BITSAT 2016 Question Paper with Answer Key pdf is available for download. BITSAT 2016 was conducted in online CBT mode by BITS Pilani. BITSAT 2016 Question Paper had 150 questions to be attempted in 3 hours.
BITSAT 2016 Question Paper with Answer Key PDF
| BITSAT 2016 Question Paper PDF | BITSAT 2016 Answer Key PDF |
|---|---|
| Download PDF | Download PDF |
What should be the velocity of rotation of earth due to rotation about its own axis so that the weight of a person becomes \( \frac{3}{5} \) of the present weight at the equator. Equatorial radius of the earth is \(6400\,km\).
Block A of mass \(m\) and block B of mass \(2m\) are placed on a fixed triangular wedge by means of a massless, inextensible string and a frictionless pulley as shown in the figure. The wedge is inclined at \(45^\circ\) to the horizontal on both the sides. If the coefficient of friction between block A and the wedge is \( \frac{2}{3} \) and that between block B and the wedge is \( \frac{1}{3} \), and both the blocks A and B are released from rest, the acceleration of A will be:

The surface charge density of a thin charged disc of radius \(R\) is \(\sigma\). The value of the electric field at the centre of the disc is \(\frac{\sigma}{2\varepsilon_0}\). With respect to the field at the centre, the electric field along the axis at a distance \(R\) from the centre of the disc:
The molecules of a given mass of a gas have r.m.s. velocity of \(200\,m s^{-1}\) at \(27^\circC\) and \(1.0 \times 10^5\,N m^{-2}\) pressure. When the temperature and pressure of the gas are respectively \(127^\circC\) and \(0.05 \times 10^5\,N m^{-2}\), the r.m.s. velocity of its molecules is:
An inductor of inductance \(L = 400\,mH\) and resistors of resistance \(R_1 = 2\Omega\) and \(R_2 = 2\Omega\) are connected to a battery of emf \(12\,V\) as shown in the figure. The internal resistance of the battery is negligible. The switch S is closed at \(t = 0\). The potential drop across \(L\) as a function of time is:

Two wires are made of the same material and have the same volume. However wire 1 has cross-sectional area \(A\) and wire 2 has cross-sectional area \(3A\). If the length of wire 1 increases by \(\Delta x\) on applying force \(F\), how much force is needed to stretch wire 2 by the same amount?
Two spheres of different materials, one with double the radius and one-fourth the thickness of the other, are filled with ice. If the time taken for complete melting of the larger sphere is 25 minutes and for the smaller one is 16 minutes, the ratio of thermal conductivities of the materials of the larger sphere to that of the smaller sphere is:
A biconvex lens has a radius of curvature of magnitude \(20\,cm\). Which of the following options best describe the image formed of an object of height \(2\,cm\) placed \(30\,cm\) from the lens?
In the figure below, what is the potential difference between the points A and B and between B and C respectively in steady state?

A radioactive element X converts into another stable element Y. Half-life of X is 2 hours. Initially only X is present. After time \(t\), the ratio of atoms of X and Y is found to be \(1:4\). Then \(t\), in hours is:
The approximate depth of an ocean is \(2700\,m\). The compressibility of water is \(45.4 \times 10^{-11}\,Pa^{-1}\) and density of water is \(10^3\,kg m^{-3}\). What fractional compression of water will be obtained at the bottom of the ocean?
A frictionless wire AB is fixed on a sphere of radius \(R\). A very small spherical ball slips on this wire. The time taken by this ball to slip from A to B is:

A string of length \(\ell\) is fixed at both ends. It is vibrating in its 3rd overtone with maximum amplitude \(a\). The amplitude at a distance \(\ell/6\) from one end is:
A deuteron of kinetic energy \(50\,keV\) is describing a circular orbit of radius \(0.5\,m\) in a plane perpendicular to the magnetic field \(B\). The kinetic energy of the proton that describes a circular orbit of radius \(0.5\,m\) in the same plane with the same \(B\) is:
In the circuit shown in the figure, find the current in the \(45\Omega\) resistor.

