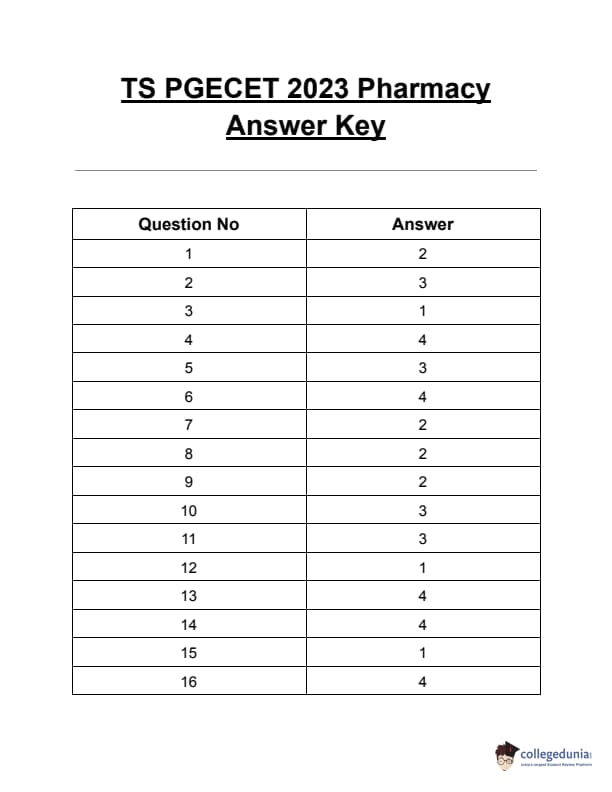
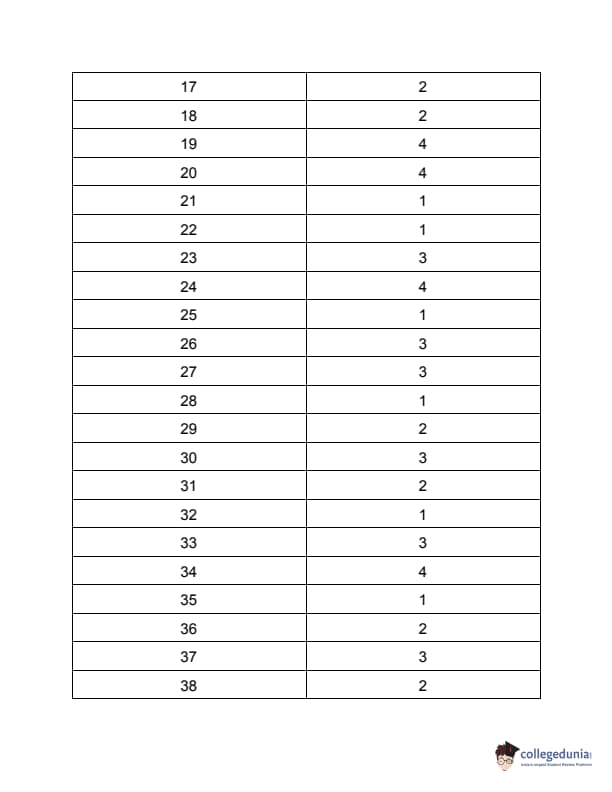
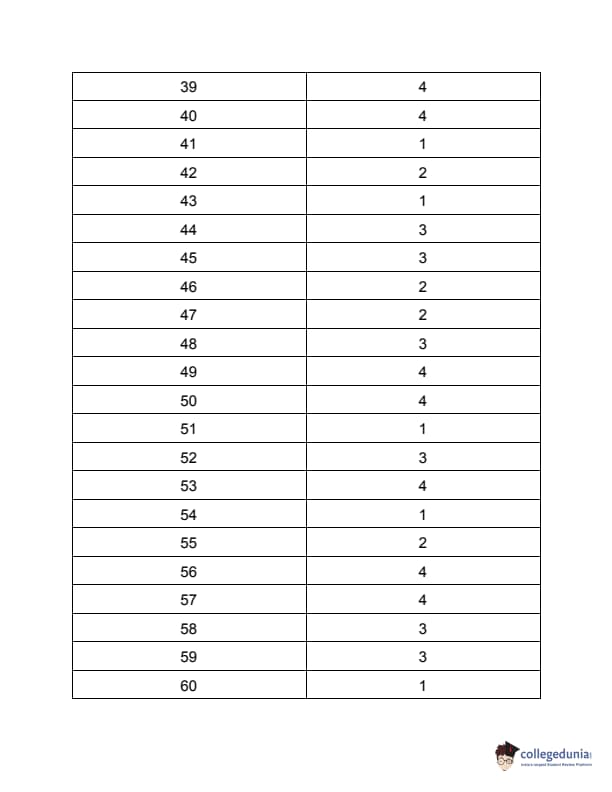
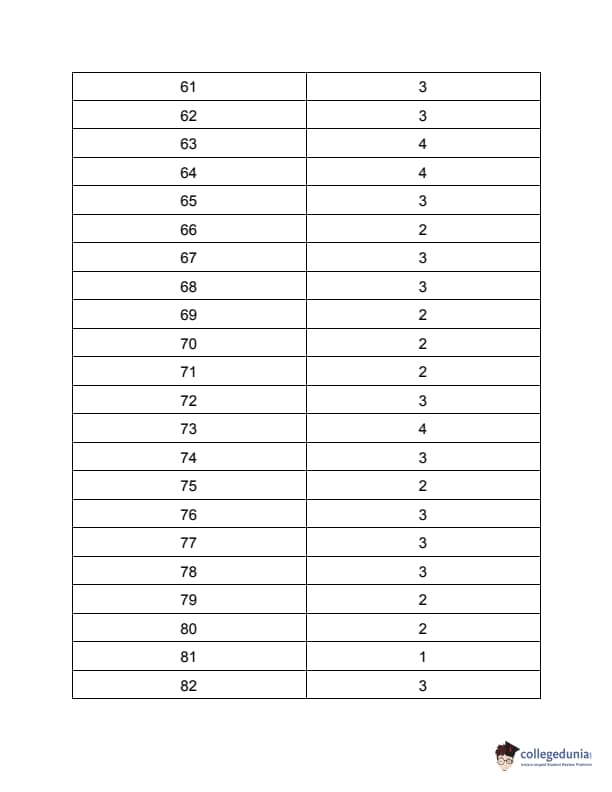
TS PGECET 2023 Pharmacy Question Paper with Answer key PDF is available here for download. TS PGECET 2023 was conducted by JNTU Hyderabad on behalf of TSCHE on May 29, 2023. TS PGECET 2023 PY Question Paper consisted of 120 questions carrying 1 mark for each.
TS PGECET 2023 Pharmacy Question Paper
| TS PGECET 2023 PY Question Paper PDF | Download PDF | Check Solution |
Who isolated morphine from opium?
View Solution
Johann Friedrich Sertuerner, a German pharmacist, was the first to isolate morphine from opium in 1804.
He named it "morphium" after Morpheus, the Greek god of dreams, due to its strong sedative properties.
Morphine became the first alkaloid to be extracted from a plant, marking a significant advancement in pharmaceutical chemistry. Quick Tip: Morphine is classified as an opioid analgesic and is widely used to treat severe pain. Its discovery paved the way for isolating other alkaloids like quinine and cocaine.
Asafoetida obtained from which species of plant?
View Solution
Asafoetida, commonly known as "hing," is obtained from plants of the genus \textit{Ferula.
The resin is collected by making incisions in the root of the plant, and it is known for its strong pungent smell.
It has been used in traditional medicine for digestive issues and as an anti-flatulent agent. Quick Tip: Asafoetida is a powerful antimicrobial and antifungal agent. It is commonly used in Indian cuisine to add flavor and aid digestion.
Which medicinal plant did India prohibit from being exported in 1998?
View Solution
Swertia chirata, commonly known as "chirata," is a medicinal herb known for its bitter taste and therapeutic properties.
India prohibited its export in 1998 due to concerns over overharvesting and its vital role in traditional medicine.
It is primarily used in Ayurvedic medicine for treating liver disorders, fever, and digestive ailments. Quick Tip: Swertia chirata is rich in antioxidants and has hepatoprotective properties, making it highly beneficial for liver health.
Reichert Meissle value is measured to determine
View Solution
Reichert Meissle value is a measure of the volatile water-soluble acid content in fats and oils.
It is used primarily in the analysis of dairy fats, particularly butter, to determine its quality.
