Education Content Expert
Group 15 Elements consist of the p-block elements which include Nitrogen, Phosphorus, Arsenic, Antimony, and Bismuth. The p-block elements are situated on the right-hand side of the modern periodic table. Group 15 elements show similar patterns in their electronic configuration, especially in their outermost shell. The elements in this group have 2 electrons in their subshell, and all of their outermost shells consist of 5 electrons. The chemical properties of the elements are determined by the oxidation states exhibited by them.
Oxidation State refers to the number of electrons an atom utilizes or receives when it forms a compound. Oxidation State denotes the electrons lost by a substance when it is oxidized, and the electrons gained when a substance is reduced. Group 15 elements have 5 valence electrons and can have variable oxidation state: +3, +3, and +5.
Read More: NCERT Solutions For Class 12 Chemistry The p-Block Elements
Key Terms: Group 15 Elements, Oxidation State, Electrons, Valence Electrons, Nitrogen, Arsenic, Electronic Configuration, Electronegativity
Group 15 Elements
[Click Here for Sample Questions]
Group 15 elements comprise 5 elements namely Nitrogen, Phosphorus, Arsenic, Antimony, and Bismuth.
- There is a transition from non-metallic to metallic through a metalloid character in Group 15 elements.
- Nitrogen and Phosphorus are non-metals, arsenic and antimony are metalloids and bismuth is a typical metal.
- The valence shell electronic configuration of Group-15 elements is ns2np3.
- The s orbital of these elements is completely filled whereas the p orbital is half-filled which makes them stable in nature.
- The oxidation state exhibited by these elements determines their chemical properties.
What is Oxidation State?
[Click Here for Previous Years' Questions]
Oxidation State of an element denotes the electrons gained by a substance when it is reduced, and the electrons lost when a substance is oxidized. Oxidation occurs when there is an increase in the oxidation number, while reduction occurs when there is a decrease in the oxidation number.
- There is a negative (-ve) oxidation number when the atom gains electrons or shares them with another atom of less electronegativity.
- There is a positive (+ve) oxidation number when an atom loses electrons or shares with another atom of more electronegativity.
Read More: MCQs on P-Block Elements
Oxidation State of Group 15 Elements
[Click Here for Sample Questions]
Group 15 elements generally exhibits -3, +3 and +5 oxidation states. The number of electrons in the outermost shell of the Group 15 elements is 5. Thus, these elements need 3 more electrons to make it an octet configuration. They need to gain 3 more electrons or share 3 electrons with the help of the covalent bonds.
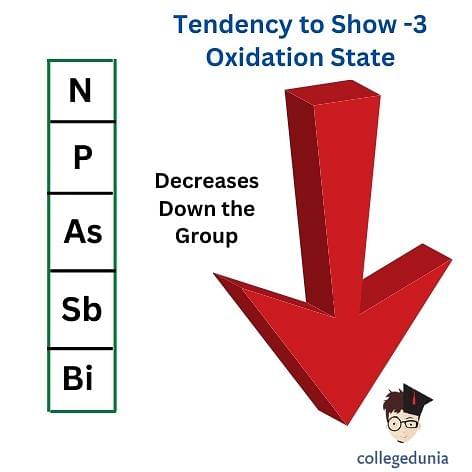
-3 Oxidation State
- The more common oxidation state of Group 15 elements is -3 oxidation which means gaining 3 more electrons.
- As one moves down the group, the tendency to produce the -3 oxidation decreases.
- It happens due to the increase in metallic character and the atomic size of the elements.
- -3 oxidation state is utilized by reacting Group 15 elements with hydrogen in order to produce ammonia, phosphane, arsane, stibane, and bismuthine.
- Going down the group, these hydrides become less stable and more toxic.
-3 Oxidation State of Group 15 Elements
Read More: Classification of Elements and Periodicity in Properties
+3 Oxidation State
- The +3 oxidation state occurs by sharing electrons.
- In +3 oxidation, the sharing is done through covalent bonds, in the case of- nitrogen, arsenic, and phosphorus. For instance, Nitrogen trichloride, phosphorus trichloride, and arsenic trichloride.
