BITSAT 2024 Question Paper PDF is available for download. BITSAT 2024 was conducted in online CBT mode by BITS Pilani. BITSAT 2024 Question Paper had 150 questions to be attempted in 3 hours.
BITSAT 2024 Question Paper with Answer Key PDF
| BITSAT 2024 Question Paper with Answer Key | Check Solution |
Question 1:
You measure two quantities as A = 1.0 m ± 0.2 m, B = 2.0 m ± 0.2 m. We should report the correct value for √(AB) as:
The dimensional formula of latent heat is:
The dimensions of the coefficient of self-inductance are:
A particle is moving in a straight line. The variation of position x as a function of time t is given as: x = t3 - 6t2 + 20t + 15
The velocity of the body when its acceleration becomes zero is:
The distance travelled by a particle starting from rest and moving with an acceleration 4/3 ms-2, in the third second is:
A projectile is projected with velocity of 40 m/s at an angle θ with the horizontal. If R is the horizontal range covered by the projectile and after t seconds its inclination with horizontal becomes zero, then the value of cot θ is:
[Take, g = 10 m/s2]
A rigid body rotates about a fixed axis with variable angular velocity ω = α - βt at time t, where α, β are constants. The angle through which it rotates before it stops is:
The range of a projectile projected at an angle of 15° with the horizontal is 50 m. If the projectile is projected with the same velocity at an angle of 45°, then its range will be:
A particle of mass m is projected with a velocity u making an angle of 30° with the horizontal. The magnitude of angular momentum of the projectile about the point of projection when the particle is at its maximum height h is:
A body is thrown with a velocity of 9.8 m/s making an angle of 30° with the horizontal. It will hit the ground after a time:
A light string passing over a smooth light pulley connects two blocks of masses m1 and m2 (where m2 > m1). If the acceleration of the system is g/√2, then the ratio of the masses m1/m2 is:
A block of mass 1 kg is pushed up a surface inclined to horizontal at an angle of 60° by a force of 10 N parallel to the inclined surface. When the block is pushed up by 10 m along the inclined surface, the work done against frictional force is:
[Given: g = 10 m/s2, μs = 0.1]
A person of mass 60 kg is inside a lift of mass 940 kg. The lift starts moving upwards with an acceleration of 1.0 m/s2. If g = 10 m/s2, the tension in the supporting cable is:
A force of F = 0.5 N is applied on the lower block as shown in the figure. The work done by the lower block on the upper block for a displacement of 3 m of the upper block with respect to the ground is (Take, g = 10 m/s2):
Diagram:
Two blocks are stacked vertically. The upper block has a mass of 1 kg, and the lower block has a mass of 2 kg. The coefficient of friction between the two blocks is 0.1. A force F = 0.5 N is applied horizontally to the lower block.
A pendulum of mass 1 kg and length l = 1 m is released from rest at an angle θ = 60°. The power delivered by all the forces acting on the bob at angle θ = 30° will be (Take, g = 10 m/s2):
An ideal massless spring S can be compressed 1 m by a force of 100 N in equilibrium. The same spring is placed at the bottom of a frictionless inclined plane inclined at 30° to the horizontal. A 10 kg block M is released from rest at the top of the
The moment of inertia of a cube of mass m and side a about one of its edges is equal to:
A body which is initially at rest at a height R above the surface of the Earth of radius R, falls freely towards the Earth. The velocity on reaching the surface of the Earth is:
The distance between the Sun and Earth is R. The duration of a year if the distance between the Sun and Earth becomes 3R will be:
For a particle inside a uniform spherical shell, the gravitational force on the particle is:
The kinetic energy of a satellite in its orbit around Earth is E. What should be the kinetic energy of the satellite to escape Earth's gravity?
Two wires of the same material (Young’s modulus Y) and same length L but radii R and 2R respectively, are joined end to end and a weight W is suspended from the combination. The elastic potential energy in the system is:
Diagram: Two wires are joined end-to-end. The first wire has radius R and length L. The second wire has radius 2R and length L. A weight W is suspended from the bottom.
With rise in temperature, the Young's modulus of elasticity:
Young's modules of materials of a wire of Length ' L ' and cross-sectional area A is Y. If the length of the wire is doubled and cross-sectional area is halved then Young's modules will be:
Pressure inside two soap bubbles are 1.01 and 1.02 atmosphere, respectively. The ratio of their volumes is:
A cube of ice floats partly in water and partly in kerosene oil. The radio of volume of ice immersed in water to that in kerosene oil (specific gravity of Kerosene oil = 0.8, specific gravity of ice = 0.9)
Diagram: A cube is shown floating partially submerged in two layers of liquid. The top layer is labeled "Kerosene oil," and the bottom layer is labeled "Water."
A solid metallic cube having total surface area 24 m2 is uniformly heated. If its temperature is increased by 10°C, calculate the increase in volume of the cube.
Given: α = 5.0 × 10-4 C-1
In the given cycle ABCDA, the heat required for an ideal monoatomic gas will be:
Diagram: A P-V diagram showing a cyclic process ABCDA. A is at (V0, P0). B is at (2V0, P0). C is at (2V0, 2P0), and D is back at (V0, 2P0).
A gas can be taken from A to B via two different processes ACB and ADB. When path ACB is used, 60 J of heat flows into the system and 30 J of work is done by the system. If path ADB is used, the work done by the system is 10 J. The heat flow into the system in path ADB is:
Diagram: A P-V diagram. Points A and B are marked. Two paths are shown between A and B: ACB (a curved path going up and then right) and ADB (a curved path going right and then up).
A source supplies heat to a system at the rate of 1000 W. If the system performs work at the rate of 200 W, the rate at which internal energy of the system increases is:
On Celsius scale, the temperature of a body increases by 40°C. The increase in temperature on Fahrenheit scale is:
In a mixture of gases, the average number of degrees of freedom per molecule is 6. The RMS speed of the molecule of the gas is c. Then the velocity of sound in the gas is:
The temperature of an ideal gas is increased from 200 K to 800 K. If the RMS speed of gas at 200 K is v0,
The temperature of an ideal gas is increased from 200 K to 800 K. If the RMS speed of gas at 200 K is v0, then the RMS speed of the gas at 800 K will be:
Two vessels A and B are of the same size and are at the same temperature. A contains 1 g of hydrogen and B contains 1 g of oxygen. PA and PB are the pressures of the gases in A and B respectively, then PA/PB is:
Five identical springs are used in the three configurations as shown in figure. The time periods of vertical oscillations in configurations (a), (b) and (c) are in the ratio:
Diagram: Three configurations of springs are shown.
(a) A single spring with a mass attached.
(b) Two springs in series with a mass attached to the bottom spring.
(c) Two springs in parallel with a mass attached below them.
