The Fourier number (Fo) is a dimensionless number that characterizes transient heat conduction. It represents the ratio of the heat conduction rate to the rate of thermal energy storage. Quick Tip: Transient heat conduction: Fourier number.
The temperature gradient in a fluid flowing over a heated plate will be
View Solution
The temperature difference between the heated plate and the fluid is greatest at the surface of the plate. This leads to a very steep temperature gradient at the surface, which decreases as you move away from the plate into the fluid. Quick Tip: Heated plate: Steepest temperature gradient at the surface.
The intensity of solar radiation on the earth is
View Solution
The average solar radiation reaching the Earth's surface is approximately 450 W/m\(^2\). It can vary depending on location, time of day, and weather conditions. Quick Tip: Solar radiation on Earth: ~450 W/m\(^2\).
Entropy decreases during
View Solution
Crystallization leads to a more ordered state (solid) from a less ordered state (solution), thus decreasing entropy. Rusting, melting, and vaporization all increase entropy as they involve transitions to more disordered states. Quick Tip: Decreased entropy: More ordered state (e.g., crystallization).
Floatability of any mineral is its tendency to attach to
View Solution
Floatability in froth flotation refers to a mineral’s ability to adhere to air bubbles, rising to the surface for separation, driven by hydrophobicity. Reagents (e.g., collectors) enhance this property, making the mineral water-repellent. Hydrophilic minerals attach to water and sink, agitators mix the slurry, and reagents are not attachment targets. For example, chalcopyrite attaches to air bubbles in copper flotation. Thus, air bubbles (option 2) is correct. Quick Tip: Floatability in froth flotation depends on hydrophobicity, enabling attachment to air bubbles. Reagents like collectors enhance this for mineral separation.
Galena is the sulfide of
View Solution
Galena (PbS) is the primary ore of lead, a lead sulfide often containing traces of silver. Zinc's sulfide is sphalerite (ZnS), copper's primary sulfide ore is chalcopyrite (CuFeS\(_2\)), and zirconium's common mineral is zircon (ZrSiO\(_4\)), a silicate. Thus, galena is specifically lead sulfide. Quick Tip: Galena (PbS) is the lead sulfide ore. Know sulfide ores: galena (lead), sphalerite (zinc), chalcopyrite (copper).
Stefan-Boltzmann law applies to
View Solution
The Stefan-Boltzmann law (\(j^* = \sigma T^4\)) gives the power radiated by a body, proportional to its temperature’s fourth power, where \(\sigma\) is the Stefan-Boltzmann constant. It applies to an ideal black body, which absorbs all radiation and emits based on temperature. White bodies reflect all radiation, and red/green bodies are not standard terms. Real bodies approximate black bodies with emissivity \(\epsilon\) (\(j^* = \epsilon \sigma T^4\)). Thus, black body (option 1) is correct. Quick Tip: Stefan-Boltzmann law (\(j^* = \sigma T^4\)) applies to black bodies, ideal absorbers/emitters. Real bodies use emissivity \(\epsilon\) to adjust.
The important factors for producing low silicon pig iron in a blast furnace are
View Solution
In a blast furnace, pig iron is produced by reducing iron ore, but silicon impurities from silica (SiO\(_2\)) in the ore can incorporate into the iron. To minimize silicon content in pig iron:
Higher basicity: Basicity is the ratio of basic oxides (e.g., CaO from limestone) to acidic oxides (e.g., SiO\(_2\)) in the slag. Higher basicity (more CaO) enhances slag formation, which captures silicon as calcium silicate, removing it from the molten iron. The reaction is:
\[ SiO_2 + CaO \rightarrow CaSiO_3 (slag) \]
Lower temperature: High temperatures favor the reduction of SiO\(_2\) to silicon, which dissolves into the iron:
\[ SiO_2 + 2C \xrightarrow{high temp} Si + 2CO \]
Lower temperatures reduce this reaction, favoring iron oxide reduction (Fe\(_2\)O\(_3\) to Fe) over silicon formation, thus lowering silicon content in the pig iron.
Thus, lower temperature and higher basicity are key, making option (3) correct. Options (1) and (4) involve higher temperatures, increasing silicon content. Option (2) with lower basicity reduces slag’s ability to remove silicon. Quick Tip: To reduce silicon in pig iron, use high basicity (more CaO in slag) to trap silicon as silicate and lower temperatures to minimize SiO\(_2\) reduction.
The process using gaseous reductant for the production of DRI is
View Solution
Direct Reduced Iron (DRI), or sponge iron, is produced by reducing iron ore (Fe\(_2\)O\(_3\) or Fe\(_3\)O\(_4\)) using a reductant without melting. The HYL process (Hojalata y Lamina) uses gaseous reductants, primarily a mixture of hydrogen (H\(_2\)) and carbon monoxide (CO), derived from reformed natural gas. The reduction occurs in a shaft furnace:
\[ Fe_2O_3 + 3H_2 \rightarrow 2Fe + 3H_2O \]
\[ Fe_2O_3 + 3CO \rightarrow 2Fe + 3CO_2 \]
SL/RN: Uses solid reductants (coal) in a rotary kiln, not gaseous.
KRUPP-RENN: Involves solid reductants and slag formation, not gaseous.
Mini Blast Furnace: Produces molten pig iron using coke, not DRI with gaseous reductants.
Thus, only the HYL process uses gaseous reductants, making option (4) correct. Quick Tip: For DRI production, identify the reductant type: HYL uses gaseous H\(_2\) and CO, while SL/RN and KRUPP-RENN use solid reductants like coal.
In which crusher is the "angle of nip" term used?
View Solution
The "angle of nip" is the angle between the two crushing surfaces in a roll crusher, where the material is gripped and crushed. It determines the maximum size of the feed material that the crusher can process efficiently without slipping. In a roll crusher, the angle of nip typically ranges from 15° to 30°, ensuring effective crushing.
Jaw crusher: Uses a different mechanism (compression between fixed and moving jaws) and does not typically refer to an angle of nip.
Cone crusher: Involves conical crushing surfaces but focuses on the "closed side setting" rather than nip angle.
Gyratory crusher: Similar to cone crushers, it uses a different terminology for crushing geometry.
Thus, the angle of nip is specific to roll crushers, making option (2) correct. Quick Tip: The angle of nip is a key parameter in roll crushers, determining the feed size and crushing efficiency.
Matte is produced during smelting of __________ ores.
View Solution
Matte is a molten mixture of metal sulfides formed during the smelting of sulfide ores, typically copper or nickel. In nickel extraction, smelting nickel sulfide ores (e.g., pentlandite, (Ni,Fe)\(_9\)S\(_8\)) produces a nickel-copper sulfide matte, which is an intermediate product further refined to extract pure nickel.
Magnesium: Extracted from oxide or chloride ores (e.g., magnesite, MgCO\(_3\)) via reduction or electrolysis, not smelting.
Uranium: Processed from oxide ores (e.g., uraninite, UO\(_2\)) through leaching, not smelting.
Titanium: Extracted from oxide ores (e.g., ilmenite, FeTiO\(_3\)) via the Kroll process, not smelting.
Only nickel sulfide ores produce matte during smelting, making option (2) correct. Quick Tip: Matte, a sulfide-rich intermediate, is formed during smelting of nickel or copper sulfide ores.
Yellow cake consists of
View Solution
Yellowcake is a uranium concentrate produced during the extraction of uranium from its ores, primarily consisting of uranium oxides, mainly triuranium octoxide (U\(_3\)O\(_8\)) and uranium dioxide (UO\(_2\)). It appears yellow due to its oxidized form and is a key intermediate in the nuclear fuel cycle.
TiO\(_2\): Titanium dioxide, used as a pigment, not related to uranium.
WO\(_2\): Tungsten dioxide, not associated with yellowcake.
MgO: Magnesium oxide, unrelated to uranium processing.
Thus, yellowcake consists of UO\(_2\) (and U\(_3\)O\(_8\)), making option (1) correct. Quick Tip: Yellowcake is uranium oxide (UO\(_2\), U\(_3\)O\(_8\)), a key product in uranium extraction for nuclear fuel.
Killed steels have the following defect mostly
View Solution
Killed steels are fully deoxidized using deoxidizers like aluminum or silicon, preventing gas evolution (e.g., CO) during solidification, which eliminates blow holes. However, deoxidation leads to significant volume shrinkage as the molten steel solidifies, forming a central shrinkage cavity known as a pipe at the top of the ingot.
Blow holes: Common in rimming steels, not killed steels, due to gas entrapment.
Segregation: Occurs in all steels but is not the primary defect in killed steels.
Longitudinal cracks: Less common, related to casting stresses, not deoxidation.
