TS PGECET 2023 Nano Technology Question Paper with Answer key PDF is available here for download. TS PGECET 2023 was conducted by JNTU Hyderabad on behalf of TSCHE on June 1, 2023. TS PGECET 2023 NT Question Paper consisted of 120 questions carrying 1 mark for each.
TS PGECET 2023 Nano Technology Question Paper
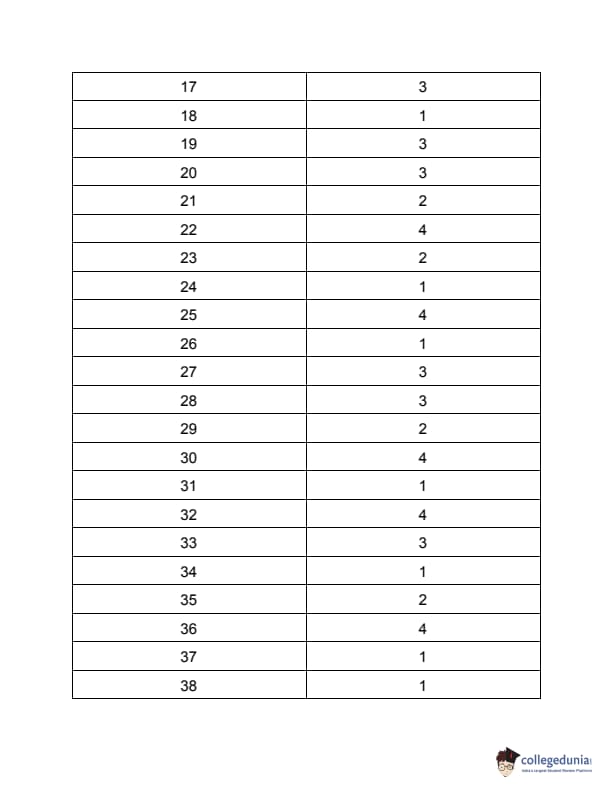
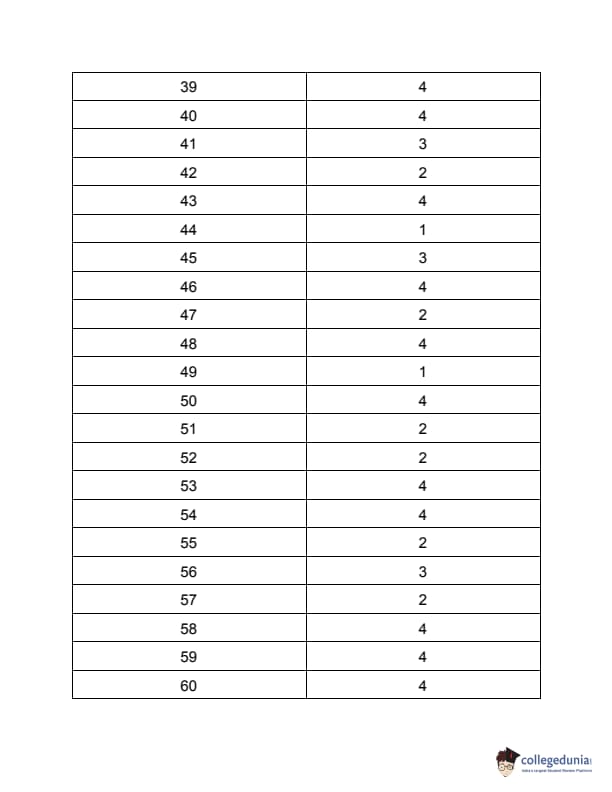
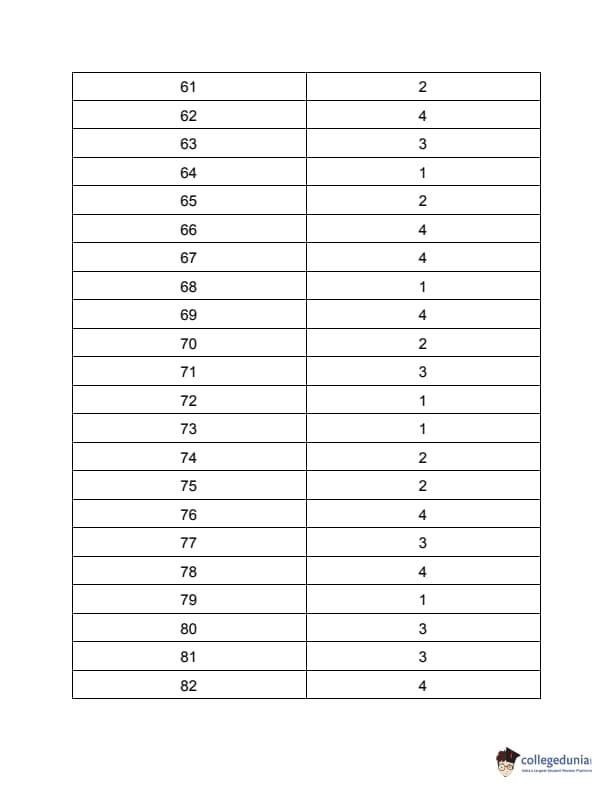
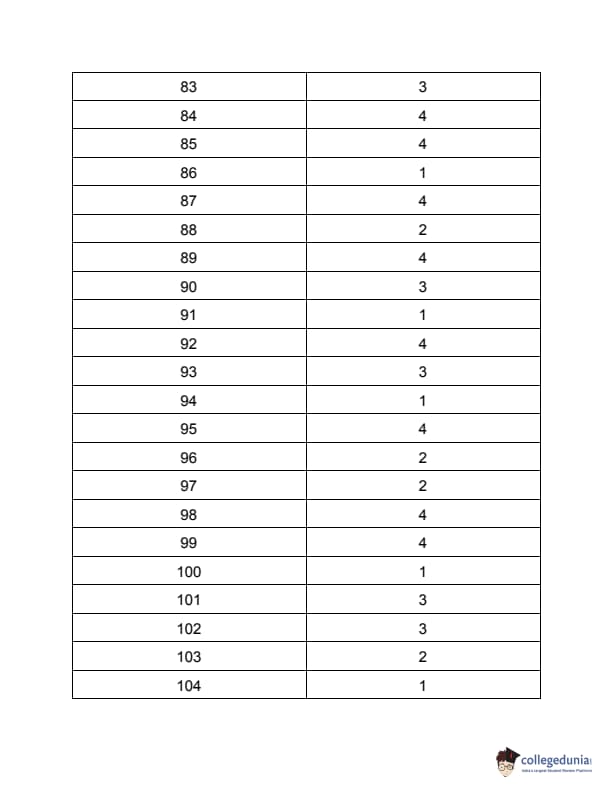
| TS PGECET 2023 NT Question Paper with Answer Key | Download PDF | Check Solution |

Question 1:
Two bars having different materials but of same area and length are subjected to same tensile force. If the bars have their axial elongation in the ratio of 4:6, then the two material’s ratio of Modulus of Elasticity is _________.
View Solution
N/A Quick Tip: When bars are geometrically identical and under the same load, their elongation is inversely proportional to their modulus of elasticity.
A metal bar of 10 mm diameter when subjected to a pull of 23.5 kN gave an elongation of 0.3mm on a gauge length of 200 mm. The metal’s Young’s modulus of elasticity is _________.
View Solution
N/A Quick Tip: Always convert force to Newtons and calculate area in mm\(^2\) to maintain unit consistency. Use \(E = \frac{FL}{A \Delta L}\) for direct substitution.
If in a crystal there exists a point defect of the interstitial atom and if solute atom is smaller than the solvent atom, then select the true statement for the edge dislocation.
View Solution
N/A Quick Tip: Remember: Small atoms create compressive fields and get attracted to tensile regions (below dislocation line). Large atoms go to compressive regions.
Which one of the following is the metallic tiny whisker?
View Solution
N/A Quick Tip: Whiskers are long, thin metallic crystals with zero dislocations. Their formation is spontaneous and can be problematic in electronics.
Select the following false statement for strain-relief crystallization.
View Solution
N/A Quick Tip: Strain-relief mechanisms reduce internal stresses by eliminating dislocations and defects — climb and annihilation play major roles.
When a material is plastically deformed at low temperature (around 25 degrees) relative to the melting temperature, the following change is not observed in ¨___________________.
View Solution
N/A Quick Tip: Cold working increases dislocation density and strengthens the material, but no healing or recovery occurs without heat.
