JNTU Hyderabad conducted the TS EAMCET 2025 exam on behalf of the Telangana Council of Higher Education. The TS EAMCET 2025 Agriculture and Pharmacy (BiPC) exam was held on 29 April 2025 in shift 1 from 9:00 AM to 12:00 PM.
The TS EAMCET 2025 Agriculture and Pharmacy exam is being held in online CBT mode with a total of 160 questions divided between the three subjects. Physics (40 questions), Chemistry (40 questions) and Biology (Zoology-40, Botany-40) to be completed in 180 minutes (3 hours) for a total of 160 marks. As per the TS EAMCET 2025 marking scheme, +1 marks is given for every correct answer with no negative marking for incorrect answers.
The TS EAMCET 2025 April 29 Shift 1 Question Paper PDF with Solution Pdf is available to download here.
Related Links:
TS EAMCET 2025 Agriculture and Pharmacy April 29 Shift 1 Question Paper with Solution PDF
| TS EAMCET 2025 April 29 Shift 1 Question Paper with Answer Key | Download PDF | Check Solution |

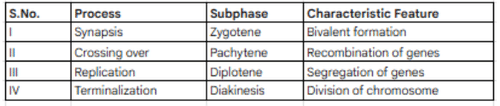
Study the following table. The correct match is.
Consider the following statements
Assertion (A): Achyranthes shows spike inflorescence
Reason (R): Spike shows basipetal arrangement of sessile flowers
Example for "runners"
I. Underground stem in Strawberry
II. Sub-aerial stem in Oxalis
III. Lateral branches of Chrysanthemum
IV. Lateral branches of Jasmine
Unilocular (one chambered) ovary becomes bilocular (two chambered) due to the formation of false septum is seen in
Anatomical characters applicable to hydrophytes and xerophytes respectively are
Which of the following is prevented by unisexual flowers that is present on different plants?
Assertion (A): In Liliaceae, flowers are homochlamydeous
Reason (R): In Liliaceae perianth is differentiated
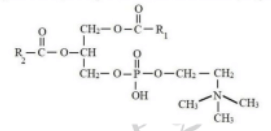
Identify the below structural formula along with its related function
Select the correct pair
Identify the characters of ovules related to Bean family.
I. Body of ovule bends
II. Ovule is placed at right angles to the funiculus
III. Embryo sac is slightly curved
IV. Micropyle comes towards the funiculus
Identify the wrong pair

Agaricus basidiocarp is having 60 basidia. Find out number of nuclear fusions, meiotic divisions and basidiospores formed from that basidiocarp
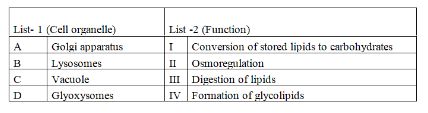
Identify the correct statements with regard to Golgi apparatus
I. Convex side is called cis or forming face
II. Concave side is called trans or maturing face
III. Cisternae of Golgi can modify proteins
IV. Materials enter through trans face and are released through cis face
Identify the true statements given below
I. Gymnosperms have albuminous cells and sieve cells
II. Phloem fibres are made up of collenchyma cells
III. Phloem parenchyma is absent in most of the dicots
IV. Most of the Gymnosperms lack vessels in their xylem
Life cycle in Rockweed of Brown Algae is
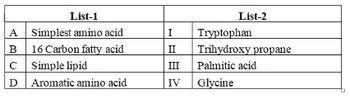
Match the following
Which of the following is quick referral systems in taxonomical studies?