A particle executes simple harmonic motion between x = -A and x = +A. If the time taken by the particle to go from x = 0 to A/2 is 2 s, then the time taken by the particle in going from x = A/2 to A is:
A simple pendulum doing small oscillations at a place R height above the Earth's surface has a time period of T1 = 4 s. T2 would be its time period if it is brought to a point which is at a height 2R from the Earth's surface. Choose the correct relation [R = radius of Earth]:
The speed of sound in oxygen at STP will be approximately:(Given, R = 8.3J(K)-1, γ = 1.4)
A plane progressive wave is given by y = 2 cos 2π(330t - x) m. The frequency of the wave is:
An oil drop of radius 1 μm is held stationary under a constant electric field of 3.65 × 104 N/C due to some excess electrons present on it. If the density of the oil drop is 1.26 g/cm3, then the number of excess electrons on the oil drop approximately is: [Take, g = 10 m/s2]
The potential of a large liquid drop when eight liquid drops are combined is 20 V. Then, the potential of each single drop was:
A dust particle of mass 4 × 10-12 mg is suspended in air under the influence of an electric field of 50 N/C directed vertically upwards. How many electrons were removed from the neutral dust particle? [Take, g = 10 m/s2]
The electric field at point (30, 30, 0) due to a charge of 0.008 μC placed at the origin will be: (coordinates are in cm)
If two charges q1 and q2 are separated with distance 'd' and placed in a medium of dielectric constant K. What will be the equivalent distance between charges in air for the same electrostatic force?
Electric potential at a point 'P' due to a point charge of 5 × 10-9 C is 50 V. The distance of 'P' from the point charge is: (Assume, 1/(4πε0) = 9 × 109 Nm2C-2)
View Solution
Five charges +q, +5q, -2q, +3q and -4q are situated as shown in the figure. The electric flux due to this configuration through the surface S is:
Diagram: A closed surface 'S' is drawn. Inside the surface, there are three point charges labeled: +q, -2q, and +5q. Outside the surface, there are two point charges labeled: +3q and -4q.
A parallel plate capacitor with plate area A and plate separation d = 2 m has a capacitance of 4μF. The new capacitance of the system if half of the space between them is filled with a dielectric material of dielectric constant K = 3 (as shown in the figure) will be:
Diagram: A parallel plate capacitor. The left half of the space between the plates is empty. The right half is filled with a material labeled "K=3". The distance between the plates is labeled 'd'.
In the given circuit, E1 = E2 = E3 = 2V and R1 = R2 = 4Ω, then the current flowing through the branch AB is:
Diagram:
A circuit diagram with three voltage sources (E1, E2, E3) and two resistors (R1, R2).
- E1 is connected in series with R1, forming a loop on the left side.
- E2 is connected in series with R2, forming a loop on the right side.
- E3 is connected between the two loops, forming a branch labeled AB.
The positive terminals of E1 and E2 face upwards. The positive terminal of E3 faces towards point A.
In the following circuit diagram, when the 3Ω resistor is removed, the equivalent resistance of the network:
Diagram: A Wheatstone bridge circuit. There are four resistors arranged in a diamond shape. The resistors in the top left and bottom left arms are labeled 3Ω. The resistors in the top right and bottom right arms are labeled 6Ω. A 3Ω resistor is connected across the middle of the bridge (between the midpoints of the left and right sides).
A conducting wire is stretched by applying a deforming force, so that its diameter decreases to 40% of the original value. The percentage change in its resistance will be:
A wire of resistance 160Ω is melted and drawn into a wire of one-fourth of its length. The new resistance of the wire will be:
Five cells each of emf E and internal resistance r send the same amount of current through an external resistance R whether the cells are connected in parallel or in series. Then the ratio R/r is:
The straight wire AB carries a current I. The ends of the wire subtend angles θ1 and θ2 at the point P as shown in the figure. The magnetic field at the point P is:
Diagram: A straight wire segment AB is shown. Point P is located such that perpendicular lines can be drawn from P to the line of the wire. The angles between these perpendicular lines and the lines connecting P to A and P to B are labeled θ1 and θ2 respectively. The perpendicular distance from P to the wire is labeled 'd'.
A long straight wire of radius a carries a steady current I. The current is uniformly distributed across its cross-section. The ratio of the magnetic field at a/2 and 2a from the axis of the wire is:
The electrostatic force F1 and magnetic force F2 acting on a charge q moving with velocity v can be written as:
Inside a solenoid of radius 0.5 m, the magnetic field is changing at a rate of 50 × 10-6 T/s. The acceleration of an electron placed at a distance of 0.3 m from the axis of the solenoid will be:
There are two long co-axial solenoids of the same length l. The inner and outer coils have radii r1 and r2 and the number of turns per unit length n1 and n2, respectively. The ratio of mutual inductance to the self-inductance of the inner coil is:
View Solution
A rectangular loop of length 2.5 m and width 2 m is placed at 60° to a magnetic field of 4 T. The loop is removed from the field in 10 sec. The average emf induced in the loop during this time is:
View Solution
Find the average value of the current shown graphically from t = 0 to t = 2 s.
Diagram: A graph of current (i) vs. time (t).
- From t=0 to t=1, the current increases linearly from 0 to 10 A.
- From t=1 to t=2, the current decreases linearly from 10 A to 0 A.
In an AC circuit, an inductor, a capacitor, and a resistor are connected in series with XL = R = XC. The impedance of this circuit is:
An alternating voltage V(t) = 220 sin 100πt volt is applied to a purely resistive load of 50Ω. The time taken for the current to rise from half of the peak value to the peak value is:
A parallel plate capacitor consists of two circular plates of radius R = 0.1 m. They are separated by a short distance. If the electric field between the capacitor plates changes as: dE/dt = 6 × 1013 V/(m · s) then the value of the displacement current is:
View Solution
Electromagnetic waves travel in a medium with speed 1.5 × 108 m/s. The relative permeability of the medium is 2.0. The relative permittivity will be:
Power of a biconvex lens is P diopter. When it is cut into two symmetrical halves by a plane containing the principal axis, the ratio of the power of two halves is:
- (A) 1:2
- (B) 2:1
- (C) 1:4
- (D) 1:1
The magnifying power of a telescope is 9. When adjusted for parallel rays, the distance between the objective and eyepiece is 20 cm. The ratio of the focal length of the objective lens to the focal length of the eyepiece is:
- (A) 8
- (B) 7
- (C) 9
- (D) 12
In normal adjustment, for a refracting telescope, the distance between the objective and eyepiece is 30 cm. The focal length of the objective, when the angular magnification of the telescope is 2, will be:
If the distance between an object and its two times magnified virtual image produced by a curved mirror is 15 cm, the focal length of the mirror must be:
- (A) 10/3 cm
- (B) -12 cm
- (C) -10 cm
- (D) 15 cm
Young's double slit experiment is performed in a medium of refractive index 1.33. The maximum intensity is I0. The intensity at a point on the screen where the path difference between the light coming out from slits is λ/4, is:
In YDSE, monochromatic light falls on a screen 1.80 m from two slits separated by 2.08 mm. The first and second order bright fringes are separated by 0.553 mm. The wavelength of light used is:
A microwave of wavelength 2.0 cm falls normally on a slit of width 4.0 cm. The angular spread of the central maxima of the diffraction pattern obtained on a screen 1.5 m away from the slit will be:
The property of light which cannot be explained by Huygen's construction of a wavefront is:
When a light ray incidents on the surface of a medium, the reflected ray is completely polarized. Then the angle between reflected and refracted rays is:
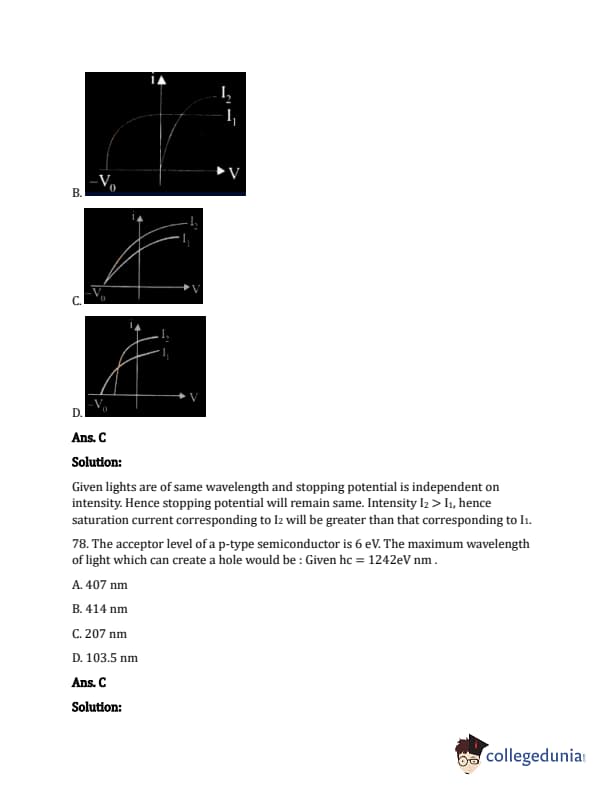
Which figure shows the correct variation of applied potential difference (V) with photoelectric current (I) at two different intensities of light (I1 < I2) of same wavelengths:
The question describes four graphs, but since I can't process images I need them converted to descriptions.