Classification of Composites can be based on __________________.
View Solution
N/A Quick Tip: Composites are broadly classified using both the matrix (binder) and reinforcement (load-bearing) components.
Which of the following is an example for Carbon fiber?
View Solution
N/A Quick Tip: Carbon fiber is a classic reinforcement material, valued for its high tensile strength and low weight.
Which of the following polymer additive is used to remove parts from molds?
View Solution
N/A Quick Tip: Lubricants are essential mold release agents in polymer processing. They reduce sticking and improve part quality.
________________ is not a characteristic trait of polymer materials.
View Solution
N/A Quick Tip: Polymers are lightweight and chemically resistant, but typically not high in strength unless reinforced.
The smallest portion of the crystal lattice is _________________.
View Solution
N/A Quick Tip: The unit cell is the basic building block of any crystal structure. Repeating it in space forms the full crystal.
Which one of the following is Packing fraction of a simple cubic structure?
View Solution
N/A Quick Tip: Simple cubic has low packing efficiency; only 52.4% of the space is occupied by atoms.
Burgers vector of edge dislocation is ------- to the dislocation line.
View Solution
N/A Quick Tip: Remember: Edge dislocation → Burgers vector is perpendicular; Screw dislocation → it’s parallel.
The ratio of lateral strain to linear strain is__________________.
View Solution
N/A Quick Tip: Poisson’s Ratio = transverse contraction / axial extension; always less than 0.5 for stable materials.
The Capacity of a material to absorb energy prior to failure is _________________.
View Solution
N/A Quick Tip: Toughness = energy absorbing capacity before breaking; NOT the same as strength or hardness.
________________ structure has amorphous solids.
View Solution
N/A Quick Tip: Amorphous = Irregular atomic arrangement → no sharp melting point, unlike crystalline solids.
Each point (position of particle) in a crystal lattice is termed as __________________.
View Solution
N/A Quick Tip: Always remember: The basic building block of a crystal structure starts with the \textbf{lattice point} which holds the basis (atom or group of atoms).
If a metal forms a FCC lattice with unit edge length of 500 pm and atomic mass of 110, calculate the density of the metal.
View Solution
N/A Quick Tip: For FCC lattices, always remember \(Z = 4\). Convert pm to meters and grams to kg carefully for SI unit consistency.
Which of the following is the advantage of using conducting polymers in place of metals?
View Solution
N/A Quick Tip: Polymers are known for being lightweight—this makes them suitable for use in aerospace, electronics, and automotive industries.
What is the average maximum temperature used for engineering ceramics?
View Solution
N/A Quick Tip: Remember: Engineering ceramics like alumina and zirconia can typically withstand temperatures around 2700–2800°C.
Example for a thermoplastic is __________.
View Solution
N/A Quick Tip: Thermoplastics can be remelted; Acetal is one example, widely used in mechanical engineering applications.
QD lasers have a very low threshold current densities ranging from ______.
View Solution
N/A Quick Tip: Quantum Dot lasers have low threshold current due to discrete energy states—ideal for efficient operation.
Buried hetero-junction (BH) device is a type of __________, where the active volume is buried in a material of wider band-gap and lower refractive index.
View Solution
N/A Quick Tip: BH devices enhance laser efficiency via strong optical confinement—think of strong index guiding.
Which of the following have anisotropic nature within their structure?
View Solution
N/A Quick Tip: Crystalline = direction-dependent = anisotropic. Snowflakes follow this rule perfectly.
_________ is a phenomenon, where the magnetic lines of force cannot penetrate the body of a superconductor.
View Solution
N/A Quick Tip: Superconductors = no magnetic entry = Meissner effect. Remember: total magnetic repulsion!
In the periodic table, number of metallic elements is ________.
View Solution
N/A Quick Tip: Periodic table: Over 3/4 are metals. 95 is the magic number!
Which property of metal is used for making strings of musical instruments like Sitar and Violin.
View Solution
N/A Quick Tip: Sound-producing metals = Sonorous = musical instruments love them!
______ metal is used for nuclear energy.
View Solution
N/A Quick Tip: Nuclear energy? Think Uranium—it’s the atomic powerhouse!
From the following, forbidden energy gap of dielectrics is ________.