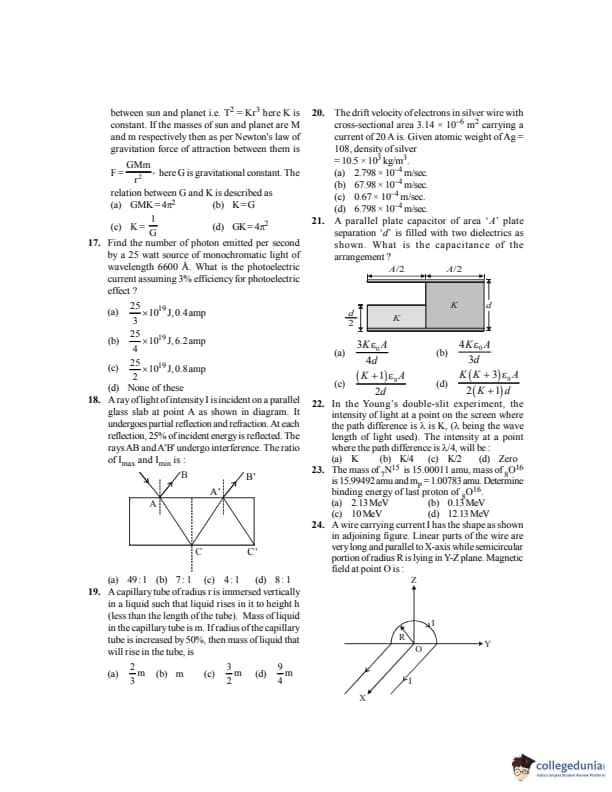
Kepler's third law states that the square of the period of revolution (\(T\)) of a planet around the sun is proportional to the third power of average distance \(r\) between sun and planet i.e. \(T^2 = Kr^3\), where \(K\) is a constant. If the masses of sun and planet are \(M\) and \(m\) respectively and as per Newton's law of gravitation the force of attraction between them is \(F=\dfrac{GMm}{r^2}\), where \(G\) is the gravitational constant. The relation between \(G\) and \(K\) is described as:
Find the number of photons emitted per second by a \(25\,W\) source of monochromatic light of wavelength \(6600\,\AA\). What is the photoelectric current assuming \(3%\) efficiency for photoelectric effect?
A ray of light of intensity \(I\) is incident on a parallel glass slab at point A as shown in the diagram. It undergoes partial reflection and refraction. At each reflection, \(25%\) of incident energy is reflected. The rays AB and A'B' undergo interference. The ratio of \(I_{\max}\) and \(I_{\min}\) is:

A capillary tube of radius \(r\) is immersed vertically in a liquid such that liquid rises in it to height \(h\) (less than the length of the tube). Mass of liquid in the capillary tube is \(m\). If radius of the capillary tube is increased by \(50%\), then mass of liquid that will rise in the tube is:
The drift velocity of electrons in a silver wire with cross–sectional area \(3.14\times10^{-6}\,m^2\) carrying a current of \(20\,A\) is to be calculated. Given: atomic weight of Ag \(=108\), density of silver \(=10.5\times10^3\,kg m^{-3}\).
A parallel plate capacitor of area \(A\) and plate separation \(d\) is filled with two dielectrics as shown. What is the capacitance of the arrangement?

