BITSAT 2018 Question Paper PDF is available for download. BITSAT 2018 was conducted in online CBT mode by BITS Pilani. BITSAT 2018 Question Paper had 150 questions to be attempted in 3 hours.
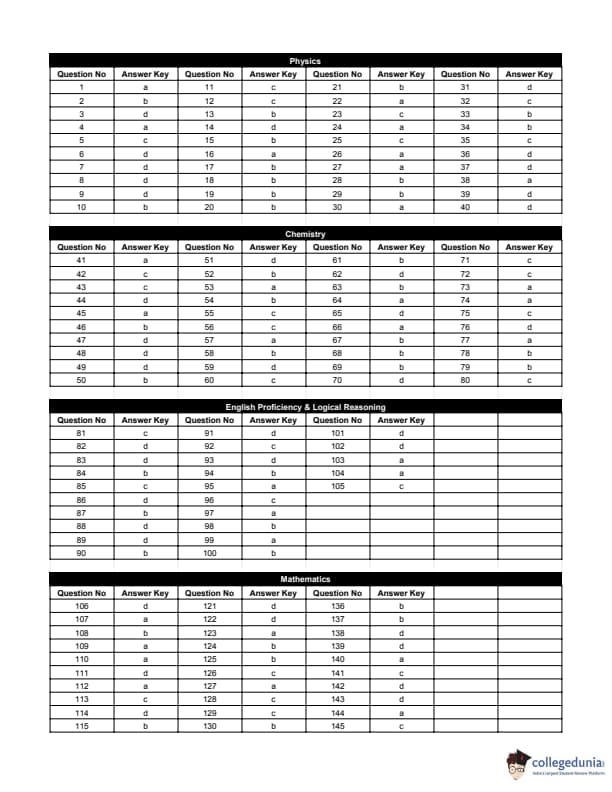
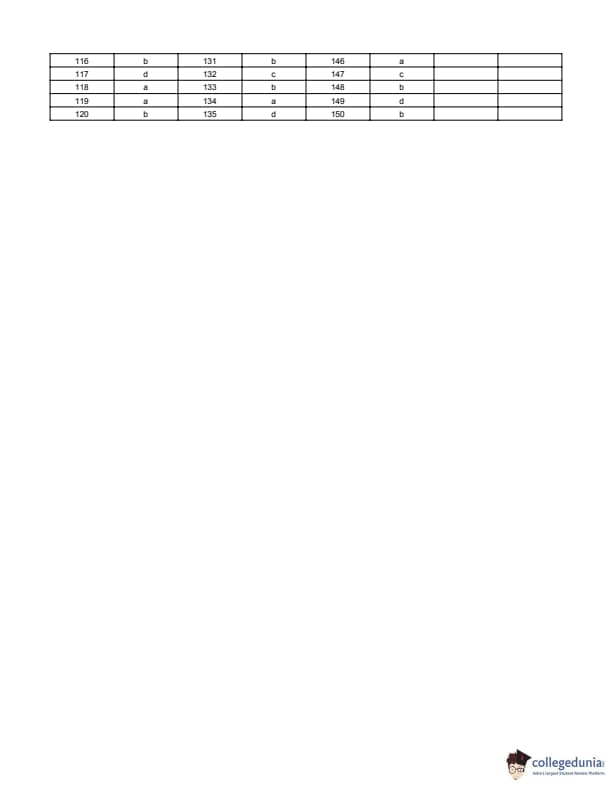
BITSAT 2018 Question Paper with Answer Key PDF
| BITSAT 2018 Question Paper PDF | BITSAT 2018 Answer Key PDF |
|---|---|
| Download PDF | Download PDF |
Four point charges \( -Q, -q, 2q \) and \( 2Q \) are placed, one at each corner of the square. The relation between \( Q \) and \( q \) for which the potential at the centre of the square is zero is:
Two long parallel wires carry equal current \( i \) flowing in the same direction and are at a distance \( 2d \) apart. The magnetic field at a point lying on the perpendicular bisector joining the wires and at a distance \( x \) from the midpoint is:
In the circuit shown, the symbols have their usual meanings. The cell has emf \( E \). \( X \) is initially joined to \( Y \) for a long time. Then, \( X \) is joined to \( Z \). The maximum charge on \( C \) at any later time will be:

