BITSAT 2017 Question Paper with Answer Key pdf is available for download. BITSAT 2017 was conducted in online CBT mode by BITS Pilani. BITSAT 2017 Question Paper had 150 questions to be attempted in 3 hours.
BITSAT 2017 Question Paper with Answer Key PDF
| BITSAT 2017 Question Paper PDF | BITSAT 2017 Answer Key PDF |
|---|---|
| Download PDF | Download PDF |
What is the minimum energy required to launch a satellite of mass \( m \) from the surface of a planet of mass \( M \) and radius \( R \) in a circular orbit at an altitude of \( R \)?
A mercury drop of radius 1 cm is sprayed into \(10^6\) drops of equal size. The energy pressed in joule is (surface tension of mercury is \(460 \times 10^{-3}\,N/m\)).
Two plano-concave lenses (1 and 2) of glass of refractive index 1.5 have radii of curvature 25 cm and 20 cm. They are placed in contact with their curved surfaces towards each other and the space between them is filled with liquid of refractive index \(4/3\). The combination is

A charged particle moves perpendicular to a magnetic field. Then
After two hours, one-sixteenth of the starting amount of a certain radioactive isotope is remained undecayed. The half-life of the isotope is
A coil of inductance 300 mH and resistance \(2\Omega\) is connected to a source of voltage 2 V. The current reaches half of its steady state value in
Two concentric conducting spherical shells \(A\) and \(B\) having radii \(r_A\) and \(r_B\) \((r_B>r_A)\) are charged to \(+2q\) and \(-q\) respectively. The electric field along a line passing through the centre is

A capillary tube of radius \(R\) is immersed in water and water rises in it to a height \(H\). Mass of water in the capillary tube is \(M\). If the radius of the tube is doubled, mass of water that will rise in the capillary tube will now be
A sonometer wire resonates with a given tuning fork forming standing waves with five antinodes between the two bridges when a mass of 9 kg is suspended from the wire. When this mass is replaced by a mass \(M\), the wire resonates with the same tuning fork forming three antinodes for the same position of the bridges. The value of \(M\) is
When a metal surface is illuminated by light of wavelength 400 nm and 250 nm, the maximum velocities of the photoelectrons ejected are \(v\) and \(2v\) respectively. The work function of the metal is (h = Planck’s constant, c = velocity of light in air)
Two conducting shells of radius \(a\) and \(b\) are connected by a conducting wire as shown in the figure. The capacity of the system is

When \(^{235}\mathrm{U}\) undergoes fission, 0.1% of its original mass is changed into energy. How much energy is released if 1 kg of \(^{235}\mathrm{U}\) undergoes fission
One mole of an ideal gas is taken from state A to state B by three different processes, (i) ACB (ii) ADB (iii) AEB as shown in the P–V diagram. The heat absorbed by the gas is

In the formula \(X = 3YZ^2\), \(X\) has dimensions of capacitance and magnetic induction respectively. The dimensional formula of \(Y\) in MKSA system is
Two long, straight, parallel wires carry steady currents \(I\) and \(-I\) respectively. The distance between the wires is \(d\). At a certain instant of time, a point charge \(q\) is at a point equidistant from the two wires. The instantaneous velocity \(v\) is perpendicular to this plane. The magnitude of the force due to the magnetic field acting on the charge is
Two projectiles A and B are thrown with speeds in the ratio \(1:\sqrt{2}\) and acquire the same heights. If A is thrown at an angle of \(45^\circ\) with the horizontal, the angle of projection of B will be
A meter bridge is set up as shown to determine an unknown resistance \(X\) using a standard 10 ohm resistor. The galvanometer shows null point at 52 cm mark. The end corrections are 1 cm and 2 cm respectively for the ends A and B. The determined value of \(X\) is

A disk of radius \(a/4\) having a uniformly distributed charge \(6\,C\) is placed in the \(x-y\) plane with its centre at \((-a/2,0,0)\). A rod of length \(a\) carrying a uniformly distributed charge \(8\,C\) is placed on the \(x\)-axis from \(x=a/4\) to \(x=5a/4\). Two point charges \(-7\,C\) and \(3\,C\) are placed at \((a/4,-a/4,0)\) and \((-3a/4,3a/4,0)\) respectively. Consider a cubical surface formed by six surfaces \(x=\pm a/2,\ y=\pm a/2,\ z=\pm a/2\). The electric flux through this cubical surface is