In Young’s double–slit experiment, the intensity of light at a point on the screen where the path difference is \(K\lambda\) is \(K\) (\(\lambda\) being the wavelength). The intensity at a point where the path difference is \(\lambda/4\) will be:
The mass of \(^{15}\mathrm{N}\) is \(15.00011\,amu\) and that of \(^{16}\mathrm{O}\) is \(15.99492\,amu\). The mass of the first proton is \(1.00783\,amu\). Determine the binding energy of the last proton of \(^{16}\mathrm{O}\).
A wire carrying current \(I\) has the shape as shown in the adjoining figure. Linear parts of the wire are very long and parallel to X–axis while the semicircular portion of radius \(R\) lies in Y–Z plane. Magnetic field at point \(O\) is:

A stone projected with velocity \(u\) at an angle \(\theta\) with the horizontal reaches maximum height \(H_1\). When it is projected with velocity \(u\) at an angle \(\left(\frac{\pi}{2}-\theta\right)\), it reaches maximum height \(H_2\). The relation between the horizontal range \(R\) of the projectile, heights \(H_1\) and \(H_2\) is:
If the series limit wavelength of the Lyman series for hydrogen atom is \(912\,\AA\), then the series limit wavelength for the Balmer series of hydrogen atom is:
In the shown arrangement of the meter bridge AC corresponding to null deflection of galvanometer is \(x\). What would be its value if the radius of the wire AB is doubled?

A \(1\,kg\) mass is attached to a spring of force constant \(600\,N m^{-1}\) and rests on a smooth horizontal surface with other end of the spring tied to a wall as shown in the figure. A second mass of \(0.5\,kg\) slides along the surface with initial speed \(3\,m s^{-1}\). If the masses make a perfectly inelastic collision, then find the amplitude and time period of oscillation of the combined mass.

The frequency of vibration of a string is given by \[ \nu=\frac{p}{2l}\left[\frac{F}{m}\right]^{1/2} \]
Here \(p\) is number of segments in the string and \(l\) is the length. The dimensional formula for \(m\) will be:
For the angle of minimum deviation of a prism to be equal to its refracting angle, the prism must be made of a material whose refractive index:
Consider elastic collision of a particle of mass \(m\) moving with a velocity \(u\) with another particle of the same mass at rest. After the collision the projectile and the struck particle move in directions making angles \(\theta_1\) and \(\theta_2\) respectively with the initial direction of motion. The sum of the angles \(\theta_1+\theta_2\) is:
A conducting circular loop is placed in a uniform magnetic field of \(0.04\,T\) with its plane perpendicular to the magnetic field. The radius of the loop starts shrinking at \(2\,mm s^{-1}\). The induced emf in the loop when the radius is \(2\,cm\) is:
Figure below shows two paths that may be taken by a gas to go from a state A to a state C. In process AB, \(400\,J\) of heat is added to the system and in process BC, \(100\,J\) of heat is added to the system. The heat absorbed by the system in the process AC will be:

Two resistances at \(0^\circC\) with temperature coefficients of resistance \(\alpha_1\) and \(\alpha_2\) joined in series act as a single resistance in a circuit. The temperature coefficient of their single resistance will be:
Two identical charged spheres suspended from a common point by two massless strings of length \(l\) are initially at a distance \(d \ll l\) apart because of their mutual repulsion. The charges begin to leak from both the spheres at a constant rate. As a result, the spheres approach each other with a velocity \(v\). Then \(v\) varies as a function of distance \(x\) between the spheres as:
A point particle of mass \(0.1\,kg\) is executing S.H.M. of amplitude \(0.1\,m\). When the particle passes through the mean position, its kinetic energy is \(8\times10^{-3}\,J\). Obtain the equation of motion of this particle if its initial phase of oscillation is \(45^\circ\).
A source of sound \(S\) emitting waves of frequency \(100\,Hz\) and an observer \(O\) are located at some distance from each other. The source is moving with a speed of \(19.4\,m s^{-1}\) at an angle of \(60^\circ\) with the source–observer line as shown. The observer is at rest. The apparent frequency observed is (velocity of sound in air \(=330\,m s^{-1}\)):