Thus, pipe formation is the most prominent defect, making option (2) correct. Quick Tip: Killed steels, fully deoxidized, avoid blow holes but form pipe cavities due to shrinkage during solidification.
The AOD process is suitable for production of __________ steels.
View Solution
The Argon Oxygen Decarburization (AOD) process is a refining method used primarily for stainless steel production. It involves blowing a mixture of argon and oxygen into molten steel to reduce carbon content to very low levels while minimizing chromium oxidation, which is critical for stainless steel’s corrosion resistance.
Tool steels: Produced via other methods (e.g., electric arc furnace) for high hardness, not typically AOD.
Ausforming: A thermomechanical process, not a steel type.
Maraging steels: High-strength steels produced differently, not via AOD.
Thus, the AOD process is suited for stainless steels, making option (2) correct. Quick Tip: The AOD process refines stainless steel by decarburizing with argon-oxygen, preserving chromium content.
Hairline cracks in steel ingots are caused by
View Solution
Hairline cracks, or flakes, in steel ingots result from hydrogen embrittlement. Hydrogen, absorbed from moisture or furnace gases during steelmaking, dissolves in molten steel. As the steel cools and solidifies, hydrogen’s solubility decreases, leading to trapped gas pockets that create internal stresses and microcracks. Vacuum degassing or controlled cooling reduces this defect.
- Sulphur: Leads to hot shortness by forming low-melting FeS, not hairline cracks.
- Carbon: Influences hardness but doesn’t cause cracks directly.
- Silicon: Acts as a deoxidizer, not a crack initiator.
Thus, hydrogen causes hairline cracks, making option (1) correct. Quick Tip: Hydrogen embrittlement causes hairline cracks in steel; mitigate with vacuum degassing or slow cooling.
The Pidgeon process is the pyrometallurgical process used in the extraction of
View Solution
The Pidgeon process extracts magnesium from dolomite (CaMg(CO\(_3\))\(_2\)) or magnesite (MgCO\(_3\)). The ore is calcined to form MgO, which is reduced with ferrosilicon (Si) at ~1200°C under vacuum:
\[ 2MgO + Si \rightarrow 2Mg + SiO_2 \]
Magnesium vapor is condensed into solid metal.
- Ti: Extracted via the Kroll process (MgCl\(_2\) reduction).
- Cu: Smelted from sulfide ores.
- In: Recovered via hydrometallurgy or electrolysis.
Thus, magnesium is extracted, making option (3) correct. Quick Tip: The Pidgeon process uses ferrosilicon to reduce MgO under vacuum for magnesium extraction.
High refractoriness of refractory bricks means that it has a
View Solution
Refractoriness is the ability of refractory bricks to resist high temperatures without melting or softening. High refractoriness indicates a high melting point (high resistance to fusion), critical for applications in furnaces and kilns.
- Spalling resistance: Resistance to thermal shock-induced cracking, distinct from refractoriness.
- Porosity: Affects insulation but not directly the melting point.
Thus, high resistance to fusion is correct, making option (3) correct. Quick Tip: Refractoriness measures a refractory’s ability to resist melting at high temperatures.
The panel test in refractories is used to investigate the following property:
View Solution
The panel spalling test evaluates a refractory’s resistance to thermal shock, or spalling, where rapid temperature changes cause cracking or flaking. The test simulates furnace conditions by cycling a refractory panel between high and low temperatures.
- Compression: Tested via strength tests.
- Corrosion: Assessed through chemical exposure.
- Thermal conductivity: Measured via heat transfer tests.
Thus, spalling resistance is correct, making option (3) correct. Quick Tip: The panel test checks refractories’ ability to withstand thermal shock, preventing spalling.
Which of the following processes is also called the Thomas-Gilchrist process?
View Solution
The Thomas-Gilchrist process, or basic Bessemer process, is a steelmaking method using a converter with a basic lining (e.g., dolomite, MgO) to remove phosphorus from pig iron. Phosphorus forms P\(_2\)O\(_5\), which reacts with the lining to form slag:
\[ P_2O_5 + 3CaO \rightarrow Ca_3(PO_4)_2 (slag) \]
- Crucible: Produces high-purity steel, not for phosphorus removal.
- Acid Bessemer: Uses acidic linings, unsuitable for phosphorus.
- Basic open hearth: A different process with a basic hearth.
Thus, basic Bessemer is correct, making option (3) correct. Quick Tip: The Thomas-Gilchrist process (basic Bessemer) removes phosphorus using a basic-lined converter.
Which of the following furnaces works on the principle of a transformer?
View Solution
An induction furnace operates on the principle of electromagnetic induction, akin to a transformer. A primary coil, carrying alternating current (AC), generates a fluctuating magnetic field that induces eddy currents in the conductive metal charge (acting as the secondary coil). These eddy currents produce heat via Joule heating (\( P = I^2 R \)), melting the metal. Key features include:
- Efficiency: High energy transfer with minimal heat loss.
- Applications: Melting steel, aluminum, and copper in foundries.
- Comparison to others:
- Blast furnace: Uses chemical reduction with coke to produce iron, no electromagnetic induction.
- Cupola furnace: Melts iron via combustion of coke, a fuel-based process.
- Salt bath furnace: Heats metal in molten salt baths, typically for heat treatment, using gas or electric resistance heating.
Thus, only the induction furnace resembles a transformer’s operation, making option (3) correct. Quick Tip: Induction furnaces use transformer-like electromagnetic induction to melt metals efficiently.
Minimum waste gases are produced in the following furnace:
View Solution
Electric arc furnaces (EAFs) produce minimal waste gases because they generate heat via electric arcs (10,000–30,000°C) between graphite electrodes and the metal charge, without fuel combustion. Waste gases are limited to:
- Small amounts of \(\ce{CO}\) from carbon electrode oxidation or charge impurities.
- Minor slag-related fumes (e.g., metal oxides).
Typical EAF emissions are <0.5 kg/ton of steel, compared to several hundred kg/ton for combustion-based furnaces. Key features include:
- Operation: Melts scrap steel or direct reduced iron, ideal for recycling.
- Environmental benefit: Lower \(\ce{CO2}\) footprint (0.4–0.6 tons CO₂/ton steel vs. 1.8–2.0 for blast furnaces).
- Comparison to other furnaces:
- Blast furnace: Emits large volumes of \(\ce{CO}\), \(\ce{CO2}\), and \(\ce{SO2}\) (e.g., 400–600 m³/ton of pig iron) from coke combustion and ore reduction.
- Open hearth furnace: Burns fossil fuels (gas/oil), producing \(\ce{CO2}\), \(\ce{CO}\), and \(\ce{NOx}\) (emissions ~200–300 kg/ton steel).
- Kiln: Combustion-driven (e.g., cement kilns release ~900 kg CO₂/ton clinker), with additional gases from material decomposition (e.g., \(\ce{CaCO3 -> CaO + CO2}\)).
The EAF’s reliance on electricity ensures the least waste gas production, making option (3) correct. Quick Tip: Pick electric arc furnaces for eco-friendly melting with minimal gas emissions.
‘Take care of the slag, it will take care of the metal’ is applicable in
View Solution
In steelmaking, slag is a molten oxide layer (e.g., \(\ce{CaO}\), \(\ce{SiO2}\)) critical for refining molten metal. Proper slag composition (basicity index ~1.5–3) removes impurities like sulfur (\(\ce{S + CaO -> CaS}\)) and phosphorus (\(\ce{2P + 5FeO + 4CaO -> Ca4P2O9}\)), protects against oxidation, and minimizes gas absorption (e.g., \(\ce{N2}\)). For example, in basic oxygen furnaces (BOF), slag reduces phosphorus from 0.1% to <0.02%. In iron making (e.g., blast furnace), slag forms but primarily handles silica and alumina, with less focus on metal quality. Annealing (heat treatment) and flame hardening (surface hardening) involve no slag. Thus, slag control is most critical in steelmaking, making option (2) correct. Quick Tip: Optimize slag in steelmaking to purify and protect the metal.
The strongest deoxidizer is
View Solution
Deoxidizers remove dissolved oxygen from molten steel to prevent porosity and inclusions. Strength is determined by affinity for oxygen, measured by the standard free energy of oxide formation (\(\Delta G^\circ\)). Calcium has the highest affinity (\(\ce{Ca + O -> CaO}\), \(\Delta G^\circ \approx -600 \, kJ/mol at 1600°C\)), followed by aluminum (\(\ce{2Al + 3O -> Al2O3}\), \(\Delta G^\circ \approx -550 \, kJ/mol\)), silicon (\(\ce{Si + 2O -> SiO2}\), \(\Delta G^\circ \approx -400 \, kJ/mol\)), and manganese (\(\ce{Mn + O -> MnO}\), \(\Delta G^\circ \approx -300 \, kJ/mol\)). Calcium’s high reactivity makes it the strongest, though it’s used sparingly due to cost and volatility. Aluminum and silicon are common, while manganese is weaker, often used with others. Thus, option (3) is correct. Quick Tip: Calcium tops deoxidizers: Ca \(>\) Al \(>\) Si \(>\) Mn for oxygen removal.