A higher Reichert Meissle value indicates a greater proportion of volatile acids, which contribute to the characteristic aroma and flavor of butter. Quick Tip: Butter typically has a high Reichert Meissle value, distinguishing it from other fats like margarine. This test helps detect adulteration in butter samples.
Saffron mixed with dried Catharanthus tinctorious flowers is which type of adulteration?
View Solution
Saffron is often adulterated by mixing it with superficially similar inferior substances, such as dried petals of \textit{Catharanthus tinctorious.
These substitutes mimic the appearance of genuine saffron but lack its distinctive aroma and medicinal properties.
Such adulteration affects the quality and efficacy of saffron used in culinary and medicinal applications. Quick Tip: Always check saffron purity by placing strands in warm water—genuine saffron releases color slowly while adulterants bleed color instantly.
Which class of compounds are widely used to treat migraine?
View Solution
Ergot alkaloids, particularly derivatives such as ergotamine, are widely used in the treatment of migraines.
These compounds work by constricting blood vessels in the brain, reducing headache severity.
They are commonly prescribed for acute migraine attacks but must be used cautiously due to potential side effects. Quick Tip: Ergot alkaloids are most effective when taken early in a migraine attack. Excessive use may lead to rebound headaches, so use them under medical supervision.
The Surface sterilizing agent for leaves is
View Solution
Mercuric chloride is a strong disinfecting agent used for surface sterilization of leaves.
A dilute concentration of 0.1% is sufficient to eliminate microbial contaminants without harming the leaf tissue.
It ensures aseptic conditions during plant tissue culture studies. Quick Tip: Always rinse leaves thoroughly after mercuric chloride treatment to remove residual chemicals that could interfere with plant growth.
Ash value of crude drug determines
View Solution
Ash value is used to determine the amount of inorganic material present in a crude drug.
It helps identify impurities such as silica, calcium oxalate, and heavy metals, ensuring drug quality.
Different types of ash values include total ash, acid-insoluble ash, and water-soluble ash, each assessing specific inorganic content. Quick Tip: Ash value testing is crucial for detecting adulteration in herbal drugs, ensuring purity and standardization in pharmaceutical formulations.
Pair of hormones required for a callus to differentiate are
View Solution
The combination of auxin and cytokinin is essential for callus differentiation in plant tissue culture.
Auxin promotes cell division and elongation, while cytokinin supports shoot formation and organ development.
The balance between these two hormones determines whether the callus will produce shoots or roots. Quick Tip: Maintaining a proper auxin-to-cytokinin ratio in plant tissue culture media is key to directing shoot or root formation in regenerated plantlets.
The Biological source of Malt is
View Solution
Malt is derived from \textit{Hordeum vulgare, commonly known as barley.
Malt is produced by germinating barley grains, followed by drying them to develop enzymes that convert starch into fermentable sugars.
It is widely used in brewing, distilling, and food production due to its enzymatic activity. Quick Tip: Malted barley contains alpha-amylase and beta-amylase enzymes, which are crucial for breaking down starch into fermentable sugars during beer production.
Ketosugar can be detected by
View Solution
Seliwanoff's test is a chemical test used to differentiate ketose sugars from aldose sugars.
Upon heating with resorcinol in hydrochloric acid, ketoses produce a deep red color, confirming their presence.
Fructose is a common ketose sugar that gives a strong positive reaction in this test. Quick Tip: Seliwanoff's test is particularly useful in detecting fructose in food products, distinguishing it from glucose and other monosaccharides.
The pH at which there is no net charge on amino acid molecules and it does not migrate to any electrode under the influence of electric current is known as
View Solution
The isoelectric point (\( pI \)) is the pH at which an amino acid carries no net charge.
At this pH, the molecule exists as a zwitterion, with balanced positive and negative charges, preventing migration in an electric field.
Each amino acid has a distinct \( pI \), which depends on its side-chain properties. Quick Tip: Proteins can be separated based on their isoelectric points using isoelectric focusing, an important technique in electrophoresis.
MAO is a
View Solution
Monoamine oxidase (MAO) is an enzyme responsible for breaking down neurotransmitters such as serotonin, dopamine, and norepinephrine.
MAO inhibitors (MAOIs) are used in psychiatric medicine to treat depression by preventing the breakdown of these neurotransmitters, increasing their availability. Quick Tip: Patients taking MAOIs should avoid tyramine-rich foods, as it may cause hypertensive crises due to excessive neurotransmitter accumulation.
Night blindness is caused by deficiency of
View Solution
Vitamin A deficiency leads to night blindness, which affects the ability to see in low-light conditions.
This is due to the role of vitamin A in maintaining the health of the retina, specifically in the production of rhodopsin, a light-sensitive pigment.
A prolonged deficiency can cause xerophthalmia, a severe eye condition that may lead to permanent blindness. Quick Tip: Carrots, sweet potatoes, and leafy green vegetables are rich sources of vitamin A and can help prevent night blindness.
Active proteolytic enzyme in gastric fluid is
View Solution
Pepsin is the main proteolytic enzyme in gastric juice, responsible for breaking down proteins into smaller peptides.
It is secreted as pepsinogen by the stomach’s chief cells and is activated into pepsin in the acidic environment of the stomach.
Pepsin plays a crucial role in the digestion of dietary proteins, facilitating their absorption in the small intestine. Quick Tip: Pepsin works optimally at a highly acidic pH (around 1.5-2), making stomach acidity essential for efficient protein digestion.
Which of the following is a Calcineurin inhibitor?
View Solution
Tacrolimus is a potent calcineurin inhibitor used as an immunosuppressant to prevent organ rejection in transplant patients.
It blocks the activation of T-cells by inhibiting calcineurin, thereby reducing the immune response.
Tacrolimus is commonly prescribed alongside other immunosuppressants to maintain organ transplant viability. Quick Tip: Patients on tacrolimus need regular monitoring for kidney function and blood pressure, as it can cause nephrotoxicity and hypertension.
What is the colour of phenolphthalein in alkali medium?
View Solution
Phenolphthalein is a pH indicator that is colorless in acidic solutions and turns pink in alkaline solutions (pH \textgreater 8.2).
Its color change occurs due to structural modifications influenced by the hydrogen ion concentration.
This property makes it widely used in acid-base titrations. Quick Tip: If an alkaline solution gradually loses its pink color, it may indicate carbon dioxide absorption from the air, forming a weak acid.
Glauber salt is used as
View Solution
Glauber salt, chemically known as sodium sulfate (\( Na_2SO_4 \)), is commonly used as a laxative.
It works by drawing water into the intestines, softening stool, and facilitating bowel movement.
Its use in medicine dates back centuries, particularly in treating constipation and digestive issues. Quick Tip: Glauber salt should be taken with ample water to avoid dehydration, as it can cause significant fluid loss during its laxative action.
Which of the following drug is a cardiovascular glycoside?
View Solution
Digoxin is a cardiovascular glycoside derived from \textit{Digitalis plants.
It is used to treat heart failure and atrial fibrillation by increasing the force of heart contractions while reducing heart rate.
Digoxin exerts its effect by inhibiting the sodium-potassium ATPase pump, leading to increased intracellular calcium levels. Quick Tip: Patients taking digoxin should have regular blood monitoring, as its therapeutic range is narrow, and toxicity can lead to severe cardiac complications.
Remimazolam is used as
View Solution
Remimazolam is an ultra-short-acting benzodiazepine used as a sedative in procedural anesthesia.
It acts on GABA\(_A\) receptors, providing rapid onset and recovery, making it preferable for short-duration sedation.
Unlike traditional benzodiazepines, remimazolam undergoes rapid metabolism, reducing prolonged sedative effects. Quick Tip: Remimazolam has a safer profile compared to other benzodiazepines, particularly in elderly patients or those with compromised liver function.
What is the name of antiviral oral medication used for Covid 19?
View Solution
Paxlovid is an oral antiviral medication developed by Pfizer for the treatment of COVID-19.
It consists of two active ingredients: nirmatrelvir, which inhibits the SARS-CoV-2 protease, and ritonavir, which boosts nirmatrelvir's effectiveness.
It is recommended for high-risk individuals to reduce the severity of symptoms and prevent hospitalization. Quick Tip: Paxlovid should be administered within five days of symptom onset to maximize efficacy in preventing severe COVID-19 outcomes.
Cisplatin is used for the treatment of
View Solution
Cisplatin is a platinum-based chemotherapy drug used to treat testicular cancer, among other types.
It works by forming DNA cross-links, preventing cancer cells from replicating and leading to cell death.