- It occurs through ionic bonds in the case of antimony and bismuth. For instance Antimony trifluoride and Bismuth trifluoride.
- The covalency character of the elements decreases as we move down the group.

+3 Oxidation State of Group 15 Elements
+5 Oxidation State
- The +5 oxidation state also occurs by sharing electrons.
- The +5 oxidation state for the element nitrogen forms the N2O5.
- The actual +5 oxidation occurs in antimony, phosphorus, and arsenic. Phosphorus even produces oxyhalides.

+5 Oxidation State of Group 15 Elements
Other Oxidation States for Group 15 Elements
The various other oxidation state exhibited by Group 15 elements are listed below:
- Nitrogen can form various oxides and exhibits +1, +2, and +4 oxidation states.
- Phosphorus exhibits +1 and +4 states in some oxo acids.
- The +3 state is stable with respect to disproportionation in the case of arsenic, antimony, and bismuth.
Periodic Trends in Group 15 Elements
[Click Here for Previous Years' Questions]
The periodic trends followed by all the Group 15 elements are as follows:
- Electronegativity: Electronegativity of an atom refers to the ability of that atom to attract electrons. It decreases down the group in the case of Group 15 elements.
- Ionization Energy: The ionization energy of an atom refers to the amount of energy it requires to remove an electron from an isolated atom or molecule. In Group 15 elements, it decreases down the group.
- Atomic Radii: The atomic radius increases in size down the group in Group 15 elements.
- Electron Affinity: Electron affinity refers to the ability of an atom to accept an electron. It decreases down the group in these elements.
- Melting Point: It refers to the amount of energy required to break bonds in order to change a substance to a liquid phase from a solid phase. The melting point increases down the group in Group 15 elements.
- Boiling Point: It is the amount of energy required to break bonds in order to change a substance to a vapour phase from the liquid phase. The boiling point increases down the group in Group 15 elements.
- Metallic Character: Metallic character refers to the level of reactivity of the metal. It increases down the group in case of Group 15 elements.
Handwritten Notes on p-Block Elements
[Click Here for Sample Questions]
Here are the handwritten notes for p-Block Elements (Group 15 elements):



































Things to Remember
- Group 15 elements consist of 5 elements namely Nitrogen, Phosphorus, Arsenic, Antimony, and Bismuth.
- Group 15 elements have 5 valence electrons (electrons in the outermost shell).
- The oxidation state of Group 15 elements are -3, +3, and +5.
- The tendency to show -3 oxidation state decreases as we down the group due to an increase in the size and metallic character.
- The stability of the +3 state increases down the group in Group 15 elements.
- The stability of the +5 state decreases down the group due to the inert pair effect.
Previous Years’ Questions (PYQs)
- Group 15 elements have more electron gain enthalpy… (VITEEE 2016)
- The basic character of hydrides of the 15 group elements… (NEET 1996)
- Which among the group-15 elements does not exists as…
- Group oxidation state of p-block elements is…
- The number of naturally occurring p-block elements that are… (KCET 2011)
- Oxidation number of…
- The highest oxidation state shown by Mn is… (JIPMER 2002)
- The oxidation state of Cr in CrO6 is… (NEET 2019)
- Oxidation state of oxygen in F2O is… (JIPMER 2009)
- More number of oxidation state are exhibited by the… (COMEDK 2015)
- The maximum oxidation state exhibited by actinide ions… (KEAM)
Sample Questions
Ques. What is the oxidation state of Group 15 elements? (3 Marks)
Ans. The oxidation states of Group 15 elements are -3, +3, and +5 oxidation states. As we move down the Group 15 of the periodic table, the tendency to exhibit a -3 oxidation state decreases due to an increase in the size of the atom and the metallic character. The stability of the +5 state also decreases on moving down the group. The stability of the +5 state decreases, and the +3 state increases as we move down the group due to the inert pair effect.