A resistor of resistance \(R\), capacitor of capacitance \(C\) and inductor of inductance \(L\) are connected in parallel to an AC power source of voltage \(\varepsilon_0\sin\omega t\). The maximum current through the resistance is half of the maximum current through the power source. Then value of \(R\) is:
A lens having focal length \(f\) and aperture of diameter \(d\) forms an image of intensity \(I\). Aperture of diameter \(d/2\) in central region of lens is covered by a black paper. Focal length of lens and intensity of image now will be respectively:
A circular disc of radius \(R\) and thickness \(\frac{R}{6}\) has moment of inertia \(I\) about an axis passing through its centre perpendicular to its plane. It is melted and recast into a solid sphere. The moment of inertia of the sphere about its diameter is:
In \(\mathrm{PO_4^{3-}}\), the formal charge on each oxygen atom and the P–O bond order respectively are:
The decreasing order of the ionization potential of the following elements is:
Knowing that the chemistry of lanthanoids (Ln) is dominated by its \(+3\) oxidation state, which of the following statements is incorrect?
Which of the following arrangements does not represent the correct order of the property stated against it?
Which of the following is paramagnetic?
The hypothetical cis–chloro–diaquatramminecobalt(III) chloride can be represented as:
The normality of \(26%\) (w/v) solution of ammonia (density \(=0.855\)) is approximately:
A \(1.25\,g\) sample of a mixture of \(Na_2CO_3\) and \(Na_2SO_4\) is dissolved in \(250\,mL\) solution. \(25\,mL\) of this solution neutralises \(20\,mL\) of \(0.1\,N\,H_2SO_4\). The % of \(Na_2CO_3\) in the sample is:
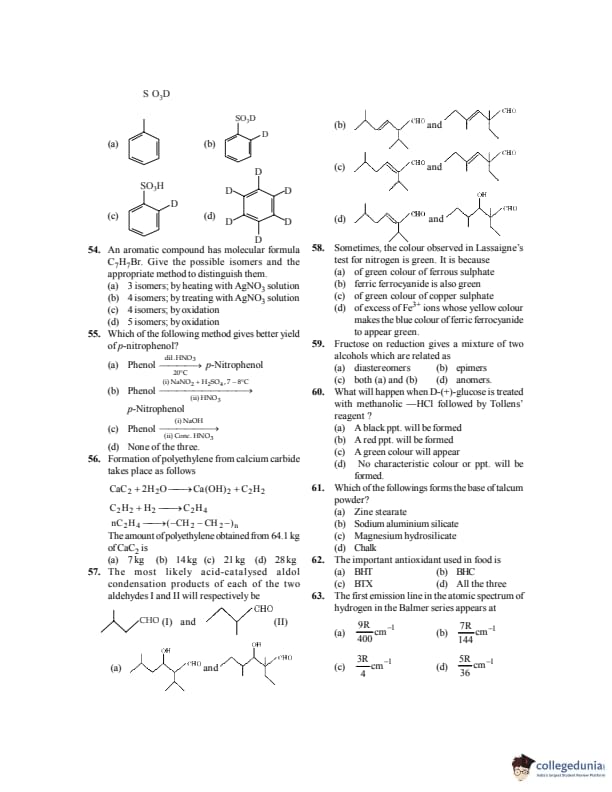
Which of the following compound has all the four types (1°, 2°, 3° and 4°) of carbon atoms?
Which of the following has two stereoisomers?
\[ CH_3C \equiv CCH_3 \xrightarrow{H_2/Pt} A \xrightarrow{D_2/Pt} B \]
The compounds \(A\) and \(B\), respectively, are:
Give the possible structure of \(X\) in the following reaction: \[ \mathrm{C_6H_6 + D_2SO_4 \xrightarrow[D_2O]{} X} \]