Cisplatin is often combined with other chemotherapy drugs to enhance treatment efficacy. Quick Tip: Cisplatin can cause nephrotoxicity; maintaining proper hydration and monitoring kidney function is essential during treatment.
What is the best anti-inflammatory drug that is not an NSAIDs?
View Solution
Acetaminophen (paracetamol) is an effective anti-inflammatory drug that is not classified as an NSAID.
Unlike NSAIDs, it primarily acts on the central nervous system to reduce pain and fever but has minimal effects on peripheral inflammation.
It is widely used as a safer alternative for individuals who are at risk of NSAID-related gastrointestinal complications. Quick Tip: Acetaminophen should not be taken in excessive doses, as overdose can lead to serious liver toxicity.
4,5-Bis (hydroxymethyl)-2-methylpyridin-3-ol is the chemical name of
View Solution
Pyridoxine, commonly known as vitamin B\(_6\), is an essential nutrient involved in various metabolic processes.
It plays a crucial role in amino acid metabolism, neurotransmitter synthesis, and hemoglobin production.
Pyridoxine deficiency can lead to neurological symptoms such as irritability, depression, and impaired immune function. Quick Tip: Vitamin B\(_6\) is abundant in foods like bananas, nuts, and whole grains, helping maintain optimal nervous system function.
The drug used for treatment of parasitic, viral and bacterial infections is
View Solution
Metronidazole is an antimicrobial agent effective against parasitic, bacterial, and some viral infections.
It is commonly used to treat anaerobic bacterial infections and protozoal diseases such as giardiasis and trichomoniasis.
Metronidazole works by disrupting the DNA structure of microbes, inhibiting their replication and growth. Quick Tip: Alcohol should be avoided while taking metronidazole, as it can cause severe nausea and vomiting due to its interaction with alcohol metabolism.
Which of the following is not a synthetic drug?
View Solution
Rifampin is a naturally derived antibiotic obtained from \textit{Amycolatopsis rifamycinica.
It is used primarily for the treatment of tuberculosis and other bacterial infections.
Rifampin inhibits bacterial RNA synthesis by targeting RNA polymerase, making it highly effective against mycobacterial species. Quick Tip: Rifampin can cause orange discoloration of bodily fluids such as urine, sweat, and tears, which is harmless but noticeable.
Which of the following drug removes the extra uric acid from blood and helps in treatment of gout?
View Solution
Allopurinol is a xanthine oxidase inhibitor that reduces uric acid production in the body.
It helps in the long-term management of gout by lowering serum uric acid levels and preventing gout attacks.
Unlike colchicine, which treats acute inflammation, allopurinol is used for long-term gout prevention. Quick Tip: Patients should increase water intake while taking allopurinol to help flush excess uric acid from the kidneys and prevent kidney stone formation.
Which of the following is an extracellular electrolyte?
View Solution
Sodium (\( Na^+ \)) is the primary extracellular electrolyte responsible for maintaining fluid balance and nerve function.
It regulates osmotic pressure and plays a crucial role in nerve impulse transmission and muscle contraction.
Sodium levels are tightly controlled by the kidneys through aldosterone and other regulatory mechanisms. Quick Tip: High sodium intake can lead to hypertension; maintaining a balanced diet with controlled sodium consumption helps prevent cardiovascular complications.
Example of alpha glucosidase inhibitor is
View Solution
Acarbose is an alpha-glucosidase inhibitor that delays carbohydrate digestion in the intestines.
It lowers postprandial blood glucose levels by slowing glucose absorption, making it useful in diabetes management.
Unlike saxagliptin or canagliflozin, acarbose specifically targets enzymatic digestion rather than insulin regulation or glucose reabsorption. Quick Tip: Acarbose should be taken with the first bite of meals to effectively reduce post-meal blood sugar spikes.
The following is used in the treatment of dental caries
View Solution
Sodium fluoride (\( NaF \)) is widely used in the treatment and prevention of dental caries.
It strengthens tooth enamel by promoting remineralization and making teeth more resistant to acid attacks from bacteria.
Fluoride ions help inhibit bacterial metabolism, reducing plaque formation and cavity development. Quick Tip: Regular use of fluoride toothpaste significantly reduces the risk of cavities by enhancing enamel strength and preventing demineralization.
Which of the following is a coumarin derivative?
View Solution
Warfarin is a coumarin derivative used as an anticoagulant to prevent blood clot formation.
It works by inhibiting vitamin K-dependent clotting factors, reducing the risk of thromboembolic events.
Warfarin is widely prescribed for patients with atrial fibrillation, deep vein thrombosis, and pulmonary embolism. Quick Tip: Patients taking warfarin should monitor their diet, as excessive vitamin K intake can reduce the drug’s effectiveness in preventing clot formation.
MOA of Gentamicin is
View Solution
Gentamicin is an aminoglycoside antibiotic that inhibits bacterial protein synthesis.
It binds to the 30S ribosomal subunit, causing misreading of mRNA and preventing bacterial protein formation, leading to cell death.
Gentamicin is effective against Gram-negative bacteria and is used for severe infections, including sepsis and pneumonia. Quick Tip: Gentamicin requires careful dosing, as prolonged use or high doses may cause nephrotoxicity and ototoxicity, affecting kidneys and hearing.
Which of the following is a desensitising agent?
View Solution
Strontium fluoride (\( SrF \)) is a well-known desensitizing agent used in dental care.
It works by occluding the dentinal tubules, reducing the transmission of stimuli to the nerve endings in teeth.
This helps alleviate sensitivity caused by enamel erosion or exposed dentin. Quick Tip: Desensitizing toothpastes often contain strontium-based compounds, providing relief for individuals with sensitive teeth.
Which of the following is a potassium sparing diuretic?
View Solution
Amiloride is a potassium-sparing diuretic that prevents potassium loss while promoting sodium excretion.
It works by inhibiting sodium channels in the renal tubules, reducing potassium secretion and maintaining electrolyte balance.
Amiloride is often prescribed alongside other diuretics to minimize hypokalemia (low potassium levels). Quick Tip: Potassium-sparing diuretics are useful for individuals at risk of electrolyte imbalances but should be used cautiously in patients with kidney disease or hyperkalemia.
The following is a hydrophilic statin
View Solution
Pravastatin is a hydrophilic statin that lowers cholesterol levels by inhibiting HMG-CoA reductase.
Unlike lipophilic statins, pravastatin has lower penetration into non-liver tissues, reducing the risk of muscle-related side effects.
It is widely used for cardiovascular disease prevention and management of hyperlipidemia. Quick Tip: Hydrophilic statins like pravastatin have a lower risk of drug interactions compared to lipophilic statins, making them preferable for certain patients.
Xanthine nucleus is seen in
View Solution
Caffeine contains a xanthine nucleus, which is characteristic of methylxanthine derivatives.
This structure allows caffeine to act as a central nervous system stimulant by inhibiting adenosine receptors.
Other xanthine derivatives include theophylline and theobromine, which are used in respiratory and cardiac therapies. Quick Tip: Xanthine derivatives like caffeine can increase alertness but excessive consumption may lead to insomnia and tachycardia.
MALOPRIM is a combination of
View Solution
MALOPRIM is a combination of pyrimethamine and dapsone, used for malaria prophylaxis.
Pyrimethamine inhibits parasitic folic acid synthesis, while dapsone enhances its effect by preventing dihydropteroate synthase activity.
This combination is particularly effective against \textit{Plasmodium falciparum, reducing the risk of malaria infection. Quick Tip: MALOPRIM is used for malaria prevention but is not suitable for treating acute malaria infections; alternative therapies should be used for treatment.
Which of the following is a second-generation Cephalosporin?
View Solution
Cefuroxime is a second-generation cephalosporin antibiotic used to treat bacterial infections.
It has increased activity against Gram-negative bacteria compared to first-generation cephalosporins.
Cefuroxime is commonly used for respiratory infections, urinary tract infections, and skin infections. Quick Tip: Second-generation cephalosporins like cefuroxime are more resistant to beta-lactamase enzymes, making them effective against penicillin-resistant strains.
The following is a Diaminopyramidine
View Solution
Pyrimethamine is a diaminopyrimidine compound used as an antimalarial and antiparasitic drug.
It works by inhibiting folic acid synthesis in \textit{Plasmodium species, preventing parasite replication.
Pyrimethamine is often combined with sulfadoxine for treating malaria, enhancing its efficacy. Quick Tip: Pyrimethamine is also used in the treatment of toxoplasmosis, often combined with sulfadiazine to increase effectiveness.
