VITEEE 2022 Question paper for is available for download. The exam was successfully organized by Vellore Institute of Technology (VIT). The question paper comprised a total of 125 questions divided among 5 sections. Candidates can download the memory-based VITEEE 2022 question paper using the link below.
Latest Update: Download VITEEE 2024 Question Paper with Solution PDF
VITEEE 2022 Question Paper PDF With Answer Key
| VITEEE 2022 Question Paper PDF With Answer Key | Check Solution |
VITEEE 2022 QuestionPaer With Solution
PHYSICS
Question 1:
The root mean square speed of smoke particles of mass \( 5 \times 10^{-17} \) kg in their Brownian motion in air at NTP is approximately. (Given \( k = 1.38 \times 10^{-23} \, \text{JK}^{-1} \))
The equation of a particle executing simple harmonic motion is given by \[ x = \sin \pi \left( t + \frac{1}{3} \right) \, \text{m}. \] At \( t = 1 \, \text{s} \), the speed of particle will be (Given \( \pi = 3.14 \)):
Following are expressions for four plane simple harmonic waves \(y_1 = A \cos 2 \pi \left( n_1 t + \frac{x}{\lambda_1} \right)\),
\(y_2 = A \cos 2 \pi \left( n_1 t + \frac{x}{\lambda_1} + \pi \right)\),
\(y_3 = A \cos 2 \pi \left( n_2 t + \frac{x}{\lambda_2} \right)\),
\(y_4 = A \cos 2 \pi \left( n_2 t - \frac{x}{\lambda_2} \right)\)
The pairs of waves which will produce destructive interference and stationary waves respectively in a medium, are
If a charge \( q \) is placed at the centre of a closed hemispherical non-conducting surface, the total flux passing through the flat surface would

The electric potential \( V(x) \) in a region around the origin is given by \( V(x) = 4x^2 \, \text{volts}. \) \text{The electric charge enclosed in a cube of 1m side with its center at the origin is (in coulomb)
A heater coil is cut into two equal parts and only one part is now used in the heater. The heat generated will now be
In a region, steady and uniform electric and magnetic fields are present. These two fields are parallel to each other. A charged particle is released from rest in this region. The path of the particle will be a
An object is thrown vertically upwards. At its maximum height, which of the following quantity becomes zero?
The self-induced emf of a coil is 25 volts. When the current in it is changed at uniform rate from 10A to 25 A in 1s, the change in the energy of the inductance is:
Alternating current cannot be measured by a D.C. ammeter because
The magnetic field of a plane electromagnetic wave is given by: \[ \vec{B} = 2 \times 10^{-8} \sin \left(0.5 \times 10^3 x + 1.5 \times 10^{11} t \right) \hat{j} \, \text{T}. The amplitude of the electric field would be:
An ideal gas is expanding such that \( P T^3 = \text{constant} \). The coefficient of volume expansion of the gas is:
Two light beams of intensities in the ratio of 9 : 4 are allowed to interfere. The ratio of the intensity of maxima and minima will be:
The de Broglie wavelength of a proton and \( \alpha \)-particle are equal. The ratio of their velocities is:
The recoil speed of a hydrogen atom after it goes from \( n = 5 \) state to \( n = 1 \) state will be:
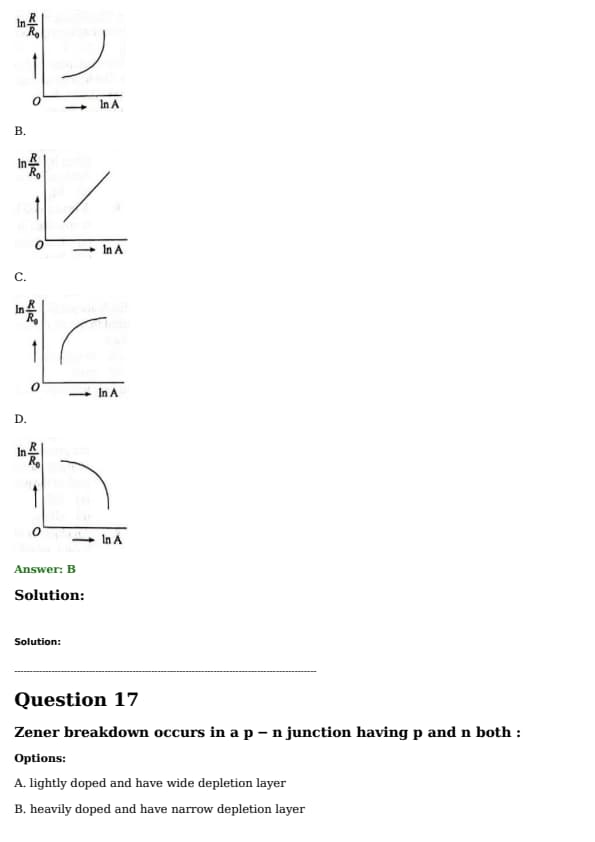
Which of the following figure represents the variation of \( \ln \left( \frac{R}{R_0} \right) \) with \( \ln A \) (If \( R \) is the radius of a nucleus and \( A \) is its mass number)?