Puddling process is for converting
View Solution
The puddling process, a 19th-century method, converts pig iron (3–4% carbon) into wrought iron (<0.1% carbon) by oxidizing carbon in a reverberatory furnace. Workers stir molten pig iron with iron oxide (\(\ce{Fe2O3}\)), which reacts: \(\ce{C + Fe2O3 -> 2Fe + CO}\), reducing carbon content. The resulting low-carbon iron is ductile and fibrous. Steelmaking (0.2–2% carbon) uses different processes (e.g., Bessemer). Alloy steel production involves adding elements (e.g., Cr, Ni) to mild steel, not puddling. Lead extraction uses smelting, not puddling. Thus, option (3) is correct. Quick Tip: Puddling turns high-carbon pig iron into low-carbon wrought iron.
For a combination of strength, ductility, and weldability, which of the following materials are chosen?
View Solution
Case hardened steels (e.g., low-carbon steels with carburized surfaces) offer high surface hardness (600–800 HV) for strength and wear resistance, while the ductile core (20–30% elongation) ensures toughness and weldability. Welding is feasible before or after hardening with proper techniques (e.g., low-hydrogen electrodes). Maraging steels (e.g., 18Ni-250) are ultra-strong (2000 MPa) but require specialized welding due to high alloy content, reducing weldability. Ceramics are brittle (low ductility, non-weldable). Plastics lack metallic strength (tensile strength <100 MPa). Thus, option (2) is correct. Quick Tip: Case hardened steels balance hard surfaces with ductile, weldable cores.
Grinding wheels use ceramic materials because of their:
View Solution
Grinding wheels use ceramics (e.g., \(\ce{Al2O3}\), \(\ce{SiC}\)) for their high hardness (2000–3000 HV) and compressive strength (>2000 MPa), enabling abrasion of metals (e.g., steel, 500–700 HV). Hardness ensures cutting efficiency, while strength resists fracture under grinding forces. Toughness and wear resistance are secondary, as ceramics are brittle (fracture toughness ~2–5 MPa\(\sqrt{m}\)). Machinability and ductility are undesirable, as they’d cause deformation or dulling. Thus, option (1) is correct. Quick Tip: Ceramic grinding wheels excel due to extreme hardness and strength.
If the concentration of point defects increases in a crystal, then its configurational entropy:
View Solution
Configurational entropy (\( S = k \ln \Omega \), where \( k \) is Boltzmann’s constant and \( \Omega \) is the number of microstates) measures disorder in a crystal. Point defects (e.g., vacancies, interstitials) increase possible atomic arrangements. For \( N \) lattice sites and \( n \) vacancies, the number of configurations is:
\[ \Omega = \frac{N!}{n!(N-n)!} \]
As \( n \) increases, \( \Omega \) grows, raising entropy. No decrease occurs unless defects order (unlikely). Thus, option (3) is correct. Quick Tip: Point defects boost crystal disorder, increasing configurational entropy.
Which of the following is the softest?
View Solution
Hardness in iron-carbon phases depends on crystal structure and composition. Ferrite (\(\alpha\)-iron, BCC) is the softest (~100–150 HV) due to low carbon content (\(<\)0.02%) and high ductility. Austenite (FCC, ~200–300 HV) is harder but less stable at room temperature. Pearlite (ferrite + cementite lamellae, ~250–350 HV) is harder due to cementite. Cementite (\(\ce{Fe3C}\), ~800–1000 HV) is brittle and hardest. Thus, option (2) is correct. Quick Tip: Ferrite is softest in iron-carbon phases; cementite is hardest.
At equilibrium, the maximum number of phases in a three-component system at constant pressure is:
View Solution
The Gibbs phase rule is \( F = C - P + 2 \), where \( F \) is degrees of freedom, \( C \) is components, and \( P \) is phases. At constant pressure, it becomes:
\[ F = C - P + 1 \]
For a three-component system (\( C = 3 \)) at an invariant point (\( F = 0 \)):
\[ 0 = 3 - P + 1 \implies P = 4 \]
Thus, the maximum number of phases at equilibrium is 4, as in a ternary eutectic. Option (4) is correct. Quick Tip: Max phases at constant pressure: \( P = C + 1 \) (ternary system, \( P = 4 \)).
For an ideal hexagonal close-packed (HCP) structure, the c/a ratio and packing efficiency, respectively, are
View Solution
In an ideal hexagonal close-packed (HCP) structure, atoms are arranged to maximize packing efficiency. The c/a ratio is the ratio of the height (\( c \)) to the basal edge length (\( a \)) of the hexagonal unit cell. For ideal HCP, atoms touch along the c-axis and in the basal plane, yielding:
\[ c/a = \sqrt{\frac{8}{3}} \approx 1.633 \]
The packing efficiency is the fraction of the unit cell volume occupied by atoms. HCP, like face-centered cubic (FCC), has a packing efficiency of 74%, calculated as:
\[ Packing efficiency = \frac{Volume of atoms in unit cell}{Volume of unit cell} = \frac{2 \cdot \frac{4}{3}\pi r^3}{Unit cell volume} \approx 0.74 or 74% \]
- Option (1): 52% is the packing efficiency of a simple cubic lattice, not HCP.
- Option (3): 1.733 is incorrect for ideal HCP; 68% corresponds to body-centered cubic (BCC).
- Option (4): 1.733 is incorrect for c/a.
Thus, option (2) is correct. Quick Tip: For ideal HCP, remember c/a = 1.633 and packing efficiency = 74%, same as FCC.
Which one of the following elements alloyed with iron is a ferrite stabilizer?
View Solution
Ferrite stabilizers expand the stability range of ferrite (BCC \(\alpha\)-iron) in the iron-carbon phase diagram. Silicon promotes ferrite formation by raising the critical transformation temperature and narrowing the austenite (\(\gamma\)-phase, FCC) field.
- Nickel: Austenite stabilizer, expands the \(\gamma\)-phase field.
- Manganese: Austenite stabilizer, lowers the \(\gamma\)-to-\(\alpha\) transition temperature.
- Carbon: Austenite former, stabilizes \(\gamma\)-phase and forms carbides.
Thus, silicon is the ferrite stabilizer, making option (4) correct. Quick Tip: Ferrite stabilizers (e.g., Si, Cr) favor BCC \(\alpha\)-iron; austenite stabilizers (e.g., Ni, Mn) favor FCC \(\gamma\)-iron.
Which one of the following is the correct decreasing sequence of quenching power for quenchants used in heat treatment of steels?
View Solution
Quenching power is the ability of a quenchant to rapidly extract heat from steel, determined by its heat transfer coefficient.
- Brine (salt water): Highest quenching power due to salt reducing vapor formation, enhancing heat transfer.
- Water: High quenching power but forms a vapor blanket, reducing efficiency compared to brine.
- Oil: Lower heat transfer rate, gentler quenching.
- Air: Lowest quenching power, slowest cooling.
The sequence Brine > Water > Oil > Air reflects decreasing heat transfer efficiency, making option (3) correct. Quick Tip: Quenching power decreases as: Brine > Water > Oil > Air, based on heat transfer efficiency.
How many effective numbers of atoms are present per unit cell in a body-centered cubic (BCC) lattice structure?
View Solution
In a body-centered cubic (BCC) lattice, atoms are located at:
- 8 corners, each shared by 8 unit cells (\( 8 \times \frac{1}{8} = 1 \)).
- 1 atom at the center, fully within the unit cell (\( 1 \)).
Total effective atoms per unit cell:
\[ 1 + 1 = 2 \]
- Option (1): Incorrect, as BCC has more than 1 atom.
- Option (3): Incorrect, no standard lattice has 3 atoms.
- Option (4): Incorrect, FCC has 4 atoms.
Thus, option (2) is correct.
Quick Tip: BCC unit cell: 1 corner atom (shared) + 1 center atom = 2 atoms.
Increasing electron beam energy in a transmission electron microscope will
View Solution
Increasing electron beam energy in a transmission electron microscope (TEM) increases the energy of incident electrons, affecting sample interactions:
- Higher energy electrons penetrate deeper, increasing the interaction volume and exciting more inner-shell electrons, leading to greater X-ray emission (characteristic X-rays).
- Magnification: Depends on lens settings, not beam energy directly.