Silver nitrate is assayed by direct titration with
View Solution
Silver nitrate is assayed using direct titration with ammonium thiocyanate (\( NH_4SCN \)).
This method is based on precipitation titration, where silver ions react with thiocyanate to form silver thiocyanate.
The endpoint is determined using ferric ammonium sulfate as an indicator, which forms a red complex when excess thiocyanate is present. Quick Tip: Silver nitrate assay is commonly performed using Volhard's method, ensuring precise quantification in pharmaceutical formulations.
Which one is non-electrolyte?
View Solution
Latex is a polymeric material that does not dissociate into ions when dissolved in water, making it a non-electrolyte.
Electrolytes, such as salts and metals, dissolve in water and conduct electricity by ionizing.
Substances like potassium chloride and sodium chloride are strong electrolytes because they completely dissociate into their respective ions. Quick Tip: Non-electrolytes do not conduct electricity in solution, whereas strong electrolytes completely ionize and enhance conductivity.
Which drug is a promotility agent?
View Solution
Metoclopramide is a promotility agent that enhances gastrointestinal movement by stimulating the motility of the upper digestive tract.
It works by blocking dopamine receptors, increasing muscle contractions in the esophagus, stomach, and intestines.
Metoclopramide is commonly used to treat conditions like gastroesophageal reflux disease (GERD) and delayed gastric emptying. Quick Tip: Promotility drugs improve digestion and prevent nausea caused by delayed gastric emptying or acid reflux.
Dental Amalgam is a mixture of
View Solution
Dental amalgam is a mixture of mercury, silver, tin, and other metals used for dental fillings.
Mercury binds the metals together, forming a durable material resistant to chewing forces.
Despite concerns about mercury exposure, dental amalgam remains widely used due to its strength and longevity. Quick Tip: Proper disposal of dental amalgam is essential to minimize environmental mercury contamination.
Which reagent is used for Lead limit test?
View Solution
Thioglycolic acid is the reagent used in the Lead limit test for detecting trace amounts of lead in pharmaceutical substances.
It forms a complex with lead ions, producing a color reaction that indicates the presence of lead contamination.
This test ensures that pharmaceutical products meet safety standards and do not exceed acceptable lead levels. Quick Tip: The Lead limit test is essential for quality control in pharmaceuticals, preventing toxic effects caused by excessive lead exposure.
What is the use of Aluminium Glycinate?
View Solution
Aluminium glycinate is used as an antacid to neutralize excess stomach acid and provide relief from conditions like acid reflux and gastritis.
It works by increasing the pH of gastric contents, reducing acidity and preventing discomfort caused by excess stomach acid.
Unlike other antacids, aluminium glycinate has a mild effect and is less likely to cause constipation compared to aluminium hydroxide. Quick Tip: Antacids should not be taken alongside certain medications, as they may interfere with drug absorption—always consult a healthcare professional.
Andreasen apparatus is used to measure
View Solution
The Andreasen apparatus is a specialized instrument used to measure particle size distribution in suspensions.
It works based on sedimentation principles, where particles settle at different rates depending on their size and density.
This method is particularly useful in pharmaceutical and industrial applications for optimizing formulation processes. Quick Tip: Particle size plays a crucial role in drug solubility, absorption, and bioavailability—controlling it is key in pharmaceutical formulation.
Porosity of porous powder is defined as
View Solution
Porosity is a measure of the void spaces within a material, particularly in porous powders.
It is defined as the ratio of void volume to bulk volume, representing the fraction of space within the material that is unoccupied.
Higher porosity indicates more air gaps, influencing material properties such as permeability and compressibility. Quick Tip: Porosity plays a crucial role in pharmaceutical formulations, affecting drug dissolution rates and tablet compression efficiency.
The type of viscosity specified in I.P (Ostwald Viscometer) is
View Solution
The Ostwald viscometer is used to measure kinematic viscosity, which is the ratio of dynamic viscosity to density.
This method relies on the flow time of a liquid through a capillary tube under gravity, making it useful for fluid characterization.
Kinematic viscosity is commonly expressed in units of centistokes (cSt) in pharmaceutical and industrial applications. Quick Tip: Kinematic viscosity measurements help determine fluid stability and consistency in drug formulations and industrial products.
Brookfield viscometer is an example of type
View Solution
The Brookfield viscometer operates on the rotating spindle principle, measuring the resistance of a fluid to shear stress.
It uses a cylindrical spindle immersed in the liquid, rotating at a set speed, with torque measured to determine viscosity.
This method is widely used in quality control for semi-solid formulations, creams, and emulsions in pharmaceutical and cosmetic industries. Quick Tip: Brookfield viscometers provide accurate viscosity measurements for non-Newtonian fluids, helping optimize product texture and stability.
Thermolabile solutions are sterilized by
View Solution
Thermolabile solutions are sensitive to heat and cannot be sterilized using high-temperature methods such as autoclaving or dry heat sterilization.
Sterilization by filtration is used to remove microorganisms without exposing the solution to heat.
This method involves passing the liquid through membrane filters with pore sizes small enough to trap bacteria while allowing the fluid to pass through. Quick Tip: Membrane filtration is commonly used for sterilizing vaccines, injectable drugs, and culture media to maintain their stability.
How moist heat destroys microorganisms?
View Solution
Moist heat sterilization destroys microorganisms by causing irreversible coagulation and denaturation of their cellular proteins and enzymes.
The presence of water enhances the penetration of heat, leading to effective microbial destruction at lower temperatures compared to dry heat sterilization.
This method is widely used for sterilizing surgical instruments, media, and medical equipment. Quick Tip: Autoclaving at 121°C for 15 minutes under pressure is one of the most effective moist heat sterilization methods used in healthcare and laboratory settings.
An antigen that stimulates the lymphocytes to produce antibody is
View Solution
An immunogen is an antigen that triggers an immune response by stimulating lymphocytes to produce antibodies.
Unlike passive immunity agents like antitoxins or immunoglobulins, immunogens actively induce immunity by interacting with immune cells.
Vaccines are examples of immunogens used to generate protective immunity against infectious diseases. Quick Tip: Immunogens are essential for vaccine development, helping the body build long-term immunity against pathogens.
The poliomyelitis virus enters the body through
View Solution
Poliovirus primarily enters the human body through the mouth via contaminated food, water, or direct contact with infected individuals.
Once inside, it multiplies in the intestines and spreads through the bloodstream, potentially attacking the nervous system.
Polio can cause paralysis, making vaccination crucial for prevention. Quick Tip: Oral polio vaccines provide strong immunity and help eliminate poliovirus transmission in communities through herd immunity.
Which test is used to determine Rickettsial infections?
View Solution
The Weil-Felix test is an agglutination test used to detect Rickettsial infections by identifying antibodies against Proteus bacteria.
Although it is an older method, it has diagnostic relevance for diseases such as typhus fever and Rocky Mountain spotted fever.
More specific tests, such as indirect fluorescent antibody testing, are preferred for accuracy in modern diagnostics. Quick Tip: Weil-Felix test works by cross-reactivity between Rickettsial bacteria and \textit{Proteus antigens, making it useful but less specific than newer tests.
What does Passive immunity refer to?
View Solution
Passive immunity refers to the transfer of preformed antibodies into an individual to provide immediate protection against infections.
Unlike active immunity, which involves stimulating the immune system to produce its own antibodies, passive immunity offers temporary defense.
Examples include maternal antibodies passed to infants and therapeutic antibody injections against diseases like rabies. Quick Tip: Passive immunity provides rapid but short-term protection, while vaccines help develop long-lasting immunity through active immune response.
Gas Gangrene is commonly caused by
View Solution
Gas gangrene is an aggressive bacterial infection commonly caused by Clostridium species, especially \textit{Clostridium perfringens.
This condition occurs when bacteria infect deep wounds, leading to rapid tissue necrosis and gas production due to anaerobic metabolism.
Immediate medical intervention, including debridement and antibiotic therapy, is critical for preventing systemic spread. Quick Tip: Hyperbaric oxygen therapy is sometimes used to treat gas gangrene, as high oxygen levels inhibit \textit{Clostridium growth.
Which food is referred to as body builder's food?
View Solution
Proteins are referred to as body builder's food due to their fundamental role in muscle repair, growth, and overall body maintenance.
They are made up of amino acids, which serve as the building blocks for tissues, enzymes, and hormones.
Adequate protein intake supports muscle recovery, making it essential for athletes and physically active individuals. Quick Tip: Lean meats, eggs, legumes, and dairy products are excellent sources of high-quality protein for muscle development and recovery.
Which one of the properties is applicable to suspensions?