Ques. Explain the periodic trends in Group 15 elements. (5 Marks)
Ans. The periodic trends followed by all the Group 15 elements are:
- Electronegativity: The electronegativity of an atom refers to the ability of that atom to attract electrons. Electronegativity decreases down the group in Group 15 elements.
- Ionization Energy: It refers to the amount of energy an atom requires to remove an electron from an isolated atom/molecule. Ionization Energy decreases down the group in Group 15 elements.
- Atomic Radii: It increases in size down the group in Group 15 elements.
- Electron Affinity: It refers to the ability of an atom to accept an electron. It decreases down the group in Group 15 elements.
- Metallic Character: It exhibits the level of reactivity of the metal. It increases down the group in Group 15 elements.
Ques. Explain the formation of Oxides, Hydrides, and Halides by Group 15 elements. (3 Marks)
Ans. The formation of Oxides, Hydrides, and Halides by Group 15 elements is as follows:
- Group 15 elements form oxides such as Nitric Oxide, Bismuth Oxychloride, and others through the oxidation process under different oxidation states of p block elements.
- Group 15 elements are used to form hydrides which are compounds of hydrogen with any other element. For instance, NH3 (Ammonia), BiH3 (Bismuthine), PH3(Phosphine), AsH3 (Arsine), and SbH3 (Stibine) occur.
- Group 15 elements form Halides too which are compounds formed with halogen atoms and any element. They are usually trihalides and pentahalides such as Phosphorus trichloride (PCl3) and Phosphorous pentachloride (PCl5).
Ques. Which all elements are included in Group 15 of the modern periodic table? (2 Marks)
Ans. There are five elements in Group 15 of the modern periodic table:
- Nitrogen (N)
- Phosphorus (P)
- Arsenic (As)
- Antimony (Sb)
- Bismuth (Bi)
Ques. What is Oxidation State? (3 Marks)
Ans. Oxidation State, also referred to as the oxidation number, exhibits the number of electrons an atom uses or receives when it forms a compound. The electrons gained by a substance when it is reduced and the electrons lost when a substance is oxidized are shown by the oxidation number. The oxidation number is denoted by Roman numbers, with a plus or minus sign. The sum of the oxidation number in a compound is equal to its overall charge.
Ques. Explain the melting and boiling points of Group 15 elements. (3 Marks)
Ans. The melting point gradually increases as the atomic size increases as we move down the group. In Group 15 elements, Nitrogen has the least melting point and as we move to Arsenic and Phosphorus, the melting point starts to increase. A decrease is observed in the trend from antimony onwards. It happens because of the loose structure of atoms in the bonding. The boiling point of Group 15 elements regularly increases as we move down. The density of the elements also increases.
Ques. Elaborate on the electron configuration of Group 15 elements. (3 Marks)
Ans. The Group 15 elements have a similar electron configuration. All these elements have 2 electrons in their subshells and 5 electrons in their outer shells. This electron configuration leads to the patterning of trends in their reactivity. As we go down the group from Nitrogen to Bismuth, the metallic character of the elements increases, and the ionization enthalpy character of the elements decreases.
Ques. List the two most important elements of Group 15 elements. (3 Marks)
Ans. Nitrogen and Phosphorus are the two most important elements of Group 15 elements. Nitrogen occurs in a free state as a gas that is diatomic. It constitutes more than 70% of the volume of air. Phosphorus is present in our RNA and DNA in various forms. Phosphorus is also there in the bone marrow.
Ques. What is the oxidation state of Nitrogen? (3 Marks)
Ans. The oxidation states of Nitrogen range from +1 to +4 and tend to be disproportionate in acidic solutions. Nitrogen exhibits a maximum covalency of 4 since only four (one s and three p) are available for bonding. The heavier elements have a vacant d orbital in the valence shell which is then used for bonding.
Ques. What is a group in the periodic table? (1 Mark)
Ans. Groups of elements refer to different columns of elements in the modern periodic table. There are in total 18 groups from left to right in the periodic table.
Check-Out:





Comments