Zener breakdown occurs in a p − n junction having p and n both:
If \( E \) and \( H \) represent the intensity of electric field and magnetizing field respectively, then the unit of \( \frac{E}{H} \) will be:
A stone of mass \( m \), tied to a string is being whirled in a vertical circle with a uniform speed. The tension in the string is:
A particle is moving with a velocity \( \vec{v} = K (y \hat{i} + x \hat{j} )\), where \( K \) is a constant. The general equation for its path is:
A particle of mass M originally at rest is subjected to a force whose direction is constant but magnitude varies with time according to the relation \[ F = F_0 \left[ 1 - \left( \frac{t - T}{T} \right)^2 \right] \] Where \( F_0 \) and \( T \) are constants. The force acts only for the time interval \( 2T \). The velocity \( v \) of the particle after time \( 2T \) is:
The magnetic moment of an electron (e) revolving in an orbit around nucleus with an orbital angular momentum is given by:
Angular momentum of the particle rotating with a central force is constant due to
The escape velocity of a body depends upon mass as:
Potential energy as a function of \(r\) is given by \( U = \frac{A}{r^{10}} - \frac{B}{r^5} \), where \(r\) is the interatomic distance, \(A\) and \(B\) are positive constants. The equilibrium distance between the two atoms will be:
If two soap bubbles of different radii are connected by a tube:
The focal length \( f \) is related to the radius of curvature \( r \) of the spherical convex mirror by:
A thermodynamic system is taken from an original state D to an intermediate state E by the linear process shown in the figure. Its volume is then reduced to the original volume from E to F by an isobaric process. The total work done by the gas from D to E to F will be:

A vertical electric field of magnitude \( 4.9 \times 10^5 \, \text{N/C} \) just prevents a water droplet of a mass \( 0.1 \, \text{g} \) from falling. The value of charge on the droplet will be: (Given \( g = 9.8 \, \text{m/s}^2 \))
In the circuit shown in the figure, the total charge is \( 750 \, \mu C \) and the voltage across capacitor \( C_2 \) is \( 20 \, \text{V} \). Then the charge on capacitor \( C_2 \) is:

For a transistor, \( \alpha \) and \( \beta \) are given as \( \alpha = \frac{I_C}{I_E} \) and \( \beta = \frac{I_C}{I_B} \). Then the correct relation between \( \alpha \) and \( \beta \) will be:
A current \( I \) flows along the length of an infinitely long, straight, thin-walled pipe. Then:
A Carnot engine has an efficiency of 50%. If the temperature of the sink is reduced by \( 40^\circ C \), its efficiency increases by 30%. The temperature of the source will be:
When you walk through a metal detector carrying a metal object in your pocket, it raises an alarm. This phenomenon works on:
An electron moving with speed \( v \) and a photon with speed \( c \), have the same de-Broglie wavelength. The ratio of kinetic energy of the electron to that of the photon is:
Assuming fully decomposed, the volume of \( \text{CO}_2 \) released will be:
Among the following, the species having the smallest bond order is:
The oxidation number of phosphorus in \( \text{Ba(H}_2\text{PO}_2)_2 \) is:
Water sample is reported to be highly polluted if BOD (Biological Oxygen Demand) value of sample becomes:
200 mL of an aqueous solution of a protein contains 1.26 g. The osmotic pressure of this solution at 300 K is found to be \( 2.57 \times 10^{-3} \, \text{bar} \). The molar mass of the protein will be: (R = 0.083 L bar mol\(^{-1}\) K\(^{-1}\))
Lyophilic sols are more stable than lyophobic sols because:
Which of the following is not a permissible arrangement of electrons in an atom?