View Solution
Stokes' law describes the rate of sedimentation in suspensions, determining how particles settle due to gravity.
It defines the relationship between particle size, density, viscosity, and velocity of sedimentation in a dispersion system.
Pharmaceutical suspensions rely on controlled sedimentation to maintain uniform drug distribution and stability. Quick Tip: Suspensions should be shaken well before use to ensure homogeneous distribution of active ingredients and prevent phase separation.
Methyl cellulose belongs to which type of polymer?
View Solution
Methyl cellulose is a non-ionic polymer derived from cellulose.
Its non-ionic nature makes it widely used as a thickening agent, emulsifier, and stabilizer in pharmaceutical and industrial applications.
Due to its water-solubility and gelling properties, it is often incorporated into controlled drug delivery formulations. Quick Tip: Methyl cellulose's temperature-sensitive gelation makes it useful in ophthalmic formulations and controlled-release systems.
What is the HLB value of Span 80?
View Solution
Span 80 (Sorbitan monooleate) has an HLB (Hydrophilic-Lipophilic Balance) value of 4.3.
Its low HLB value indicates that it is primarily a lipophilic surfactant, making it suitable for stabilizing water-in-oil emulsions.
It is widely used in pharmaceutical, cosmetic, and food formulations to enhance emulsion stability. Quick Tip: HLB values help determine the suitability of emulsifiers for different formulations, with lower values favoring water-in-oil emulsions.
The term "room temperature" refers to which range?
View Solution
Room temperature is commonly defined as the range between 15°C and 30°C.
This temperature range is suitable for storing many pharmaceutical and food products without requiring refrigeration or excessive heat protection.
It provides an optimal environment for chemical stability and biological preservation. Quick Tip: Controlled room temperature storage is crucial for maintaining drug efficacy, as extreme temperatures can alter the stability of active ingredients.
Partial or complete removal of the crown of a tablet from the main body is called----
View Solution
Capping refers to the partial or complete detachment of the top or bottom surface of a tablet from its main body.
It occurs due to insufficient bonding between granules, excessive air entrapment, or improper compression force during tablet manufacturing.
Preventive measures include optimizing compression pressure, ensuring proper binder usage, and improving granule flow properties. Quick Tip: Capping in tablets can be minimized by controlling moisture content and adjusting die settings during tablet compression.
Which test ensures complete removal of microorganisms from parenteral preparations?
View Solution
Sterility testing is performed to ensure that parenteral preparations are free from viable microorganisms.
It involves incubating samples in suitable media and monitoring microbial growth under controlled conditions.
This test is critical for injectable drugs and intravenous fluids to prevent infections in patients. Quick Tip: Sterility testing follows strict guidelines under pharmacopoeial standards, using methods like membrane filtration or direct inoculation for accurate detection.
Which of the following hypnotic agents is absorbed slowly?
View Solution
Phenobarbital is a long-acting barbiturate hypnotic that is absorbed slowly from the gastrointestinal tract.
Its delayed absorption leads to a prolonged onset of action, making it effective for sustained sedation and seizure control.
Unlike short-acting hypnotics, phenobarbital has a longer half-life, resulting in extended effects over time. Quick Tip: Phenobarbital should be used cautiously, as prolonged use may lead to tolerance, dependence, and withdrawal symptoms upon discontinuation.
Castor oil is used in lipsticks for
View Solution
Castor oil is widely used in lipsticks to prevent bleeding of color beyond the lip lines.
Its thick and cohesive nature helps keep the pigment contained within the application area, ensuring sharp definition.
Additionally, castor oil contributes to smooth texture and hydration, making lipsticks more comfortable to wear. Quick Tip: Lipsticks with castor oil provide better longevity and prevent feathering, making them ideal for precise application.
The unit of elimination rate constant for first order kinetics is
View Solution
In first-order kinetics, the elimination rate constant (\( k \)) has the unit \( Time^{-1} \), indicating that drug elimination is proportional to the remaining concentration.
The equation governing first-order elimination is:
\[ C_t = C_0 e^{-kt} \]
where \( C_t \) is the concentration at time \( t \), \( C_0 \) is the initial concentration, and \( k \) is the elimination rate constant. Quick Tip: First-order drug elimination follows an exponential decay pattern, meaning a constant fraction of the drug is removed per unit time.
Which of the following represents the correct order of bioavailability of dosage forms?
View Solution
The correct order of bioavailability is:
\[ Solutions > Suspensions > Tablets > Enteric coated tablets \]
Solutions have the highest bioavailability as the drug is already dissolved, enabling immediate absorption.
Suspensions require minimal dissolution before absorption, followed by tablets which undergo disintegration and dissolution.
Enteric-coated tablets have delayed absorption as they must first dissolve in the intestines rather than the stomach. Quick Tip: Liquid dosage forms generally offer faster drug absorption compared to solid forms due to immediate dissolution and availability for systemic circulation.
In nonlinear pharmacokinetics, the Y-intercept of Lineweaver – Burke graph represents which quantity?
View Solution
The Lineweaver-Burke plot is a linear transformation of the Michaelis-Menten equation, used to analyze enzyme kinetics and nonlinear pharmacokinetics.
In this plot, the equation is expressed as:
\[ \frac{1}{V} = \frac{Km}{Vmax} \cdot \frac{1}{[S]} + \frac{1}{Vmax} \]
where \( V \) is the reaction velocity, \( Vmax \) is the maximum rate of reaction, \( Km \) is the Michaelis constant, and \( [S] \) is the substrate concentration.
The Y-intercept (\( \frac{1}{Vmax} \)) represents the inverse of the maximum reaction rate, providing insight into drug metabolism and saturation kinetics. Quick Tip: Lineweaver-Burke plots are useful for estimating kinetic parameters but may distort error distribution—nonlinear regression methods provide more accurate analysis.
Calculate the loading dose required to attain a steady state of 10mg/L, if volume of distribution is 10L.
View Solution
The loading dose (\( LD \)) is calculated using the formula:
\[ LD = C_{ss} \times V_d \]
where \( C_{ss} \) is the desired steady-state concentration and \( V_d \) is the volume of distribution.
Substituting the given values:
\[ LD = 10 mg/L \times 10 L = 100 mg \]
Thus, the required loading dose to achieve a steady-state concentration of 10 mg/L is 100 mg. Quick Tip: A loading dose helps quickly achieve therapeutic drug levels, particularly for drugs with long half-lives—maintenance dosing then sustains the steady state.
Equation of zero order half-life is given by the equation
View Solution
In zero-order kinetics, the rate of drug elimination is constant and independent of drug concentration.
The half-life (\( t_{1/2} \)) is given by the equation:
\[ t_{1/2} = \frac{A_0}{K_0} \]
where \( A_0 \) is the initial drug concentration and \( K_0 \) is the zero-order rate constant.
Unlike first-order kinetics, where half-life remains constant, in zero-order kinetics, half-life depends on the initial drug concentration. Quick Tip: Drugs exhibiting zero-order kinetics, like alcohol and phenytoin, show a constant elimination rate regardless of plasma concentration.
When the enzymes get saturated during active transport, the rate of the process becomes
View Solution
During active transport, when enzyme systems responsible for drug transport become fully saturated, the rate of transport follows zero-order kinetics.
This means the transport rate becomes independent of drug concentration and proceeds at a constant rate.
Such saturation occurs in carrier-mediated absorption, enzymatic metabolism, and renal tubular secretion processes. Quick Tip: Drugs like phenytoin exhibit zero-order elimination kinetics when metabolic enzymes reach saturation, leading to dose-dependent elimination rates.
Passive diffusion follows
View Solution
Passive diffusion follows Fick's first law of diffusion, which states that the rate of diffusion is directly proportional to the concentration gradient.
Mathematically, it is expressed as:
\[ J = -D \frac{dC}{dx} \]
where \( J \) is the diffusion flux, \( D \) is the diffusion coefficient, and \( \frac{dC}{dx} \) represents the concentration gradient.
Passive diffusion allows drugs to move across biological membranes without requiring energy, driven solely by differences in concentration. Quick Tip: Lipophilic drugs exhibit higher passive diffusion due to their ability to cross lipid membranes easily, influencing their absorption rates.
Which of the following factors does not affect the rate of excretion of drugs by kidneys?
View Solution
Blood pH does not directly impact drug excretion by the kidneys.
Renal excretion is primarily influenced by renal blood flow, urine pH, and kidney function, which affect drug filtration, reabsorption, and secretion.