The acceptor level of a p-type semiconductor is 6 eV. The maximum wavelength of light which can create a hole would be: Given hc = 1242 eV nm.
When light is incident on a metal surface, the maximum kinetic energy of emitted electrons:
If the kinetic energy of a free electron doubles, its de-Broglie wavelength changes by the factor:
Which of the following transitions of He+ ion will give rise to a spectral line that has the same wavelength as the spectral line in a hydrogen atom?
The ratio of the shortest wavelength of the Balmer series to the shortest wavelength of the Lyman series for the hydrogen atom is:
View Solution
The minimum excitation energy of an electron revolving in the first orbit of hydrogen is:
View Solution
The atomic mass of 6C12 is 12.000000 u and that of 6C13 is 13.003354 u. The required energy to remove a neutron from 6C13, if the mass of the neutron is 1.008665 u, will be:
View Solution
The nucleus having highest binding energy per nucleon is:
View Solution
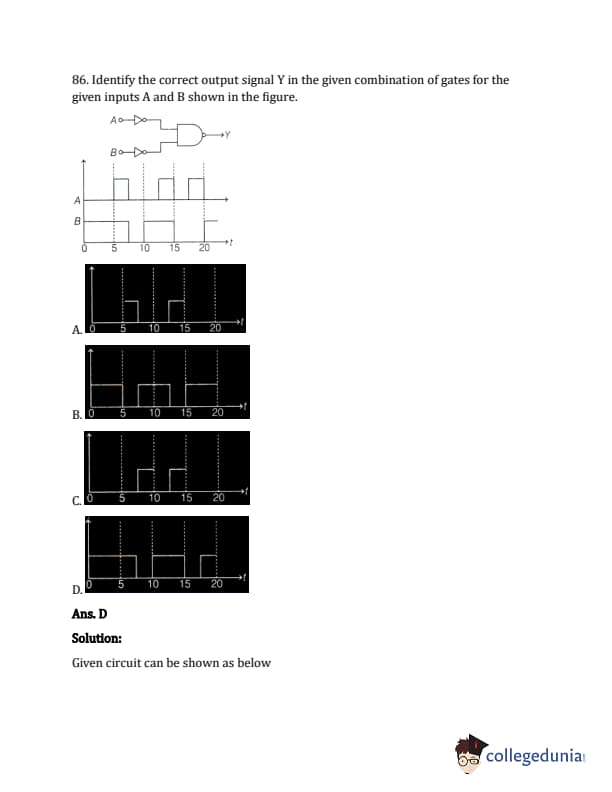
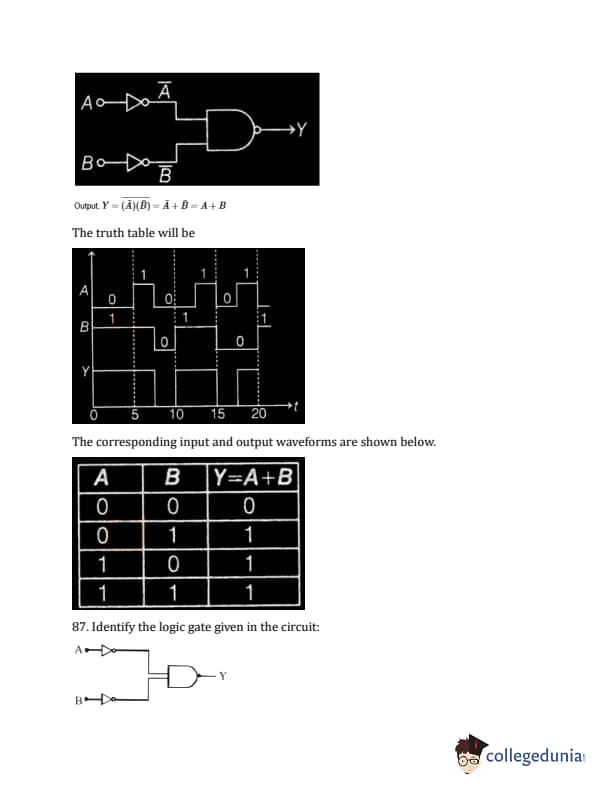
Identify the correct output signal Y in the given combination of gates for the given inputs A and B shown in the figure.
The question describes four waveforms as options, labelled (A), (B), (C), and (D) showing output Y. The circuit is a combination of logic gates with inputs A and B. From the problem description in the original document, it should have 2 NOT gates (one on A, one on B) and the output of the not gates connected to an AND gate. The correct output waveform will match that operation.
View Solution
Identify the logic gate given in the circuit:
Diagram: Two NOT gates whose outputs are connected to the inputs of a NAND gate.
View Solution
A reverse biased zener diode when operated in the breakdown region works as:
View Solution
Identify the logic operation performed by the following circuit.
Diagram: A logic circuit with two inputs, A and B. Each input goes into a NOR gate. The outputs of these two NOR gates are then fed as inputs into a third NOR gate. The output of this final NOR gate is labeled Y.
View Solution
One main scale division of a vernier caliper is equal to m units. If the mth division of main scale coincides with the (n+1)th division of vernier scale, the least count of the vernier caliper is:
A 1 L closed flask contains a mixture of 4 g of methane and 4.4 g of carbon dioxide. The pressure inside the flask at 27°C is (Assume ideal behaviour of gases):
A 1 L closed flask contains a mixture of 4 g of methane and 4.4 g of carbon dioxide. The pressure inside the flask at 27°C is (Assume ideal behaviour of gases):
In which mode of expression, the concentration of a solution remains independent of temperature?
The degeneracy of hydrogen atom that has energy equal to -RH/9 is (where RH = Rydberg constant)
If the de-Broglie wavelength of a particle of mass (m) is 100 times its velocity, then its value in terms of its mass (m) and Planck constant (h) is:
The energy of the second orbit of a hydrogen atom is -5.45 × 10-19 J. What is the energy of the first orbit of Li2+ ion (in J)?
A photon of wavelength 3000 Å strikes a metal surface. The work function of the metal is 2.13 eV. What is the kinetic energy of the emitted photoelectron? (h = 6.626 × 10-34 Js)
A stream of electrons from a heated filament was passed between two charged plates at a potential difference V volt. If e and m are the charge and mass of an electron, then the value of h/λ is:
Electron affinity is positive when:
The ionic radii in (Å) of N3-, O2- and F- are respectively.