The value of van der Waals constant ' a ' for gases \( \text{O}_2 \), \( \text{N}_2 \), \( \text{NH}_3 \), and \( \text{CH}_4 \) are 1.360, 1.390, 4.170, and 2.253 L\(^2\)atm/mol\(^2\) respectively. The gas which can most easily be liquefied is:
Which one of the following does not have a pyramidal shape?
Boric acid is polymeric due to:
Which of the following order is not correct?
A catalyst is a substance which:
Which of the following is a non-reducing sugar?
An ideal gas expands against a constant external pressure of 2.0 atmosphere from 20 litre to 40 litre and absorbs 10 kJ of heat from the surrounding. What is the change in internal energy of the system? (given: \( 1 \, \text{atm-litre} = 101.3 \, \text{J} \))
The polymer used for optical lenses is:
Which of the following order is not correct for the ionic radii of the given species: \( \text{O}^{2-} \), \( \text{S}^{2-} \), \( \text{N}^{3-} \), \( \text{P}^{3-} \)?

The IUPAC name of the following compound is:

In CsCl type structure, the co-ordination number of \( \text{Cs}^+ \) and \( \text{Cl}^- \) respectively are:
Which one of the following reactions will not result in the formation of a carbon-carbon bond?
Water is:
Carboxylic acids are more acidic than phenol and alcohol because of:
The order of increasing sizes of atomic radii among the elements O, S, Se, and As is:
Bauxite ore is generally contaminated with impurity of oxides of two elements X and Y. Which of the following statement is correct?
The partial pressure of \(CH_3\) OH(g), CO(g) and \(H_2(g)\) in equilibrium mixture for the reaction, CO(g) + (2H_2(g)) \rightleftharpoons \(CH_3OH(g)) ] are 2.0, 1.0 and 0.1 atm respectively at \(427^\circ C\). The value of \(K_p\) for the decomposition of \(CH_3OH\) to CO and \(H_2\) is: \flushleft
The conjugate base of (CH_3)_2 NH_2^+ { is:
Which of the following is not present in a nucleotide?
The shape of [Cu(NH_3{)_4]^{2+ is:
Heroin is a derivative of:
The limiting equivalent conductivity of NaCl, KCl and KBr are 126.5, 150.0 and 151.5 S cm^2 { eq^{-1 {, respectively. The limiting equivalent ionic conductivity for Br is 78 S cm^2 eq^{-1. The limiting equivalent ionic conductivity for Na^+ \text{ ions would be:
Rate of dehydration of alcohols follows the order:
An alkene having molecular formula ( {C}_7{H}_{14} ) was subjected to ozonolysis in the presence of zinc dust. An equimolar amount of the following two compounds was obtained. The IUPAC name of the alkene is:

Lanthanoid contraction can be observed in:
The form of iron obtained from blast furnace is:
A class has 175 students. The following data shows the number of students opting for one or more subjects. Maths = 100, Physics = 70, Chemistry = 40, Maths and Physics = 30, Maths and Chemistry = 28, Physics and Chemistry = 23, Maths, Physics, and Chemistry = 18.
How many have offered Maths alone?