- Contrast: May decrease slightly due to reduced scattering, but not the primary effect.
- Resolution: Improves (smaller de Broglie wavelength, \( \lambda \propto \frac{1}{\sqrt{E}} \)), contradicting option (3).
Thus, increased X-ray emission is the most direct consequence, making option (4) correct. Quick Tip: Higher TEM beam energy boosts X-ray emission due to increased electron-sample interactions.
X-rays are
View Solution
X-rays are electromagnetic waves, consisting of photons with no electric charge. Unlike charged particles (e.g., electrons, protons), which experience Lorentz forces in electric or magnetic fields (\( \vec{F} = q(\vec{E} + \vec{v} \times \vec{B}) \)), X-rays are unaffected by these fields and travel in straight lines.
- Options (1)–(3): Incorrect, as only charged particles are deflected by electric and/or magnetic fields.
Thus, option (4) is correct. Quick Tip: X-rays, being neutral electromagnetic waves, are unaffected by electric or magnetic fields.
The strength to density ratio is called
View Solution
Specific strength is defined as the ratio of a material’s strength (e.g., ultimate tensile strength, \(\sigma\)) to its density (\(\rho\)):
\[ Specific strength = \frac{\sigma}{\rho} \]
It quantifies a material’s strength per unit weight, critical for lightweight applications (e.g., aerospace).
- Specific gravity: Ratio of a material’s density to water’s density, dimensionless.
- Specific stiffness: Young’s modulus (\(E\)) divided by density (\(E/\rho\)).
- Specific modulus: Often synonymous with specific stiffness.
Thus, option (2) is correct. Quick Tip: Specific strength (\(\sigma/\rho\)) measures a material’s strength relative to its weight.
Martensite has a crystal lattice structure
View Solution
Martensite, a hard phase formed in steel during rapid quenching, has a body-centered tetragonal (BCT) crystal structure. The BCT structure is a distorted version of body-centered cubic (BCC), caused by carbon atoms trapped in the lattice, elongating one axis. This distortion contributes to martensite’s high hardness.
- BCC: Ferrite’s structure, less distorted.
- FCC: Austenite’s structure.
- HCP: Found in some metals (e.g., Mg), not steel phases.
Thus, option (1) is correct. Quick Tip: Martensite’s BCT structure, a distorted BCC, causes its high hardness in quenched steel.
Cyaniding is a process of
View Solution
Cyaniding is a case hardening process where steel is heated in a cyanide salt bath (~800–900°C), allowing carbon and nitrogen to diffuse into the surface, forming a hard, wear-resistant layer.
- Tempering: Reduces brittleness in quenched steel.
- Normalizing: Refines grain structure via controlled cooling.
- Spherodizing: Produces spheroidal carbides for machinability.
Thus, option (2) is correct. Quick Tip: Cyaniding hardens steel surfaces by diffusing carbon and nitrogen.
The name of the test used to evaluate the hardenability of steel is
View Solution
The Jominy end quench test measures steel’s hardenability, the ability to form martensite upon quenching. A standardized steel bar is heated to the austenitizing temperature, quenched at one end with water, and hardness is measured along its length to assess the depth of hardening.
- Fatigue test: Evaluates resistance to cyclic loading.
- Creep test: Measures deformation under constant stress at high temperatures.
- Tensile test: Determines strength and ductility.
Thus, option (1) is correct. Quick Tip: Jominy test measures steel hardenability via hardness gradient after end quenching.
Frank-Read source
View Solution
A Frank-Read source is a mechanism for dislocation multiplication in crystals during plastic deformation. A dislocation segment, pinned at two points (e.g., by defects), bows out under shear stress, forming a loop that generates new dislocations, enabling continued plastic flow.
- Hinders movement: Applies to obstacles like precipitates, not Frank-Read sources.
- Polygonization: Rearrangement of dislocations into low-energy walls, unrelated to Frank-Read.
- Dislocation climb: Involves vacancy diffusion, not multiplication.
Thus, option (1) is correct. Quick Tip: Frank-Read source multiplies dislocations, enabling plastic deformation in crystals.
Hume-Rothery’s rule predicts the existence of solid solubility. Which of the following factors is NOT included?
View Solution
Hume-Rothery rules determine conditions for significant solid solubility in metallic alloys:
1. Crystal structure: Solute and solvent must have similar crystal structures.
2. Atomic size: Atomic radii difference \(<\) 15%.
3. Electronegativity: Similar electronegativity minimizes compound formation.
4. Valence: Similar valence electron counts favor solubility.
Atomic weight is not a direct factor, as solubility depends on structural and electronic properties, not mass.
Thus, option (2) is correct. Quick Tip: Hume-Rothery rules for solid solubility: same crystal structure, similar atomic size, electronegativity, and valence (not atomic weight).
When the wavelength of the incident X-ray increases, then the angle of diffraction
View Solution
Bragg’s law governs X-ray diffraction:
\[ n\lambda = 2d\sin\theta \]
where \( \lambda \) is the X-ray wavelength, \( d \) is the interplanar spacing, \( \theta \) is the diffraction angle, and \( n \) is the order (constant). For fixed \( d \) and \( n \), if \( \lambda \) increases, \( \sin\theta \) must increase to maintain equality, thus \( \theta \) increases.
- Decreases: Incorrect, as \( \sin\theta \propto \lambda \).
- Constant: Incorrect, as \( \theta \) varies with \( \lambda \).
- No systematic variation: Incorrect, as the relationship is systematic.
Thus, option (2) is correct. Quick Tip: Per Bragg’s law, longer X-ray wavelength increases the diffraction angle (\( \theta \)).
Alnico alloy is a
View Solution
Alnico alloys, composed of aluminum (Al), nickel (Ni), cobalt (Co), and iron (Fe), with minor additives, are permanent magnetic alloys known for high coercivity and strong, stable magnetic fields, used in magnets for motors and sensors.
- Soft magnetic alloy: Low coercivity, easily magnetized/demagnetized (e.g., permalloy).
- Semi-magnetic alloy: Not a standard term.
- Nanomagnetic alloy: Refers to nanoscale magnetic materials, not Alnico.
Thus, option (2) is correct. Quick Tip: Alnico alloys are permanent magnets due to high coercivity.
Quartz is a
View Solution
Quartz (SiO\(_2\)) is a piezoelectric material, generating an electric charge under mechanical stress due to its non-centrosymmetric crystal structure. It’s widely used in oscillators, sensors, and watches.
- Superplastic: Exhibits high ductility at specific conditions (e.g., some alloys).
- Ferroelectric: Shows spontaneous polarization (e.g., BaTiO\(_3\)), unlike quartz.
- Creep-resistant: Resists deformation under constant stress, not a primary quartz property.
Thus, option (2) is correct. Quick Tip: Quartz generates electric charge under stress, making it piezoelectric.
Diamond can be classified as a good
View Solution
Pure diamond, a crystalline form of carbon, is an excellent electrical insulator due to its wide band gap (~5.5 eV), which prevents electron conduction under normal conditions.
- Conductor: Requires free electrons (e.g., metals like Cu).
- Superconductor: Exhibits zero resistance at low temperatures (e.g., NbTi), not diamond.
- Semiconductor: Has a smaller band gap (e.g., Si, ~1.1 eV); doped diamond can be semiconducting, but pure diamond is not.
Thus, option (4) is correct. Quick Tip: Pure diamond’s wide band gap makes it an electrical insulator.
The Curie temperature of Nickel is
View Solution
The Curie temperature is the point above which a ferromagnetic material loses its permanent magnetism, becoming paramagnetic. For nickel, this occurs at approximately 358°C (631 K).
- 770°C, 910°C, 1123°C: Far exceed nickel’s Curie temperature; iron’s is ~770°C.
Thus, option (4) is correct. Quick Tip: Nickel loses ferromagnetism above its Curie temperature, 358°C.
SmCo\(_5\) is an example of a
View Solution
SmCo\(_5\) (samarium-cobalt) is a rare-earth permanent magnetic material with high coercivity and magnetic strength, used in high-performance magnets for motors and sensors.
- Ceramic: Non-metallic, inorganic materials (e.g., Al\(_2\)O\(_3\)).
- Refractory: Heat-resistant materials (e.g., zirconia).
- Insulator: Non-conductive materials; SmCo\(_5\) is conductive.
Thus, option (3) is correct. Quick Tip: SmCo\(_5\) is a high-strength permanent magnet material.
Which of the following materials is easily shattered by a hammer?
View Solution
Cast iron is brittle due to its high carbon content (~2–4 % C), often in the form of graphite flakes in gray cast iron, causing it to shatter under impact.
- Copper: Ductile, deforms plastically.