Intramolecular hydrogen bonding is found in
View Solution
The hybridisation scheme for the central atom includes a d-orbital contribution in
View Solution
In the following species, how many species have the same magnetic moment? (i) Cr2+ (ii) Mn3+ (iii) Ni2+ (iv) Sc2+ (v) Zn2+ (vi) V3+ (vii) Ti4+
View Solution
The spin only magnetic moment of Fe3+ ion (in BM) is approximately.
View Solution
Which one of the following compounds is having maximum 'lone pair-lone pair' electron repulsions?
View Solution
Identify the species having one π-bond and maximum number of canonical forms from the following:
View Solution
sp3 d2 hybridisation is not displayed by:
What would be the amount of heat absorbed in the cyclic process shown below?
Diagram: A P-V diagram showing a cyclic process. The cycle is a circle. The center of the circle is at (15, unknown P value). The circle extends horizontally from V=5 to V=25.
The bond dissociation energy of X2, Y2 and XY are in the ratio of 1 : 0.5 : 1. ΔH for the formation of XY is -200 kJ/mol. The bond dissociation energy of X2 will be
Which of the following relation is not correct?
The standard Gibbs energy (ΔG°) for the following reaction is A(s) + B2+(aq) ⇌ A2+(aq) + B(s), Kc = 1012 at
(Kc = equilibrium constant)
View Solution
The combustion of benzene (L) gives CO2 (g) and H2O (L). Given that heat of combustion of benzene at constant volume is -3263.9 kJ/mol at 25°C, heat of combustion (in kJ/mol) of benzene at constant pressure will be: (R = 8.314J K-1 mol-1)
Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following:
Le-Chatelier's principle is not applicable to
The ratio Kp/Kc for the reaction CO(g) + 1/2 O2(g) ⇌ CO2(g) is:
The pH of 1 N aqueous solutions of HCl, CH3COOH and HCOOH follows the order:
20 mL of 0.1 M acetic acid is mixed with 50 mL of potassium acetate. Ka of acetic acid = 1.8 × 10-5 at 27°C. Calculate the concentration of potassium acetate if the pH of the mixture is 4.8.
What is the stoichiometric coefficient of SO2 in the following balanced reaction?
MnO4-(aq) + SO2(g) → Mn2+(aq) + HSO4-(aq) (in acidic solution)
View Solution
Volume of M/8 KMnO4 solution required to react completely with 25.0 cm3 of M/4 FeSO4 in acidic medium is:
View Solution
Which of the following is only a redox reaction but not a disproportionation reaction?
View Solution
Among the following, the correct statements are:
I. LiH, BeH2 and MgH2 are saline hydrides with significant covalent character
II. Saline hydrides are volatile
III. Electron - precise hydrides are Lewis bases
IV. The formula for chromium hydride is CrH
View Solution
In which of the following reactions of H2O2 acts as an oxidizing agent (either in acidic, alkaline, or neutral medium)?
Given Reactions
(i) 2Fe2+ + H2O2 →
(ii) 2MnO4- + 6H+ + 5H2O2 →
(iii) I2 + H2O2 + 2OH- →
(iv) Mn2+ + H2O2 →
View Solution
The strongest reducing agent among the following is:
View Solution
The correct order of melting points of the following salts is:
LiCl (I)
LiF (II)
LiBr (III)
Which among the following is used in detergent?
Thermal decomposition of lithium nitrate gives:
The number of geometrical isomers possible for the compound, CH3CH = CH - CH = CH2 is:
View Solution
Correct order of stability of carbanion is:
Diagram: Four structures of carbanions are provided:
(A) A phenyl ring with a negative charge on a carbon directly attached to the ring.
(B) A simple carbanion with three single bonds and a negative charge, R3C-.
(C) A carbanion with two single bonds, one double bond, and a negative charge, R2C=CR-.
(D) A cyclic structure (cyclopentadienyl anion) with five carbons in a ring, alternating single and double bonds, and a negative charge on one carbon.
View Solution
Which of the following is not correct about Grignard reagent?
View Solution
The IUPAC name of the following molecule is:
Diagram: A benzene ring with three substituents: a methyl group (CH3), a chlorine atom (Cl), and a nitro group (NO2). The relative positions need to be determined from the answer choices, as the original was an image.
Choose the correct stability order of the given free radicals.
Diagram: Four free radical structures.
I. A phenyl ring with a CH2 radical directly attached.
II. A simple primary free radical (CH3)3C•
III. A primary free radical with an NO2 group nearby, CH2=CH-CH2•
IV. A primary free radical with a CN group nearby. CH3-CH(CN)-CH2•
Which of the following is the strongest Bronsted base?
Four structures were given. I'll convert them to text based on descriptions from solutions in your original text.
Structure 1: Aniline (a benzene ring with an NH2 group).
Structure 2: A pyridine ring (a six-membered ring with one nitrogen replacing a carbon).
Structure 3: A pyrrole ring (five membered ring with NH)
Structure 4: A simple amine, like cyclohexylamine
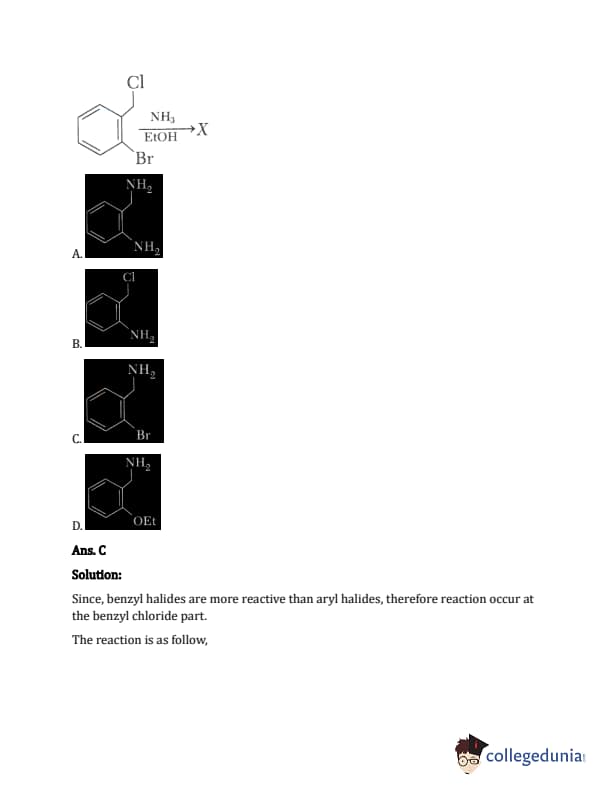
The major product X in the following given reaction is:
Diagram: o-bromobenzyl chloride reacting with NH3
The options are variations of benzylamine, with the Br group in different positions (ortho, meta, para) or with multiple substitutions or no substitution on the ring.
The major product of the reaction between CH3CH2ONa and (CH3)3CCl in ethanol is:
Dinitrogen is a robust compound, but reacts at high altitude to form oxides. The oxide of nitrogen that can damage plant leaves and retard photosynthesis is:
A decimolar solution potassium ferrocyanide is 50% dissociated at 300 K. The osmotic pressure of solution is (R = 8.314 J K-1 mol-1):
58.5 g of NaCl and 180 g of glucose were separately dissolved in 1000 mL of water. Identify the correct statement regarding the elevation of boiling point of the resulting solution.
One molar concentration of a solution represents:
Which of the following substances show the highest colligative properties?