View Solution
N/A Quick Tip: Dielectrics have large band gaps—usually 3 eV or more—making them excellent insulators.
The torque induced on a dipole when placed in an electric field \(E\) is ________.
View Solution
N/A Quick Tip: Torque on a dipole is \(pE \sin \theta\)—maximum when perpendicular to the field.
The value of 1 Debye in cm is ________.
View Solution
N/A Quick Tip: Remember: 1 Debye \(\approx 3.33 \times 10^{-30}\) cm — a common constant in dipole problems.
In a water drop of radius 1 mm all the molecular dipole points are in the same direction. If the dipole moment of a water molecule is \(6 \times 10^{-30}\) m, what is the polarization in the water drop?
View Solution
N/A Quick Tip: Polarization = dipole moment per volume. Use \(P = \frac{pN}{V}\) formula carefully with unit conversions.
In the formula, \(P = \chi_e E\), \(\chi_e\) is called as ___________________.
View Solution
N/A Quick Tip: Electric susceptibility relates \(P\) and \(E\): \(P = \chi_e E\). It tells us how much the material gets polarized under an electric field.
In Ferroelectric materials, Polarization is___________________.
View Solution
N/A Quick Tip: Ferroelectrics can flip their polarization direction with an electric field — so polarization is reversible.
The temperature characteristic of ferroelectric crystal is called __________________.
View Solution
N/A Quick Tip: Ferroelectrics become non-ferroelectric above the transition (Curie) temperature.
___\ Part of the curve shows Spontaneous Polarization.
View Solution
N/A Quick Tip: Spontaneous polarization is shown where \(E = 0\) and \(P \neq 0\) — typically the top intercept on the P-axis.
___________ is the symmetry of BaTiO\textsubscript{3 in ferroelectric form.
View Solution
N/A Quick Tip: Remember: BaTiO\textsubscript{3} becomes tetragonal in its ferroelectric phase — this distortion enables spontaneous polarization.
The minimum amount of current passed through the body of superconductor in order to destroy the superconductivity is called ______.
View Solution
N/A Quick Tip: Superconductivity breaks when current > critical value. This maximum current is called **critical current**.
Solid that offers no _______ for the passage of electricity is called superconductors.
View Solution
N/A Quick Tip: Zero resistance is the hallmark of superconductors — current flows without energy loss.
The shifting of electrons in superconductors is prevented by _______.
View Solution
N/A Quick Tip: Superconductivity is a quantum effect due to formation of Cooper pairs — electrons shift collectively, not individually.
An ideal superconductor exhibit _________.
View Solution
N/A Quick Tip: Superconductors expel magnetic fields due to the Meissner effect — this is key to identifying a true superconductor.
The Preparation of superconductors by ceramic method with homogeneous mixture of the oxides in their molar ratios _______.
View Solution
N/A Quick Tip: YBCO superconductors are typically made using Y\(_2\)O\(_3\), BaCO\(_3\), and CuO in ceramic synthesis methods.
Which of the following is relation between transition temperature (T\(_c\)) and isotopic mass (M)?
View Solution
N/A Quick Tip: In BCS superconductors, heavier isotopes lower \(T_c\) due to reduced phonon interaction — classic isotope effect.
The sequence of the colour change during the polymerisation of polyaniline, is _________.
View Solution
N/A Quick Tip: Polyaniline synthesis shows characteristic color shifts — track oxidation by observing color change pattern.
Conductivity of a material is defined as ________.
View Solution
N/A Quick Tip: Conductivity is inversely related to resistance and directly related to length per area: \(\sigma = \dfrac{l}{RA}\).
The material's insulating capacity against high voltages is called as ________.
View Solution
N/A Quick Tip: Dielectric strength measures how much voltage an insulating material can endure before breaking down.
The nature of the coefficient of resistance of an insulator is ________.
View Solution
N/A Quick Tip: Insulators have extremely high resistance, often considered infinite for ideal cases.
Which one is the most commonly used for making magnetic recording tape?
View Solution
N/A Quick Tip: Ferric oxide is the standard magnetic material used in audio and video recording tapes.
Example of piezoelectric material is ________.
View Solution
N/A Quick Tip: Quartz is a natural piezoelectric crystal that produces voltage under mechanical stress.
Which one of the following is the correct classification of the conducting materials?