A point object \( O \) is placed in front of a glass rod having spherical end of radius of curvature \( 30\,cm \). The refractive index of glass is \( \mu = 1.5 \). The image would be formed at:

In Young’s double slit experiment, \( \lambda = 500\,nm \), \( d = 1\,mm \), \( D = 1\,m \). Minimum distance from the central maximum for which intensity is half of the maximum intensity is:
What is the voltage gain in a common emitter amplifier where input resistance is \( 3\,\Omega \), load resistance \( 24\,\Omega \), and \( \beta = 0.6 \)?
The acceleration due to gravity on the surface of the moon is \( \frac{1}{6} \) that on earth and the diameter of the moon is one-fourth that of earth. The ratio of escape velocities on earth and moon will be:
Given \( \vec{P} = 2\hat{i} - 3\hat{j} + 4\hat{k} \) and \( \vec{Q} = \hat{j} - 2\hat{k} \). The magnitude of their resultant is:
A particle of mass \( m \) executes SHM with amplitude \( a \) and frequency \( \nu \). The average kinetic energy during its motion from the position of equilibrium to the end is:
The dipole moment of the given charge distribution is:

At a place, if the earth’s horizontal and vertical components of magnetic field are equal, the angle of dip will be:
The third line of Balmer series of an ion equivalent to hydrogen atom has wavelength 108.5 nm. The ground state energy of an electron of this ion will be:
The binding energy per nucleon of \( ^{10}X \) is 9 MeV and that of \( ^{11}X \) is 7.5 MeV. The minimum energy required to remove a neutron from \( ^{11}X \) is:
If \( C \) is the velocity of light, \( g \) the acceleration due to gravity and \( P \) the atmospheric pressure, then the dimensions of length will be same as that of:
Figure shows a capillary rise \( H \). If air is blown through the horizontal tube as shown, then rise in capillary tube will be:

A boy running on a horizontal road at \( 8\,km/h \) finds rain falling vertically. He increases his speed to \( 12\,km/h \) and finds the drops make \( 30^\circ \) with the vertical. The speed of rain with respect to the road is:
A hunter aims his gun directly at a monkey on a tree. At the instant the bullet leaves the barrel, the monkey drops. Pick the correct statement regarding the situation.
A particle of mass \( m_1 \) moving with velocity \( v \) collides with a mass \( m_2 \) at rest and they get embedded. Just after collision, velocity of the system:
The ratio of specific heats of a gas is \( \dfrac{C_p}{C_v} = 1.66 \). The gas may be:
Two oscillators are started simultaneously in same phase. After 50 oscillations of one, they get out of phase by \( \pi \). The half oscillation. The percentage difference of frequencies of the two oscillators is nearest to:
A juggler keeps on moving four balls in the air throwing the balls after intervals. When one ball leaves his hand (speed = \( 20\,m/s \)), the position of other balls (height in m) will be (Take \( g = 10\,m/s^2 \)):
If a stone of mass \(0.05\,kg\) is thrown out of a window of a train moving at a constant speed of \(100\,km/h\), the magnitude of the net force acting on the stone is:
A body of mass \(M\) hits normally a rigid wall with velocity \(V\) and bounces back with the same velocity. The impulse experienced by the body is:
A hoop rolls down an inclined plane. The fraction of its total kinetic energy associated with rotational motion is:
Infinite number of masses, each \(1\,kg\), are placed along the \(x\)-axis at \(x=\pm1\,m, \pm2\,m, \pm4\,m, \pm8\,m, \ldots\). The magnitude of the resultant gravitational potential at the origin is:
Water of volume \(2\,litre\) in a container is heated with a coil of \(1\,kW\) at \(27^\circC\). The lid is open and energy dissipates at a rate of \(160\,J/s\). In how much time will the temperature rise from \(27^\circC\) to \(77^\circC\)?
(Given specific heat of water \(= 4.2\,kJ/kgK\))
In the following \(P\)-\(V\) diagram of an ideal gas, two adiabates and two isotherms at \(T_1=300\,K\) and \(T_2=200\,K\) are shown. Values are \(V_A=2\), \(V_B=8\), \(V_C=16\). Find \(V_D\).