View Solution
The pH of 0.5 L of 1.0 M NaCl solution after electrolysis for 965 s using 5.0 A current is:
View Solution
Calculate the molarity of a solution containing 5 g of NaOH dissolved in the product of H2 – O2 fuel cell operated at 1 A current for 595.1 hours.(Assume F = 96500C/mol of electron and molecular weight of NaOH as 40 g/mol).
View Solution
When the same quantity of electricity is passed through the aqueous solutions of the given electrolytes for the same amount of time, which metal will be deposited in maximum amount on the cathode?
View Solution
For the reaction 2SO2 + O2 ⇌ 2SO3, the rate of disappearance of O2 is 2 × 10-4 mol L-1 s-1. The rate of appearance of SO3 is:
View Solution
If for a first-order reaction, the value of A and Ea are 4 × 1013 s-1 and 98.6 kJ mol-1 respectively, then at what temperature will its half-life be 10 minutes?
View Solution
In the chemical reaction A → B, what is the order of the reaction? Given that, the rate of reaction doubles if the concentration of A is increased four times.
View Solution
Calculate the activation energy of a reaction, whose rate constant doubles on raising the temperature from 300 K to 600 K.
View Solution
In the reaction, A → products, if the concentration of the reactant is doubled but the rate of reaction remains unchanged, what is the order of the reaction with respect to A?
View Solution
In a first-order reaction, the concentration of the reactant decreases from 0.8 M to 0.4 M in 15 minutes. The time taken for the concentration to change from 0.1 M to 0.025 M is:
The charge on colloidal particles is due to:
The chemical composition of 'slag' formed during the smelting process in the extraction of copper is:
Calamine, malachite, magnetite, and cryolite, respectively, are:
In which of the following molecules, all bond lengths are not equal?
View Solution
The sol formed in the following unbalanced equation is:
As2O3 + H2S → ?
View Solution
Which of the following has least tendency to liberate H2 from mineral acids?
View Solution
The metal that shows highest and maximum number of oxidation states is:
View Solution
Hybridisation and geometry of [Ni(CN)4]2- are:
Match List I with List II.
| List I (Complex) | List II (Oxidation Number of Metal) |
|---|---|
| A. Ni(CO)4 | I. +1 |
| B. [Fe(H2O)5NO]2+ | II. Zero |
| C. [Co(CO)5]2- | III. -1 |
| D. [Cr2(CO)10]2- | IV. -2 |
View Solution
Which of the following is the correct order of ligand field strength?
View Solution
The correct statement among the following is:
View Solution
The type of isomerism present in nitropentammine chromium (III) chloride is:
View Solution
Identify, from the following, the diamagnetic, tetrahedral complex:
View Solution
Ferrocene is:
View Solution
The chemical name of calgon is:
View Solution
The complex with the highest magnitude of crystal field splitting energy (Δ0) is:
View Solution
IUPAC name of [Pt(NH3)2Cl(NH2CH3)]Cl is:
View Solution
Which of the following complexes will exhibit maximum attraction to an applied magnetic field?
View Solution
In an SN2 substitution reaction of the type: R - Br + Cl- → R - Cl + Br-, Which one of the following has the highest relative rate?
View Solution
The final product in the following reaction Y is:
Reaction: Benzene ring with
* (A) o-bromoaniline
* (B) m-bromoaniline
* (C) o-bromobenzylamine
* (D) p-bromoaniline
Question 168:
In the Victor-Meyer test, the color given by 1°, 2°, and 3° alcohols are respectively:
What is X in the following reaction?
CO + 2H2 &xrightarrow{X} CH3OH
An unknown alcohol is treated with "Lucas reagent" to determine whether the alcohol is primary, secondary, or tertiary. Which alcohol reacts fastest and by what mechanism?
Which of the following compounds will undergo self aldol condensation in the presence of cold dilute alkali?
An alkene X on ozonolysis gives a mixture of Propan-2-one and methanal. What is X?
Cheilosis and digestive disorders are due to deficiency of:
A tetrapeptide is made of naturally occurring alanine, serine, glycine, and valine. If the C-terminal amino acid is alanine and the N-terminal amino acid is chiral, the number of possible sequences of the tetrapeptide is:
Which one of the following is a water-soluble vitamin that is not excreted easily?
Glycosidic linkage between C1 of α-glucose and C2 of β-fructose is found in:
The naturally occurring amino acid that contains only one basic functional group in its chemical structure is:
Which of the following is not a semi-synthetic polymer?
Zinc acetate - antimony trioxide catalyst is used in the preparation of which polymer?
........ is a potent vasodilator.
If \( z, \bar{z}, -z, -\bar{z} \) forms a rectangle of area \( 2\sqrt{3} \) square units, then one such \( z \) is:
If \( z_1, z_2, \dots, z_n \) are complex numbers such that \( |z_1| = |z_2| = \dots = |z_n| = 1 \), then \( |z_1 + z_2 + \dots + z_n| \) is equal to:
If \( |z_1| = 2, |z_2| = 3, |z_3| = 4 \) and \( |2z_1 + 3z_2 + 4z_3| = 4 \), then the absolute value of \( 8z_2z_3 + 27z_3z_1 + 64z_1z_2 \) equals:
A person invites a party of 10 friends at dinner and places so that 4 are on one round table and 6 on the other round table. Total number of ways in which he can arrange the guests is:
How many different nine-digit numbers can be formed from the number 223355888 by rearranging its digits so that the odd digits occupy even positions?
If \( 22 P_{r+1} : 20 P_{r+2} = 11 : 52 \), then \( r \) is equal to:
At an election, a voter may vote for any number of candidates not exceeding the number to be elected. If 4 candidates are to be elected out of the 12 contested in the election and voter votes for at least one candidate, then the number of ways of selections is:
The number of arrangements of all digits of 12345 such that at least 3 digits will not come in its position is:
If \( a > 0, b > 0, c > 0 \) and \( a, b, c \) are distinct, then \( (a + b)(b + c)(c + a) \) is greater than:
If \( \sum_{k=1}^{n} k(k+1)(k-1) = pn^4 + qn^3 + tn^2 + sn \), where \( p, q, t, s \) are constants, then the value of \( s \) is equal to:
There are four numbers of which the first three are in GP and the last three are in AP, whose common difference is 6. If the first and the last numbers are equal, then the two other numbers are:
If \( A = 1 + r^a + r^{2a} + r^{3a} + \dots \infty \) and \( B = 1 + r^b + r^{2b} + r^{3b} + \dots \infty \), then \( \frac{a}{b} \) is equal.