- Steel: Tough and ductile, resists fracturing.
- Magnetite: A brittle mineral (Fe\(_3\)O\(_4\)), but less relevant in this context compared to cast iron, a common engineering material.
Thus, option (3) is correct. Quick Tip: Cast iron’s brittleness makes it prone to shattering under impact.
The Rockwell hardness tester on the C scale uses
View Solution
The Rockwell C scale (HRC), used for hard materials like tempered steel, employs a 120° diamond cone indenter (Brale indenter) with a 150 kg major load. Hardness is calculated based on indentation depth.
- Square base diamond pyramid: Used in Vickers hardness testing.
- 10 mm steel ball: Used in Rockwell B scale for softer materials.
- 3 mm steel ball: Not standard in Rockwell testing.
Thus, option (1) is correct. Quick Tip: Rockwell C uses a 120° diamond cone for testing hard materials.
Nanomaterials have a large ratio between
View Solution
Nanomaterials, with dimensions in the 1–100 nm range, have a high surface area to volume ratio due to their small size. For a sphere of radius \( r \), surface area (\( A = 4\pi r^2 \)) and volume (\( V = \frac{4}{3}\pi r^3 \)) yield:
\[ \frac{A}{V} = \frac{3}{r} \]
As \( r \) decreases, \( A/V \) increases, enhancing properties like reactivity and catalysis.
- Volume to surface area: Inverse ratio, decreases with smaller size.
- Length to surface area, surface area to length: Not standard metrics for nanomaterials.
Thus, option (2) is correct. Quick Tip: Nanomaterials’ high surface area to volume ratio drives their unique properties.
Devices used to operate at high temperatures require
View Solution
Creep is the slow, permanent deformation of a material under constant stress at high temperatures. Devices like turbine blades require high creep resistance to maintain shape and function.
- Abrasion resistance: Relevant for wear, not high-temperature deformation.
- Thermal conductivity: Important for heat transfer, not structural stability.
- Ductility and machinability: Useful for forming but not critical for high-temperature operation.
Thus, option (3) is correct. Quick Tip: High-temperature devices need creep resistance to prevent deformation.
Which of the following materials exhibit high specific strength and fracture toughness?
View Solution
Specific strength (strength-to-density ratio, \( \sigma/\rho \)) and fracture toughness (resistance to crack propagation, \( K_{Ic} \)) are critical for structural applications. Composites, such as carbon-fiber-reinforced polymers or metal-matrix composites, are engineered to combine high specific strength (e.g., from fibers) and improved fracture toughness (e.g., via matrix-fiber interactions).
- Ceramics: High strength but low fracture toughness due to brittleness.
- Plastics: Low specific strength and moderate toughness.
- Metals: Good toughness and moderate specific strength, but composites often outperform in specific strength.
The provided answer (4) Metals is incorrect, as composites better satisfy both criteria in modern applications (e.g., aerospace). Thus, option (3) is correct. Quick Tip: Composites excel in specific strength and fracture toughness for lightweight applications.
Dispersion hardening techniques are done mainly by __________ process.
View Solution
Dispersion hardening strengthens materials by dispersing fine, hard particles (e.g., oxides) in a metal matrix to impede dislocation motion. Powder metallurgy is the primary method, involving mixing, compacting, and sintering powders to achieve uniform particle distribution.
- Casting: Produces coarse microstructures, unsuitable for fine dispersions.
- Welding: Joins materials, not used for hardening.
- Rolling: Deforms materials, not for particle dispersion.
Thus, option (3) is correct. Quick Tip: Powder metallurgy enables uniform dispersion hardening for strength.
The most common carbide powders used in cemented carbides are
View Solution
Cemented carbides, or hardmetals, are composites of hard carbide particles in a metal binder (e.g., Co). Tungsten carbide (WC) and titanium carbide (TiC) are the most common carbides due to their extreme hardness and wear resistance, used in cutting tools and wear-resistant parts.
- SiC, CaC: SiC is used in abrasives, CaC is not standard in cemented carbides.
- B\(_4\)C, V\(_3\)C: B\(_4\)C is for armor, V\(_3\)C is rare.
- MoC, B\(_4\)C: MoC is less common, B\(_4\)C is not typical.
Thus, option (1) is correct. Quick Tip: Cemented carbides rely on WC and TiC for hardness and wear resistance.
Stiffness is the property of a material by virtue of which it resists
View Solution
Stiffness, quantified by Young’s modulus (\( E \)), measures a material’s resistance to elastic deformation under load, following Hooke’s law: \( \sigma = E\epsilon \), where \( \sigma \) is stress and \( \epsilon \) is strain. A higher \( E \) indicates less deformation for a given load.
- Dendritic growth: Occurs during solidification, unrelated to mechanical properties.
- Decay: Refers to chemical or radioactive breakdown, not mechanical resistance.
- Doping: Alters semiconductor properties, irrelevant to stiffness.
Thus, option (1) is correct. Quick Tip: Stiffness (Young’s modulus) resists elastic deformation under load.
The respective units for dislocation density and stress intensity factor are
View Solution
- Dislocation density: Defined as the number of dislocation lines per unit area, with units m\(^{-2}\) (e.g., 10\(^{12}\) m\(^{-2}\) in deformed metals).
- Stress intensity factor (\( K \)): Measures stress concentration at a crack tip, with units MPa.m\(^{1/2}\), combining stress (MPa) and crack length (m\(^{1/2}\)).
- Option (1), (2): m\(^2\) is incorrect (area, not inverse area).
- Option (4): MPa.m is incorrect for stress intensity factor.
Thus, option (3) is correct. Quick Tip: Dislocation density: m\(^{-2}\); stress intensity factor: MPa.m\(^{1/2}\).
How is Burgers vector related to the edge dislocation line?
View Solution
The Burgers vector (\( \vec{b} \)) of an edge dislocation defines the lattice distortion’s magnitude and direction. For an edge dislocation, \( \vec{b} \) is perpendicular to the dislocation line. In contrast, for a screw dislocation, \( \vec{b} \) is parallel to the dislocation line.
- Inclined: Occurs in mixed dislocations, not pure edge.
- Parallel: Incorrect for edge dislocations.
- Either: Incorrect, as edge dislocations have a fixed orientation.
Thus, option (1) is correct. Quick Tip: Edge dislocation: Burgers vector perpendicular; screw dislocation: parallel.
With respect to the matrix of Al-Cu alloys, G-P zones are
View Solution
Guinier-Preston (G-P) zones in Al-Cu alloys are coherent precipitates, meaning their crystal lattice aligns continuously with the aluminum matrix, creating strain fields that enhance strength through precipitation hardening.
- Incoherent: No lattice continuity, typical of larger precipitates.
- Semi-coherent: Partial matching, seen in later stages (e.g., \(\theta'\)).
- Chemically indistinguishable: Incorrect, as G-P zones are Cu-rich.
Thus, option (1) is correct.
Quick Tip: G-P zones are coherent, strengthening Al-Cu alloys via lattice strain.
As compared to the engineering stress-engineering strain curve, the true stress-true strain curve for a given material is
View Solution
- Engineering stress (\( \sigma_e = F/A_0 \)) and strain (\( \epsilon_e = \Delta L/L_0 \)) use original area (\( A_0 \)) and length (\( L_0 \)).
- True stress (\( \sigma_t = F/A \)) and strain (\( \epsilon_t = \ln(1 + \epsilon_e) \)) account for instantaneous area (\( A \)) and length. As necking reduces \( A \), \( \sigma_t > \sigma_e \), and \( \epsilon_t < \epsilon_e \) (since \( \ln(1 + x) < x \)).
Thus, the true stress-true strain curve lies above (higher stress) and to the left (lower strain) of the engineering curve.
- Below/right: Incorrect, as true stress is higher.
- Crosses: Incorrect, as curves diverge.
- Identical: Only at negligible strains.
Thus, option (1) is correct. Quick Tip: True stress-strain curve lies above and left due to necking effects.
A plastically deformed metal crystal at low temperature exhibits a wavy slip line pattern due to
View Solution
Wavy slip lines in plastically deformed metals at low temperatures result from cross-slip, where dislocations move between intersecting slip planes. This is prevalent in metals with many slip systems (e.g., BCC and FCC metals like Fe, Cu), allowing dislocations to glide across multiple planes, creating wavy patterns.
- Dislocation pile-up: Causes stress concentration, not wavy lines.
- Low stacking fault energy: Hinders cross-slip (e.g., in brass), leading to planar slip.
- Dislocation climb: Requires high temperatures and vacancy diffusion, irrelevant at low temperatures.
Thus, option (2) is correct.
Quick Tip: Wavy slip lines indicate cross-slip in metals with multiple slip systems.
