
Content Curator
Citric acid chemical formula is represented as C6H8O7. The IUPAC name of this compound is 2-hydroxypropane-1,2,3-tricarboxylic acid. This weak acid consists of 6 Carbon atoms, 8 Hydrogen atoms, and 7 Oxygen atoms. There are two types of citrus acid: monohydrate and anhydrous (without water).
- Citrus fruits like lemons, limes, and other citrus fruits naturally contain citric acid, a mild organic acid.
- Artificial citric acid is added by producers to processed foods to preserve them.
- It is a tribasic acid, a chemical that totally dissociates in water to create three hydrogen ions per acid molecule.
Read More: Acid, Bases and Salts
| Table of Content |
Key Terms: Citric Acid, Acid, Properties of Citric Acid, Applications of Citric Acid, 3D- Structure
Citric Acid Structure
[Click Here for Sample Questions]
Oxaloacetic acid and Acetyl CoA are combined to create citric acid. Additionally, a chemical process transfers the acetyl group CH3COO from CoA to oxaloacetic acid at the carbon of the ketone. Citric acid is a tasteless, odorless, white, crystalline chemical. A monoclinic crystal structure describes it.

This is the 2D chemical structure of the Citric Acid.

This is the 3D confirmer structure of the acid.

This is the Crystal structure form of Citric Acid.
Read More: Acid Vs Bases
Chemical Formula of Citric Acid
[Click Here for Sample Questions]
Eight hydrogen atoms, six carbon atoms, and Seven oxygen atoms make up citric acid. Citric acid is essential for the human body's energy production and for maintaining the biosynthetic process. Additionally, citric acid functions in glycolysis as a sugar breaker and supplies energy for ATP synthesis.
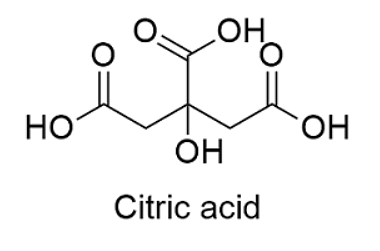
Citric Acid
- Citric acid chemical formula is represented as C6H8O7.
- The longer formula for citric acid is CH2 COOH-COHCOOH-CH2COOH.
- Its molar mass is 192.12 g mol-1.
Read More: Ammonium Chloride
Physical Properties of Citric Acid
[Click Here for Sample Questions]
A few important physical properties of Citric Acid are–
- Crystals of citric acid are of sour acidic taste, colorless, and have no odor. It's also denser than water.
- Rhombic crystals formed from water with a crystallization rate of 1 mol of water.
- The melting point of this compound is 307 °F.
- Easily soluble in ethanol, very soluble in water, and soluble in ether, and insoluble in benzene, and chloroform.
- In the air, stable; at 150 °C becomes anhydrous.
- Its anhydrous form has a density of 1.665 g ml-1 whereas its monohydrate form has a density of 1.542 g ml-1.
Read More: Acid Strengths
Chemical Properties of Citric Acid
[Click Here for Sample Questions]
A few important chemical properties of Citric Acid are–
- Citric acid is used as a pH buffer regulator in many industrial processes.
- Citric acid also has the ability to bind metals, producing chelate metals that are similar to EDTA complexes.
- The pH of the related aqueous solution ranges from pH 2 to pH 8, depending on how negatively charged the anion is.
- In an aqueous solution, the anionic -1, -2, and -3 forms of this weak acid and the neutral form are constantly in equilibrium with one another.
Read More: Salt Analysis
Applications of Citric Acid
[Click Here for Sample Questions]
Some significant uses of Citric Acid are–
- The basic pH of soaps and detergents is softened using citric acid.
- Juices, soft drinks, and other liquids frequently include citrus acids or citric acid, giving them an acidic flavor.
- It functions as a food preservative because the food is protected against deterioration by its acidic pH, which inhibits the growth of several microorganisms.
- By chelating calcium in the blood, citric acid acts as an anticoagulant and as an acidulant to control pH levels.
- Citric acid is used as a blood preservative in the pharmaceutical industry.
- To maintain the proper pH balance between acid and base, it is also used as a preservative in many cosmetics. In cosmetics, it is used to remove dead skin cells, lessen wrinkles, and level out skin tone.
- Citric acid is used in caramel to stop sugar from crystallizing.
- Citric acid is sometimes used as an emulsifier in the ice industry to avoid utilizing fat globules.
Read More: pH for Weak Acids
Things to Remember
- Citric acid chemical formula is represented as C6H8O7.
- Oxaloacetic acid and Acetyl CoA are combined to create citric acid.
