On 13th February 2021, GATE 2021 Chemical Engineering (CH) Question Paper was held. The exam was conducted by IIT Bombay in the Forenoon Session, from 9:30 AM to 12:30 PM. As instructed by the aspirants, the difficulty level of the question paper was moderate.
The General Aptitude section was easy to attempt. The Mass Transfer section was the most difficult section in GATE 2021 CH. A candidate could easily attempt 40+ questions in the exam. 70 or more than 70 marks would be considered a good score in the exam.
GATE 2021 Chemical Engineering (CH) Question Paper with Solutions
| GATE 2021 Chemical Engineering (CH) Question Paper | Check Solutions |

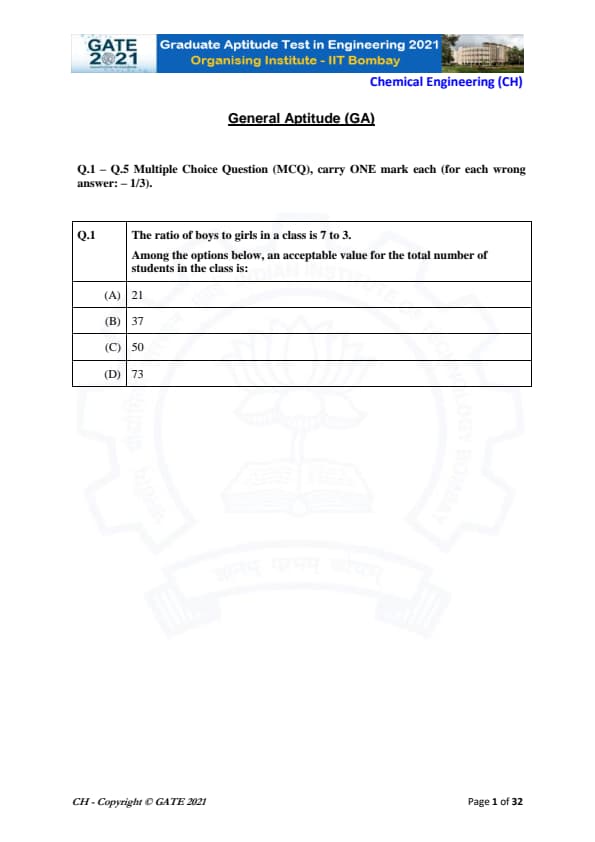
The ratio of boys to girls in a class is 7 to 3. Among the options below, an acceptable value for the total number of students in the class is:
A polygon is convex if, for every pair of points inside the polygon, the line segment joining them lies completely inside or on the polygon. Which one of the following is NOT a convex polygon?

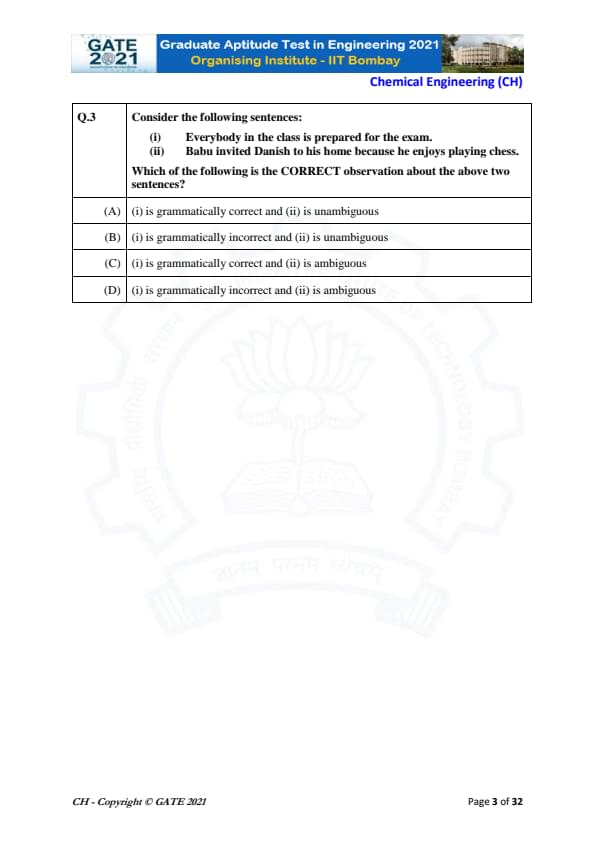
Consider the following sentences:
(i) Everybody in the class is prepared for the exam.
(ii) Babu invited Danish to his home because he enjoys playing chess.
Which of the following is the CORRECT observation about the above two sentences?
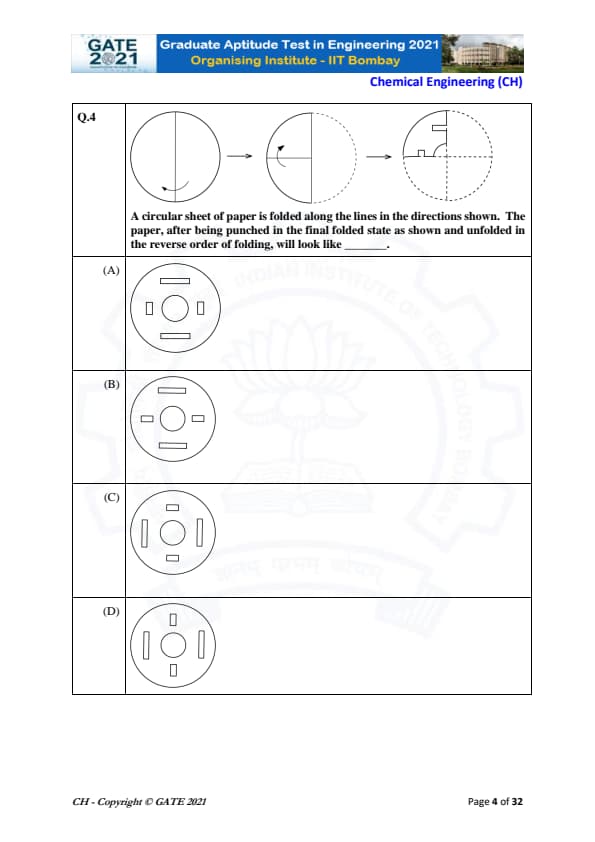
A circular sheet of paper is folded along the lines in the directions shown. The paper, after being punched in the final folded state as shown and unfolded in the reverse order of folding, will look like \hspace{2cm}.


