
Education Journalist | Study Abroad Strategy Lead
Saponification is derived from the Latin word 'Sapo' which actually means soap. It is the process of making soap. Soaps are composed of a long chain of fatty acids, potassium salts and sodium. This is a process in which an ester reacts with water and the base. For example, Sodium hydroxide or Potassium hydroxide reacts with the ester to produce the alcohol and Sodium or Potassium for acid. Basically, soaps are used to clean the body in order to maintain the hygiene of the person. The dirt which is stuck to the surface of the skin can be cleaned after the use of soap. Soaps are used for many purposes like bathing, washing utensils, clothes and other household works.
| Table of Content |
Key Terms: Saponification, Soaps, Chemical Reactions, Ester, Sodium Hydroxide, Fatty Acids, Alcohol, Potassium Hydroxide, Glycerol
What is Saponification?
[Click Here for Sample Questions]
The process of manufacturing soaps is called Saponification. It is a chemical process which undergoes various steps to form soaps. Soaps are defined as the long chain of fatty acids made from Sodium and Potassium salts.
When this process occurs, the ester reacts with the water and the base to form alcohol and soaps. To produce glycerol and soaps, the triglycerides are reacted with Sodium and Potassium hydroxide which is also known as 'lye'. Triglycerides are produced from the vegetable oils and fats of animals.

Saponification
There are two types of soaps. The hard and soft soaps are used for other purposes. The hard soap is formed when sodium hydroxide is used in the process of saponification. Whereas the use of Potassium hydroxide results in the formation of soft soap.
Read More: Cleaning Capacity of Soap in Soft & Hard Water
Reactions of Saponification
[Click Here for Sample Questions]
Saponification is the process in which the ester group reacts with water and bases like NaOH and KoH to form alcohol and carboxylate ions. Thus the carboxylate ions are converted to carboxylic acid. Glycerol is an alcohol which is used in the process of saponification. The mechanism of the saponification reaction involves three steps. They are given below.
In the very first step of saponification reaction, the product is formed which is named orthoester. This is because nucleophile hydroxide ion attacks the ester group.
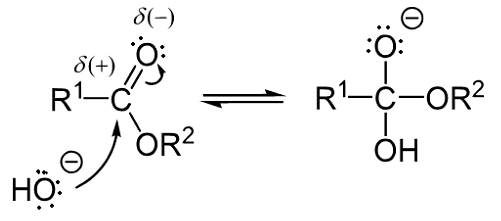
Orthoester Formation
Then comes the second step, the orthoester is broken into a carboxylic acid and the alkoxide group. Hence the alkoxide group is removed.

Expulsion of Carboxylic Acid and Alkoxide
The third and last step involves the deprotonation of carboxylic acid and alkoxide groups. Therefore, alcohol and carboxylate ions are produced or formed.

Creation of Alcohol
Effects of Saponification
[Click Here for Sample Questions]
The Saponification process shows desirable and undesirable effects. They are given below.
- This process is mainly used in fire extinguishers to convert fats and oil into non-combustible soaps. Further, it will lead to extinguishing the fire. It generally decreases the temperature of the flames because it grabs heat from the surrounding.
- It can damage the oil paintings because bulk metals used in the pigments react with the oil. Which are used in pigments that react with oil as it contains fatty acid. Because of this, the oil paintings get damaged.
- Soaps are the part of our life which are used daily to clean the surface of an object or the skin of the human body. There are different types of soaps like sodium soaps which are used for clothes, potassium soaps for cleaning purposes and lithium soaps are used to lubricate the grease.
Read More: Alcohols Phenols and Ethers
Uses of Saponification
[Click Here for Sample Questions]
Some major uses of saponification are as follows:
- Saponification is a process used to extinguish cooking oil and fats.
- Through the Saponification process, we can produce a variety of soaps. For that, different alkalis are used.
- Potassium hydroxide results in the formation of soft soaps.
- Sodium hydroxide leads to the formation of hard soaps.
Things to Remember
- Soaps are an important part of our lives. It is widely used for cleaning purposes. We use different types of soaps.
- Soaps are produced by the process of Saponification.
- It is widely applicable for cleaning the clothes, utensils and bathing and fire extinguishers.
- Saponification is a chemical process which is used to manufacture different types of soaps.
- In this process, the ester gets hydrolysed with Sodium hydroxide or Potassium hydroxide in order to form alcohol and Sodium or Potassium salt.
Read More: Properties of Bases
Sample Questions
Ques. What is the saponification process? (3 Marks)
Ans. Saponification can be characterized as a "hydration reaction where free hydroxide breaks the ester bonds between the unsaturated fats and glycerol of a fatty oil, bringing about free unsaturated fats and glycerol," which are each solvent in aqueous solutions.
Ques. How is soap made from fat? (3 Marks)
Ans. Soap is produced using a chemical reaction between fat and a soluble base ingredient. Fat soaps are made by blending animal fat with sodium hydroxide, all the more generally known as lye. Lye is exceptionally corrosive, yet when it blends in with fat it goes through a response called saponification.
Ques. What is the importance of saponification? (3 Marks)
Ans. Saponification is important to the industrial user for it assists with knowing how much free unsaturated fat is available in a food material. The amount of free unsaturated fat can be recognized by deciding the amount of soluble base that should be added to the fat or oil to make it neutral.
Ques. Where is saponification used? (3 Marks)
Ans. The main application of saponification is in the production of soaps. There are various types of soaps filling various needs like clothing, cleaning, and oil. Soaps might be hastened by salting them out with soaked sodium chloride. Saponification additionally works with fire extinguishers.
Ques. Saponification is important. Why? (3 Marks)
Ans. Saponification is very important for commercial uses. It is beneficial for industrial users to learn about the knowledge of fatty acids which are present in food items. The amount of free fatty acid can be differentiated by analyzing the amount of alkali which is to be added to the fat or oil in order to make it neutral.
Ques. What are the things required for this process? (3 Marks)
Ans. The important things required for the process are given below:
Vegetable oil, Common salt, NaOH solution, beaker, the cylinder that can be measurable, Litmus papers, glass rod, burner, wire gauze, funnel for filtering, spatula and knife are needed to perform this process of Saponification.
Ques. What is the unit of saponification value? (3 Marks)
Ans. The number of milligrams of KOH needed to neutralize the fatty acids is obtained by complete hydrolysis of 1gram of an oil sample. It depends on the molecular weight along with the percentage concentration of the components of fatty acid present in the oil.
Also Read:
Also Read:





Comments