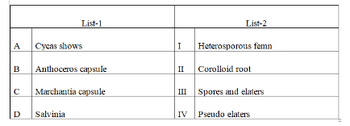
Match the following
Function not related to Peroxysomes
Match the following
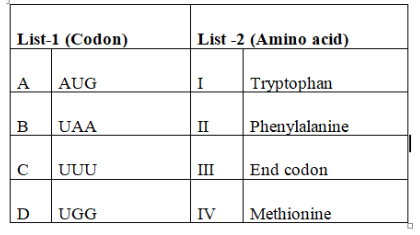
Match the following
In Pea plant, Mendel studied seven pairs of contrasting characters. The ratio of pairs of characters that are related to flower, pod and seed respectively:
Find out mismatched pair from the following:
Final acceptor of electrons in light reaction:
The length of DNA is 510 Å. It has 20% of 6-aminopurines. Find out the total number of nucleotides and total hydrogen bonds in that DNA:
Transduction was discovered by 'X' in 'Y'. Identify 'X' and 'Y' respectively:
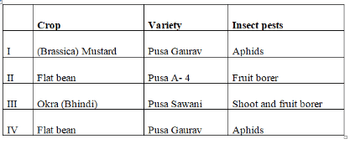
Crop varieties bred by hybridization, selection for disease resistance to fungi, bacteria and viral disease are given below. Identify mismatch:

Identify the incorrect match:
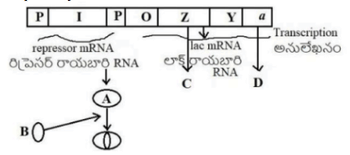
The figure given below shows lac operon and its function. Identify A, B, C and D respectively:
"A" cell is placed in hypertonic solution. After some time, its osmotic potential is measured as -0.5 MPa. Then its water potential would be:
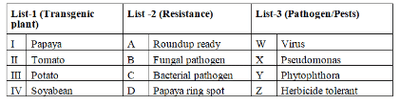
Study the List-1, List-2 and List-3 and identify correct pair:
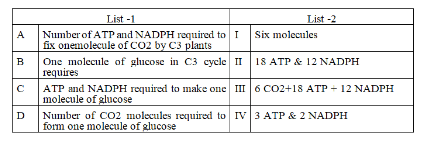
Identify the correct matching pair to Calvin cycle:
The combination of frequency between the genes a and c is 5%; b and c is 15%; b and d is 9%; a and b is 20%; c and d is 24%; and a and d is 29%. Identify the sequence of genes on a linear chromosome:
Which seeds cannot germinate and establish without the presence of mycorrhizae?
Tetracycline resistance gene of PBR322 shows recognition site for which restriction enzyme?
Consider the following statements:
Assertion (A): Seed dormancy may be caused by hard seed coats in Fabaceae
Reason (R): Such type of seed dormancy can be broken by stratification
Chromosome maps / Genetic maps were first prepared by:
Identify the incorrect pair:
I. Zinc - ABA Synthesis
II. Boron - Cell elongation
III. Nickel - Urease activator
IV. Molybdenum - Carbohydrate translocation
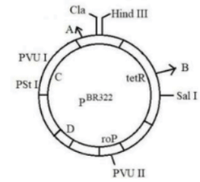
Identify A, B, C and D in the diagram of E.coli cloning vector of pBR322:
Observe the chemical reaction and identify the enzyme and its prosthetic group:

The study of fossils of animals is
Pick up the sacred groves from the following I. Aravali hills II. Seshachalam hills III. Papi kondalu IV. Eturunagaram V. Chanda
Consider the following statements.
Assertion (A): Bilaterally symmetrical animals are more efficient than other animals in seeking food, locating the mate, escape from enemies etc.