A particle of mass \(m\) moving in the \(x\)-direction with speed \(2v\) is hit by another particle of mass \(2m\) moving in the same direction with speed \(v\). If the collision is perfectly inelastic, the percentage loss in the energy during the collision is close to
A coil is suspended in a uniform magnetic field with the plane of the coil parallel to the magnetic lines of force. When a current is passed through the coil it starts oscillating; it is very difficult to stop. But if an aluminium plate is placed near the coil, it stops. This is due to
A steel wire of length \(L\) at \(40^\circC\) is suspended from the ceiling and carries a mass \(m\) from its free end. The wire is cooled from \(40^\circC\) to \(30^\circC\) to regain its original length \(L\). The coefficient of linear expansion is \(10^{-5}\,/^\circC\), Young’s modulus of steel is \(10^{11}\,N m^{-2}\) and the radius of the wire is \(1\,mm\). The value of \(m\) in kg is nearly
On a hypotenuse of a right angled prism of liquid is placed as shown in the figure. Light is allowed to fall normally on the short face of the prism. In order that the ray of light may be totally reflected, the maximum value of refractive index is

A tuning fork of frequency 392 Hz resonates with 50 cm length of a string under tension \(T\). If the mass of the string is decreased by 2%, keeping the tension constant, the number of beats heard when the string and the tuning fork are made to vibrate simultaneously is
Hydrogen (H), deuterium (D), singly ionized helium (\(He^+\)) and doubly ionized lithium (\(Li^{2+}\)) all have one electron around the nucleus. Consider \(n=2\) to \(n=1\) transition. The wavelengths of emitted radiations are \(\lambda_1, \lambda_2, \lambda_3\) and \(\lambda_4\) respectively. Then approximately
The following figure depicts a circular motion. The radius of the circle, the period of revolution, the initial position and the sense of revolution are indicated in the figure. The simple harmonic motion of the \(x\)-projection of the radius vector of the rotating particle \(P\) can be shown as

There are two sources kept at distances \(2\lambda\). A large screen is perpendicular to the line joining the sources. Number of maxima on the screen in this case is (\(\lambda=\) wavelength of light)

In the circuit shown in the figure the current through the resistor is

A telescope has an objective lens of 10 cm diameter and is situated at a distance of one kilometer from two objects. The minimum distance between these two objects which can be resolved by the telescope, when the wavelength of light is \(5000\,\AA\), is of the order of
During vaporisation:
I. change of state from liquid to vapour state occurs.
II. temperature remains constant.
III. both liquid and vapour states coexist in equilibrium.
IV. specific heat of substance increases.
Correct statements are
A wire is connected to a battery between the points \(M\) and \(N\) as shown in figure (1). The same wire is bent in the form of a square and then connected to the battery between the points \(M\) and \(N\) as shown in figure (2). Which of the following quantities increases?

A body moves in a circular orbit of radius \(R\) under the action of a central force. Potential due to the central force is given by \(V(r)=kr\) (where \(k\) is a positive constant). Period of revolution of the body is proportional to
Two equal heavy spheres, each of radius \(r\), are in equilibrium within a smooth cup of radius \(3r\). The ratio of reaction between the cup and one sphere and that between the two spheres is

A long, hollow conducting cylinder is kept coaxially inside another long, hollow conducting cylinder of larger radius. Both the cylinders are initially electrically neutral.
A thin but rigid semicircular wire frame of radius \(r\) is hinged at \(O\) and can rotate in its own vertical plane. A smooth peg \(P\) starts from \(O\) and moves horizontally with constant speed \(v_0\), lifting the frame upwards as shown in the figure. Find the angular velocity \(\omega\) of the frame when its diameter makes an angle of \(60^\circ\) with the vertical.