View Solution
N/A Quick Tip: A good conductor has low resistivity and hence high conductivity, since \(\sigma = \frac{1}{\rho}\).
With increase in temperature, the electrical conductivity of intrinsic semiconductor _______.
View Solution
N/A Quick Tip: In intrinsic semiconductors, conductivity increases with temperature due to thermal generation of carriers.
What is the net charge on n-type material?
View Solution
N/A Quick Tip: n-type semiconductors have excess electrons but remain neutral overall due to balanced charges.
The ________ statement is true for a dielectric.
View Solution
N/A Quick Tip: Some dielectrics can become superconductors at cryogenic temperatures—zero resistance flow!
A material with one dimension in Nano range and the other two dimensions are large is known as
View Solution
N/A Quick Tip: A quantum well has one nano-dimension and two large dimensions—free motion in a plane but confined perpendicularly!
Product design belongs to ________.
View Solution
N/A Quick Tip: Think top-down as designing the whole product idea first, then zooming into the parts!
A special case of non-inverting amplifier in which all of the output voltage is feedback to the inverting input of the op-amp is known as ______.
View Solution
N/A Quick Tip: A voltage follower gives the same output as input—perfect for buffering without altering the signal!
______ effects are exhibited by Schmitt trigger.
View Solution
N/A Quick Tip: Schmitt triggers use hysteresis to make switching smooth and stable—even in noisy signals!
Frequency response improves with __________ in an RC coupled amplifier.
View Solution
N/A Quick Tip: Higher \(C_c\) improves low-frequency gain by reducing the lower cutoff frequency in RC coupled amplifiers.
_________ transistor array is essential in construction of a mirror circuit where the formation of diode takes place by an adjacent transistor.
View Solution
N/A Quick Tip: Use CA3086 for precise current mirrors—its 5 matched NPN transistors are ideal for analog IC design.
An increase in operating frequency also increases _____ between input and output and decreases _____ for a compensating network of an amplifier.
View Solution
N/A Quick Tip: High frequencies reduce amplifier gain and increase phase shift—compensate using frequency compensation networks.
______ transistor is connected in parallel with feedback capacitor for termination of each ramp at a prescribed level in a Voltage Controlled Sawtooth Oscillator (VCO).
View Solution
N/A Quick Tip: PUTs help reset timing capacitors in sawtooth VCOs—they discharge capacitors after hitting a threshold.
The below stated condition applicable for Non-inverting amplifier is ______.
View Solution
N/A Quick Tip: Non-inverting amplifiers maintain phase with the input and offer gain \(\geq\) 1, making them ideal for signal buffering and amplification.
Choose a correct order from the following steps to be done in one of the algorithm of divide and conquer method:
i) Store the signal column wise
ii) Compute the M-point DFT of each row
iii) Multiply the resulting array by the phase factors \(W_N^{kn}\)
iv) Compute the L-point DFT of each column
v) Read the result array row wise
View Solution
N/A Quick Tip: 2D FFTs split the transform into simpler 1D DFTs along rows and columns using twiddle factors for efficiency.
The photoresist layer is exposed to _______ in CMOS fabrication
View Solution
N/A Quick Tip: Photolithography uses UV light to expose the photoresist because its shorter wavelength allows finer feature resolution on chips.
Which of the following is sputtered on the whole wafer?
View Solution
N/A Quick Tip: Aluminium is uniformly sputtered on wafers to create conductive paths in CMOS circuits during metallization.
The Boolean equation \(Y = \overline{A}BC + \overline{A}B\overline{C} + AB\overline{C} + ABC\) is to be implemented using only two-input NAND gates. The minimum number of gates required is
View Solution
N/A Quick Tip: Use K-map and gate-sharing logic to reduce the number of required NAND gates. NAND gates can implement any logic by rearranging with De Morgan's laws.
A thermoelectric refrigerator works on the principle of
View Solution
N/A Quick Tip: Thermoelectric cooling relies on the Peltier effect, which absorbs heat at one junction and releases it at another due to electric current.
In a bipolar junction transistor, the current gain \(\beta\) ________
View Solution
N/A Quick Tip: In BJTs, current gain \(\beta\) typically increases with temperature due to enhanced carrier mobility and reduced recombination in the base.
The number of depletion layers in a transistor is ________.