The mass of an \(H_2\) molecule is \(3.32\times10^{-27}\,kg\). If \(10^{23}\) hydrogen molecules per second strike \(2\,cm^2\) of a wall at an angle of \(45^\circ\) with the normal while moving at a speed of \(10^5\,cm/s\), the pressure exerted on the wall is nearly:
The wavelengths of two waves are \(50\,cm\) and \(51\,cm\) respectively. If the temperature of the room is \(20^\circC\), then what will be the number of beats produced per second by these waves, when the speed of sound at \(0^\circC\) is \(332\,m/s\)?
The figure shows the interference pattern obtained in a double slit experiment using light of wavelength \(600\,nm\). Fingers marked 1, 2, 3, 4 and 5 are shown. The third order bright fringe is:
Electric potential at any point is \( V = -5x + 3y + \sqrt{15}\,z \). The magnitude of the electric field is:
Seven resistances, each of value \(20\,\Omega\), are connected to a \(2\,V\) battery as shown. The ammeter reading will be:

The variation of magnetic susceptibility \( \chi \) with temperature \( T \) for a diamagnetic substance is best represented by:

A copper rod of length \( \ell \) rotates about its end with angular velocity \( \omega \) in a uniform magnetic field \( B \). The emf developed between the ends of the rod if the field is normal to the plane of rotation is:
A \(10\,V\) battery with internal resistance \(1\,\Omega\) and a \(15\,V\) battery with internal resistance \(0.6\,\Omega\) are connected in parallel to a voltmeter (see figure). The reading in the voltmeter will be close to:

Ten tuning forks are arranged in increasing order of frequency such that any two nearest tuning forks produce 4 beats/s. The highest frequency is twice of the lowest. The possible highest and lowest frequencies (in Hz) are:
A charged particle enters a uniform magnetic field with a certain velocity. The power delivered to the particle by the magnetic field depends on:
A resistor and an inductor are connected to an AC supply of \(120\,V\) and \(50\,Hz\). The current in the circuit is \(3\,A\). If the power consumed is \(108\,W\), then the resistance in the circuit is:
In an electron gun, the potential difference between the filament and plate is \(3000\,V\). What will be the velocity of electron emitting from the gun?
A radioactive substance decays with decay constant \(0.5\,s^{-1}\) and is being produced at a constant rate of 50 nuclei per second. If there are no nuclei present initially, the time (in s) after which 25 nuclei will be present is:
The \(25\,mL\) of a \(0.15\,M\) solution of lead nitrate, \( Pb(NO_3)_2 \), reacts with all the aluminium sulphate, \( Al_2(SO_4)_3 \), present in \(20\,mL\) of solution. What is the molar concentration of the solution of \( Al_2(SO_4)_3 \)? \[ 3Pb(NO_3)_2 + Al_2(SO_4)_3 \rightarrow 3PbSO_4 + 2Al(NO_3)_3 \]
100 mL \( O_2 \) and \( H_2 \) are kept at the same temperature and pressure. What is true about their number of molecules?
If Planck’s constant \( h = 6.6\times10^{-34}\,J\,s \), the de Broglie wavelength of a particle having momentum \( 3.3\times10^{-24}\,kg m s^{-1} \) will be:
Amongst the elements with following electronic configurations, which one may have the highest ionisation energy?
Which of the following is the correct and increasing order of lone pairs of electrons on the central atom?
According to molecular orbital theory which statement about the magnetic character and bond order is correct regarding \( O_2 \)?
If \(V\) is the volume of one molecule of a gas under given conditions, the van der Waals constant \(b\) is:
For vaporization of water at 1 atmospheric pressure, the values of \( \Delta H \) and \( \Delta S \) are \(40.63\,kJ mol^{-1}\) and \(108.8\,J K^{-1}mol^{-1}\), respectively. The temperature when Gibbs free energy change (\(\Delta G\)) for this transformation will be zero, is:
For the reaction taking place at certain temperature \[ NH_2COONH_4(s) \rightleftharpoons 2NH_3(g) + CO_2(g), \]
if equilibrium pressure is \(3X\) bar, then \( \Delta G^\circ \) would be:
The pH of \(0.1\,M\) solution of the following salts increases in order:
When \( N_2O_5 \) is heated at certain temperature, it dissociates as \[ N_2O_5(g) \rightleftharpoons N_2O_3(g) + O_2(g);\quad K_1=2.5 \]
At the same time \[ N_2O_3(g) \rightleftharpoons N_2O(g) + O_2(g) \]
If initially \(4.0\) moles of \( N_2O_5 \) are taken in a \(1.0\,L\) flask and equilibrium concentration of \( O_2 \) is \(2.5\,M\), equilibrium concentration of \( N_2O_5 \) is:
Consider the reactions \[ (A)\quad H_2O_2 + 2HI \rightarrow I_2 + 2H_2O \] \[ (B)\quad HOCl + H_2O_2 \rightarrow H_3O^+ + Cl^- + O_2 \]
Which statement is correct about \( H_2O_2 \) with reference to these reactions?
Following are colours shown by some alkaline earth metals in flame test. Which of the following are not correctly matched?
\begin{tabular{|c|c|
\hline
Metal & Colour
\hline
(i) Calcium & Apple green
(ii) Strontium & Crimson
(iii) Barium & Brick red
\hline
\end{tabular
Beryllium shows diagonal relationship with aluminium. Which of the following similarity is incorrect?
An element \(X\) occurs in short period having configuration \(ns^2np^1\). The formula and nature of its oxide is:
Which of the following is the strongest nucleophile?
The IUPAC name of the compound is:

Which of the following will have a meso-isomer also?
In a set of reactions, ethylbenzene yielded a product \(D\). The reaction sequence is: \[ Ethylbenzene \xrightarrow[KOH]{KMnO_4} B \xrightarrow[\ ]{Br_2/FeCl_3} C \xrightarrow[\ ]{C_2H_5OH,\,H^+} D \]
The correct structure of product \(D\) is:


Identify the incorrect statement from the following:
Each edge of a cubic unit cell is \(400\,pm\) long. If atomic mass of the element is 120 and its density is \(6.25\,g cm^{-3}\), the crystal lattice is: (Use \(N_A=6\times10^{23}\))
Chloroform, \( CHCl_3 \), boils at \(61.7^\circC\). If \(K_b\) for chloroform is \(3.63^\circC/molal\), what is the boiling point of a solution of \(15.0\,g\) of \( CHCl_3 \) and \(0.616\,kg\) of acenaphthalene (\(C_{12}H_{10}\))?
The pH of a \(0.1\,M\) monobasic acid is found to be 2. Hence, its osmotic pressure at a given temperature \(T\) is:
On passing a current of \(1.0\,A\) for 16 min and 5 s through one litre of \( CuCl_2 \) solution, all copper of the solution was deposited at cathode. The strength of the \( CuCl_2 \) solution was (Molar mass of Cu = 63.5; Faraday constant = \(96{,}500\,C mol^{-1}\)):
A \(100.0\,mL\) dilute solution of \( Ag^+ \) is electrolysed for \(15.0\) minutes with a current of \(1.25\,mA\) and the silver is removed completely. What was the initial \([Ag^+]\)?
The accompanying figure depicts a change in concentration of species \(A\) and \(B\) of the reaction \(A \rightarrow B\), as a function of time. The point of intersection of the two curves represents:

The rate constant of a reaction is \(1.5\times10^{-3}\) at \(25^\circC\) and \(2.1\times10^{-2}\) at \(60^\circC\). The activation energy is:
Freundlich equation for adsorption of gases (in amount of \(x\) g) on a solid (in amount of \(m\) g) at constant temperature can be expressed as:
Which of the following feature of catalysts is described in reactions given below? \[ \begin{aligned} (i)\;& CO(g)+2H_2(g)\xrightarrow{Cu/ZnO–Cr_2O_3}CH_3OH(g)
(ii)\;& CO(g)+H_2(g)\xrightarrow{Cu}HCHO(g)
(iii)\;& CO(g)+3H_2(g)\xrightarrow{Ni}CH_4(g)+H_2O(g) \end{aligned} \]
Which of the following is not a member of chalcogens?
Pick out the wrong statement.
Which of the following element do not form complex with EDTA?
Which one of the following cyano complexes would exhibit the lowest value of paramagnetic behaviour?
(At. Nos.: Cr = 24, Mn = 25, Fe = 26, Co = 27)
When an aqueous solution of copper(II) sulphate is saturated with ammonia, the blue compound crystallises on evaporation. The formula of this blue compound is:
Phenol reacts as shown: \[ Phenol\xrightarrow{NaOH}[X]\xrightarrow{CH_2=CHCH_2Cl}[Y] \]
Here \([Y]\) is:

Following compounds are given: \[ (1)\ CH_3CH_2OH\quad (2)\ CH_3COCH_3\quad (3)\ CH_3\!-\!CHOH\!-\!CH_3\quad (4)\ CH_3OH \]
Which of the above compound(s), on being warmed with iodine solution and NaOH, will give iodoform?
Arrange the following alcohols in increasing order of their reactivity towards reaction with HCl: \[ (1)\ CH_3CH_2OH,\quad (2)\ (CH_3)_2CHOH,\quad (3)\ (CH_3)_3COH \]
Thirty percent of the bases in a sample of DNA extracted from eukaryotic cells is adenine. What percentage of cytosine is present in this DNA?
The blue colour of snail is due to presence of:
Which of the following is a diamine?
Choose the word which is most similar in meaning to the word \emph{‘Optimistic’}.
Choose the word which is most opposite in meaning to the word \emph{‘Drowsy’}.
He is really feeling under the weather today; he has a terrible cold.
(I) feeling like the weather
(II) feeling over the weather
(III) feeling in the weather
By working part-time and looking after his old mother, he managed to get the best for both worlds.
(I) the best of both worlds
(II) the best on both worlds
(III) the best in both worlds
Hey, Nanny, speak about the devil and you are here.
(I) speak at the devil
(II) speak of the devil
(III) speak on the devil
According to the WHO Global Burden of Disease study which of the following is/are pollution linked health impacts?
(I) Infection of the lower respiratory system
(II) Chronic obstructive pulmonary disease
(III) Stroke and ischaemic heart disease
The conclusion regarding the deaths attributed to particulate matter 2.5 micrometers is considered to be caveated because
Which of the following is/are not true in the context of the passage?
As per the given passage, which of the measure(s) is/are suggested for lowering particulate matter in the atmosphere?
(I) Making cleaner fuels available
(II) Landscaping open areas
(III) Providing cooking stoves designed scientifically
If sentence (B) "The Finance Ministry's warning to potential investors in bitcoin and other cryptocurrencies has come at a time when a new, seemingly attractive investment area has opened up that few have enough information about." is to be placed after rearrangement, the correct order of sentences is:
(A) One of the main reasons for this volatility is speculation and the entry into the market of a large number of people lured by the prospect of quick and easy profits.
(B) The Finance Ministry's warning to potential investors in bitcoin and other cryptocurrencies has come at a time when a new, seemingly attractive investment area has opened up that few have enough information about.
(C) A number of investors, daunted by the high price of bitcoin, have put their money into less well-established and often spurious cryptocurrencies, only to lose it all.
(D) Investment in bitcoin and other cryptocurrencies increased tremendously in India over the past year, but most new users know close to nothing of the technology or how to verify the genuineness of a particular cryptocurrency.
(E) The price of bitcoin, the most popular of all cryptocurrencies, not only shot up by well over 1000% over the course of the last year, but also fluctuated wildly.
(F) The government's caution comes on top of three warnings issued by the Reserve Bank of India since 2013.
If sentence (C) “Clinical trials involving human subjects have long been a flashpoint between bioethicists and clinical research organisations (CROs) in India.” is the first sentence, what is the order of other sentences after rearrangement?
(A) Such over-volunteering occurs more frequently in bioequivalence studies, which test the metabolism of generics in healthy subjects.
(B) Landmark amendments to the Drugs and Cosmetics Act in 2013 led to better protection of vulnerable groups such as illiterate people, but more regulation is needed to ensure truly ethical research.
(C) Clinical trials involving human subjects have long been a flashpoint between bioethicists and clinical research organisations (CROs) in India.
(D) The big problem plaguing clinical research is an over-representation of low-income groups among trial subjects.
(E) While CROs have argued that more rules will stifle industry, the truth is that ethical science is often better science.
(F) Sometimes CROs recruit them selectively, exploiting financial need and medical ignorance; at other times people volunteer for the money.
Despite being (a)/ a good teacher, (b)/ he has no influence on his pupil. (c)/ No error (d)
Yesterday, when we were returning from the party, (a)/ our car met with an accident, (b)/ but we were fortunate to reach our home safely. (c)/ No error (d)
A group of sheep is known as:
A group of trees is known as:
In a code language, if REGAINS is coded as QDFZHKM, then the word PERIODS will be coded as:
If \(5+6=121\) and \(10+8=324\), then find the value of \(23+14\):
Which of the following cube in the answer figure cannot be made based on the unfolded cube in the question figure?