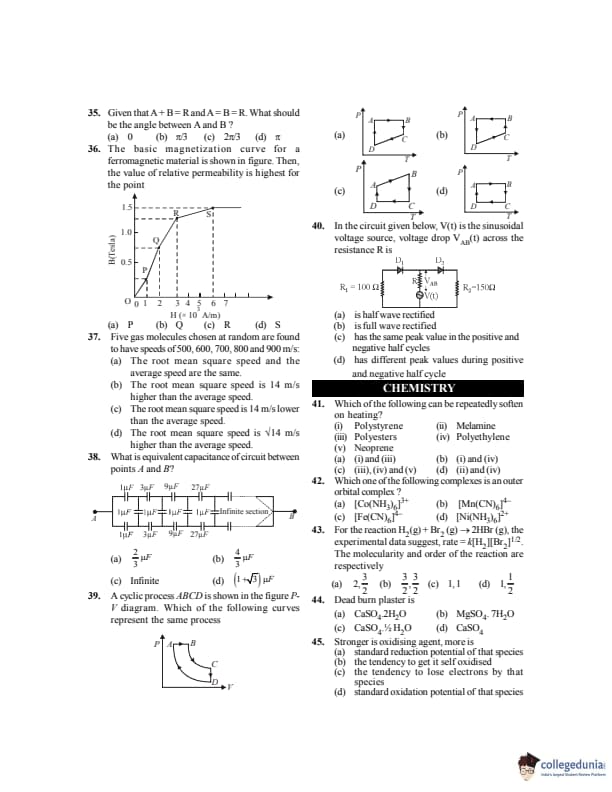
Given that \( \vec A + \vec B = \vec R \) and \( A = B = R \). What should be the angle between \( \vec A \) and \( \vec B \)?
The basic magnetization curve for a ferromagnetic material is shown in figure. Then, the value of relative permeability is highest for the point

Five gas molecules chosen at random are found to have speeds of 500, 600, 700, 800 and 900 m/s.
What is equivalent capacitance of the circuit between points A and B?

A cyclic process ABCD is shown in the P–V diagram. Which of the following curves represent the same process?


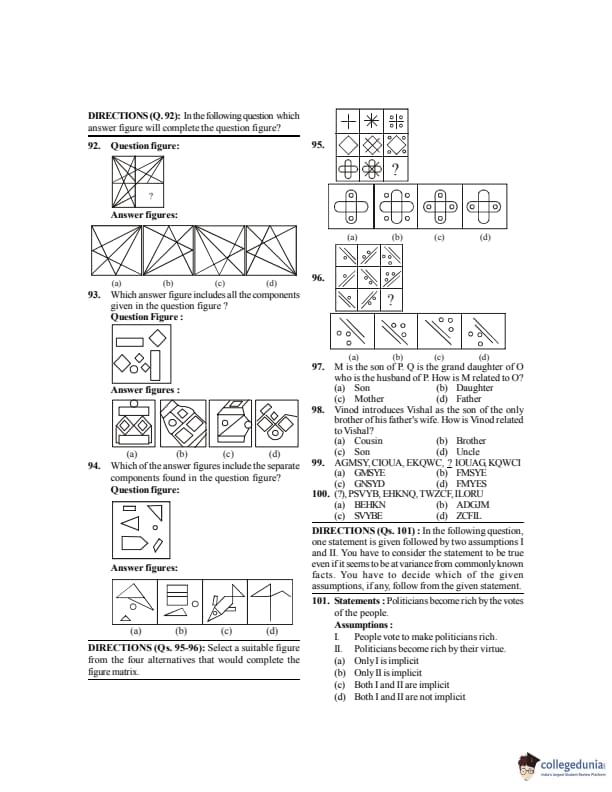
In the circuit shown below, \(V(t)\) is the sinusoidal voltage source, the voltage drop \(V_{AB}(t)\) across the resistance \(R\) is