Fatigue cracks mostly initiate at the
View Solution
Fatigue cracks typically initiate at the surface due to higher stress concentrations from cyclic loading, often at surface imperfections (e.g., scratches, notches). Surfaces experience maximum tensile stresses in bending or torsion, common fatigue modes.
- Inclusions, pores: Can initiate cracks internally, but surface initiation dominates due to environmental exposure and stress.
- Soft spots: Not standard; may imply weaker regions, but less common than surface defects.
Thus, option (1) is correct.
Quick Tip: Fatigue cracks usually start at surface imperfections under cyclic stress.
Which one of the following is FALSE for creep deformation?
View Solution
- Option (1): False. The minimum creep rate occurs in the secondary stage (stage II), where a steady-state balance exists, not in the primary stage (stage I), where the rate decreases.
- Option (2): False. Creep resistance typically *increases* with smaller grain sizes, as more grain boundaries impede grain boundary sliding and diffusion, key creep mechanisms at high temperatures.
- Option (3): True. Coble creep involves diffusion along grain boundaries, dominant at lower temperatures.
- Option (4): True. Nabarro-Herring creep involves vacancy diffusion through the lattice, dominant at higher temperatures.
Since the question asks for the false statement, and option (2) is incorrect (creep resistance increases with smaller grains), it is the answer. Note: Option (1) is also false, but option (2) is the provided correct answer, likely due to context or emphasis on grain size effects.
Thus, option (2) is correct. Quick Tip: Smaller grains enhance creep resistance by hindering grain boundary sliding.
Fibrous fracture occurs in
View Solution
Fibrous fracture, characterized by a rough, fibrous appearance (e.g., cup-and-cone in tensile tests), occurs in ductile materials (e.g., low-carbon steel, Al) due to extensive plastic deformation and microvoid coalescence before failure.
- Hard materials: Often brittle, showing cleavage or granular fractures.
- Brittle materials: Exhibit smooth, cleavage fractures (e.g., ceramics).
- Viscous materials: Typically polymers or liquids, not associated with fibrous fracture.
Thus, option (1) is correct. Quick Tip: Fibrous fracture signals ductility with significant plastic deformation.
Critical resolved shear stress in a single crystal is calculated by applying
View Solution
Schmid’s law calculates the critical resolved shear stress (\( \tau_R \)) in a single crystal, stating:
\[ \tau_R = \sigma \cos\phi \cos\lambda \]
where \( \sigma \) is the applied stress, \( \phi \) is the angle between the normal to the slip plane and the stress axis, and \( \lambda \) is the angle between the slip direction and the stress axis. This determines the stress required to initiate slip.
- Bragg’s law: Applies to X-ray diffraction.
- Hooke’s law: Relates stress and strain in elastic deformation.
- Coulomb’s law: Governs electrostatic forces, irrelevant here.
Thus, option (4) is correct. Quick Tip: Schmid’s law links applied stress to shear stress for crystal slip.
Deformation twins are formed in metals when deformed at
View Solution
Deformation twinning occurs when slip (dislocation glide) is restricted, favoring twinning as an alternative plastic deformation mechanism. High strain rates and low temperatures suppress dislocation motion (e.g., in BCC or HCP metals like Fe, Ti), promoting twinning.
- High strain rate, high temperature: Slip dominates due to thermal activation.
- Low strain rate, low temperature: Twinning possible in some cases, but high strain rate enhances twinning likelihood.
- Low strain rate, high temperature: Slip and climb dominate, not twinning.
Thus, option (1) is correct. Quick Tip: Twinning occurs at high strain rates and low temperatures when slip is restricted.
The patenting process is a
View Solution
Patenting is a heat treatment process for cold-drawn steel wires, involving austenitization (heating to ~850–950°C) followed by rapid cooling (e.g., in a lead bath at ~500°C) to form a fine pearlite microstructure, enhancing strength and ductility for applications like springs.
- Wire drawing: Mechanical forming, not heat treatment.
- Coating: Surface treatment (e.g., galvanizing), unrelated to patenting.
- Deep drawing: Sheet metal forming, not wire processing.
Thus, option (2) is correct. Quick Tip: Patenting enhances wire strength and ductility via heat treatment.
Coble creep is related to
View Solution
Coble creep is a creep mechanism dominated by vacancy diffusion along grain boundaries, prevalent at lower temperatures and smaller grain sizes compared to Nabarro-Herring creep. Its creep rate is inversely proportional to grain size cubed (\( \dot{\epsilon} \propto 1/d^3 \)).
- Lattice diffusion: Drives Nabarro-Herring creep.
- Pipe diffusion: Occurs along dislocation cores, less relevant.
- Dislocation climb: Involves lattice diffusion, not grain boundaries.
Thus, option (3) is correct. Quick Tip: Coble creep relies on grain boundary diffusion, favored at lower temperatures.
The plane strain fracture toughness parameter, \( K_{Ic} \), has the units
View Solution
The plane strain fracture toughness (\( K_{Ic} \)) quantifies a material’s resistance to crack propagation under plane strain conditions, with units derived from stress (\( MPa = Pa \)) and crack length (\( \sqrt{m} \)):
\[ K_{Ic} units = Pa \cdot m^{1/2} = MPa \cdot m^{1/2} \]
- MPa.m: Incorrect, implies stress times length.
- MPa.m\(^2\): Incorrect, not physically meaningful.
- \(\sqrt{MPa.m}\): Incorrect unit form.
Thus, option (1) is correct. Quick Tip: \( K_{Ic} \) units: MPa\(\sqrt{m}\), measuring fracture toughness.
Fatigue failure may occur commonly in
View Solution
Fatigue failure occurs under cyclic loading, and all options are susceptible. However, airplanes are particularly prone due to frequent cyclic stresses (e.g., pressurization cycles, wing vibrations) and critical safety requirements, making fatigue a primary design concern.
- Rail bridges: Experience cyclic loads but are designed for long-term durability.
- Ships: Face cyclic wave loads, but fatigue is less frequent than in airplanes.
- Tool bits: Undergo cyclic loading in machining, but their smaller scale and frequent replacement make fatigue less critical than in airplanes.
The provided answer (4) tool bits is less accurate, as airplanes’ high-stakes cyclic loading makes them more commonly associated with fatigue failure. Thus, option (1) is correct. Quick Tip: Airplanes are highly susceptible to fatigue due to cyclic stresses.
A notch may be the main cause of fatigue failure due to
View Solution
Notches act as stress concentrators, significantly reducing fatigue life by initiating cracks. The Griffith theory explains brittle fracture and crack propagation, stating that cracks grow when the energy release rate exceeds the material’s fracture energy, which is applicable to fatigue crack initiation at notches under cyclic loading.
- Orowan theory: Relates to plastic deformation and dislocation strengthening, not directly to notches or fatigue.
- Wood’s concept: Not a standard term in fatigue; possibly a misreference to notch effects, but less specific than Griffith.
- Jungham principle: Not recognized in materials science.
The provided answer (3) Wood’s concept is incorrect, as Griffith’s theory is the established framework for crack-related fatigue failure. Thus, option (2) is correct. Quick Tip: Notches cause fatigue failure via stress concentration, explained by Griffith’s theory.
A jet engine turbine block is normally manufactured by
View Solution
Jet engine turbine blades (often referred to as “blocks” in context) require complex geometries, high-temperature superalloys (e.g., Ni-based), and precise tolerances. Investment casting (lost-wax casting) is ideal, producing intricate shapes with excellent surface finish and dimensional accuracy.
- Forging: Used for simpler shapes, less suitable for intricate blades.
- Shell molding: Less precise, used for larger castings.
- Rolling: Produces sheets or bars, not complex components.
Thus, option (3) is correct. Quick Tip: Investment casting produces precise turbine blades for jet engines.
The ductile to brittle transition for steels depends on
View Solution
The ductile-to-brittle transition temperature (DBTT) in steels marks the shift from ductile (plastic deformation) to brittle (cleavage fracture) behavior. Higher strain rates increase the DBTT, promoting brittle fracture by limiting dislocation motion and plastic flow.
- Tensile strength: Influences strength but not directly DBTT.
- Shear modulus: Relates to elastic properties, not fracture mode.
- Damping capacity: Absorbs vibration energy, unrelated to DBTT.
Thus, option (3) is correct. Quick Tip: Higher strain rates raise the DBTT, favoring brittle fracture in steels.
The widest application of laser welding is in
View Solution
Laser welding’s high precision, minimal heat-affected zone, and ability to join small components make it ideal for the electronics industry (e.g., circuit boards, microelectronics).
- Heavy industry: Uses arc or resistance welding for large structures.
- Process industry: Focuses on chemical processing, less welding-intensive.