- The IUPAC name of this compound is 2-hydroxypropane-1,2,3-tricarboxylic acid.
- This weak acid consists of 6 Carbon atoms, 8 Hydrogen atoms, and 7 Oxygen atoms.
- There are two types of citrus acid: monohydrate and anhydrous (without water).
- The longer formula for citric acid is CH2 COOH-COHCOOH-CH2COOH.
- Crystals of citric acid are of sour acidic taste, colorless, and have no odor. It's also denser than water.
- Citric acid is used as a pH buffer regulator in many industrial processes.
Sample Questions
Ques. Where in nature can you find citric acid? (3 Marks)
Ans. Citric acid is a natural preservative used in the food industry to impart a sour flavor to meals and soft beverages. Contrarily, citric acid is artificially added and is not a naturally occurring component of any of these meals.
Lemons, limes, oranges, grapefruits, pomelos, and tangerines are just a few examples of fruits and vegetables that naturally contain citric acid. However, almost all fruits contain modest levels of citric acid. These include fruits like pineapple, strawberries, cherries, and tomatoes. These fruits encourage healthy metabolism and help people avoid bloating and constipation.
Ques. Is citric acid a poison? (2 Marks)
Ans. Citrus fruits naturally contain citric acid, but synthetic versions are frequently used in foods, medications, vitamins, and cleaning goods. Citric acid is usually regarded as a safe component, although mold spores left over from the manufacturing process may cause allergies in certain people.
Ques. What are the applications of citric acid? (3 Marks)
Ans. Mentionable uses of Citric Acid are–
- The basic pH of soaps and detergents is softened using citric acid.
- Juices, soft drinks, and other liquids frequently include citrus acids or citric acid, giving them an acidic flavor.
- It functions as a food preservative because the food is protected against deterioration by its acidic pH, which inhibits the growth of several microorganisms.
- By chelating calcium in the blood, citric acid acts as an anticoagulant and as an acidulant to control pH levels.
- Citric acid is used as a blood preservative in the pharmaceutical industry.
Ques. Is it true that citric acid is a disinfectant? (2 Marks)
Ans. It is helpful for general disinfection and cleaning since it eliminates bacteria, mold, and mildew. It is possible to eliminate hard water stains, soap scum, calcium deposits, lime, rust, and rust. It is a common preservative used in cleaning goods.
Ques. What are the dangers of citric acid? (3 Marks)
Ans. The Food and Drug Administration (FDA) states that businesses use chemical procedures to produce citric acid. There is currently no proof that artificially swallowing citric acid—when ingested in big doses over time—is bad for one's health or physique. There have been several instances of allergic reactions and individuals becoming unwell as a result of the inclusion. Some individuals have complained of edema, stiffness, and joint pain in addition to gastrointestinal and muscular issues. Breathlessness was another issue for several people. Contrarily, there is no harm associated with consuming citric acid from fruits and vegetables like lemons and limes.
Ques. Which microorganism is helpful in the synthesis of citric acid? (2 Marks)
Ans. To generate citric acid, a fungus called Aspergillus niger can be employed. Additionally, it is employed in the production of the organic acid gluconic acid. Under particular circumstances, it causes black mold sickness and various plants and fruits.
Ques. Identify citric acid as basic or acidic. (1 Mark)
Ans. With the molecular formula C6H8O7, citric acid is a powerful organic molecule but a weak organic acid. Because it turns blue litmus paper red, the substance is acidic. The organic acid is weak.
Ques. What are the substitutes for citric acid? (1 Mark)
Ans. Lemon juice serves as a replacement for citric acid and is a common household item. Tartaric acid is another option.
Ques. Can the body absorb citric acid? (1 Mark)
Ans. After being consumed orally, citric acid is absorbed by the stomach. It also increases the bioavailability of minerals and aids in their effective absorption by the body.
Ques. What is the boiling and melting point of Citric Acid? (1 Mark)
Ans. Citric acid reaches its boiling point at 310°C.
153°C is the melting point.
For Latest Updates on Upcoming Board Exams, Click Here: https://t.me/class_10_12_board_updates
Check-Out:





Comments