An aromatic compound has molecular formula \(\mathrm{C_7H_7Br}\). Give the possible isomers and appropriate method to distinguish them.
Which of the following method gives better yield of \(p\)-nitrophenol?
Formation of polyethylene from calcium carbide takes place as follows: \[ \begin{aligned} \mathrm{CaC_2 + 2H_2O} &\rightarrow \mathrm{Ca(OH)_2 + C_2H_2}
\mathrm{C_2H_2 + H_2} &\rightarrow \mathrm{C_2H_4}
n\mathrm{C_2H_4} &\rightarrow (-\mathrm{CH_2-CH_2}-)_n \end{aligned} \]
The amount of polyethylene obtained from \(64.1\,kg\) of \(\mathrm{CaC_2}\) is:
The most likely acid–catalysed aldol condensation products of each of the two aldehydes I and II will respectively be:


Sometimes, the colour observed in Lassaigne’s test for nitrogen is green. It is because:
Fructose on reduction gives a mixture of two alcohols which are related as:
What will happen when D-(+)-glucose is treated with methanolic HCl followed by Tollen’s reagent?
Which of the following forms the base of talcum powder?
The important antioxidant used in food is:
The first emission line in the atomic spectrum of hydrogen in the Balmer series appears at:
An electron has magnetic quantum number \(m_l=-3\). What is its principal quantum number?
At what temperature is the rate of diffusion of \(SO_2\) double that at \(50^\circC\)?
The average kinetic energy of an ideal gas molecule in SI unit at \(25^\circC\) will be:
The degree of dissociation of \(PCl_5(g)\) obeying the equilibrium \[ PCl_5 \rightleftharpoons PCl_3 + Cl_2 \]
is related to the equilibrium pressure by:
In a closed system, \[ A(s) \rightleftharpoons 2B(g)+3C(g) \]
if the partial pressure of \(C\) is doubled, then partial pressure of \(B\) will be:
For a particular reversible reaction at temperature \(T\), \(\Delta H\) and \(\Delta S\) were found to be both positive. If \(T_e\) is the temperature at equilibrium, the reaction would be spontaneous when:
Given the following data:
\begin{tabular{l c
Reaction & Energy change (kJ)
\hline
Li(s) \(\rightarrow\) Li(g) & 161
Li(g) \(\rightarrow\) Li\(^{+}\)(g) & 520
\(\tfrac{1}{2}\)F\(_2\)(g) \(\rightarrow\) F(g) & 77
F(g) + e\(^{-}\) \(\rightarrow\) F\(^{-}\)(g) & ?
Li\(^{+}\)(g) + F\(^{-}\)(g) \(\rightarrow\) LiF(s) & \(-1047\)
Li(s) + \(\tfrac{1}{2}\)F\(_2\)(g) \(\rightarrow\) LiF(s) & \(-617\)
\end{tabular
Based on the data provided, the value of electron gain enthalpy of fluorine would be:
The percentage hydrolysis of \(0.15\) M solution of ammonium acetate.
Given: \(K_a\) for CH\(_3\)COOH \(=1.8\times10^{-5}\) and \(K_b\) for NH\(_3\) \(=1.8\times10^{-5}\).
For a sparingly soluble salt \(A_pB_q\), the relationship of its solubility \(S\) with its solubility product \(K_{sp}\) is:
Consider the reaction: \[ Cl_2(aq)+H_2S(aq)\rightarrow S(s)+2H^+(aq)+2Cl^-(aq) \]
The rate equation is \(rate=k[Cl_2][H_2S]\).
Which of the following mechanisms is/are consistent with this rate equation?
In the reaction \(P+Q\rightarrow 2R+S\), the time taken for \(75%\) reaction of \(P\) is twice the time taken for \(50%\) reaction of \(P\). The concentration of \(Q\) varies with time as shown. The overall order of the reaction is:

The EMF of the cell
Ti\(|\)Ti\(^{+}\)(0.001 M)\(||\)Cu\(^{2+}\)(0.01 M)\(|\)Cu\( is \)0.83\( V.
The cell EMF can be increased by:
Electrolysis is carried out in three cells:
(A) \(1.0\,M\) CuSO\(_4\) using Pt electrodes
(B) \(1.0\,M\) CuSO\(_4\) using Cu electrodes
(C) \(1.0\,M\) KCl using Pt electrodes
If volume of electrolyte solution is maintained constant in each cell, which is the correct set of change of pH in the (A), (B) and (C) cell respectively?
The equilibrium constant for the disproportionation reaction \[ 2Cu^+(aq) \rightleftharpoons Cu(s)+Cu^{2+}(aq) \]
at \(25^\circC\) is given that \(E^\circ_{Cu^+/Cu}=0.52\,V\), \(E^\circ_{Cu^{2+}/Cu^+}=0.16\,V\).
The non-stoichiometric compound \(\mathrm{Fe_{0.94}O}\) is formed when \(x%\) of \(\mathrm{Fe^{2+}}\) ions are replaced by as many \(\tfrac{2}{3}\mathrm{Fe^{3+}}\) ions. The value of \(x\) is:
Al (atomic weight = 27) crystallizes in the cubic system with a cell edge of \(4.05\,\AA\). Its density is \(2.7\,g cm^{-3}\). Determine the unit cell type and radius of the Al atom.
A compound of Xe and F is found to have \(53.5%\) Xe. What is the oxidation number of Xe in this compound?
CORPULENT
EMBEZZLE
ARROGANT
EXODUS
According to the author of “Mentality of a given nation” is mainly produced of its
The need for a greater understanding between nations
The character of a nation is the result of its
According to the author his countrymen should
S1: A force exists between everybody in the universe.
P: Normally it is very small but when one of the bodies is a planet, like earth, the force is considerable.
Q: It has been investigated by many scientists including Galileo and Newton.
R: Everything on or near the surface of the earth is attracted by the mass of earth.
S: This gravitational force depends on the mass of the bodies involved.
S6: The greater the mass, the greater is the earth’s force of attraction on it. We can call this force of attraction gravity.
The proper sequence should be:
S1: Calcutta unlike other cities keeps its trams.
P: As a result there horrendous congestion.
Q: It was going to be the first in South Asia.
R: They run down the centre of the road.
S: To ease in the city decided to build an underground railway line.
S6: The foundation stone was laid in 1972.
The proper sequence should be:
The miser gazed ____ at the pile of gold coins in front of him.
I saw a ____ of cows in the field.
We discussed about the problem so thoroughly on the eve of the examination that I found it very easy to work it out.
An Indian ship laden with merchandise got drowned in the Pacific Ocean.
I could not put up in a hotel because the boarding and lodging charges were exorbitant.
Select a suitable figure from the four alternatives that would complete the figure matrix.

Select a suitable figure from the four alternatives that would complete the figure matrix.

Select a suitable figure from the four alternatives that would complete the figure matrix.

Choose the correct alternative that will continue the same pattern and replace the question mark in the given series: \(3, 4, 7, 13, 13, 21, 31, 34, ?\)
Introducing a boy, a girl said, “He is the son of the daughter of the father of my uncle.” How is the boy related to the girl?
QAR, RAS, SAT, TAU, ___
DEF, DEE\(_2\), DE\(_2\)F\(_2\), ___
View Solution
Step 1: Count subscripts of letters D, E, F.
Step 2: Powers increase cyclically: \[ D:1\rightarrow1\rightarrow2\rightarrow2,\quad E:1\rightarrow2\rightarrow2\rightarrow3,\quad F:1\rightarrow1\rightarrow2\rightarrow1 \]
Step 3: Hence next term is \(D_2E_3F\). Quick Tip: Check numerical progression of subscripts carefully.
Statements: Raman is always successful. No fool is always successful.
Conclusions:
I. Raman is a fool.
II. Raman is not a fool.
Statements: Some desks are caps. No cap is red.
Conclusions:
I. Some caps are desks.
II. No desk is red.
Choose the set of figures which follows the given rule.
Rule: Closed figures losing their sides and open figures gaining their sides.