Reason (R): Cephalisation. The correct answer is
Vagina is lined by
Consider the following statements Statement I: Bones are homeostatic reservoirs of calcium, magnesium and phosphorus Statement II: Bones are highly vascular. The correct answer is
Study the following and choose the correct combinations:

Tape worms belong to
These animals are commonly called scavengers of the sea
Match the following
Number of flagellae in Trichomonas
Choose the hyperparasite
Consider the following statements Statement I: In the crop of female Anopheles, microgametes of Plasmodium are separated from the microgametocyte by ex-flagellation. Statement II: Schuffner's dots are believed to be the antigens released by Plasmodium. The correct answer is
Match the following

Heroin is obtained from
These structures hold the food and bring it to the mandibles in cockroach
Consider the following statements Assertion (A): Respiratory system of cockroach is polypneustic type. Reason (R): All spiracles are functional. The correct answer is
This part of ommatidium of cockroach is a refractive region
Consider the following statements Statement I: Cyclical seasonal morphological variations in certain organisms is called cyclomorphosis. Statement II: Allen's rule states that with the increase of every \(10^{\circ}C\) the rate of metabolic activities double in organisms. The correct answer is
Ranatra and Notonecta in an ecosystem are described as
Choose the greenhouse gas
Study the following and identify the correct statements:
I. Aminopeptidases are secreted by the pancreas
II. Enamel on teeth is secreted by ameloblasts
III. Dental formula of human milk dentition is 2102/2102
IV. Liver is the largest endocrine gland in the body of a man
Volume of air remained in lungs even after forcible expiration:
Assertion (A): In amphibians and reptiles the blood circulation is described as incomplete double circulation.
Reason (R): They have a single ventricle in the heart, where oxygenated and deoxygenated blood are mixed and then circulated to the body parts as mixed blood.
Match the following:
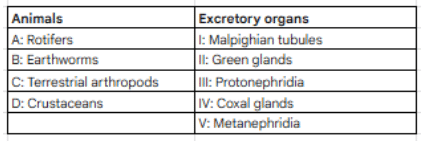
This bone of skull is modified into incus in the middle ear
Observe the following and find out the related parts of brain:
I. Foramen ovale
II. Foramen Manro
III. Foramen magnum
IV. Ductus Sylvius
V. Ductus arteriosus
VI. Metacoel
Assertion (A): Thymosins secreted by thymus gland play significant role in strengthening of immune system.
Reason (R): They play a major role in differentiation of T-lymphocytes.
Match the following:
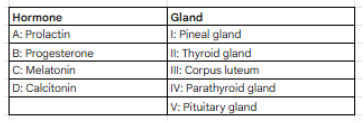
In the connective tissue, monocytes become
Study the following and choose the correct combinations:

Choose the mismatched pair:
Assertion (A): In hymenopteran insects, sperms are produced by mitosis.
Reason (R): Male hymenopteran insects are haploid.
If one parent has A blood group (homozygous) and the other has B blood group (homozygous), these blood groups are not expected in their children
Study the following and identify the correct statements:
I. The alleles I\textsubscript{A} and I\textsubscript{B} regarding blood groups are dominant over I\textsubscript{O}, and I\textsubscript{A} and I\textsubscript{B} are co-dominant.
II. In some fishes, reptiles and birds, females are heterogametic (ZW) and males are homogametic (ZZ).
III. Genic balance theory states that Y-chromosome is essential for determination of sex in male Drosophila.
IV. Person with O blood group are called universal donors because their RBC contain both antigens A and B.
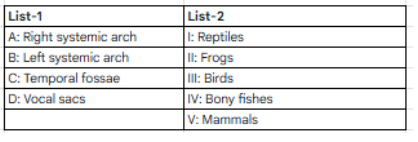
Intermediate form between reptiles and birds
Study the following and choose the correct statements:
I. Darwinism explains the survival of the fittest but not the arrival of the fittest.
II. Mutations are subjected to natural selection.
III. Hardy Weinberg equilibrium is applicable to small populations.
IV. Industrial melanism was best studied in Drosophila melanogaster.
It was more man like
The practice of mating of animals within the same breed, but have no common ancestors on either side of the pedigree for 4-6 generations is
Number of amino acids in polypeptide chain A and polypeptide chain B of insulin
Tall T-wave in ECG indicates
In the \(\beta^{+}\) decay, the particle emitted along with neutron and positron is
If the relative error in the determination of area of a body is 0.08, then the percentage error in the determination of its volume is
A motor cyclist is travelling towards north with a uniform speed of 10 ms\textsuperscript{−1} and a train is travelling towards north-west with a uniform speed of 102 ms\textsuperscript{−1}. The direction of motion of the motor cyclist as observed by a passenger in the train is
A cannon on a cliff 55 m above the ground fires a shell with a velocity of \(50\hat{i}+50\hat{j}\) ms\textsuperscript{−1. The displacement vector of the shell when it hits the ground is (Acceleration due to gravity = 10 ms\textsuperscript{−2)
A ball of mass 250 g moving with a speed of 72 kmph is deflected by a batsman by an angle of 120\(^\circ\) without changing its initial speed. The impulse imparted to the ball is:
A bullet of mass \(m\) moving with a velocity \(u\) gets embedded in a block of mass \(M\) initially at rest. If \(\mu\) is the coefficient of friction between the block and the ground, then the displacement of the block due to the impact is:
The blades of a windmill generating electrical energy sweep out an area of 20 m\(^2\). If the efficiency of the windmill is 25% and wind speed is 36 kmph, then the electrical power generated is (Density of air = 1.2 kg m\(^{-3}\)):
A uniform metre scale of mass 700 g is suspended horizontally using two strings tied at 20 cm and 70 cm marks on the scale. The tension in the string at 70 cm mark is (Acceleration due to gravity = 10 m/s\(^2\)):
A uniform circular disc of mass \(\dfrac{\pi}{40}\) kg is rotating about an axis passing through its center and perpendicular to its plane with an angular speed of 150 rev/min. If the angular momentum of the disc is 6.25 Js, then its radius is:
The displacement of a body executing simple harmonic motion is \(y = 8\cos(\pi t)\) cm. The displacement of the body in the time interval between 1.5 s and 2.5 s is:
The weight of a body at a height of 3\(R_E\) from the surface of the earth is 90 N, where \(R_E\) is the radius of the earth. The weight of the same body at a height of \(R_E\) from the surface of the earth is:
When a metal wire of area of cross-section \(1.5\times10^{-6}\) m\(^2\) is subjected to a tension of 45 N, the decrease in its area of cross-section is \(3\times10^{-10}\) m\(^2\). If the Poisson’s ratio of the material of the wire is 0.4, the Young’s modulus of the material of the wire is:
A tank is filled with water to a height of 80 cm. The speed of efflux of water through a hole on the side wall near its bottom is (g = 10 m/s\(^2\)):
The energy required to increase the radius of a soap bubble from 3 cm to 4 cm is (Surface tension = \(3\times10^{-2}\) N/m):
A metal plate of mass 50 g is heated to a temperature of \(200^\circ\mathrm{C}\) and then immersed in 150 g of water at \(20^\circ\mathrm{C}\). If the temperature of the water is \(50^\circ\mathrm{C}\) at equilibrium, then the specific heat capacity of the metal is (specific heat capacity of water \(=4200\ \mathrm{J\,kg^{-1}K^{-1}}\)):
A pendulum clock gains 6 s per day when the temperature is \(296\ \mathrm{K}\) and gains 9 s per day when the temperature is \(291\ \mathrm{K}\). The temperature at which the pendulum clock loses 12 s per day is:
The efficiency of a Carnot heat engine is 25%. If the absolute temperature of the sink is increased by 10%, then the efficiency of the engine becomes:
The specific heat capacity of one mole of water is (R is the universal gas constant):
In a medium, a source produces 60 crests and 60 troughs in a time of 0.2 s. If the distance between a crest and its adjacent trough is 100 cm, then the speed of sound in the medium is:
An observer moves towards a source at rest with a speed of 25% of the speed of sound in air. If the frequency of the sound emitted by the source is 200 Hz, then the frequency of sound heard by the observer is:
A convex lens produces clear images when placed at two positions between an object and a screen that are 1 m apart. If the distance between the two positions of the lens at which clear images are formed is 20 cm, the focal length of the lens is:
The distance between the objective and eyepiece of an astronomical telescope when the final image forms at infinity is 62 cm. If the magnification of the telescope is 30, the focal lengths of the objective and eyepiece respectively are:
In an experiment, the angular width of interference fringes for a light of wavelength 5896 Å is found to be \(3.5\times10^{-3}\) radian. The wavelength of light for which the angular width of the fringes becomes 10% greater is:
A proton and an alpha particle enter a uniform electric field perpendicular to the direction of the field. The ratio of distances travelled by the proton and the alpha particle in the direction of the field after a time of \(t\) seconds is:
If a charge of 3 nC is placed at each vertex of a cube of side 3 m, then the electric potential at the centre of the cube is:
If the magnitude of electric field in a conductor is \(200\ NC^{-1}\) and the current density is \(10^9\ Am^{-2}\), then the resistivity of the material of the conductor is:
If a 20 W bulb and a 100 W fan are used for 5 and 15 hours a day respectively, then the electrical energy consumed in a period of 30 days is:
Two charged particles enter a uniform magnetic field normally. If the ratio of the specific charges of the two particles is 2:3, then the ratio of the times taken by the two particles to complete one revolution is:
The magnetic field (in \(10^{-4}\) T) at the centre of a toroid of mean radius 10 cm with 200 turns and carrying a current of 2.5 A is:
The properties required for a material to be used as the core of an electromagnet are:
A train is moving with a speed of 198 kmph. If the distance between the rails is 120 cm and the vertical component of earth's magnetic field is \(0.25 \times 10^{-4}\) T, the induced emf between the ends of the axle of the train is:
A resistor of resistance 160 \(\Omega\), an inductor of 280 mH, and a capacitor are connected in series to an AC source of 80 V, 50 Hz. If the circuit is in resonance, the potential difference across the capacitor is:
The potential difference between the plates of a parallel plate capacitor is changing at the rate of \(3.5\times10^6\) V/s. If the displacement current is 7 A, the capacitance of the capacitor is:
If the de Broglie wavelength of a proton accelerated through a potential difference \(V_1\) is same as the de Broglie wavelength of an alpha particle accelerated through \(V_2\), then \(V_1 : V_2 = \)
The ratio of frequencies of second line of Lyman series and third line of Balmer series of hydrogen atom is:
If the half-life of a radioactive material is 5 days, then the ratio of the number of atoms disintegrated in 10 and 15 days is:
If the energy released in the fission of one uranium nucleus is 200 MeV, then the number of fissions per second required to produce 20 MW is:
In a common emitter amplifier, the collector resistance is 3 times the base resistance. If the common emitter current amplification factor is 60, then the power gain of the amplifier is:
The intrinsic carrier concentration of a semiconductor is \(1.2\times10^{16}\) m\(^{-3}\). On doping with an impurity, the electron concentration becomes \(20\times10^5\) times initial concentration. The concentration of holes in the doped semiconductor is:
Optical communication using fibres is performed in the frequency range of:
According to Bohr's theory of hydrogen atom, the approximate angular momentum of electron in H atom in the ground state is (\(h = 6.62 \times 10^{-34}\) Js):
Observe the elements from H (Z=1) to Ca (Z=20). How many elements have no unpaired electrons in their ground state?
Among Li, Na, O, S, the elements having the most negative electron gain enthalpy and least negative electron gain enthalpy respectively are:
Ionization enthalpies of the elements X, Y, Z having successive atomic numbers respectively are 2080, 496, 737 kJ mol\(^{-1}\). X, Y, Z are:
Which of the following statements are correct?
I. The increasing order of covalent character of NaCl, RbCl, MgCl\(_2\) is RbCl \(<\) NaCl \(<\)\ MgCl\(_2\)
II. The high boiling point of water is due to intramolecular hydrogen bonding
III. H\(_2\)O has more dipole moment among NH\(_3\), H\(_2\)O, and HF
Identify the pairs in which both molecules do not possess same type of hybridization:
I. H\(_2\)O, SO\(_2\)
II. PCl\(_3\), NH\(_3\)
III. SO\(_2\), SO\(_3\)
IV. BCl\(_3\), NCl\(_3\)
Isotherms (\(p\)-\(V\) lines) of one mole of an ideal gas at \(T_1\) and \(T_2\) have slope ratio 1:2. If \(T_1=1000\) K, find \(T_2\).