While urine pH can alter drug ionization and renal clearance, blood pH remains relatively stable and does not significantly impact excretion processes. Quick Tip: Weak acids are excreted faster in alkaline urine, while weak bases are excreted more efficiently in acidic urine due to ionization effects.
Deficiency of which neurotransmitter causes Parkinson's disease?
View Solution
Parkinson's disease is caused by the deficiency of dopamine, a neurotransmitter responsible for regulating movement and coordination.
Dopamine-producing neurons in the substantia nigra of the brain progressively degenerate, leading to symptoms like tremors, stiffness, and impaired motor function.
Treatment strategies often include dopamine replacement therapy, such as levodopa, to restore dopamine levels and alleviate symptoms. Quick Tip: Exercise and a healthy diet may help slow Parkinson's progression by promoting dopamine function and neuroprotection.
Testosterone is an example of
View Solution
Testosterone is the primary male sex hormone responsible for the development of male reproductive tissues and secondary sexual characteristics.
It plays a crucial role in muscle growth, bone density, and the regulation of libido.
Although present in both sexes, males have significantly higher levels compared to females. Quick Tip: Testosterone production declines with age, impacting muscle mass and energy levels in older individuals.
"Pink puffers" is a term used for
View Solution
"Pink puffers" refers to patients with emphysema, a chronic lung condition characterized by the destruction of alveoli and difficulty in gas exchange.
These individuals often appear thin, with pursed-lip breathing and a pink complexion due to the increased effort required to maintain oxygen levels.
Emphysema is a subtype of COPD (Chronic Obstructive Pulmonary Disease), primarily caused by smoking and environmental pollutants. Quick Tip: Unlike "blue bloaters" associated with chronic bronchitis, "pink puffers" rely on increased respiratory efforts to compensate for poor oxygen exchange.
Which mediators are primarily released in the late and early stages of bronchial asthma?
View Solution
Bronchial asthma involves an early phase mediated by histamine release, causing airway constriction and inflammation.
In the late phase, immune cells such as macrophages and neutrophils contribute to sustained inflammation and tissue damage.
This biphasic response leads to recurring episodes of airway obstruction, mucus production, and bronchial hyperresponsiveness. Quick Tip: Anti-inflammatory medications such as corticosteroids are often used to manage the late phase of asthma and prevent long-term lung damage.
Which anti-hypertensive drug is mostly preferred in pregnancy?
View Solution
Methyldopa is the preferred anti-hypertensive drug for managing hypertension during pregnancy due to its safety profile.
It acts centrally as an alpha-2 adrenergic agonist, reducing blood pressure by decreasing sympathetic outflow from the brain.
Unlike ACE inhibitors such as captopril, methyldopa does not pose a risk of fetal toxicity, making it suitable for long-term use in pregnancy. Quick Tip: Methyldopa is frequently recommended for pregnancy-induced hypertension, but close monitoring is essential to prevent excessive blood pressure reduction.
The anti-hypertensive prodrug converted into active metabolite to exert its action is
View Solution
Enalapril is an anti-hypertensive prodrug that is metabolized into its active form, enalaprilat, in the liver.
As an ACE inhibitor, enalapril blocks the conversion of angiotensin I to angiotensin II, leading to vasodilation and reduced blood pressure.
The prodrug formulation enhances its oral bioavailability, while enalaprilat itself is available only as an injectable form. Quick Tip: ACE inhibitors like enalapril should be avoided during pregnancy due to their potential to cause fetal toxicity and developmental complications.
A 65-year-old male is suffering from chest pain, is taken to hospital, and was diagnosed with classical angina. What is the first-line drug administered immediately to relieve the symptom?
View Solution
Sublingual nitroglycerin is the first-line treatment for relieving acute angina symptoms due to its rapid onset of action.
It works by dilating blood vessels, reducing myocardial oxygen demand, and improving coronary circulation.
Administering nitroglycerin sublingually allows direct absorption into the bloodstream, bypassing first-pass metabolism, leading to faster relief. Quick Tip: Patients experiencing chest pain should sit down and take sublingual nitroglycerin as prescribed—if pain persists, seek emergency medical attention immediately.
First step of atherosclerosis is
View Solution
The first step in atherosclerosis is oxidation of low-density lipoprotein (LDL), which leads to the accumulation of modified LDL particles in arterial walls.
Oxidized LDL triggers an inflammatory response, attracting immune cells such as macrophages, which engulf LDL and form foam cells—key components of plaque buildup.
This process initiates progressive narrowing of arteries, increasing the risk of heart disease and stroke over time. Quick Tip: Managing LDL cholesterol through a balanced diet, exercise, and medication helps prevent the early stages of atherosclerosis and cardiovascular complications.
The prominent symptom in chronic renal failure is
View Solution
Chronic renal failure results in a gradual decline in kidney function, leading to the accumulation of waste products in the blood.
One of the most prominent indicators is increased serum creatinine, reflecting impaired kidney filtration capacity.
Elevated creatinine levels signal reduced glomerular filtration rate (GFR), a key diagnostic marker for chronic kidney disease (CKD). Quick Tip: Regular monitoring of serum creatinine and kidney function tests helps assess disease progression and treatment effectiveness.
Which of the following symptom initiates haemodialysis for patients with acute renal failure?
View Solution
Persistent pulmonary edema is a serious complication of acute renal failure, warranting urgent haemodialysis to remove excess fluid and restore balance.
When the kidneys fail to filter waste and fluid effectively, accumulation leads to life-threatening respiratory distress.
Dialysis helps clear excess fluid, electrolytes, and toxins, stabilizing the patient’s condition. Quick Tip: Immediate dialysis is required in cases of volume overload, electrolyte imbalances, or severe metabolic acidosis in acute renal failure.
According to the principles of ICH GCP, what is the most important consideration when conducting a Clinical Trial?
View Solution
According to ICH Good Clinical Practice (GCP) guidelines, the protection of trial subjects is the highest priority in clinical trials.
Ensuring participant safety, informed consent, and ethical treatment are fundamental aspects of responsible trial conduct.
While accuracy, adherence, and statistical quality are important, safeguarding human rights and well-being is paramount in all research studies. Quick Tip: Compliance with ICH GCP principles ensures ethical research practices, maintaining credibility and trust in clinical studies.
The neurotransmitter associated specifically with bipolar disorder is
View Solution
Bipolar disorder is strongly linked to dopamine dysregulation, affecting mood stability and cognitive functions.
Fluctuations in dopamine levels contribute to manic episodes, characterized by heightened energy, impulsivity, and reduced need for sleep.
Additionally, imbalances in serotonin and norepinephrine also play a role in depressive phases of bipolar disorder. Quick Tip: Mood stabilizers such as lithium help regulate dopamine levels, reducing extreme mood swings in individuals with bipolar disorder.
Inflammatory bowel disease is commonly seen among the people of
View Solution
Inflammatory bowel disease (IBD), including Crohn’s disease and ulcerative colitis, is more prevalent among Northern European populations.
Genetic predisposition, environmental triggers, and dietary habits contribute to the higher incidence of IBD in these regions.
While IBD can affect individuals worldwide, Westernized diets and lifestyles are believed to play a role in increasing cases in developed nations. Quick Tip: Maintaining a balanced diet, managing stress, and following prescribed treatments help reduce IBD symptoms and prevent flare-ups.
Gray Baby syndrome occurs in newborns with
View Solution
Gray Baby Syndrome occurs in newborns due to chloramphenicol toxicity, resulting from immature liver enzymes unable to metabolize the drug effectively.
The condition leads to symptoms such as cyanosis, low blood pressure, vomiting, and circulatory collapse, hence the characteristic gray skin tone.
This syndrome underscores the importance of cautious drug administration in neonates, particularly those with underdeveloped metabolic pathways. Quick Tip: Due to the risk of toxicity, chloramphenicol use in neonates is avoided, and safer alternatives are preferred for bacterial infections.
The basic principle of Therapeutic Drug Monitoring is to measure
View Solution
Therapeutic Drug Monitoring (TDM) is a clinical practice that measures blood drug concentration to ensure optimal dosing and efficacy.
By assessing plasma drug levels, clinicians can prevent toxicity, optimize therapeutic outcomes, and adjust dosing for individual patients.
TDM is commonly applied to drugs with narrow therapeutic indices, such as anticonvulsants, antibiotics, and immunosuppressants. Quick Tip: Regular monitoring of plasma drug levels helps maintain efficacy while minimizing adverse effects, improving patient safety.
Which one of the following drugs is not a 5-HT uptake inhibitor but used as an antidepressant?