- Structural work: Employs conventional welding for robustness.
Thus, option (1) is correct. Quick Tip: Laser welding excels in electronics for precision and minimal heat effects.
A riser is not required for the casting of
View Solution
Grey cast iron’s graphitization during solidification (forming graphite flakes) causes a volume expansion that compensates for shrinkage, often eliminating the need for a riser.
- White cast iron: Solidifies with shrinkage, requiring a riser.
- Al-4% Cu: Exhibits significant shrinkage, needing a riser.
- Al-12% Si: Near-eutectic, with less shrinkage, but typically requires a riser.
Thus, option (1) is correct. Quick Tip: Grey cast iron’s graphitization reduces the need for casting risers.
Draft allowance given to a pattern is for
View Solution
Draft allowance is a slight taper (typically 0.5°–2°) applied to vertical surfaces of a pattern, enabling easy withdrawal from the mold without damaging the sand or mold cavity.
- Liquid state/solidification shrinkage: Compensated by risers or pattern oversizing, not draft.
- Core placement support: Handled by core prints, not draft allowance.
Thus, option (2) is correct. Quick Tip: Draft allowance ensures smooth pattern removal in casting molds.
In which process is a wax pattern used?
View Solution
Investment casting (lost-wax casting) uses a wax pattern, which is coated with a ceramic slurry to form a mold. The wax is melted out, leaving a cavity for molten metal.
- Shell molding: Uses resin-coated sand, no wax.
- Die casting: Uses metal molds, no patterns.
- Sand casting: Uses wooden or metal patterns, not wax.
Thus, option (3) is correct. Quick Tip: Wax patterns define investment casting’s precision molds.
Which of the following shapes of powder particles would tend to have the lowest interparticle friction?
View Solution
Spherical particles minimize interparticle friction due to their low surface area-to-volume ratio and smooth, rounded surfaces, reducing contact points during flow or compaction in powder metallurgy.
- Flakes: High surface area, high friction due to interlocking.
- Cubic, triangular: Angular shapes increase friction via edge contacts.
Thus, option (1) is correct. Quick Tip: Spherical powders reduce friction for better flow in powder metallurgy.
Which of the following techniques involves melting of the materials?
View Solution
Gas atomization involves melting a material (e.g., metal) and disintegrating the molten stream into fine droplets using a high-velocity gas jet, solidifying into powders for powder metallurgy. The provided option “General atomization” likely refers to “Gas atomization,” a common technique.
- Thermal decomposition: Breaks compounds via heat, no melting.
- Precipitation: Forms solids from solutions, no melting.
- Attrition: Mechanical grinding, no melting.
Thus, option (4) is correct. Quick Tip: Gas atomization melts materials for powder production.
The riser is designed such that the melt in the riser solidifies
View Solution
A riser supplies molten metal to the casting during solidification to compensate for shrinkage. It is designed (via size, shape, or insulation) to solidify after the casting, ensuring a continuous feed of liquid metal.
- Before casting: Would deplete the riser prematurely.
- Same time: Insufficient to fully compensate shrinkage.
- Irrespective: Incorrect, as riser timing is critical.
Thus, option (3) is correct. Quick Tip: Risers solidify after castings to feed shrinkage.
Among the welding processes:
{[1] Laser Beam Welding
{[2] Submerged Arc Welding
{[3] Metal Inert Gas Welding
The width of the heat-affected zone in decreasing order is
View Solution
The heat-affected zone (HAZ) width depends on the welding process’s heat input and concentration.
- Submerged Arc Welding (SAW): High heat input (large current, slow travel speed) creates a wide HAZ, suitable for thick sections.
- Metal Inert Gas Welding (MIG): Moderate heat input (arc welding with filler wire) results in a narrower HAZ than SAW.
- Laser Beam Welding (LBW): Highly focused, low heat input (high energy density, fast welding) produces the narrowest HAZ, ideal for precision welds.
Thus, the order of decreasing HAZ width is SAW \(>\) MIG \(>\) LBW.
Thus, option (2) is correct. Quick Tip: HAZ width decreases with focused heat: SAW > MIG > LBW.
Core prints are used to
View Solution
Core prints are extensions on a pattern that create recesses in the mold to anchor and position cores accurately, ensuring they remain stable during casting.
- Produce cores: Cores are made separately, not by prints.
- Help removal: Core removal relies on shakeout or chemical means, not prints.
- Supply heat: Irrelevant, as prints are for positioning.
Thus, option (3) is correct. Quick Tip: Core prints anchor cores in casting molds.
Zipper cracks occur
View Solution
Zipper cracks are internal defects in continuously cast strips, forming in the central portion due to tensile stresses during solidification, often from uneven cooling or shrinkage.
- Edges: May have edge cracks, not zipper cracks.
- Anywhere: Incorrect, as zipper cracks are central.
- Roll surface: Relates to roll damage, not cast defects.
Thus, option (1) is correct. Quick Tip: Zipper cracks form centrally in continuously cast strips.
Typical products made by impact extrusion are
View Solution
Impact extrusion forms hollow, thin-walled tubular components (e.g., aluminum cans, toothpaste tubes) by striking a metal slug with a high-velocity punch, forcing material to flow around the punch.
- Strips/sheets: Made by rolling, not extrusion.
- Wires/rods: Produced by wire drawing or conventional extrusion.
- Circular rings: Typically forged or machined, not extruded.
Thus, option (3) is correct.
Quick Tip: Impact extrusion creates hollow tubular products.
Springback in a sheet metal bending operation is due to
View Solution
Springback occurs when a bent sheet metal partially returns to its original shape after the bending force is removed, due to the elastic recovery of the material within the elastic portion of its stress-strain response.
- Overstraining: Implies excessive plastic deformation, not elastic recovery.
- Overbending: A technique to compensate for springback, not its cause.
- Thick component: Influences springback magnitude but isn’t the cause.
Thus, option (2) is correct. Quick Tip: Springback results from elastic recovery in sheet metal bending.
Which one of the following is difficult to solder?
View Solution
Stainless steel’s surface chromium oxide layer resists solder wetting, making it difficult to solder without aggressive fluxes (e.g., acid-based) or surface preparation (e.g., abrasion).
- Copper, Brass: High solderability due to clean, oxide-free surfaces with standard fluxes.
- Silver: Readily soldered, often used in solder alloys.
Thus, option (2) is correct. Quick Tip: Stainless steel soldering requires special fluxes to remove oxide layers.
The common flux used in brazing is
View Solution
Borax (Na\(_2\)B\(_4\)O\(_7\)) is a widely used brazing flux, dissolving oxides on metal surfaces (e.g., copper, steel) and promoting filler metal flow at high temperatures (\(\sim\)600–900°C).
- Sodium nitrate, Silver nitrate: Used in chemical or soldering processes, not brazing.
- Barium: Not a flux; likely a typo for barium compounds, which are rare in brazing.
Thus, option (4) is correct. Quick Tip: Borax flux cleans surfaces for brazing filler flow.
Alligatoring is a defect commonly observed in
View Solution
Alligatoring is a surface defect in rolled products (e.g., slabs, sheets), resembling alligator skin, caused by non-uniform deformation, high frictional stresses, or internal defects (e.g., segregation) splitting the material.
- Extrusion, Drawing: Produce defects like surface cracks or center bursts, not alligatoring.
- Forging: May cause laps or folds, not alligatoring.
Thus, option (1) is correct. Quick Tip: Alligatoring signals non-uniform rolling stresses.
Segregation is decreased by
View Solution
Segregation, the non-uniform distribution of alloying elements in castings, is reduced by soaking the ingot at high temperatures (e.g., ~1100–1200°C) for extended periods, allowing diffusion to homogenize the composition.
- Riser: Compensates shrinkage, not segregation.
- Chaplets: Support cores, unrelated to segregation.
- Sulphur: Can increase segregation or embrittlement in steels.
Thus, option (4) is correct. Quick Tip: Soaking homogenizes ingots to reduce segregation.
Solidification is slower when the casting has
View Solution
Solidification rate depends on heat dissipation, primarily through the casting’s surface. A high volume-to-surface area ratio (e.g., thick castings) reduces the relative surface area available for heat loss, slowing solidification.
- Low volume to surface area ratio: Increases surface area (e.g., thin sections), speeding solidification.
- Ratio of one: Neutral, not specific.
- High surface area to volume: Equivalent to low volume-to-surface area, speeds solidification.
Thus, option (1) is correct. Quick Tip: High volume-to-surface area slows casting solidification.
Swaging is an operation of
View Solution
Swaging is a forging process that reduces the diameter of rods, tubes, or wires by hammering or pressing with rotating dies, often performed hot or cold to shape tapered or stepped profiles.