Let \(f(x)=\dfrac{ax+b}{cx+d}\). Then \(f(x)=x\), provided that:
Two finite sets have \(m\) and \(n\) elements. The number of subsets of the first set is 112 more than that of the second set. The values of \(m\) and \(n\) respectively are:
If \(A\) and \(B\) are positive acute angles satisfying \(3\cos^2A+2\cos^2B=4\) and \(\dfrac{3\sin A}{\sin B}=\dfrac{2\cos B}{\cos A}\), then the value of \(A+2B\) is equal to:
If \(\sin\theta_1+\sin\theta_2+\sin\theta_3=3\), then \(\cos\theta_1+\cos\theta_2+\cos\theta_3=\):
If \(\tan(\cot x)=\cot(\tan x)\), then \(\sin 2x\) is equal to:
The general solution of the equation \(\sin2x+2\sin x+2\cos x+1=0\) is:
In a \(\triangle ABC\), if \(\dfrac{\cos A}{a}=\dfrac{\cos B}{b}=\dfrac{\cos C}{c}\), and the side \(a=2\), then area of triangle is:
If \[ \sin^{-1}\!\left(\frac{2a}{1+a^2}\right) -\cos^{-1}\!\left(\frac{1-b^2}{1+b^2}\right) =\tan^{-1}\!\left(\frac{2x}{1-x^2}\right), \]
then the value of \(x\) is:
The arithmetic mean of numbers \(a, b, c, d, e\) is \(M\). What is the value of \((a-M)+(b-M)+(c-M)+(d-M)+(e-M)\)?
The fourth term of an A.P. is three times of the first term and the seventh term exceeds the twice of the third term by one. Then the common difference of the progression is
The sum to \(n\) terms of the series \(\dfrac12+\dfrac34+\dfrac78+\dfrac{15}{16}+\cdots\) is
If \(\log a,\log b,\log c\) are in A.P. and also \(\log a-\log 2b,\;\log 2b-\log 3c,\;\log 3c-\log a\) are in A.P., then
\[ \left(x+\frac1x\right)^2+\left(x^2+\frac1{x^2}\right)^2+\cdots+\left(x^n+\frac1{x^n}\right)^2 \]
upto \(n\) terms is
If \(z_1=\sqrt3+i\sqrt3\) and \(z_2=\sqrt3+i\), then the complex number \(\left(\dfrac{z_1}{z_2}\right)^{50}\) lies in the
If the matrix \[ \begin{pmatrix} 1 & 3 & \lambda+2
2 & 4 & 8
3 & 5 & 10 \end{pmatrix} \]
is singular, then \(\lambda=\)
Let \(\alpha_1,\alpha_2\) and \(\beta_1,\beta_2\) be the roots of \(ax^2+bx+c=0\) and \(px^2+qx+r=0\) respectively. If the system \[ \alpha_1 x+\alpha_2 y=0,\qquad \beta_1 y+\beta_2 z=0 \]
has a non-trivial solution, then
If \([x]\) denotes the greatest integer less than or equal to \(x\), and \(-1
[x] & [y] & [z]+1 \end{vmatrix} \]
is
If \(\alpha,\beta\) are the roots of the equation \(x^2-2x-1=0\), then the value of \(\alpha^2\beta^2+\alpha-\alpha^2\beta^2\) is
If \(a,b,c\) are real numbers then the roots of \((x-a)(x-b)+(x-b)(x-c)+(x-c)(x-a)=0\) are always
If \(a>b>1\), then \[ \lim_{n\to\infty}\frac{a^n+b^n}{a^n-b^n} \]
is equal to
The number of points at which the function \[ f(x)=\frac{1}{\log|x|} \]
is discontinuous is
If \[ f(x)= \begin{cases} \dfrac{x\log(\cos x)}{\log(1+x^2)}, & x\ne0
0, & x=0 \end{cases} \]
then \(f(x)\) is
For any differentiable function \(y\) of \(x\), \[ \frac{d^2x}{dy^2}\left(\frac{dy}{dx}\right)^3+\frac{d^2y}{dx^2}= \]
The set of all values of \(a\) for which the function \[ f(x)=(a^2-3a+2)(\cos^2x/4-\sin^2x/4)+(a-1)x \]
is decreasing is
Match List I with List II and select the correct answer using the code given below the lists.
List I