View Solution
Maprotiline is an antidepressant that does not function as a selective serotonin reuptake inhibitor (SSRI).
Instead, it is a tetracyclic antidepressant that mainly inhibits norepinephrine reuptake, distinguishing it from SSRIs like fluoxetine, paroxetine, and sertraline.
Maprotiline is often prescribed for major depressive disorders, particularly in patients who may not respond well to SSRI treatment. Quick Tip: Unlike SSRIs, maprotiline targets norepinephrine pathways, providing alternative treatment options for depression.
Heparin is contraindicated in
View Solution
Heparin is contraindicated in bleeding disorders due to its potent anticoagulant properties, which can exacerbate bleeding tendencies.
It works by inhibiting thrombin and factor Xa, making it useful for preventing clot formation but dangerous in patients with active bleeding conditions.
Other contraindications include thrombocytopenia, severe hypertension, and conditions with a high risk of hemorrhage. Quick Tip: Patients on heparin therapy should undergo regular clotting profile tests to avoid excessive bleeding complications.
Which is the first-line drug for a patient with Type-2 diabetes and obesity?
View Solution
Metformin is the first-line drug for Type-2 diabetes, particularly in patients with obesity, due to its insulin-sensitizing and weight-neutral properties.
It works by decreasing hepatic glucose production, enhancing insulin sensitivity, and reducing intestinal glucose absorption.
Metformin does not induce weight gain or hypoglycemia, making it an optimal choice for diabetic patients with obesity. Quick Tip: Metformin is commonly prescribed as the initial therapy for Type-2 diabetes, but gastrointestinal side effects can occur—starting with a low dose improves tolerance.
Which of the following antidepressants is a selective short-acting MAO-A inhibitor?
View Solution
Moclobemide is a selective short-acting MAO-A inhibitor, used as an antidepressant to enhance serotonin and norepinephrine levels.
Unlike non-selective MAO inhibitors, moclobemide has a lower risk of dietary tyramine interactions, improving its safety profile.
It is commonly used in depressive disorders, especially where conventional antidepressants may not be effective. Quick Tip: Moclobemide requires dietary precautions but is safer than irreversible MAO inhibitors like tranylcypromine, reducing hypertensive crisis risk.
Which of the following is not a selective muscarinic receptor stimulant?
View Solution
Oxotremorine is a non-selective muscarinic receptor stimulant that affects both central and peripheral cholinergic pathways.
Unlike methacholine, pilocarpine, and carbachol, which selectively activate muscarinic receptors in peripheral tissues, oxotremorine also influences neuronal pathways.
It has been used in research settings to induce tremors, mimicking Parkinson's disease symptoms for neuropharmacological studies. Quick Tip: Selective muscarinic stimulants are clinically used for bronchial challenge tests (methacholine), glaucoma treatment (pilocarpine), and urinary retention (carbachol).
The shape of a polarographic curve is
View Solution
The polarographic curve follows an S-shape (sigmoidal curve) due to the diffusion-controlled electrochemical reaction at the dropping mercury electrode.
Initially, current is low, but as the voltage increases, a rapid rise occurs due to ion reduction at the electrode surface.
This technique is widely applied in quantitative analysis of metal ions, biological compounds, and pharmaceutical formulations. Quick Tip: Polarography measures oxidation-reduction reactions in solutions, helping analyze metal ions and organic compounds efficiently.
Estimation of the amount of biological activity in unit quantity is known as
View Solution
Bioassay is the quantitative measurement of biological activity present in unit quantities of a drug or biological substance.
It is used to determine potency, therapeutic effects, and toxicity levels of pharmaceutical compounds by assessing their effects on living cells or organisms.
Bioassays play a crucial role in drug development, vaccine efficacy testing, and clinical pharmacology. Quick Tip: Bioassays help establish the biological potency of drugs when chemical assays are insufficient for determining their efficacy.
Potentiometric method is used for the detection of
View Solution
The potentiometric method is used to determine the accurate endpoint in titrations by measuring electrode potential changes.
Unlike visual indicators, potentiometric titration relies on an electrode system that detects changes in voltage corresponding to reaction completion.
This technique is widely used in acid-base, redox, and precipitation titrations for precise endpoint determination. Quick Tip: Potentiometric titration eliminates subjective errors in endpoint detection, making it ideal for high-precision chemical analysis.
Faradic current is due to
View Solution
Faradic current arises due to electrochemical reactions occurring at the dropping mercury electrode (DME) in polarography.
This current results from electron transfer between electroactive species and the electrode, enabling precise analytical measurements.
The controlled flow of mercury drops provides a renewed electrode surface, ensuring consistent and reproducible readings. Quick Tip: Faradic current plays a vital role in electrochemical analysis, helping determine concentration and reaction kinetics of electroactive compounds.
Which one of the following is assayed by complexometric method?
View Solution
Piperazine is assayed using the complexometric method, which relies on metal-ion complex formation for quantitative analysis.
This technique involves titration with chelating agents like EDTA, which form stable ligand-metal complexes for precise measurement.
Complexometric assays are extensively used for determining metal content in pharmaceuticals, water analysis, and industrial applications. Quick Tip: Complexometric titration is widely used in pharmaceutical quality control for metal ion quantification and impurity assessment.
Thickness of TLC Plate is around
View Solution
Thin-layer chromatography (TLC) plates typically have a thickness ranging from 0.1 mm to 2 mm, depending on their intended application.
This thin layer consists of a stationary phase, usually silica gel, alumina, or cellulose, facilitating separation of compounds by capillary action.
Thinner plates provide higher resolution, while thicker plates allow for greater sample loading capacity. Quick Tip: The thickness of the TLC plate affects separation efficiency, with thinner layers offering faster migration and better resolution.
Ion Exchange Chromatography is used for separation of
View Solution
Ion exchange chromatography is widely employed for the separation of amino acids, as it exploits differences in their charge properties.
Amino acids interact with a charged stationary phase, where they are selectively retained and eluted based on their isoelectric points (pI).
This technique is particularly valuable in protein purification, biomolecular analysis, and pharmaceutical applications. Quick Tip: Ion exchange chromatography is a powerful tool in biochemistry, enabling precise separation and quantification of amino acids and proteins.
Gas cell which is used for sampling of gases in IR is made up of
View Solution
Potassium bromide (KBr) gas cells are commonly used for sampling gases in infrared (IR) spectroscopy, as KBr is transparent to IR radiation.
KBr allows optimal transmission of IR wavelengths, ensuring accurate absorption spectra measurement without interference.
Other materials like NaCl and CaF\(_2\) may also be used depending on specific spectral requirements. Quick Tip: KBr windows in IR spectroscopy provide high-quality transmission, making them a preferred choice for gas-phase sample analysis.
Bathochromic shift depends upon
View Solution
Bathochromic shift, also known as the red shift, occurs when the absorption wavelength of a molecule shifts to a longer wavelength due to increased conjugation.
Conjugated double bonds extend delocalization, lowering the energy required for electronic transitions and moving absorption peaks to the red region of the spectrum.
This phenomenon is commonly observed in organic chromophores, dyes, and UV-visible spectroscopy applications. Quick Tip: The bathochromic shift is exploited in drug design, material science, and spectroscopy for tuning molecular absorption properties.
Which of the following is an aprotic solvent?
View Solution
An aprotic solvent is one that does not donate protons (hydrogen ions) in solution and lacks acidic hydrogen atoms.
Benzene is an example of an aprotic solvent, as it is non-polar and does not engage in hydrogen bonding.
In contrast, ethyl alcohol and acetic acid are protic solvents, since they have hydroxyl (-OH) or carboxyl (-COOH) functional groups capable of donating protons. Quick Tip: Aprotic solvents such as benzene, acetone, and dimethyl sulfoxide (DMSO) are commonly used in organic synthesis to dissolve non-polar compounds.
Which of the following is a chelating agent?
View Solution
Ethylenediaminetetraacetic acid (EDTA) is a powerful chelating agent that binds metal ions by forming stable coordinate complexes.
It is widely used in medicine (heavy metal detoxification), analytical chemistry, and industrial applications to remove unwanted metal contaminants.
Chelating agents function by sequestering metal ions, preventing their interaction with other molecules and stabilizing solutions. Quick Tip: EDTA is commonly used in biochemistry, water treatment, and pharmaceuticals for controlling metal ion concentrations.
Which Isotope is used in RLA?
View Solution
Radioimmunoassay (RIA), a sensitive analytical technique used for detecting substances in biological samples, commonly employs Iodine-125 (\(^{125}I\)) as a radioactive isotope. \(^{125}I\) is used due to its long half-life and low-energy gamma emissions, making it highly effective for labeling antigens or antibodies in immunoassays.