Which of the following can be repeatedly softened on heating?
(i) Polystyrene \quad (ii) Melamine
(iii) Polyesters \quad (iv) Polyethylene \quad (v) Neoprene
Which one of the following complexes is an outer orbital complex?
For the reaction \[ H_2(g) + Br_2(g) \rightarrow 2HBr(g), \]
the experimental data suggest the rate law \[ Rate = k[H_2][Br_2]^{1/2}. \]
The molecularity and order of the reaction are respectively
Plaster of Paris is
Stronger is oxidising agent, more is
Which of the following relation represents correct relation between standard electrode potential and equilibrium constant?
I.\quad \( \log K = \dfrac{nF E^\circ}{2.303\,RT} \)
II.\quad \( K = e^{\frac{nF E^\circ}{RT}} \)
III.\quad \( \log K = -\dfrac{nF E^\circ}{2.303\,RT} \)
IV.\quad \( \log K = 0.4342\,\dfrac{nF E^\circ}{RT} \)
Choose the correct statements.
Which of the following shows nitrogen with its increasing order of oxidation number?
Raoult’s law becomes a special case of Henry’s law when
The standard electrode potentials are: \(E^\circ(Zn^{2+}/Zn) = -0.76\ V\), \(E^\circ(Cu^{2+}/Cu) = +0.34\ V\).
The equilibrium constant for the reaction \[ Zn + Cu^{2+} \rightleftharpoons Cu + Zn^{2+} \]
at \(25^\circC\) is of the order of
Which of the following represents Gay Lussac’s law?
I.\quad \( \dfrac{P}{T} = constant \)
II.\quad \( P_1T_2 = P_2T_1 \)
III.\quad \( P_1V_1 = P_2V_2 \)
Choose the correct option.
For the reaction \[ CO(g) + \frac{1}{2}O_2(g) \rightarrow CO_2(g), \]
which one of the statements is correct at constant \(T\) and \(P\)?
The energy of an electron in second Bohr orbit of hydrogen atom is:
Which of the following order is wrong?
Which of the following is not involved in the formation of photochemical smog?
Which of the following is not present in Portland cement?
Which of the following can form buffer solution?
Which of the following complex shows \(sp^3d^2\) hybridization?
Which has glycosidic linkage?
Which of the following represents Schotten-Baumann reaction?
In the following structures, which two forms are staggered conformations of ethane?

Which of the following shows correct order of bond length?
The number of radial nodes of 3s and 2p orbitals are respectively
If a 25.0 mL sample of sulfuric acid is titrated with 50.0 mL of 0.025 M sodium hydroxide to a phenolphthalein endpoint, what is the molarity of the acid?
Find which of the following compound can have mass ratio of C:H:O as 6:1:24
The number of atoms per unit cell of bcc structure is
Which of these doesn’t exist?
Which of these compounds are directional?
For a given reaction, \(\Delta H = 35.5\) kJ mol\(^{-1}\) and \(\Delta S = 83.6\) J K\(^{-1}\) mol\(^{-1}\). The reaction is spontaneous at
Specific conductance of 0.1 M HA is \(3.75\times10^{-4}\ \Omega^{-1}cm^{-1}\). If \(\Lambda_m^\infty(HA)=250\ \Omega^{-1}cm^2mol^{-1}\), the dissociation constant \(K_a\) of HA is
The rate of reaction between two reactants A and B decreases by a factor of 4 if the concentration of reactant B is doubled. The order of this reaction with respect to reactant B is
A compound of molecular formula C\(_7\)H\(_{16}\) shows optical isomerism. Compound will be
Which of the following does not contain plane of symmetry?
Cadmium is used in nuclear reactors for?
Which reagent converts nitrobenzene to N-phenyl hydroxylamine?
Which of the following can act as both Brønsted acid and Brønsted base?
Identify the structure of water in the gaseous phase.
Electrometallurgical process is used to extract
The correct statement about the compounds A, B and C is

Correct formula of the complex formed in the brown ring test for nitrates is
Which one of the following is an amine hormone?
Loquacious
Meticulous
To write well, a person must train himself in
Initially it is difficult to write because
According to the passage, writing style can be improved by
Famous writers have achieved success by
China is a big country, area it is bigger than any other country _____ Russia.
The treasure was hidden _____ a big shore.
My father gave me (a) / a pair of binocular (b) / on my birthday. (c) / No error. (d)
The teacher as well as his students, (a) / all left (b) / for the trip. (c) / No error. (d)
Which answer figure completes the form in the question figure?

Which answer figure will complete the question figure?


Which answer figure includes all the components given in the question figure?

Which of the answer figures includes the separate components found in the question figure?

Select a suitable figure from the four alternatives that would complete the given figure matrix.

Select a suitable figure from the four alternatives that would complete the given figure matrix.

