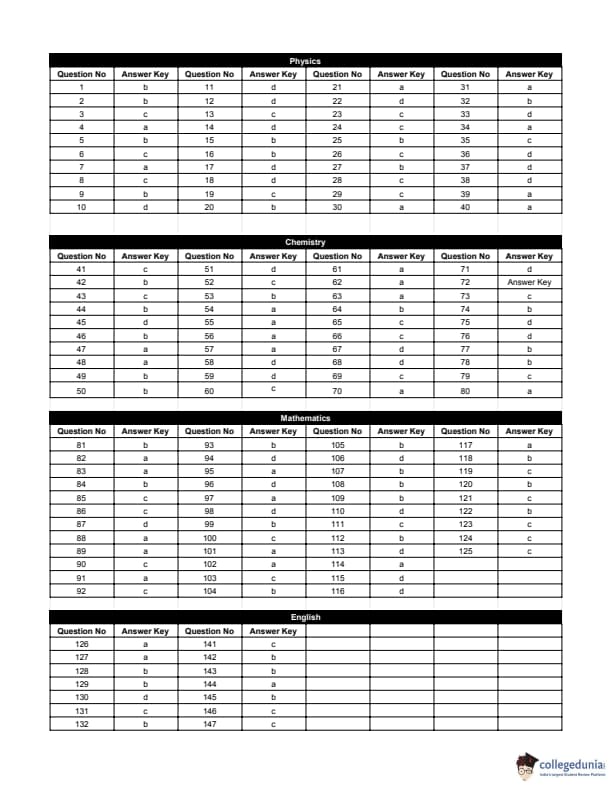
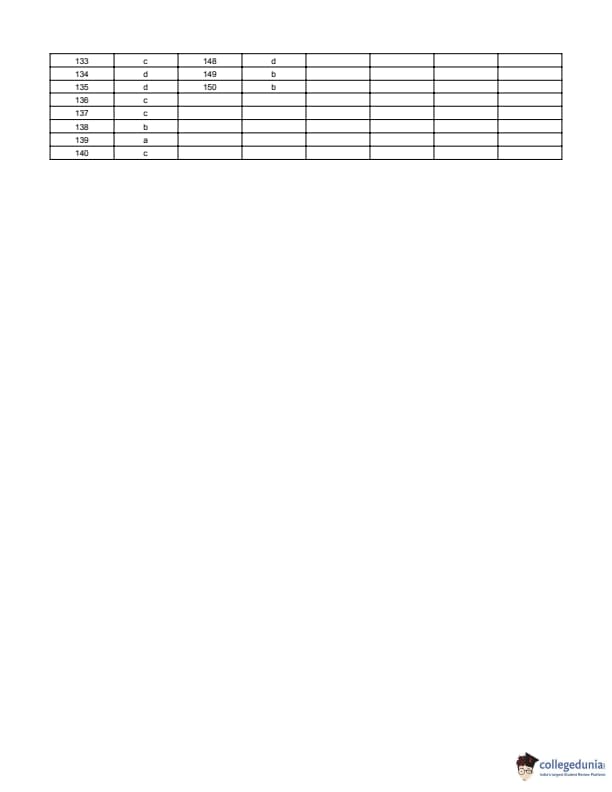
BITSAT 2014 Question Paper with Answer Key pdf is available for download. BITSAT 2014 was conducted in online CBT mode by BITS Pilani. BITSAT 2014 Question Paper had 150 questions to be attempted in 3 hours.
BITSAT 2014 Question Paper with Answer Key PDF
| BITSAT 2014 Question Paper PDF | BITSAT 2014 Answer Key PDF |
|---|---|
| Download PDF | Download PDF |
A rifle man, who together with his rifle has a mass of 100 kg, stands on a smooth surface and fires 10 shots horizontally. Each bullet has a mass 10 g and a muzzle velocity of 800 m s\(^{-1}\). The velocity which the rifle man attains after firing 10 shots is:
A train accelerating uniformly from rest attains a maximum speed of \(40 m s^{-1}\) in 20 s. It travels at this speed for 20 s and is brought to rest with uniform retardation in further 40 s. What is the average velocity during the period?
A projectile is fired with a velocity \(u\) making an angle \(\theta\) with the horizontal. What is the magnitude of change in velocity when it is at the highest point?
For the equation of force \(F = Aa^b d^c\), where \(F\) is the force, \(A\) is the area, \(v\) is the velocity and \(d\) is the density, the values of \(a, b\) and \(c\) are respectively:
A person with his hand in his pocket is skating on ice at the rate of \(10 m s^{-1}\) and describes a circle of radius \(50 m\). What is his inclination to vertical? \((g = 10 m s^{-2})\)
A small block of mass \(m\) is kept on a rough inclined surface of inclination \(\theta\) fixed in an elevator. The elevator goes up with a uniform velocity \(v\) and the block does not slide on the wedge. The work done by the force of friction on the block in time \(t\) will be:
An equilateral prism of mass \(m\) rests on a rough horizontal surface with coefficient of friction \(\mu\). A horizontal force \(F\) is applied on the prism as shown. If the coefficient of friction is sufficiently high so that the prism does not slide before toppling, then the minimum force required to topple the prism is:

A spherically symmetric gravitational system of particles has a mass density \[ \rho = \begin{cases} \rho_0 & for r \le R
0 & for r > R \end{cases} \]
where ρ0 is a constant. A test mass can undergo circular motion under the influence of
the gravitational field of particles. Its speed v as a function of distance r (0 < r < ∞)
from the centre of the system is represented by:

The load versus elongation graph for four wires is shown. The thinnest wire is:

The work done in blowing a soap bubble of surface tension \(0.06 N m^{-1}\) from radius \(2 cm\) to \(5 cm\) is:
The wavelength of radiation emitted by a body depends upon:
One mole of \(O_2\) gas having a volume equal to \(22.4 litres\) at \(0^\circ C\) and 1 atm is compressed isothermally so that its volume reduces to \(11.2 litres\). The work done in this process is:
In a thermodynamic process, the pressure of a fixed mass of a gas is changed in such a manner that the gas releases 20 J of heat and 8 J of work is done on the gas. If the initial internal energy of the gas is 30 J, the final internal energy will be:
In the kinetic theory of gases, which of these statements is/are true?
(i) The pressure of a gas is proportional to the mean speed of the molecules.
(ii) The root mean square speed of the molecules is proportional to the pressure.
(iii) The rate of diffusion is proportional to the mean speed of molecules.
(iv) The mean translational kinetic energy of a gas is proportional to its kelvin temperature.
Two balloons are filled with pure gases and other with air respectively. If the pressure and temperature of these balloons are same, then the number of molecules per unit volume is:
Two particles P and Q describe S.H.M. of same amplitude and same frequency along the same straight line. The maximum distance between the two particles is \(2A\). The initial phase difference between the particles is:
A tunnel is dug through the centre of the earth and a ball is released in it. It executes S.H.M. with time period:
A sound source, emitting sound of constant frequency, moves with a constant speed and crosses a stationary observer. The frequency \(n\) of sound heard by the observer is plotted against time \(t\). Which of the following graphs represents the correct variation?

When a string is divided into three segments of length \(l_1, l_2,\) and \(l_3\), the fundamental frequencies of these three segments are \(v_1, v_2,\) and \(v_3\) respectively. The original fundamental frequency of the string is:
Two point dipoles \(p\hat{k}\) and \(\frac{p}{2}\hat{k}\) are located at \((0,0,0)\) and \((1\,m,0,2\,m)\) respectively. The resultant electric field due to the two dipoles at the point \((1\,m,0,0)\) is:
Electric field in the region is given by \( \vec{E} = \left(\dfrac{M}{x^3}\right)\hat{i} \). The correct expression for the potential in the region is [assume potential at infinity is zero]:
Three capacitors \(C_1 = 1\,\muF,\; C_2 = 2\,\muF\) and \(C_3 = 3\,\muF\) are connected as shown in the figure. The equivalent capacitance between points A and B is:

Two coaxial non-conducting cylinders of radius \(a\) and \(b\) are separated by a material of conductivity \(\sigma\) and a constant potential difference \(V\) is maintained between them by a battery. The current per unit length flowing from one cylinder to the other is:
A wire X is half the diameter and half the length of a wire Y of similar material. The ratio of resistance of X to that of Y is:
A narrow beam of protons and deuterons, each having the same momentum, enters a region of uniform magnetic field directed perpendicular to their direction of momentum. The ratio of the radii of the circular paths described by them is:
For the circuit shown, the current is to be measured. The ammeter shown is a galvanometer with resistance \(R_g = 60.00\,\Omega\) converted into an ammeter by a shunt resistance \(R_s = 0.02\,\Omega\). The value of the current is:

The susceptibility of a magnetism at 300 K is \(1.2 \times 10^{-5}\). The temperature at which the susceptibility increases to \(1.8 \times 10^{-5}\) is:
A coil of 10 turns and resistance \(20\,\Omega\) is connected in series with a battery of resistance \(30\,\Omega\). The coil is placed with its plane perpendicular to a uniform magnetic field of induction \(10^{-2}\,T\). If it is now turned through an angle of \(60^\circ\) about an axis in its plane, the charge induced in the coil is (Area of coil \(=10^{-2}\,m^2\)):
Voltage and current in an AC circuit are given by \(V = 50\sin(50t)\,V\) and \(i = 50\sin(50t + \frac{\pi}{3})\,mA\).
The power dissipated in the circuit is:
Resolving power of a telescope will be more, if the diameter of the objective is:
The magnifying power of a telescope is 9. When it is adjusted for parallel rays, the distance between the objective and the eye piece is found to be 20 cm. The focal length of lenses are:
The angular size of the central maxima due to a single slit diffraction (a = slit width) is:
Find the final intensity of light (\(I''\)), if the angle between the axes of two polaroids is \(60^\circ\).

The threshold wavelength of tungsten is \(2300\,\AA\). If ultraviolet light of wavelength \(1800\,\AA\) is incident on it, then the maximum kinetic energy of photoelectrons would be about:
Graph between stopping potential of most energetic emitted photoelectrons (\(V_s\)) and frequency (\(\nu\)) of incident radiation on metal is given. Value of slope AB/BC in graph is [where \(h\) = Planck’s constant, \(e\) = electronic charge]:

If hydrogen atom, an electron jumps from bigger orbit to smaller orbit so that radius of smaller orbit is one-fourth of radius of bigger orbit. If speed of electron in bigger orbit was \(v\), then speed in smaller orbit is:
A nucleus of uranium decays at rest into nuclei of thorium and helium. Then:
Let binding energy per nucleon of nucleus be denoted by \(E_{bn}\) and radius of nucleus be denoted as \(r\). If mass number of nuclei A and B are 64 and 125 respectively then:
For a CE transistor amplifier, the audio signal voltage across the collector resistance of \(2.0\,k\Omega\) is \(2.0\,V\). Suppose the current amplification factor of the transistor is 100. What should be the value of \(R_B\) in series with \(V_{BB}\) supply of \(2.0\,V\) if the base current has to be 10 times the signal current?
The combination of gates shown below yields:

The formation of CO and CO\(_2\) illustrates the law of:
The wave number of the limiting line in Lyman series of hydrogen is \(109678\,cm^{-1}\). The wave number of the limiting line in Balmer series of He\(^+\) would be:
The valency shell of element A contains 3 electrons while the valency shell of element B contains 6 electrons. If A combines with B, the probable formula of the compound formed will be:
The enthalpy of sublimation of aluminium is \(330\,kJ mol^{-1}\). Its I\(^st\), II\(^nd\) and III\(^rd\) ionization enthalpies are 580, 1820 and \(2740\,kJ mol^{-1}\) respectively. How much heat must be supplied in kJ to convert 13.5 g of aluminium into Al\(^{3+}\) ions and electrons at 298 K?
Which one of the following pairs is isostructural (i.e., having the same shape and hybridization)?
N\(_2\) and O\(_2\) are converted into mono anions N\(_2^-\) and O\(_2^-\) respectively. Which of the following statements is wrong?
If the enthalpy of vaporization of water is \(18.6\,kJ mol^{-1}\), the entropy of its vaporization will be:
The heats of neutralisation of CH\(_3\)COOH, HCOOH, HCN and H\(_2\)S are \(-13.2, -13.4, -2.9\) and \(-3.8\) kcal per equivalent respectively. Arrange the acids in increasing order of acidic strength.
\(K_c\) for the reaction \([Ag(CN)_2]^-\rightleftharpoons Ag^+ + 2CN^-\) is \(4.0\times10^{-19}\) at \(25^\circC\). The equilibrium concentration of silver cation in a solution which was originally \(0.1\) molar in KCN and \(0.03\) molar in AgNO\(_3\) is:
The ratio of oxidation states of Cl in potassium chloride to that in potassium chlorate is:
Which of the following among alkali metal is most reactive?
Which of the following compounds has wrong IUPAC name?
The compound which gives the most stable carbocation on dehydration is:
The correct order of increasing C–O bond length in CO, CO\(_3^{2-}\), CO\(_2\) is:
An organic compound A (C\(_4\)H\(_9\)Cl) on reaction with Na/diethyl ether gives a hydrocarbon which on monochlorination gives only one chloro derivative. Then A is:
When rain is accompanied by a thunderstorm, the collected rain water will have a pH value:
An elemental crystal has a density of \(8570\,kg m^{-3}\). The packing efficiency is 0.68. The closest distance of approach between neighbouring atoms is \(2.86\,\AA\). What is the mass of one atom approximately?
Identify the correct order of solubility of Na\(_2\)S, CuS and ZnS in aqueous medium:
In the cell reaction \(Cu(s) + 2Ag^+(aq) \rightarrow Cu^{2+}(aq) + 2Ag(s)\), \(E^\circ_{cell} = 0.46\,V\). By doubling the concentration of \(Cu^{2+}\), \(E_{cell}\) is:
Cu\(^+\) is unstable in solution and undergoes simultaneous oxidation and reduction according to the reaction \(2Cu^+(aq) \rightarrow Cu^{2+}(aq) + Cu(s)\).
Choose the correct \(E^\circ\) for the above reaction if \(E^\circ_{Cu^{2+}/Cu} = +0.34\,V\) and \(E^\circ_{Cu^+/Cu} = +0.15\,V\).
The reduction of peroxodisulphate ion by I\(^{-}\) ion is expressed by \(S_2O_8^{2-} + 3I^- \rightarrow 2SO_4^{2-} + I_3^-\).
If rate of disappearance of I\(^{-}\) is \(9/2 \times 10^{-3}\,mol L^{-1}s^{-1}\), what is the rate of formation of \(2SO_4^{2-}\) during same time?
A gaseous reaction \(X_2(g) \rightarrow Y + \frac{1}{2}Z(g)\).
There is an increase in pressure from 100 mm to 120 mm in 5 minutes. The rate of disappearance of \(X_2\) is:
Two substances R and S decompose in solution independently, both following first order kinetics. The rate constant of R is twice that of S. Initially 0.5 millimoles of R and 0.25 of S. The molarities of R and S will be equal at the end of time equal to:
The isoelectric point of a colloidally dispersed material is the pH value at which:
Which of the following halogens exhibit only one oxidation state in its compounds?
Starch can be used as an indicator for the detection of traces of:
Which one of the following arrangements represents the correct order of electron gain enthalpy (with negative sign) of the given atomic species?
Which forms coloured salts:
The correct order of magnetic moments (spin only values in B.M.) is:
(Atomic nos.: Mn = 25, Fe = 26, Co = 27)
The number of double bonds in gammaxene is:
\[ Ph–CH(OH)–CHO \xrightarrow[H_2O]{HO^-} Q \]
P and Q are isomers. Identify Q.