This technique enables accurate quantification of hormones, drugs, and proteins by measuring radiation emitted from labeled molecules. Quick Tip: Radioimmunoassays provide high sensitivity in detecting trace biomolecules but require special handling and disposal due to radioactive material usage.
What is the tracer in Radio immuno assay?
View Solution
In radioimmunoassay (RIA), a radioactive antigen acts as the tracer, allowing precise measurement of analyte concentration through competitive binding.
When mixed with a specific antibody, the labeled antigen competes with the sample antigen for binding sites, and the amount of radioactivity detected correlates with antigen levels.
This method is extensively used for hormone assays, drug testing, and clinical diagnostics due to its high sensitivity and specificity. Quick Tip: RIA enables ultrasensitive detection but requires specialized laboratories due to the handling of radioactive materials.
For a given sample, ELISA allows for rapid screening and quantification of the presence of the following
View Solution
Enzyme-linked immunosorbent assay (ELISA) is a powerful analytical tool designed for rapid screening and quantification of antigens in biological samples.
By utilizing antibody-antigen interactions, ELISA can detect the presence and concentration of specific proteins, pathogens, and biomarkers.
This method is extensively applied in medical diagnostics, drug development, and immunological research due to its high sensitivity and specificity. Quick Tip: ELISA is widely used for infectious disease detection, including HIV, hepatitis, and COVID-19 antigen testing, ensuring reliable diagnostics.
Thermal conductivity detector is known as
View Solution
A thermal conductivity detector (TCD) is commonly referred to as a Katharometer, an instrument used in gas chromatography for detecting analytes based on thermal conductivity differences.
TCD operates by measuring changes in heat transfer as different gases pass through the detector, allowing non-destructive and universal analysis of compounds.
Unlike flame ionization detectors (FID), TCD can analyze both organic and inorganic species, making it versatile for various applications. Quick Tip: Katharometers are useful in industrial gas analysis, environmental monitoring, and pharmaceutical research due to their broad detection capabilities.
Ligands bearing more than one electron donating group are called
View Solution
Chelating agents are ligands that contain multiple electron-donating groups, allowing them to form stable ring-like metal complexes with metal ions.
These agents effectively sequester metal ions, preventing undesirable interactions, making them useful in medicine, water treatment, and analytical chemistry.
Common chelating agents include EDTA, citrate, and aminopolycarboxylates, which bind strongly to metals. Quick Tip: Chelating agents are widely used in pharmaceutical formulations, heavy metal detoxification, and industrial processes to control metal ion activity.
Triethanolamine is used as a
View Solution
Triethanolamine is commonly used as a masking agent in analytical chemistry to prevent interference from specific metal ions during titrations.
It works by binding interfering ions, allowing the target analyte to react selectively in complexometric and spectrophotometric analyses.
Triethanolamine also finds applications in cosmetics, emulsifiers, and pH adjustment in formulations. Quick Tip: Masking agents improve analytical accuracy by neutralizing unwanted chemical reactions that interfere with main analyte detection.
If the Mobile phase is gas in partition Chromatography, it is called as
View Solution
Gas-Liquid Chromatography (GLC) is a type of partition chromatography where the mobile phase is a gas, and the stationary phase is a liquid-coated support.
GLC is widely used in analytical chemistry, particularly in volatile compound separation, drug testing, and environmental analysis.
This method enables precise quantification and identification of components in complex mixtures based on their affinity for the stationary phase. Quick Tip: Gas-liquid chromatography provides high-resolution separation of volatile compounds and is extensively used in food, pharmaceutical, and forensic analysis.
The eluent strength is a measure of
View Solution
Eluent strength refers to a solvent's ability to displace analytes from the stationary phase in chromatographic separation.
It is determined by the adsorption energy of the solvent, affecting retention time and resolution in chromatographic techniques.
Higher eluent strength results in faster analyte migration, optimizing separations in liquid chromatography and thin-layer chromatography (TLC). Quick Tip: Proper selection of solvent strength enhances chromatographic resolution, ensuring effective separation and analysis of complex mixtures.
Which of the following is a part of the quality system covering the manufacturing and testing of active ingredients and finished products?
View Solution
Good Manufacturing Practice (GMP) ensures that pharmaceuticals, food products, and medical devices are consistently produced and controlled according to quality standards.
GMP guidelines regulate manufacturing, testing, documentation, and safety protocols, preventing contamination, errors, and deviations in product quality.
This framework plays a critical role in ensuring consumer safety and efficacy of medicinal products worldwide. Quick Tip: Adherence to GMP standards ensures that pharmaceutical and healthcare products meet regulatory requirements and safety benchmarks.
Standard Operating Procedure (SOP) refers to
View Solution
A Standard Operating Procedure (SOP) is a detailed, written document that outlines step-by-step instructions for performing a specific task or process.
SOPs ensure uniformity, consistency, and compliance in operational activities across industries, including pharmaceuticals, healthcare, and manufacturing.
They serve as a guideline for employees, preventing errors and maintaining efficiency in workflows. Quick Tip: Strict adherence to SOPs improves quality control, regulatory compliance, and operational safety, making them essential in high-precision industries.
The following are the main components of GMP except
View Solution
Good Manufacturing Practice (GMP) ensures quality, safety, and consistency in manufacturing processes for pharmaceuticals, food, and cosmetics.
Its key components include Products, People, and Processes, focusing on proper formulation, personnel training, and standardized production techniques.
The term "Perfect" is not a formal GMP component, as the emphasis is on continuous improvement and compliance rather than absolute perfection. Quick Tip: GMP guidelines help prevent contamination, errors, and inconsistencies, ensuring that products meet strict regulatory standards before distribution.
The Narcotic Drugs and Psychotropic Substances Act was passed in the year
View Solution
The Narcotic Drugs and Psychotropic Substances Act was enacted in 1985 to regulate and control the production, distribution, and consumption of narcotic substances in India.
This act establishes legal guidelines for preventing drug abuse, controlling illegal trafficking, and promoting medical and scientific use of narcotics and psychotropic drugs.
Severe penalties are imposed for offenses related to drug possession, smuggling, and unauthorized distribution. Quick Tip: The NDPS Act categorizes narcotics into different schedules, ensuring strict regulation to prevent misuse while enabling their medical applications.
Schedule H of Pharmacy Act comprises of the standards of
View Solution
Schedule H of the Pharmacy Act specifies the list of prescription drugs that require a valid medical prescription before dispensing.
These drugs are considered potentially hazardous if misused, necessitating regulatory control to prevent self-medication and drug abuse.
Pharmacists are required to maintain records of dispensing, ensuring compliance with healthcare regulations. Quick Tip: Schedule H drugs include antibiotics, hormonal therapies, psychotropic medicines, and controlled substances, helping regulate their distribution and usage.
How many nominated members are present in the state pharmacy council?
View Solution
The State Pharmacy Council consists of six nominated members as part of its governing body.
These members are selected to ensure compliance with pharmacy regulations, oversee professional standards, and contribute to policy-making for pharmaceutical affairs.
The council also includes elected representatives, ensuring a balanced and regulated framework for pharmacy governance within the state. Quick Tip: State Pharmacy Councils regulate pharmaceutical practice, professional ethics, and licensing to maintain healthcare integrity.
Which authority determines prices of formulations?
View Solution
The National Pharmaceutical Pricing Authority (NPPA) is responsible for determining and regulating drug prices in India.
NPPA ensures that essential medicines remain affordable, preventing excessive pricing by pharmaceutical manufacturers while maintaining quality.
It enforces price controls under the Drug Price Control Order (DPCO), ensuring fair market pricing for life-saving medications. Quick Tip: NPPA regulates the maximum retail price (MRP) of scheduled drugs, balancing consumer affordability and industry sustainability.
Ganja is obtained from
View Solution
Ganja is derived from the flower and fruit tops of the Cannabis plant, containing high concentrations of psychoactive compounds such as THC (tetrahydrocannabinol).
It is used for recreational, medicinal, and industrial purposes, with regulations varying across different countries.
Cannabis-based substances are subject to strict legal controls, ensuring responsible usage in healthcare and research. Quick Tip: Cannabis derivatives, including ganja and hashish, are regulated under the NDPS Act, governing production, distribution, and medical applications.








































































Comments