View Solution
N/A Quick Tip: A BJT has two PN junctions, each forming a depletion layer — one between emitter-base and the other between base-collector.
The following are the functions of a transistor ___________.
View Solution
N/A Quick Tip: Transistors are essential in electronics for switching operations and analog signal control, effectively behaving as variable resistors and switches.
A BJT with \(\beta = 50\) has a base to collector leakage current \(I_{CBO}\) of \(2.5\ \mu A\). If the transistor is connected in CE configuration, the collector current for \(I_B = 0\) is
View Solution
N/A Quick Tip: When \(I_B = 0\), leakage current dominates. Use \(I_C = (1 + \beta) I_{CBO}\) to compute collector current in CE configuration.
What is true with regard to the cut-off region of npn BJT?
View Solution
N/A Quick Tip: In the cut-off region of a BJT, \(V_B < V_E\), ensuring no base current and hence no collector current flows.
Binary ladder network is better than resistive divider for D/A conversion, because __________.
View Solution
N/A Quick Tip: The R-2R ladder network simplifies D/A converters by using only two resistor values, improving consistency and scalability.
In a binary ladder (R-2R), D/A converter, the input resistance for each input is
View Solution
N/A Quick Tip: In an R-2R DAC, the input resistance seen at every bit input is constant and equals \(3R\), which simplifies circuit interfacing and improves linearity.
A/D converter which does not use D/A converter is
View Solution
N/A Quick Tip: Dual slope A/D converters avoid D/A conversion by using time-based integration, offering high noise rejection and accuracy for slow-changing signals.
__________ is the main disadvantage of a dual slope integrator A/D converter
View Solution
N/A Quick Tip: Dual slope converters are accurate and immune to noise, but their conversion is time-intensive, making them unsuitable for high-speed applications.
The gate voltage in a JFET at which drain current becomes zero is called ________.
View Solution
N/A Quick Tip: In a JFET, the pinch-off voltage is the key control point where the conducting channel closes and the drain current stops flowing.
JFET is a ________ device.
View Solution
N/A Quick Tip: In JFETs, only one type of charge carrier (electron or hole) is involved in conduction — making them unipolar devices.
Junction Field Effect Transistor is a __________ controlled device.
View Solution
N/A Quick Tip: JFETs control current flow using input voltage at the gate terminal — making them voltage-controlled with high input impedance.
In FET, the np region exists between source and gate and is __________.
View Solution
N/A Quick Tip: In JFETs, the gate-to-source junction is reverse biased to control current without drawing gate current.
The wave-particle duality of light is defined as __________.
View Solution
N/A Quick Tip: Light behaves as both a wave (interference/diffraction) and a particle (photons in photoelectric effect) — this is called wave-particle duality.
Uncertainty principle is easily understood with the help of __________.
View Solution
N/A Quick Tip: Compton’s Effect provides experimental backing for the Uncertainty Principle by showing that measurement disturbs quantum systems.
Heisenberg’s uncertainty principle is significant only for __________.
View Solution
N/A Quick Tip: Heisenberg’s uncertainty principle is relevant only for very small (microscopic) particles, not for large everyday objects.
The uncertainty principle is applicable to __________.
View Solution
N/A Quick Tip: Uncertainty Principle is universal for all microscopic particles like electrons, photons, and protons — not just one of them.
Which one of the following can be explained by the wave nature of particles?
View Solution
N/A Quick Tip: Diffraction patterns observed in electron beams confirm the wave nature of matter — a core idea in quantum physics.
The central force motion is based on __________.
View Solution
N/A Quick Tip: In central force motion, the force always points towards or away from a fixed center — this helps conserve angular momentum.
Two particles A and B, initially at rest, move towards each other under mutual force of attraction. At the instant when the speed of A is \(v\) and the speed of B is \(2v\), the speed of mass of the system is __________.
View Solution
N/A Quick Tip: If no external force acts on a system, the center of mass remains at rest or moves with constant velocity.
________ Laws governs mechanical waves.
View Solution
N/A Quick Tip: Mechanical waves obey Newton's laws — force and motion relationships are key in understanding wave propagation.
The wave function of the particle lies in ________ region.
View Solution
N/A Quick Tip: In a 1D infinite potential well, the particle’s wave function exists only between the walls — \(0 < X < L\).