Let \( R \) be a relation on the set \( \mathbb{N} \) defined by \[ \{(x, y) \mid x, y \in \mathbb{N}, \, 2x + y = 41\}. \] Then, \( R \) is:
The function \( f : \mathbb{R} \to \mathbb{R} \) defined by \( f(x) = x^2 + x \) is:
If \( 12\cot^2 \theta - 31\csc \theta + 32 = 0 \), then the value of \( \sin \theta \) is:
The modulus of \( (1 + i\sqrt{3})(2 + 2i) \) / \( (\sqrt{3} - i) \) is:
If \( \alpha, \beta \) are the roots of the equation \( ax^2 + bx + c = 0 \), then \[ \frac{\alpha}{a\beta + b} + \frac{\beta}{a\alpha + b} = \]
The solution set of the inequality \[ 37 - (3x + 5) \geq 9x - 8(x - 3) \text{ is:} \]
If \[ \frac{n + 2C8}{n - 2P4} = \frac{57}{16}, \text{ then the value of } n \text{ is:} \]
The middle term in the expansion of \[ (10x + x^{10})^{10} \] \text{is:
The fourth, seventh, and tenth terms of a G.P. are \( p, q, r \) respectively, then:
The point \( (t^2 + 2t + 5, 2t^2 + t - 2) \) \text{ lies on the line \( x + y = 2 \) \text{ for:
The equations of the lines which cut off an intercept 1 from the y-axis and are equally inclined to the axes are:
The distance between the parallel lines \[ 3x - 4y + 7 = 0 \quad \text{and} \quad 3x - 4y + 5 = 0 \text{ is } \frac{a}{b}. \text{ Value of } a + b \text{ is:} \]
For what value of \( k \), does the equation \[ 9x^2 + y^2 = k(x^2 - y^2 - 2x) \] \text{represent the equation of a circle?
A parabola has the origin as its focus and the line \( x = 2 \) \text{ as the directrix. Then the vertex of the parabola is at:
Equation of the ellipse whose axes are the axes of coordinates and which passes through the point (-3, 1) and has eccentricity \( \sqrt{\frac{2}{5}} \) is:
The coordinates of the point which divides the line segment joining the points \( (2, -1, 3) \) \text{ and \( (4, 3, 1) \text{ in the ratio 3 : 4 \text{ internally are:
The relationship between \( a \) and \( b \) so that the function \( f(x) \) \text{ defined by \[ f(x) = \begin{cases} ax + 1 & \text{if } x \leq 3
bx + 3 & \text{if } x > 3 \end{cases} \] \text{is continuous at \( x = 3 \), is:
The function \( f(x) \) is given by: \[ f(x) = \begin{cases} x \sin \left( \frac{1}{x} \right) & \text{for } x \neq 0
0 & \text{for } x = 0 \end{cases} \]
The variance of the data \( 2, 4, 6, 8, 10 \) \text{ is:
Find the probability of getting the sum as a perfect square number when two dice are thrown together.
The principal value of \( \sin^{-1 \left( \sin \frac{5\pi{3 \right) \text{ is:
If the system of linear equations \[ x + ky + 3z = 0, \quad 3x + ky - 2z = 0, \quad 2x + 4y - 3z = 0 \] \text{has a non-zero solution \( (x, y, z) \), then \( \frac{xz}{y^2} \) is equal to:
The value of the definite integral \[ \int_0^{\frac{\pi{2 \log(\tan x) \, dx \text{ is:
The area enclosed between the graph of \( y = x^3 \) \text{ and the lines \[ x = 0, \, y = 1, \, y = 8 \text{ is:} \]
The total number of 3-digit numbers, the sum of whose digits is even, is equal to:
To fill 12 vacancies, there are 25 candidates of which five are from the scheduled caste. If 3 of the vacancies are reserved for scheduled caste candidates while the rest are open to all, then the number of ways in which the selection can be made is:
If \[ \frac{1}{q + r}, \quad \frac{1}{r + p}, \quad \frac{1}{p + q} \] \text{are in A.P., then:
The sum of the first n terms of the series \[ 1^2 + 2.2^2 + 3^2 + 2.4^2 + 5^2 + 2.6^2 + \cdots \] \text{is \[ \frac{n(n + 1)^2}{2} \text{ when n is even. When n is odd the sum is} \]
The locus of a point that is equidistant from the lines \[ x + y - 2\sqrt{2} = 0 \quad \text{and} \quad x + y - \sqrt{2} = 0 \text{ is:} \]
The point diametrically opposite to the point \( P(1, 0) \) \text{ on the circle \[ x^2 + y^2 + 2x + 4y - 3 = 0 \text{ is:} \]
For the parabola \( y^2 = -12x \), \text{the equation of the directrix is \( x = a \). \text{ The value of \( a \) \text{ is:
The eccentricity of the curve \[ 2x^2 + y^2 - 8x - 2y + 1 = 0 \text{ is:} \]
The equation of the hyperbola with vertices at \( (0, \pm 6) \text{ and e = \frac{5{3 \text{ is:
The following determinant is equal to: \[ \begin{vmatrix} \sin^2 x & \cos^2 x & 1
\cos^2 x & \sin^2 x & 1
-10 & 12 & 2 \end{vmatrix} \]
The function \( f(x) \) \text{ is given by: \[ f(x) = \begin{cases} x[x] & \text{if } 0 \leq x \(<\) 2
(x - 1)x & \text{if } 2 \leq x \(<\) 3 \end{cases} \] \text{The function is:
The local minimum value of the function \[ f(x) = 3 + |x|, \quad x \in \mathbb{R} \] \text{is:
The value of the integral \[ \int_0^{\frac{\pi}{2}} \frac{\sqrt{\sin x}}{\sqrt{\sin x} + \sqrt{\cos x}} \, dx \] \text{is:
The equation of the plane which bisects the angle between the planes \[ 3x - 6y + 2z + 5 = 0 \quad \text{and} \quad 4x - 12y + 3z - 3 = 0 \text{ which contains the origin is:} \]
An urn contains five balls. Two balls are drawn and found to be white. The probability that all the balls are white is:
What is the total marks obtained by Meera in all the subjects?
What is the average marks obtained by these seven students in History? (rounded off to two digits)
How many students have got 60% or more marks in all the subjects?
A series is given, with one term missing. Choose the correct alternative from the given ones that will complete the series. \[ 5, 11, 24, 51, 106, \_ ? \]
In a certain code, BANKER is written as LFSCBO. How will CONFER be written in that code?
Kailash faces towards north. Turnings to his right, he walks 25 metres. He then turns to his left and walks 30 metres. Next, he moves 25 metres to his right. He then turns to the right again and walks 55 metres. Finally, he turns to the right and moves 40 metres. In which direction is he now from his starting point?
An accurate clock shows 8 O'clock in the morning. Through how many degrees will the hour hand rotate when the clock shows 20:00 (8 O'clock in the evening)?
Two statements are given followed by three conclusions numbered I, II, and III. Assuming the statements to be true, even if they seem to be at variance with commonly known facts, decide which of the conclusions logically follow(s) from the statements. Statements: 1. All utensils are spoons. 2. All bowls are spoons. Conclusions: I. No utensil is a bowl. II. Some utensils are bowls. III. No spoon is a utensil.
What was the ratio between the ages of P and Q four years ago? I. The ratio between the present ages of P and Q is 3 : 4. II. The ratio between the present ages of Q and R is 4 : 5.
What was the cost price of the suitcase purchased by Samir? I. Samir got a 25 percent concession on the labelled price. II. Samir sold the suitcase for Rs.2000 with 25 percent profit on the labelled price.
Read the following passage and answer the question that follows. \textit{"His instrument struck against something hard, dangerously near the kidney.... 'It is not quite at the kidney, my friend,' Sadao murmured.... 'My friend,' he always called his patients and so he did now, forgetting that this was his enemy." To whom does Sadao attend to in the lines above?
Choose the correct pronunciation for the word 'sorbet' from the following options:
What is the correct syllable division of the word 'indomitable'?
Read the following passage and the question below. Choose the correct answer. \textit{Gandhi never contented himself with large political or economic solutions. He saw the cultural and social backwardness in the Champaran villages and wanted to do something about it immediately. He appealed to teachers. Which of the following statements is true about the passage?
Choose the correct meaning of the idiom 'a bolt out of the blue' from the given options:






























































Comments