Which one of the following diagram represents the correct relationship among Professor, Male and Female?

Select the related letter/word/number from the given alternatives.
Distance : Odometer :: ? : Barometer
Find the odd word/letter/number pair from the given alternatives.
Choose the correct alternatives from the given ones that will complete the series. \[ L\ N\ O\ \_\ \ M\ L\ M\ \_\ \ O\ O\ M\ L \]
Choose the correct alternatives that will complete the series. \[ 22,\ 26,\ 53,\ 69,\ 194,\ ? \]
Select the missing number from the given responses.

Identify the figure that will complete the pattern shown.

The domain of the function \( f(x)=\sqrt{x^2-[x]^2} \), where \([x]\) denotes the greatest integer less than or equal to \(x\), is:
If \(m\sin\theta=n\sin(\theta+2\alpha)\), then \(\tan(\theta+\alpha)\) is equal to:
Number of solutions of the equation \(\sin 9\theta=\sin\theta\) in the interval \([0,2\pi]\) is:
A pole stands vertically inside a triangular park \(ABC\). If the angle of elevation of the top of the pole from each corner of the park is the same, then the foot of the pole is at the:
Let \(A,B,C\) be the angles of a plain triangle. If \(\tan\frac{A}{2}=\frac{1}{3}\) and \(\tan\frac{B}{2}=\frac{2}{3}\), then \(\tan\frac{C}{2}\) is equal to:
If the amplitude of \(z-2-3i\) is \(\pi/4\), then the locus of \(z=x+iy\) is:
The roots of the equation \(x^4-2x^3+x=380\) are:
Roots of the equation \(x^2+bx-c=0\) \((b,c>0)\) are:
In how many ways can 12 gentlemen sit around a round table so that three specified gentlemen are always together?
The number of ways in which three dice can be thrown so as to get a sum of 15 is:
The coefficient of \(x^3\) in the expansion of \((x-\frac{1}{x})^7\) is:
If \(x>0\), then \[ 1+\frac{\log x}{1!}+\frac{(\log x)^2}{2!}+\cdots = \]
If \(a,b,c\) are in G.P., then
The locus of the point of intersection of the lines \[ x=\frac{1-t^2}{1+t^2},\qquad y=\frac{2at}{1+t^2} \]
represents:
The equation of the circle which passes through the point \((4,5)\) and has its centre at \((2,2)\) is:
Eccentricity of ellipse \( \dfrac{x^2}{a^2}+\dfrac{y^2}{b^2}=1 \) if it passes through points \((9,5)\) and \((12,4)\) is:
Consider the equation of parabola \(y^2+4ax=0\) where \(a>0\). Which of the following is correct?
The value of \[ \lim_{n\to\infty}\frac{1+2+3+\cdots+n}{n^2+100} \]
is equal to:
Evaluate \[ \lim_{x\to0}\sqrt{\frac{x-\sin x}{x+\sin^2 x}} \]
The probability of getting 10 in a single throw of three fair dice is:
Number of solutions of the equation \[ \tan^{-1}(1+x)+\tan^{-1}(1-x)=\frac{\pi}{2} \]
are:
If \[ A=\frac{1}{3} \begin{bmatrix} 1 & 2 & 2
2 & 1 & -2
a & 2 & b \end{bmatrix} \]
is an orthogonal matrix, then:
The points represented by the complex numbers \(1+i,\,-2+3i,\;\frac{5}{3}i\) on the Argand plane are:
If matrix \[ A=\begin{bmatrix} 3 & -2 & 4
1 & 2 & -1
0 & 1 & 1 \end{bmatrix} \quadand\quad A^{-1}=\frac{1}{k}\,adj(A), \]
then \(k\) is:
If \(x,y,z\) are complex numbers, and \[ \Delta= \begin{vmatrix} 0 & -y & -z
\bar{y} & 0 & -x
\bar{z} & \bar{x} & 0 \end{vmatrix}, \]
then \(\Delta\) is:
If \[ f(x)= \begin{cases} \sin x, & when x is rational
\cos x, & when x is irrational \end{cases} \]
then the function is:
If \[ f(x)= \begin{cases} 1, & 0
The value of \(c\in(0,2)\) satisfying the mean value theorem for the function \(f(x)=x(x-1)^2\) on \([0,2]\) is:
If \( y=\dfrac{x}{x+1}+\dfrac{x+1}{x} \), then \( \dfrac{d^2y}{dx^2} \) at \(x=1\) is equal to:
Let \(y=e^{2x}\). Then \( \left(\dfrac{d^2y}{dx^2}\right)\left(\dfrac{d^2x}{dy^2}\right) \) is:
A ball is dropped from a platform \(19.6\) m high. Its position function is:
The value of the integral \[ \int_a^b \frac{\sqrt{x}\,dx}{\sqrt{x}+\sqrt{a+b-x}} \]
is:
Evaluate \[ \int \frac{e^{x^2}(2x+x^3)}{(3+x^2)^2}\,dx \]
If \[ \int_0^a f(2a-x)\,dx=m \quadand\quad \int_0^a f(x)\,dx=n, \]
then \[ \int_0^{2a} f(x)\,dx \]
is equal to:
The integrating factor of the differential equation \[ \sin x\,\frac{dy}{dx}+2y\cos x=1 \]
is:
The expression satisfying the differential equation \((x^2-1)\dfrac{dy}{dx}+2xy=1\) is:
Let \(\vec a=i-k,\ \vec b=xi+\!j+(1-x)k,\ \vec c=yi+xj+(1+x-y)k\). Then \([\vec a,\vec b,\vec c]\) depends on:
If \(\hat i+\hat j,\ \hat j+\hat k,\ \hat i+\hat k\) are position vectors of vertices of triangle \(ABC\) taken in order, then \(\angle A\) is equal to:
The projection of the line joining \((3,4,5)\) and \((4,6,3)\) on the line joining \((-1,2,4)\) and \((1,0,5)\) is:
Which of the following statements is correct?
If the constraints in a linear programming problem are changed then:
In a binomial distribution, the mean is \(4\) and variance is \(3\). Then its mode is:
The sum \(1+\dfrac{1+a}{2!}+\dfrac{1+a+a^2}{3!}+\cdots\) is equal to:
The Boolean expression \(\sim(p\vee q)\vee(\sim p\wedge q)\) is equivalent to:
In a frequency distribution, the mean and median are \(21\) and \(22\) respectively. Then its mode is approximately:

































Comments