The sum of the infinite series \(1 + \frac{5}{6} + \frac{12}{6^2} + \frac{22}{6^3} + \frac{35}{6^4} + \dots\) is equal to:
If \(\tan^{-1}\left(\frac{1}{1+1\cdot2}\right) + \tan^{-1}\left(\frac{1}{1+2\cdot3}\right) + \ldots + \tan^{-1}\left(\frac{1}{1+n(n+1)}\right) = \tan^{-1}(x)\), then \(x\) is equal to:
If the arithmetic mean of two distinct positive real numbers \(a\) and \(b\) (where \(a > b\)) is twice their geometric mean, then \(a : b\) is:
If \[ y = \tan^{-1} \left( \frac{1}{x^2 + x + 1} \right) + \tan^{-1} \left( \frac{1}{x^2 + 3x + 3} \right) + \tan^{-1} \left( \frac{1}{x^2 + 5x + 7} \right) + \cdots \text{ (to n terms)} \], then \(\frac{dy}{dx}\) is:
The coefficient of \(x^2\) term in the binomial expansion of \(\left(\frac{1}{3}x^{\frac{1}{2}} + x^{-\frac{1}{4}}\right)^{10}\) is:
The coefficient of \(x^n\) in the expansion of \[\frac{e^{7x} + e^x}{e^{3x}}\] is:
The coefficient of the highest power of \(x\) in the expansion of \((x + \sqrt{x^2 - 1})^8 + (x - \sqrt{x^2 - 1})^8\) is:
If the 17th and the 18th terms in the expansion of \((2 + a)^{50}\) are equal, then the coefficient of \(x^{35}\) in the expansion of \((a + x)^{-2}\) is:
Let \( A, B \) and \( C \) are the angles of a triangle and \(\tan \frac{A}{2} = 1/3\), \(\tan \frac{B}{2} = \frac{2}{3}\). Then, \(\tan \frac{C}{2}\) is equal to:
The sum of all values of \(x\) in \([0, 2\pi]\), for which \(\sin(x) + \sin(2x) + \sin(3x) + \sin(4x) = 0\) is equal to:
Number of solutions of equations \(\sin(9\theta) = \sin(\theta)\) in the interval \([0,2\pi]\) is:
The range of \((8\sin(\theta) + 6\cos(\theta))^2 + 2\) is:
The locus of the point of intersection of the lines \(x = a(1 - t^2)/(1 + t^2)\) and \(y = 2at/(1 + t^2)\) (t being a parameter) represents:
If the straight line $2x + 3y - 1 = 0$, $x + 2y - 1 = 0$ and $ax + by - 1 = 0$ form a triangle with origin as orthocentre, then $(a,b)$ is equal to:
The distance from the origin to the image of $(1,1)$ with respect to the line $x + y + 5 = 0$ is:
A(3,2,0), B(5,3,2), C(-9,6,-3) are three points forming a triangle. AD, the bisector of angle $BAC$ meets BC in D. Find the coordinates of D:
The locus of the mid-point of a chord of the circle $x^2 + y^2 = 4$ which subtends a right angle at the origin is:
If \( p \) and \( q \) be the longest and the shortest distance respectively of the point (-7,2) from any point (\(\alpha, \beta\)) on the curve whose equation is \[ x^2 + y^2 - 10x - 14y - 51 = 0 \] then the geometric mean (G.M.) of \( p \) is:
From a point A(0,3) on the circle \[ (x + 2)^2 + (y - 3)^2 = 4 \] a chord AB is drawn and extended to a point Q such that AQ = 2AB. Then the locus of Q is:
If the focus of the parabola \[ (y - k)^2 = 4(x - h) \] always lies between the lines \(x + y = 1\) and \(x + y = 3\) then:
Let \(L_1\) be the length of the common chord of the curves \[ x^2 + y^2 = 9 \quad \text{and} \quad y^2 = 8x \] and let \(L_2\) be the length of the latus rectum of \(y^2 = 8x\). Then:
The foci of the hyperbola \[ 4x^2 - 9y^2 - 1 = 0 \] are:
Given a real-valued function \( f \) such that: \[ f(x) = \begin{cases} \frac{\tan^2\{x\}}{x^2 - \lfloor x \rfloor^2}, & \text{for } x > 0
1, & \text{for } x = 0
\sqrt{\{x\} \cot\{x\}}, & \text{for } x < 0 \end{cases} \] Then:
Let \( f(x) = \sin x \), \( g(x) = \cos x \), and \( h(x) = x^2 \). Then, evaluate: \[ \lim\limits_{x \to 1} \frac{f(g(h(x))) - f(g(h(1)))}{x - 1} \]
The Boolean expression: \[ \sim (p \vee q) \vee (\sim p \wedge q) \] is equivalent to:
If \( p \): 2 is an even number, \( q \): 2 is a prime number, and \( r \): \( 2 + 2 = 2^2 \), then the symbolic statement \( p \rightarrow (q \vee r) \) means:
Consider the following statements:
\( A \): Rishi is a judge.
\( B \): Rishi is honest.
\( C \): Rishi is not arrogant.
The negation of the statement "If Rishi is a judge and he is not arrogant, then he is honest" is:
If \( p \): It is raining today, \( q \): I go to school, \( r \): I shall meet my friends, and \( s \): I shall go for a movie, then which of the following represents: \[ \text{"If it does not rain or if I do not go to school, then I shall meet my friend and go for a movie?"} \]
Let \( p, q, r \) be three logical statements. Consider the compound statements: \[ S_1: (\sim p \vee q) \vee (\sim p \vee r) \] \[ S_2: p \rightarrow (q \vee r) \] Which of the following is NOT true?
Consider the following two propositions: \[ P_1: \sim (p \rightarrow \sim q) \] \[ P_2: (p \wedge \sim q) \wedge ((\sim p) \vee q) \] If the proposition \( p \rightarrow ((\sim p) \vee q) \) is evaluated as FALSE, then:
If the variance of the data \( 2,3,5,8,12 \) is \( \sigma^2 \) and the mean deviation from the median for this data is \( M \), then \( \sigma^2 - M \) is:
The mean of \( n \) items is \( X \). If the first item is increased by 1, second by 2, and so on, the new mean is:
The variance of 20 observations is 5. If each observation is multiplied by 2, then the new variance of the resulting observation is:
If the function \( f(x) \), defined below, is continuous on the interval \([0,8]\), then: \[ f(x) = \begin{cases} x^2 + ax + b, & 0 \leq x < 2
3x + 2, & 2 \leq x \leq 4
2ax + 5b, & 4 < x \leq 8 \end{cases} \]