M is the son of P. Q is the grand daughter of O who is the husband of P. How is M related to O?
Vinod introduces Vishal as the son of the only brother of his father’s wife. How is Vinod related to Vishal?
AGMSY, CIOUA, EKQWC, ? IOUAG, KQWCI
?, PSVYB, EHKNQ, TWZCF, ILORU
Statements: Politicians become rich by the votes of the people.
Assumptions:
I. People vote to make politicians rich.
II. Politicians become rich by their virtue.
Two statements are given followed by four conclusions I, II, III and IV. You have to consider the statements to be true, even if they seem to be at variance from commonly known facts. You have to decide which of the given conclusions can definitely be drawn from the given statements.
Statements:
(A) No cow is a chair.
(B) All chairs are tables.
Conclusions:
I. Some tables are chairs
II. Some tables are cows
III. Some chairs are cows
IV. No table is a cow
Statements:
1. Temple is a place of worship.
2. Church is also a place of worship.
Conclusions:
I. Hindus and Christians use the same place for worship.
II. All churches are temples.
Statement:
The human organism grows and develops through stimulation and action.
Conclusions:
I. Inert human organism cannot grow and develop.
II. Human organisms do not react to stimulation and action.
Choose the set of figure which follows the given rule.
Rule: Closed figures gradually become open and open figures gradually become closed.