Consider the following phenols as shown in the figure. The decreasing order of acidity of the above phenols is:

The ionization constant of phenol is higher than that of ethanol because:
The reaction \[ \mathrm{CH_3-CH=CH_2 \xrightarrow[H^+]{CO+H_2O} CH_3-CH(COOH)-CH_3} \]
is known as:
Aniline reacts with phosgene and KOH to form:

Which one of the following monomers gives the polymer neoprene on polymerization?
Which of the following can possibly be used as analgesic without causing addiction and modification?
Which among the following is not an antibiotic?
Which of the following ions can be separated by aq. NH\(_4\)OH in presence of NH\(_4\)Cl?
3.92 g of ferrous ammonium sulphate reacts completely with 50 ml of \(\frac{N}{10}\) KMnO\(_4\) solution. The percentage purity of the sample is:
The set \((A\setminus B)\cup(B\setminus A)\) is equal to:
The domain of the function \[ f(x)=\log_2\!\left(-\log_{\sqrt{2}}\!\left(1+\frac{1}{x^4}\right)-1\right) \]
is:
\(\cos^2\!\left(\frac{\pi}{6}+\theta\right)-\sin^2\!\left(\frac{\pi}{6}-\theta\right)=\)
The solution of \(\cos(2x)-1(3+2\cos x)=0\) in the interval \(0\le x\le2\pi\) is:
\(2^{3n}-7n-1\) is divisible by:
The greatest positive integer which divides \(n(n+1)(n+2)(n+3)\) for all \(n\in\mathbb{N}\), is:
If \(z=x+iy,\; z^{1/3}=a-ib\), then \(\dfrac{x}{a}-\dfrac{y}{b}=k(a^2-b^2)\), where \(k\) is equal to:
\(i^{57}+\dfrac{1}{i^{25}}\), when simplified has the value:
The complex number \(z=z+iy\) which satisfies the equation \[ \left|\frac{z-3i}{z+3i}\right|=1 \]
lies on:
The number of all three element subsets of the set \(\{a_1,a_2,a_3,\ldots,a_n\}\) which contain \(a_3\) is:
In how many ways can a committee of 5 be formed out of 6 men and 4 women containing at least one woman?
The coefficient of \(x^4\) in the expansion of \((1+x+x^2+x^3)^{11}\) is:
If \(T_0,T_1,T_2,\ldots,T_n\) represent the terms in the expansion of \((x+a)^n\), then \((T_0-T_2+T_4-\cdots)^2+(T_1-T_3+T_5-\cdots)^2=\)
If the \((2p)^{th}\) term of a H.P. is \(q\) and the \((2q)^{th}\) term is \(p\), then the \(2(p+q)^{th}\) term is:
If \(\frac{1}{a}, \frac{1}{b}, \frac{1}{c}\) are in A.P., then \[ \left(\frac{1}{a}+\frac{1}{b}+\frac{1}{c}\right)\left(\frac{1}{b}+\frac{1}{c}-\frac{1}{a}\right) \]
is equal to:
The product of \(n\) positive numbers is unity, then their sum is:
If \(P_1\) and \(P_2\) be the lengths of perpendiculars from the origin upon the straight lines \(x\sec\theta+y\cosec\theta=a\) and \(x\cos\theta-y\sin\theta=a\cos2\theta\) respectively, then the value of \(4P_1^2+P_2^2\) is:
The angle of intersection of the two circles \(x^2+y^2-2x-2y=0\) and \(x^2+y^2=4\) is:
An arch of a bridge is semi-elliptical with major axis horizontal. If the length of the base is \(9\) m and the highest part of the bridge is \(3\) m from the centre of the horizontal axis, the best approximation of the height of the arch \(2\) m from the centre of the base is:
\(\displaystyle \lim_{x\to0}\left(\csc x\right)^{1/\log x}\) is equal to:
If M.D. is 12, the value of S.D. will be:
A bag contains 5 brown and 4 white socks. A man pulls out 2 socks. Find the probability that they are of the same colour.
Let \(R=\{(3,3),(6,6),(9,9),(12,12),(6,12),(3,9),(3,12),(3,6)\}\) be a relation on the set \(A=\{3,6,9,12\}\). Then, the relation is:
Let \(f:\mathbb{R}\to\mathbb{R}\) be a function defined by \(f(x)=\dfrac{x-m}{x-n}\), where \(m\neq n\). Then
Find the value of \(\tan^{-1}\!\left(\dfrac{1-\frac{\pi}{5}}{2}\right)\).
If \(\begin{bmatrix}\alpha & \beta
\gamma & -\alpha\end{bmatrix}\) is a square root of identity matrix of order 2, then
The value of \(\lambda\), for which the lines \(3x-4y=13\), \(8x-11y=33\) and \(2x-3y+\lambda=0\) are concurrent is
Let \[ f(x)= \begin{cases} (x-1)\sin\!\left(\dfrac{1}{x-1}\right), & x\neq1
0, & x=1 \end{cases} \]
Then which one of the following is true?
The interval in which the function \(2x^3+15\) increases less rapidly than the function \(9x^2-12x\), is
The fuel charges for running a train are proportional to the square of the speed generated in miles per hour and cost
(48 per hour at 16 miles per hour. The most economical speed if the fixed charges (i.e., salaries etc.) amount to
)30 per hour is:
Evaluate: \[ \int \frac{1}{1+3\sin^2 x+8\cos^2 x}\,dx \]
\[ \int_{0}^{10}\frac{x^{10}}{(10-x)^{10}+x^{10}}\,dx \]
is equal to
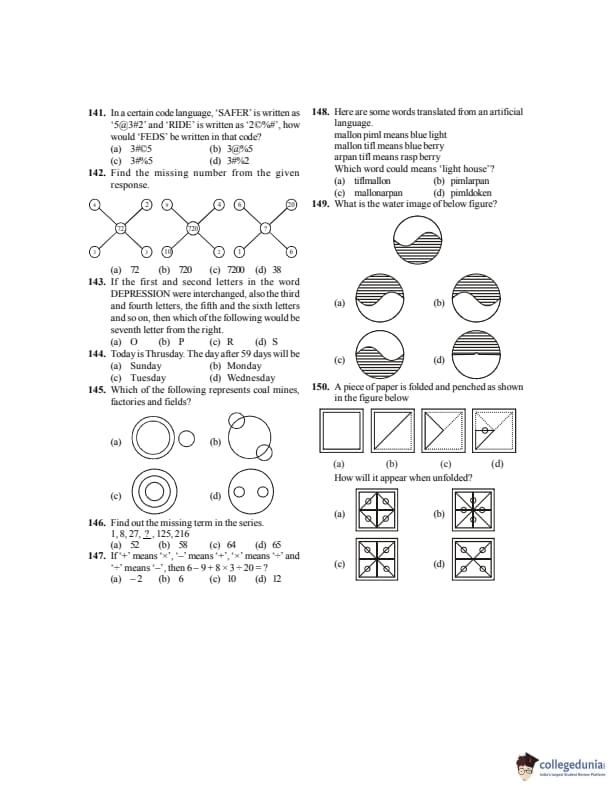
The area bounded by the x-axis, the curve \(y=f(x)\) and the lines \(x=1,\;x=b\) is equal to \(\sqrt{b^2+1}-\sqrt{2}\) for all \(b>1\). Then \(f(x)\) is
Solution of differential equation \[ x^2-1+\left(\frac{x}{y}\right)^{-1}\frac{dy}{dx} +\frac{x^2}{2!}\left(\frac{dy}{dx}\right)^2 +\frac{x^3}{3!}\left(\frac{dy}{dx}\right)^3+\cdots=0 \]
is
If the middle points of sides BC, CA and AB of triangle ABC are respectively D, E, F. If the position vectors of A, B, C are \(\hat{i}+\hat{j},\;\hat{j}+\hat{k},\;\hat{k}+\hat{i}\) respectively, then the position vector of the centre of triangle DEF is
The angle between any two diagonals of a cube is
Find the angle between the line \[ \frac{x+1}{2}=\frac{y}{3}=\frac{z-3}{6} \]
and the plane \(10x+2y-11z=3\).
The equation of the right bisector plane of the segment joining \((2,3,4)\) and \((6,7,8)\) is
A bag contains \(n+1\) coins. It is known that one of these coins shows heads on both sides, whereas the other coins are fair. One coin is selected at random and tossed. If the probability that toss results in heads is \(\frac{7}{12}\), then the value of \(n\) is
A coin is tossed 7 times. Each time a man calls head. Find the probability that he wins the toss on more occasions.
Consider \(\dfrac{x}{2}+\dfrac{y}{4}\ge1\) and \(\dfrac{x}{3}+\dfrac{y}{2}\le1,\; x,y\ge0\). Then number of possible solutions are