From the top of a cliff 50 m high, the angles of depression of the top and bottom of a tower are observed to be \(30^\circ\) and \(45^\circ\). The height of the tower is:
ABC is a triangular park with \( AB = AC = 100 \) m. A TV tower stands at the midpoint of \( BC \). The angles of elevation of the top of the tower at \( A, B, C \) are \( 45^\circ, 60^\circ, 60^\circ \) respectively. The height of the tower is:
In a statistical investigation of 1003 families of Calcutta, it was found that 63 families have neither a radio nor a TV, 794 families have a radio, and 187 have a TV. The number of families having both a radio and a TV is:
Let R be the relation "is congruent to" on the set of all triangles in a plane. Is R:
Number of subsets of set of letters of word 'MONOTONE' is:
In an examination, 62% of the candidates failed in English, 42% in Mathematics and 20% in both. The number of those who passed in both the subjects is:
If \( A = \frac{1}{3} \begin{bmatrix} 1 & 2 & 2
2 & 1 & -2
a & 2 & b \end{bmatrix} \) is an orthogonal matrix, then
If matrix \( A = \begin{bmatrix} 3 & -2 & 4
1 & 2 & -1
0 & 1 & 1 \end{bmatrix} \) and \( A^{-1} = \frac{1}{k} adj(A) \), then \( k \) is
If A and B are symmetric matrices of the same order such that \( AB + BA = X \) and \( AB - BA = Y \), then \( (XY)^T = \)
If \( A = \begin{bmatrix} 1 & 0
0 & -1 \end{bmatrix} \), \( P = \begin{bmatrix} 1 & 1
0 & 1 \end{bmatrix} \) and \( X = A P A^T \), then \( A^T X^{50} A \) is:
View Solution
If \( A \) is a square matrix of order 3, then \( | \text{Adj}(\text{Adj } A^2) | \) is:
Suppose \( p, q, r \neq 0 \) and the system of equations:
\[ (p + a)x + by + cz = 0 \] \[ ax + (q + b)y + cz = 0 \] \[ ax + by + (r + c)z = 0 \] has a non-trivial solution, then the value of \[ \frac{a}{p} + \frac{b}{q} + \frac{c}{r} \] is:
If \( x \) is a complex root of the equation
\[ \begin{vmatrix} 1 & x & x
x & 1 & x
x & x & 1 \end{vmatrix} + \begin{vmatrix} 1 - x & 1 & 1
1 & 1 - x & 1
1 & 1 & 1 - x \end{vmatrix} = 0, \] then \( x^{2007} + x^{-2007} \) is:
The system of equations:
\[ x - y + 2z = 4 \] \[ 3x + y + 4z = 6 \] \[ x + y + z = 1 \] has:
If the system of linear equations:
\[ 2x + y - z = 7 \] \[ x - 3y + 2z = 1 \] \[ x + 4y + \delta z = k \] has infinitely many solutions, then \( \delta + k \) is:
If \( \cot(\cos^{-1} x) = \sec \left( \tan^{-1} \left( \frac{a}{\sqrt{b^2 - a^2}} \right) \right) \), then:
If \( \cos \cot^{-1} \left( \frac{1}{2} \right) = \cot (\cos^{-1} x) \), then the value of \( x \) is:
Let \( [x] \) denote the greatest integer \( \leq x \). If \( f(x) = [x] \) and \( g(x) = |x| \), then the value of:
\[ f \left( g \left( \frac{8}{5} \right) \right) - g \left( f \left( \frac{-8}{5} \right) \right) \] is:
The number of real solutions of
\[ \sqrt{5 - \log_2 |x|} = 3 - \log_2 |x| \] is:
The function \[ f(x) = \frac{\cos x}{\left\lfloor \frac{2x}{\pi} \right\rfloor + \frac{1}{2}}, \] where \( x \) is not an integral multiple of \( \pi \) and \( \lfloor \cdot \rfloor \) denotes the greatest integer function, is:
The function f: R\(\rightarrow\) R is defined by \[ f(x) = \frac{x}{\sqrt{1 + x^2}} \] is:
If \( f: \mathbb{R} \to \mathbb{R} \), \( g: \mathbb{R} \to \mathbb{R} \) are defined by \( f(x) = 5x - 3 \), \( g(x) = x^2 + 3 \), then \( g \circ f^{-1}(3) \) is equal to
The domain of the real-valued function
\[ f(x) = \sqrt{\frac{2x^2 - 7x + 5}{3x^2 - 5x - 2}} \] is:
If a function \( f: \mathbb{R} \setminus \{1\} \rightarrow \mathbb{R} \setminus \{m\} \) defined by \( f(x) = \frac{x+3}{x-2} \) is a bijection, then \( 3/l + 2m = \)
Given that \( f(x) = \sin x + \cos x \) and \( g(x) = x^2 - 1 \), find the conditions under which \( g(f(x)) \) is invertible.
Let the function \( g: (-\infty, -0) \rightarrow (-\frac{\pi}{2}, \frac{\pi}{2}) \) be given by \( g(u) = 2 \tan^{-1}(e^u) - \frac{\pi}{2} \). Determine the properties of \( g \).
Let \( f \) be the function defined by:
\[ f(x) = \begin{cases} \frac{x^2 - 1}{x^2 - 2|x-1| - 1}, & \text{if } x \neq 1,
\frac{1}{2}, & \text{if } x = 1. \end{cases} \] The function is continuous at:
If \[ f(x) = \begin{cases} \frac{x^2 \log(\cos x)}{\log(1+x)}, & x \ne 0
0, & x = 0 \end{cases} \] then at \( x = 0 \), \( f(x) \) is .
If \( f(x) \) is defined as follows:
\[ f(x) = \begin{cases} 4, & \text{if } -\infty < x < -\sqrt{5},
x^2 - 1, & \text{if } -\sqrt{5} \leq x \leq \sqrt{5},
4, & \text{if } \sqrt{5} \leq x < \infty. \end{cases} \] If \( k \) is the number of points where \( f(x) \) is not differentiable, then \( k - 2 = \)
If \( x\sqrt{1 + y} + y\sqrt{1 + x} = 0 \), then find \( \frac{dy}{dx} \).
If \( y = \tan^{-1}\left( \frac{\sqrt{x} - x}{1 + x^{3/2}} \right) \), then \( y'(1) \) is equal to:
At \( x = \frac{\pi^2}{4} \), \( \frac{d}{dx} \left( \tan^{-1}(\cos\sqrt{x}) + \sec^{-1}(e^x) \right) = \)
The maximum area of a rectangle inscribed in a circle of diameter \( R \) is:
Consider the function \( f(x) = \frac{|x-1|}{x^2} \). Then \( f(x) \) is:
The maximum volume (in cu. units) of the cylinder which can be inscribed in a sphere of radius 12 units is:
If the angle made by the tangent at the point \((x_0, y_0)\) on the curve \(x = 12(t + \sin t \cos t)\), \(y = 12(1 + \sin t)^2\), with \(0 < t < \frac{\pi}{2}\), with the positive x-axis is \(\frac{\pi}{3}\), then \(y_0\) is equal to:
The altitude of a cone is 20 cm and its semi-vertical angle is \(30^\circ\). If the semi-vertical angle is increasing at the rate of \(2^\circ\) per second, then the radius of the base is increasing at the rate of:
The point of inflexion for the curve \(y = (x - a)^n\), where \(n\) is odd integer and \(n \ge 3\), is:
The population \( p(t) \) at time \( t \) of a certain mouse species satisfies the differential equation:
\[ \frac{d p(t)}{dt} = 0.5p(t) - 450. \] If \( p(0) = 850 \), then the time at which the population becomes zero is:
Evaluate the integral: \[ \int \frac{x^3 - 1}{x^3 + x} dx \]
Evaluate the integral: \[ \int \sqrt{x + \sqrt{x^2 + 2}} \, dx. \]
The value of \( \int \(e^\tan \theta\) (\sec \theta - \sin \theta) d\theta \) is:
The value of \( \int_0^\infty \frac{dx}{(x^2 + a^2)(x^2 + b^2)} \) is:
The value of definite integral \( \int_0^{\pi/2} \log(\tan x) dx \) is:
Evaluate the integral: \[ \int_{5}^{9} \frac{\log 3x^2}{\log 3x^2 + \log (588 - 84x + 3x^2)} dx \]
Evaluate the integral: \[ \int \frac{x^2 (x \sec^2 x + \tan x)}{(x \tan x + 1)^2} dx \]
Evaluate the following limit: \[ \lim_{n \to \infty} \prod_{r=3}^n \frac{r^3 - 8}{r^3 + 8}. \]
The value of \( \int_0^{\frac{\pi}{2}} \frac{\sin\left( \frac{\pi}{4} + x \right) + \sin\left( \frac{3\pi}{4} + x \right)}{\cos x + \sin x} dx \) is:
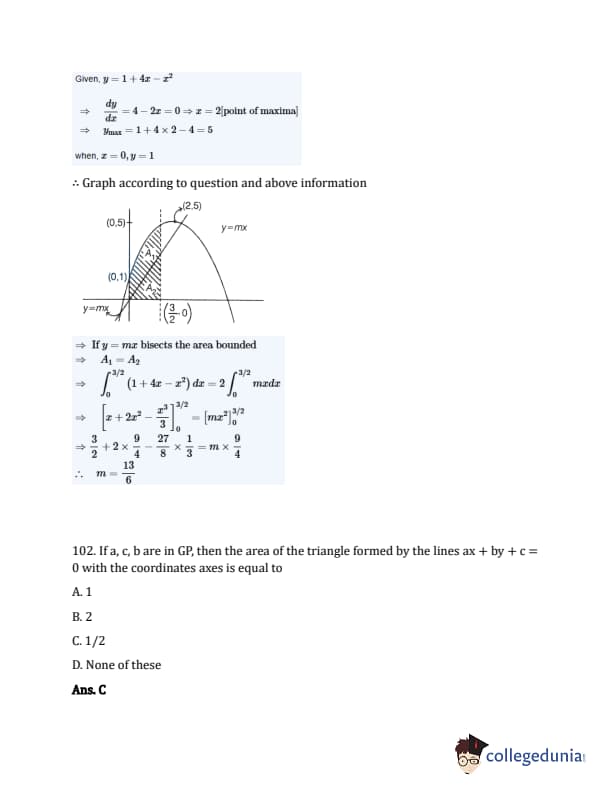
The line \(y = mx\) bisects the area enclosed by lines \(x = 0\), \(y = 0\), and \(x = \frac{3}{2}\) and the curve \(y = 1 + 4x - x^2\). Then, the value of \(m\) is:
If \( a, c, b \) are in GP, then the area of the triangle formed by the lines \( ax + by + c = 0 \) with the coordinate axes is equal to:
The area enclosed by the curves \( y = x^3 \) and \( y = \sqrt{x} \) is:
The area of the region bounded by the curves \( x = y^2 - 2 \) and \( x = y \) is:
If the area bounded by the curves \( y = ax^2 \) and \( x = ay^2 \) (where \( a > 0 \)) is 3 sq. units, then the value of \( a \) is:
The solution of the differential equation \( (x + 1)\frac{dy}{dx} - y = e^{3x}(x + 1)^2 \) is:
If \( \frac{dy}{dx} - y \log_e 2 = \(2^\sin x\) (\cos x - 1) \log_e 2 \), then \( y \) is:
Let \( \mathbf{a} = \hat{i} - \hat{k}, \mathbf{b} = x\hat{i} + \hat{j} + (1 - x)\hat{k} \), and \( \mathbf{c} = y\hat{i} + x\hat{j} + (1 + x - y)\hat{k} \). Then, \( [\mathbf{a} \, \mathbf{b} \, \mathbf{c}] \) depends on:
Let \( ABC \) be a triangle and \( \vec{a}, \vec{b}, \vec{c} \) be the position vectors of \( A, B, C \) respectively. Let \( D \) divide \( BC \) in the ratio \( 3:1 \) internally and \( E \) divide \( AD \) in the ratio \( 4:1 \) internally. Let \( BE \) meet \( AC \) in \( F \). If \( E \) divides \( BF \) in the ratio \( 3:2 \) internally then the position vector of \( F \) is:
If \( \vec{a} = 2\hat{i} + \hat{j} + 2\hat{k} \), then the value of \( |\hat{i} \times (\vec{a} \times \hat{i})|^2 + |\hat{j} \times (\vec{a} \times \hat{j})|^2 + |\hat{k} \times (\vec{a} \times \hat{k})|^2 \) is equal to:
The magnitude of projection of the line joining \( (3,4,5) \) and \( (4,6,3) \) on the line joining \( (-1,2,4) \) and \( (1,0,5) \) is:
The angle between the lines whose direction cosines are given by the equations \( 3l + m + 5n = 0 \) and \( 6nm - 2nl + 5lm = 0 \) is:
Let the acute angle bisector of the two planes \( x - 2y - 2z + 1 = 0 \) and \( 2x - 3y - 6z + 1 = 0 \) be the plane \( P \). Then which of the following points lies on \( P \)?
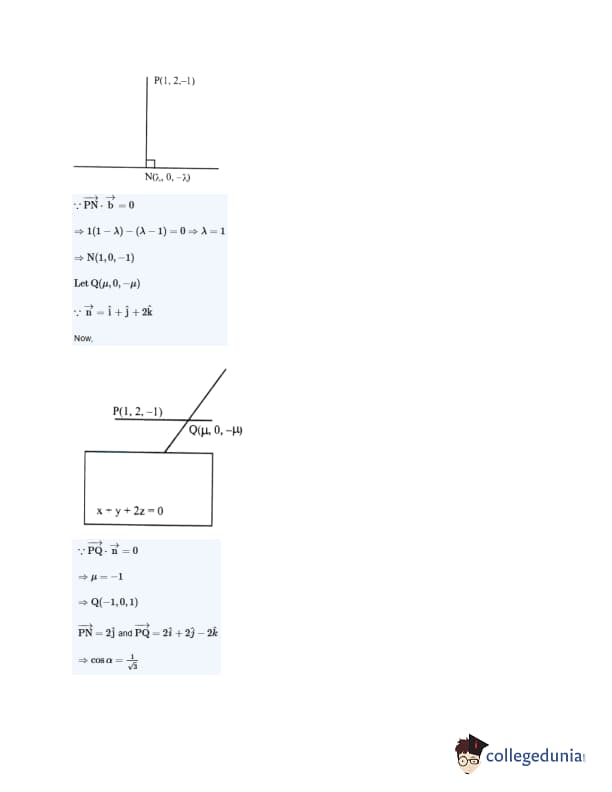
Let the foot of perpendicular from a point \( P(1,2,-1) \) to the straight line \( L : \frac{x}{1} = \frac{y}{0} = \frac{z}{-1} \) be \( N \). Let a line be drawn from \( P \) parallel to the plane \( x + y + 2z = 0 \) which meets \( L \) at point \( Q \). If \( \alpha \) is the acute angle between the lines \( PN \) and \( PQ \), then \( \cos \alpha \) is equal to:
If the number of available constraints is 3 and the number of parameters to be optimised is 4, then
The probability of getting 10 in a single throw of three fair dice is:
In a binomial distribution, the mean is 4 and variance is 3. Then, its mode is:
The probability that certain electronic component fails when first used is 0.10. If it does not fail immediately, the probability that it lasts for one year is 0.99. The probability that a new component will last for one year is
BITSAT 2025 Question Paper Analysis
Harshal Sir discusses that BITSAT exam was balanced with easy to moderate questions. The overall paper was similar to JEE level but with some exceptions.
- Physics was moderate, with basic topics like Electrostatics, Current Electricity, and Mechanics; less Optics questions compared to usual.
- Chemistry: The most challenging part due to questions from removed syllabus portions and more profound concepts.
- Mathematics: Mostly easy with some challenging Linear Programming questions; Calculus dominated, less coordinate geometry.














































































































































































































.png?h=35&w=35&mode=stretch)



Comments