The energy of the particle is proportional to ________.
View Solution
N/A Quick Tip: For a particle in a 1D box, the energy levels are quantized and increase as \(n^2\) — not linearly with \(n\).
The line of action of concurrent forces joins at ________.
View Solution
N/A Quick Tip: Concurrent forces always act through the same point — making them easier to analyze using vector rules.
Forces meeting at one point but having lines of action, not in one plane are known as ________.
View Solution
N/A Quick Tip: When forces neither lie in a plane nor meet at a point, they are called non-coplanar non-concurrent forces.
A spring, when compressed by 4 cm, has 2 J of energy stored in it. The force required to extend it by 8 cm is:
View Solution
N/A Quick Tip: Use the energy formula \( U = \frac{1}{2}kx^2 \) to find the spring constant, then apply \( F = kx \) for force.
Which of the following equation of motion represents simple harmonic motion? Where \( k, k_0, k_1 \) and \( a \) are all positive.
View Solution
N/A Quick Tip: For SHM, remember that the restoring force is always proportional to the displacement, and the acceleration is in the opposite direction to the displacement.
Which one of the following principles states that the inertia forces, couples, external forces and torques on a body together give statical equilibrium?
View Solution
N/A Quick Tip: D'Alembert's principle helps in solving problems involving dynamics and statics, where both inertial forces and external forces are at play.
A wedge of mass 104 N is sliding on a smooth inclined plane of an elevator which is moving down with a constant acceleration of \( 2 \, m/s^2 \) as shown in the figure below. What will be the net normal reaction force acting on the wedge exerted by the inclined plane of an elevator? Take \( g = 10 \, m/s^2 \). Inclination angle of the lift plane is \( 30^\circ \).
{
View Solution
N/A Quick Tip: When solving problems with inclined planes and accelerations, always break down the forces into their components along the direction of the incline and the perpendicular direction.
If a rigid body rotates with an angular momentum \( L \) and its kinetic energy is halved, then the angular momentum is
View Solution
N/A Quick Tip: The relationship between kinetic energy and angular momentum is inversely proportional to the square of the angular velocity. Halving the kinetic energy will halve the angular momentum.
A particle moves in a circular motion that is consistent. The particle’s angular momentum will be conserved at __________.
View Solution
N/A Quick Tip: Remember that in rotational motion, the axis of rotation (usually the center of the circle) is crucial for calculating angular momentum. Any external force acting on the system would alter the conservation of angular momentum.
The relationship between the load lifted (W) and the effort required (P) to lift the load often is called as the law of the machine is ________.
View Solution
N/A Quick Tip: In simple machines, the effort needed to lift a load is determined by the mechanical advantage and velocity ratio. The law of the machine gives a clear mathematical relationship between these factors.
A certain weight lifting machine of velocity ratio 30 can lift a load of 1500 N with the help of 125 N effort. What is the efficiency of the machine?
View Solution
N/A Quick Tip: Efficiency is a measure of how well a machine converts input energy into useful work. The higher the efficiency, the less energy is wasted in the form of heat, friction, or other losses.
Radiation heat transfer is depicted by __________.
View Solution
N/A Quick Tip: Radiation is the only mode of heat transfer that can occur in a vacuum, as it involves electromagnetic waves that do not require a medium.
The literature of heat transfer generally recognizes distinct modes of heat transfer.
The number of modes are ______.
View Solution
N/A Quick Tip: Remember, radiation is the only mode of heat transfer that can occur in a vacuum.
Consider system A at uniform temperature t and system B at another uniform temperature T (t > T) as shown in figure. Let the two systems be brought into contact and be thermally insulated from their surroundings but not from each other. Energy will flow from system A to system B due to ___\ .
{
View Solution
N/A Quick Tip: The primary cause of energy transfer between two objects at different temperatures is the temperature difference.
An oil cooler in a high performance engine has an outside surface area 0.12 m and surface temperature of 65°C. At any intermediate time, air moves over the surface of the cooler at a temperature of 30°C, gives rise to a surface coefficient equal to 45.4 W/m²K. The heat transfer rate is ______.
View Solution
N/A Quick Tip: To calculate the heat transfer rate, always ensure you have the correct values for the convective heat transfer coefficient, surface area, and temperature difference.