If \(A=\begin{bmatrix}1&1
1&1\end{bmatrix}\), then \(A^{100}\) is
If \[ \begin{vmatrix} p & q-r & r-z
p-x & q & r-z
p-x & q-y & r \end{vmatrix}=0, \]
then the value of \(\dfrac{p}{x}+\dfrac{q}{y}+\dfrac{r}{z}\) is
Through the vertex \(O\) of parabola \(y^2=4x\), chords OP and OQ are drawn at right angles to one another. The locus of the midpoint of PQ is
Let \[ f(x)= \begin{cases} \dfrac{1-\sin^3 x}{3\cos^2 x}, & x<\dfrac{\pi}{2}
[6pt] p, & x=\dfrac{\pi}{2}
[6pt] \dfrac{q(1-\sin x)}{(\pi-2x)^2}, & x>\dfrac{\pi}{2} \end{cases} \]
If \(f(x)\) is continuous at \(x=\dfrac{\pi}{2}\), then \((p,q)=\)
AUGMENT
CONSOLATION
AUXILIARY
AUSPICIOUS
RECOMPENSE
IMPEDE
They requested me to follow them.
She did not believed me.
I am fine, what about you?
They were afraid ____ the lion, so they dropped the idea of hunting in jungle.
Our company signed a profitable ____ last month.
What is your ____ for tonight?
Arrange the following sentences in the correct order:
I. Today we live in modern technology era.
P. We want to get everything in one day.
Q. We have a lot of problems now.
R. Ancient time was quite pleasant.
S. We had no problems then.
6. Perhaps greed is the main cause for this.
Arrange the following sentences in the correct order:
I. He is a common man.
P. Yesterday our city saw a brutal crime.
Q. Police is trying to arrest innocent persons.
R. The criminals are well known.
S. Police as well as whole system is corrupt.
6. Police will arrest him as he is an easy target because of being a common man.
Arrange the following sentences in the correct order:
I. I want to change the room.
P. Last month I got a job.
Q. I had been living there for six months.
R. The office is far from the room.
S. I want to cut expenses of travelling.
6. Hopefully I will do this next week.
In a certain code language, ‘SAFER’ is written as ‘5@3#2’ and ‘RIDE’ is written as ‘2C%#’, how would ‘FEEDS’ be written in that code?
Find the missing number from the given response.

If the first and second letters of the word DEPRESSION were interchanged, also the third and fourth letters, the fifth and the sixth letters and so on, then which of the following would be the seventh letter from the right?
Today is Thursday. The day after 59 days will be
Which of the following represents coal mines, factories and fields?

Find out the missing term in the series:
1, 8, 27, ?, 125, 216
If ‘+’ means ‘×’, ‘−’ means ‘+’, ‘×’ means ‘÷’ and ‘÷’ means ‘−’, then \(6 − 9 + 8 × 3 ÷ 20 = ?\)
Here are some words translated from an artificial language.
mallon pimm means blue light
mallon tiff means blue berry
arpan tiff means rasp berry
Which word could mean ‘light house’?
What is the water image of the below figure?

A piece of paper is folded and pinched as shown. How will it appear when unfolded?



































Comments