Let \(f\) and \(g\) be functions from \(\mathbb{R}\) to \(\mathbb{R}\) defined as
\[ f(x)= \begin{cases} 7x^2+x-8, & x\le 1
4x+5, & 1
0, & -3\le x<2
x^2+4, & x\ge 2 \end{cases} \]
Then
How many different nine digit numbers can be formed from the number 223355888 by rearranging its digits so that odd digits occupy even positions?
If \(\displaystyle \sum_{k=1}^{n} k(k+1)(k-1)=pn^4+qn^3+tn^2+sn\), where \(p,q,t,s\) are constants, then the value of \(s\) is equal to
The length of the semi-latus rectum of an ellipse is one third of its major axis. Its eccentricity would be
If \(\alpha\) and \(\beta\) are roots of the equation \[ x^2+px+\frac{3p}{4}=0, \]
such that \(|\alpha-\beta|=\sqrt{10}\), then \(p\) belongs to the set
Given the system of straight lines \[ a(2x+y-3)+b(3x+2y-5)=0, \]
the line of the system situated farthest from the point \((4,-3)\) has the equation
One mapping is selected at random from all mappings of the set \[ S=\{1,2,3,\dots ,n\} \]
into itself. The probability that it is one–one is \(\frac{3}{32}\). Then the value of \(n\) is
The integer just greater than \((3+\sqrt5)^{2n}\) is divisible by
The domain of the function \[ f(x)=\sin^{-1}\!\left\{\log_2\!\left(\frac12 x^2\right)\right\} \]
is
The marks obtained by 60 students are given. The median of the data is
If \(A,B,C\) are the angles of a triangle and \[ e^{iA},\; e^{iB},\; e^{iC} \]
are in A.P., then the triangle must be
An observer on the top of a tree finds the angle of depression of a car moving towards the tree to be \(30^\circ\). After 3 minutes this angle becomes \(60^\circ\). After how much more time will the car reach the tree?
After striking the floor a certain ball rebounds \(\frac{4}{5}\)th of its height from which it has fallen. The total distance that the ball travels before coming to rest if it is gently released from a height of \(120 m\) is
An equilateral triangle is inscribed in the circle \(x^2+y^2=a^2\) with one of the vertices at \((a,0)\). What is the equation of the side opposite to this vertex?
The function \(f(x)=x-|x-x^2|\), \(-1\le x\le1\) is continuous on
If \[ \frac{4^n}{n+1}<\frac{(2n)!}{(n!)^2}, \]
then \(P(n)\) is true for
If a system of equations \[ ax+y+z=0,\quad x-by+z=0,\quad x+y-cz=0 \quad (a,b,c\ne-1) \]
has a non-zero solution, then \[ \frac{1}{1+a}+\frac{1}{1+b}+\frac{1}{1+c}= \]
If \(f(x)=x^x\), then \(f(x)\) is increasing in interval
If \(x\) is real number, then \[ \frac{x}{x^2-5x+9} \]
must lie between
The value of \[ \lim_{x\to\infty}\left(\frac{a_1^{1/x}+a_2^{1/x}+\cdots+a_n^{1/x}}{n}\right)^{nx}, \quad a_i>0 \]
The value of \(\cot^{-1}7+\cot^{-1}8+\cot^{-1}18\) is
If \(\displaystyle \int \frac{e^x\cos x}{1+\sin x}\,dx\) is equal to
A random variable \(X\) has the probability distribution given.
For the events \(E=\{X is a prime\}\) and \(F=\{X<4\}\), then \(P(E\cup F)\) is
The number of roots of the equation \(\cos x+\cos 3x=0\) in \(0\le x\le2\pi\) is
The area under the curve \(y=|\cos x-\sin x|\), \(0\le x\le\frac{\pi}{2}\), and above the x-axis is
If \[ f(x)= \begin{cases} \dfrac{x\log(\cos x)}{\log(1+x^2)}, & x\ne0
0, & x=0 \end{cases} \]
then \(f(x)\) is
The maximum value of \(z=3x+2y\) subject to \(x+2y\ge2,\ x+2y\le8,\ y\ge0\) is
A cylindrical gas container is closed at the top and open at the bottom. If the iron plate of the top is \(\frac54\) time as thick as the plate forming the cylindrical sides, the ratio of the radius to the height of the cylinder using minimum material for the same capacity is
Let \(A,B,C\) be finite sets. Suppose that \(n(A)=10,\ n(B)=15,\ n(C)=20,\ n(A\cap B)=8\) and \(n(B\cap C)=9\). Then the possible value of \(n(A\cup B\cup C)\) is
If \(f(z)=\dfrac{7-z}{1-z^2}\), where \(z=1+2i\), then \(|f(z)|\) is equal to
If \[ f(x)=\cos^{-1}\!\left[\frac{1-(\log x)^2}{1+(\log x)^2}\right], \]
then the value of \(f'(e)\) is equal to
Statement–1: A five digit number divisible by 3 is to be formed using the digits 0,1,2,3,4 and 5 with repetition. The total number formed are 216.
Statement–2: If sum of digits of any number is divisible by 3 then the number must be divisible by 3.
The equation of one of the common tangents to the parabola \(y^2=8x\) and \(x^2+y^2-12x+4=0\) is
If \[ R(t)= \begin{pmatrix} \cos t & \sin t
-\sin t & \cos t \end{pmatrix}, \]
then \(R(s)R(t)\) equals
If \[ \int x\log\!\left(1+\frac1x\right)\,dx=f(x)\log(x+1)+g(x)x^2+Lx+C, \]
then
Let \(\vec a,\vec b,\vec c\) be non-coplanar unit vectors equally inclined to one another at an acute angle \(\theta\). Then \(|\vec a\cdot(\vec b\times\vec c)|\) in terms of \(\theta\) is equal to
\(2^{1/4}\cdot 2^{2/8}\cdot 2^{3/16}\cdot 2^{4/32}\cdots\) is equal to
If \(\displaystyle \sum_{r=0}^{n}\binom{n}{r}\frac{{}^{n}C_r}{{}^{\,r+3}C_r}=\frac{3}{a+3}\), then \(a-n\) is equal to
If \[ \begin{vmatrix} p&q-y&r-z
p-x&q&r-z
p-x&q-y&r \end{vmatrix}=0, \]
then the value of \(\dfrac{p}{x}+\dfrac{q}{y}+\dfrac{r}{z}\) is
An urn contains five balls. Two balls are drawn and found to be white. The probability that all the balls are white is
The ratio in which the join of \((2,1,5)\) and \((3,4,3)\) is divided by the plane \(x+y-z=\tfrac12\) is
Value of \(\displaystyle \int_{0}^{\pi/2}\frac{\sqrt{\sin x}}{\sqrt{\sin x}+\sqrt{\cos x}}\,dx\) is
The dot product of a vector with the vectors \(\hat i+\hat j-3\hat k\), \(\hat i+3\hat j-2\hat k\) and \(2\hat i+\hat j+4\hat k\) are \(0,5,8\) respectively. The vector is
The angle between the two lines whose intercepts on the axes are \(a,-b\) and \(b,-a\) respectively is
If the line through the points \(A(k,1,-1)\) and \(B(2k,0,2)\) is perpendicular to the line through the points \(B\) and \(C(2+2k,k,1)\), then the value of \(k\) is
View Solution
Direction vectors: \[ \vec{AB}=(k, -1, 3),\quad \vec{BC}=(2, k, -1). \]
Perpendicular \(\Rightarrow \vec{AB}\cdot\vec{BC}=0\): \[ 2k+k(-1)-3=0\Rightarrow k=-1. \] Quick Tip: Perpendicular lines have zero dot product of direction vectors.

































Comments