Convective heat transfer coefficient doesn't depend on _
___\.
View Solution
N/A Quick Tip: Convective heat transfer coefficient is a property that depends on the system's physical and flow conditions, but it is independent of time.
Thermal conductivity is maximum for the following substance __________.
View Solution
N/A Quick Tip: Diamond has the highest thermal conductivity among natural materials due to its strong covalent bonds and crystalline structure. It's often used in applications requiring superior heat dissipation.
A designer chooses the values of fluid flow rates and specific heats in such a manner that the heat capacities of the two fluids are equal. A hot fluid enters the counter flow heat exchanger at 100°C and leaves at 60°C. A cold fluid enters the heat exchanger at 40°C. What is the mean temperature difference between the two fluids?
View Solution
N/A Quick Tip: In counter-flow heat exchangers, the mean temperature difference is used to estimate the efficiency of heat transfer between fluids. A larger MTD generally indicates higher heat transfer efficiency.
The unit of overall coefficient of heat transfer is __________.
View Solution
N/A Quick Tip: The overall heat transfer coefficient is important for determining the heat transfer rate in heat exchangers. A higher \( U \) value indicates more efficient heat transfer.
LMTD in case of counter flow heat exchanger as compared to parallel flow heat exchanger will be __________.
View Solution
N/A Quick Tip: Counter-flow heat exchangers are more efficient than parallel-flow ones because they maintain a higher temperature gradient along the length of the exchanger.
When heat is transferred by molecular collision, then heat transfer is referred as __________.
View Solution
N/A Quick Tip: Conduction occurs in solids when molecules transfer heat by vibration, while convection and radiation occur in fluids and gases.
What is the latent heat of steam at atmospheric pressure?
View Solution
N/A Quick Tip: Latent heat of steam at atmospheric pressure is a standard thermodynamic constant: 2257 kJ/kg.
Carnot cycle comprises of ______
View Solution
N/A Quick Tip: The Carnot cycle has 4 steps: 2 isothermal and 2 adiabatic processes — all are reversible.
A refrigerator has a performance coefficient of 5. What is the ambient heat discharged if the temperature inside the freezer is -20°C?
View Solution
N/A Quick Tip: Convert all temperatures to Kelvin when using formulas involving COP, efficiency, or entropy.
If 10 g of ice at 0°C is converted to water at the same temperature, What will be the change in entropy? (Latent heat 80 cal/g)
View Solution
N/A Quick Tip: Entropy change during phase change = heat absorbed ÷ temperature (in Kelvin).
Gibbs phase rule defines the relationship between degrees of freedom (F) of a system, number of phases (P) and the number of components (c). The equation is _________.
View Solution
N/A Quick Tip: Use the formula \(F = C - P + 2\) to determine the independent variables in a multiphase system.
Which one of the following is correct statement?
[A = Helmholtz function (Helmholtz Free Energy), G = Gibbs function (Gibbs free energy),
U = Internal energy, H = Enthalpy, T = Absolute temperature, S = Entropy]
View Solution
N/A Quick Tip: Remember: \(A = U - TS\) for constant volume systems, and \(G = H - TS\) for constant pressure systems.
The standard heat of combustion of ethanol (C\textsubscript{2H\textsubscript{5OH) is 1372 kJ/mol. By completely burning a 20g sample, how much heat (in kJ) will be liberated?
View Solution
N/A Quick Tip: Always convert grams to moles using molar mass before applying combustion or reaction enthalpy.
A system suffers an increase in internal energy of 80 J and at the same time has 50 J of work done on it. The heat change of the system is _______.
View Solution
N/A Quick Tip: Use \( Q = \Delta U + W \) when work is done on the system; be careful with the sign convention.
If 30 J of energy is added to water in the form of heat at 27\degree C, the change in entropy of water is _____
View Solution
N/A Quick Tip: Use \( \Delta S = \frac{q_{rev}}{T} \), making sure temperature is in Kelvin and heat is in joules. Always check units!
Which one of the following thermodynamic quantities is not a state function?
View Solution
N/A Quick Tip: State functions depend only on the initial and final state, not the path. Work and heat are not state functions.











































































Comments