At 27\(^\circ\)C, 100 mL of 0.05 M Cu\(^{2+}\) solution is added to 1 L of 0.1 M KI. Find [KI] in resultant solution.
Identify the correct statements:
I. Energy of an isolated system is constant
II. In a closed system, exchange of both energy and matter is possible
III. Enthalpy is extensive
Given \(K_p\) for \(2AO_2 + O_2 \rightleftharpoons 2AO_3\) is \(4 \times 10^{10}\). Find \(K_p'\) for \(3AO_3 \rightleftharpoons 3AO_2 + \frac{3}{2} O_2\).
Which of the following is used as stabilizer for H\(_2\)O\(_2\)?
Identify correctly matched sets:
I. NaHCO\(_3\) - fire extinguisher
II. Na\(_2\)CO\(_3\) - water softening
III. NaOH - purification of bauxite
Among alkaline earth metals, metal with least melting point is:
The reaction/s which represent the production of diborane on an industrial scale is/are:
I. 4BF\(_3\) + 3LiAlH\(_4\) → 2B\(_2\)H\(_6\) + 3LiF + 3AlF\(_3\)
II. 2NaBH\(_4\) + I\(_2\) → B\(_2\)H\(_6\) + 2NaI + H\(_2\)
III. 2BF\(_3\) + 6NaH → B\(_2\)H\(_6\) + 6NaF
Correct statements regarding halides of group 14 elements:
I. Stability of dihalides: SiX\(_2\) \(>\) GeX\(_2\) \(>\) SnX\(_2\) \(>\) PbX\(_2\)
II. SnF\(_4\), PbF\(_4\) are ionic
III. PbI\(_4\) does not exist
Increase in concentration of 'X' in blood of pregnant women who smoke may induce premature delivery. What is 'X'?
IUPAC names of benzene derivatives are respectively:
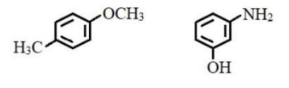
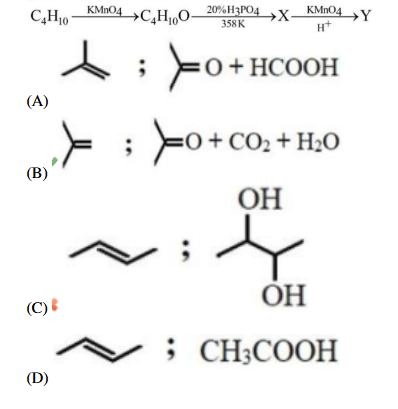
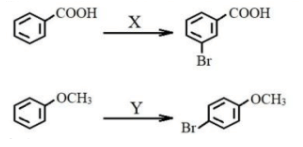
X and Y in reaction sequence:
X and Y in reactions:
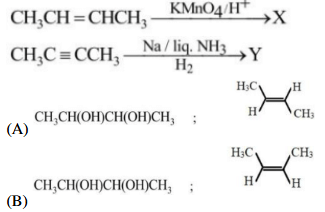
An alcohol 'X' (C\(_4\)H\(_{10}\)O) reacts with conc. HCl at room temperature and gives chloride. 'X' on dehydration followed by ozonolysis gives 'Y' and 'Z'. Find 'Y' and 'Z'.
Identify the crystal system in which body-centered lattice is not present.
Mole fraction of H\(_2\)SO\(_4\) in aqueous solution is 0.9. Find mass % of H\(_2\)SO\(_4\). (H=1, S=32, O=16 u)
At 298 K, equilibrium: M(s)+2Ag\(^+\)(aq)→M\(^{2+}\)(aq)+2Ag(s), K=10\(^{15}\), ΔS=10 J/K. Find ΔrH (kJ/mol). FRT×2.303=0.06, F=96500 C/mol.
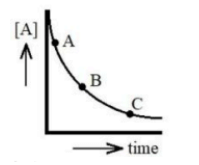
First order reaction (A→P) with slopes mA, mB, mC at points A, B, C. Identify correct order.