(A) \(f(x)=\cos x\)
(B) \(f(x)=\ln x\)
(C) \(f(x)=x^2-5x+4\)
(D) \(f(x)=e^x\)
List II
1. The graph cuts \(y\)-axis in infinite number of points
2. The graph cuts \(x\)-axis in two points
3. The graph cuts \(y\)-axis in only one point
4. The graph cuts \(x\)-axis in only one point
5. The graph cuts \(x\)-axis in infinite number of points
% Codes
(A)\;(a)\;1\;4\;5\;3 \quad
(b)\;1\;3\;5\;4 \quad
(c)\;5\;4\;2\;3 \quad
(d)\;5\;3\;2\;4
What is the \(x\)-coordinate of the point on the curve \[ f(x)=\sqrt{x}\,(7x-6), \]
where the tangent is parallel to the \(x\)-axis?
A wire \(34\) cm long is to be bent in the form of a quadrilateral of maximum area in which each angle is \(90^\circ\). What is the maximum area enclosed?
Consider the following statements in respect of the function \(f(x)=x^3-1,\; x\in[-1,1]\):
I. \(f(x)\) is continuous in \([-1,1]\).
II. \(f(x)\) has no root in \((-1,1)\).
Which of the statements given above is/are correct?
At an extreme point of a function \(f(x)\), the tangent to the curve is
The curve \(y=xe^x\) has minimum value equal to
A ray of light coming from the point \((1,2)\) is reflected at a point \(A\) on the \(x\)-axis and then passes through the point \((5,3)\). The coordinates of the point \(A\) are
The equation \[ x^2-2\sqrt{3}xy+3y^2-3x+3\sqrt{3}y-4=0 \]
represents
The line joining \((5,0)\) to \((10\cos\theta,10\sin\theta)\) is divided internally in the ratio \(2:3\) at \(P\). If \(\theta\) varies, the locus of \(P\) is
The number of integral values of \(\lambda\) for which \[ x^2+y^2+\lambda x+(1-\lambda)y+5=0 \]
is the equation of a circle whose radius exceeds \(5\), is
The lengths of the tangent drawn from any point on the circle \(15x^2+15y^2-48x+64y=0\) to the circles \(5x^2+5y^2-24x+32y+75=0\) and \(5x^2+5y^2-48x+64y+300=0\) are in the ratio
The length of the chord \(x+y=3\) intercepted by the circle \(x^2+y^2-2x-2y-2=0\) is
The locus of the point of intersection of two tangents to the parabola \(y^2=4ax\), which are at right angle to one another is
The parabola having its focus at \((3,2)\) and directrix along the \(y\)-axis has its vertex at
The number of values of \(r\) satisfying \({}^{39}C_{3r-1}={}^{39}C_{r^2-1}={}^{39}C_{3r}\) is
If \[ \sum_{r=0}^{n+2}{}^{n}C_r=2^8-1, \]
then \(n=\)
All the words that can be formed using alphabets A, H, L, U and R are written in a dictionary (no alphabet is repeated). Rank of the word RAHUL is
If the sum of odd numbered terms and even numbered terms in the expansion of \((x+a)^n\) are \(A\) and \(B\) respectively, then the value of \((x^2-a^2)^n\) is
If the third term in the expansion of \([x+x^{\log_{10}x}5]\) is \(10^6\), then \(x\) may be
If three vertices of a regular hexagon are chosen at random, then the chance that they form an equilateral triangle is
A man takes a step forward with probability \(0.4\) and backward with probability \(0.6\). The probability that at the end of ten steps he is one step away from the starting point is




































Comments