- Hot rolling: Produces sheets or plates, not localized diameter reduction.
- Extrusion: Forces material through a die, not by hammering.
- Piercing: Creates holes, typically in seamless tube production.
Thus, option (2) is correct. Quick Tip: Swaging forges reduced diameters in rods or tubes.
Directional properties can be obtained in
View Solution
Cold working (deformation below recrystallization temperature) aligns grains and develops crystallographic texture, inducing anisotropy (directional properties) in strength and ductility.
- Hot working: Above recrystallization, minimizes texture, yielding isotropic properties.
- Warm working: Partial recrystallization, less anisotropy than cold working.
- Quenching: Heat treatment for hardness, not deformation-induced texture.
Thus, option (3) is correct. Quick Tip: Cold working creates anisotropic properties via texture.
A foil is a
View Solution
A foil is an extremely thin sheet of metal (e.g., aluminum foil, thickness < 0.2 mm), produced by rolling.
- Thin slab: Thicker, semi-finished product (~50–100 mm).
- Thin plate: Thicker than foil (~0.5–6 mm).
- Thin billet: Cylindrical or square, not sheet-like.
Thus, option (1) is correct. Quick Tip: Foil is an ultra-thin metal sheet.
Hemming is an operation in
View Solution
Hemming folds a sheet metal edge over itself (e.g., in automotive panels) to create a smooth, reinforced edge, a common sheet metal forming process.
- Rolling: Produces sheets, not edge folding.
- Forging: Shapes bulk metal, not thin sheets.
- Impact extrusion: Forms tubular components, not edges.
Thus, option (3) is correct. Quick Tip: Hemming folds sheet metal edges for strength and smoothness.
Flashless precision forging occurs in
View Solution
Coining is a closed-die, flashless forging process that applies high pressure to shape metal precisely within the die cavity, producing detailed components (e.g., coins, medallions) with minimal excess material.
- Swaging: Reduces diameter, often open-die, with flash.
- Punching: Cuts holes, not forging.
- Upsetting: Increases diameter, typically with flash.
Thus, option (3) is correct. Quick Tip: Coining achieves flashless precision forging.


































































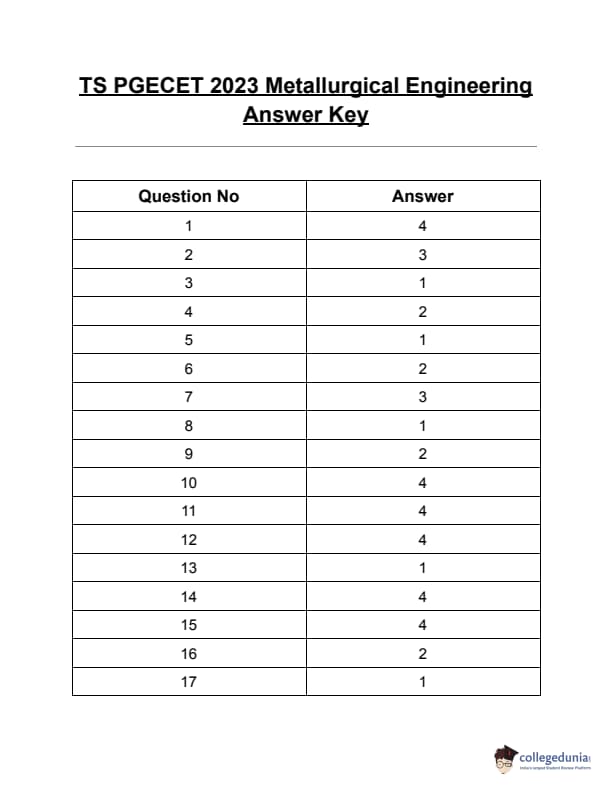
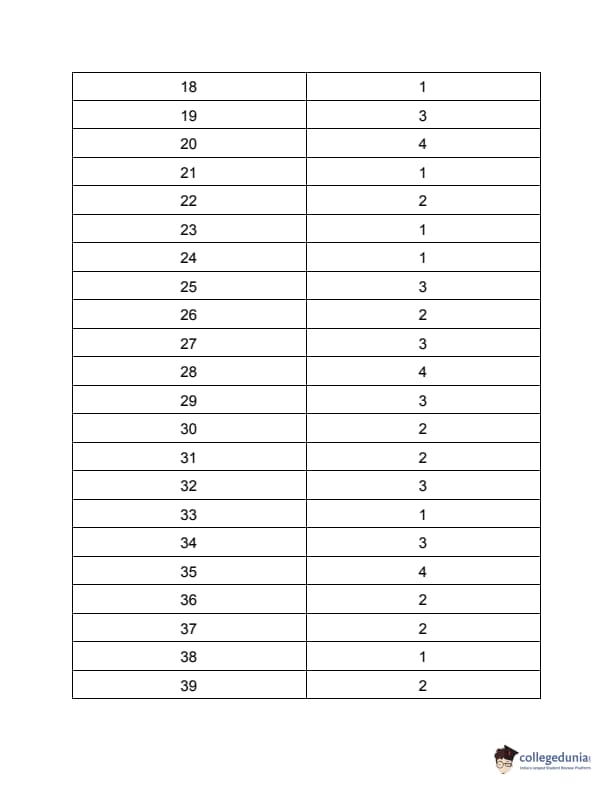
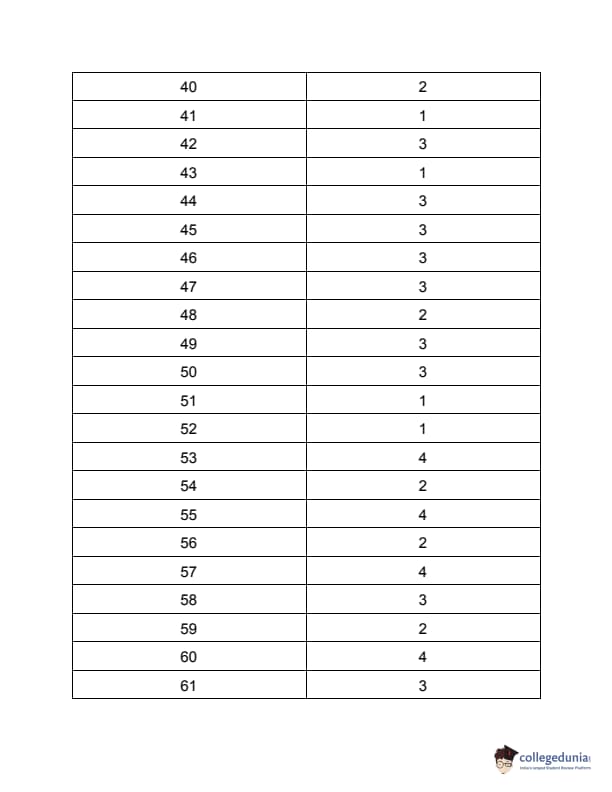
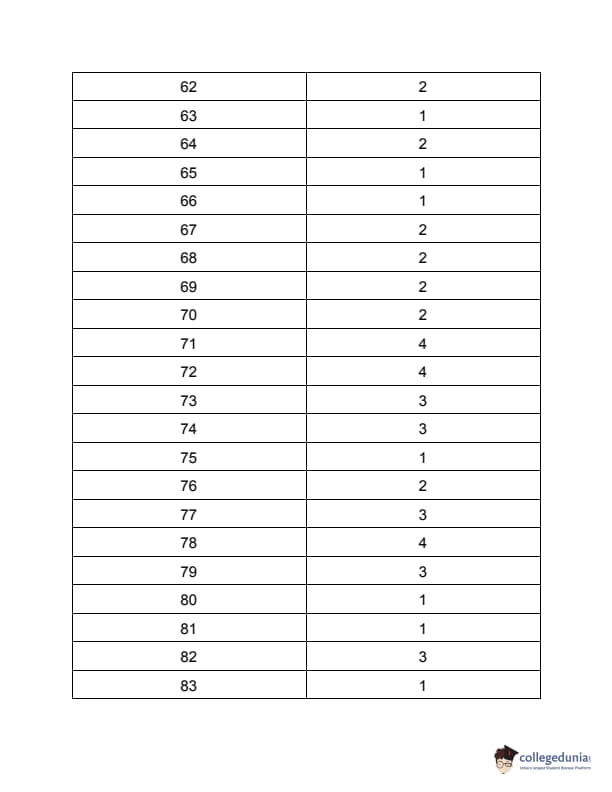







Comments