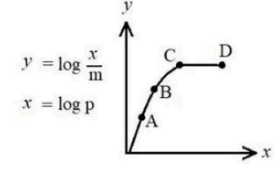
Freundlich adsorption isotherm: log(mx) vs log(p) increases then flattens (points C-D). Experimental condition represented by C-D?
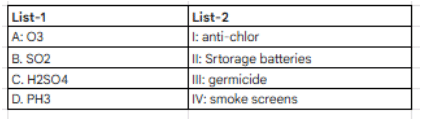
In which of the following, metal is not correctly matched with its refining process?
Match the following compounds with their uses:
Assertion (A): The order of acidic strength of hydrogen halides in aqueous solution is HF \(<\) HCl \(<\) HBr \(<\) HI.
Reason (R): The order of bond dissociation enthalpy of hydrogen halides is HF \(>\) HCl \(>\) HBr \(>\) HI.
Among noble gases, the most abundant one in atmosphere is:
Change in oxidation state of sulphur during oxidation of thiosulphate ion in neutral/alkaline solution by KMnO\(_4\) is:
IUPAC name of [Co(NH\(_3\))\(_4\)(H\(_2\)O)Cl]Cl\(_2\) is:
Arrange the following polymers in increasing order of intermolecular forces:
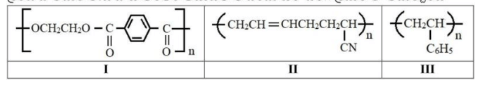
Source for vitamin C and vitamin E respectively are:
Identify the correct antibiotic set:
What are X and Y in the following reaction sequence?
Statement I: Boiling point of diethyl ether \(<\) 1-butanol.
Statement II: Diethyl ether miscible with water to almost same extent as 1-butanol.
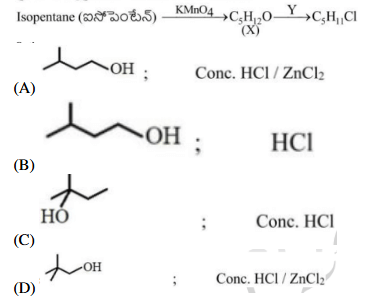
Isopentyl alcohol reacts with reagent X → carboxylic acid → decarboxylation → Y. Reaction of Y with Z → t-butyl alcohol. Identify X and Z.
X and Y in reactions:
Reagents and compound to convert ethyl bromide → 2-butanone:
An amine X (C4H11N) on heating with CHCl3 in ethanolic KOH forms a foul-smelling substance. Product of other amine Y (C4H11N) and benzenesulphonyl chloride is insoluble in alkali. What are X and Y respectively?













































TS EAMCET 2025 BiPC Exam Difficulty Level (Expected)
According to TG EAPCET 2025 past year trends:
Botany and Zoology (Biology) is expected to be of moderate difficulty level with mostly NCERT based questions. Plant Physiology, Human Anatomy, Genetics and Animal Diversity will be some of the most asked topics.
Physics is expected to be tough and lengthy due to the numerical problems. The most asked questions are expected from Thermodynamics and Mechanics and they might be slightly tough.
Chemistry is expected to be the easiest section and the most scoring. The questions are expected to be mostly direct with some tricky questions from Organic Chemistry. Most asked topics include Chemical Bonding, Hydrocarbons and Thermodynamics.
Quick Links:
| Subject | Expected Difficulty Level | Most Asked Topics |
| Botany | Easy to Moderate | Plant Physiology, Genetics, Cell Structure, Ecology |
| Zoology | Moderate | Human Physiology, Reproduction, Genetics, Structural Organisation in Animals |
| Physics | Moderate to Tough | Thermodynamics, Work-Energy-Power, Laws of Motion, Oscillations |
| Chemistry | Easy to Moderate | Atomic Structure, Chemical Bonding, Thermodynamics, Organic Chemistry (Hydrocarbons, Alcohols, Phenols) |










Comments