_____ is to surgery as writer is to _____
Which one of the following options maintains a similar logical relation in the above sentence?
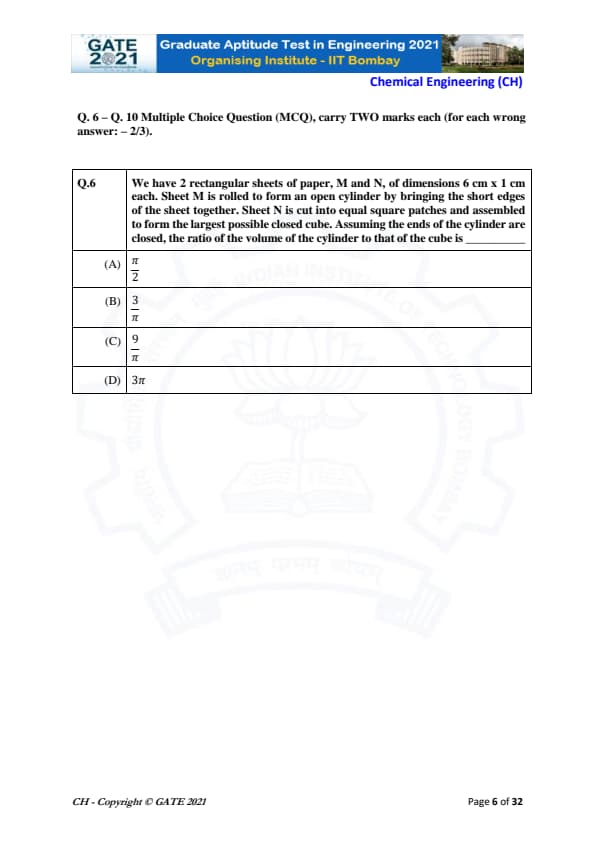
We have 2 rectangular sheets of paper, M and N, of dimensions 6 cm × 1 cm each. Sheet M is rolled to form an open cylinder by bringing the short edges of the sheet together. Sheet N is cut into equal square patches and assembled to form the largest possible closed cube. Assuming the ends of the cylinder are closed, the ratio of the volume of the cylinder to that of the cube is:
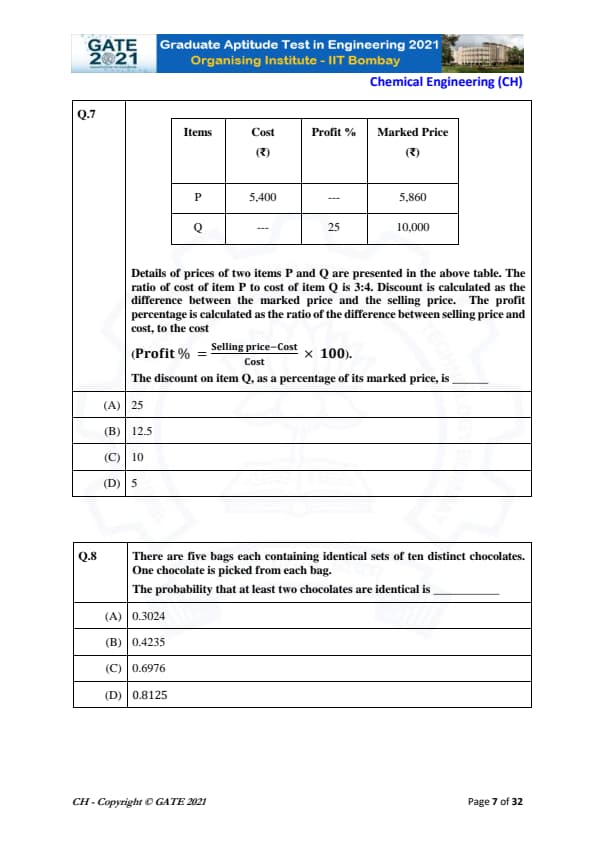
Details of prices of two items P and Q are presented in the above table. The ratio of cost of item P to cost of item Q is 3:4. Discount is calculated as the difference between the marked price and the selling price. The profit percentage is calculated as the ratio of the difference between selling price and cost, to the cost.
The formula for Profit Percentage is:
\[ Profit % = \frac{Selling Price - Cost}{Cost} \times 100 \]
The discount on item Q, as a percentage of its marked price, is:
There are five bags each containing identical sets of ten distinct chocolates. One chocolate is picked from each bag. The probability that at least two chocolates are identical is:
Based on the given statements and conclusions, which option is logically correct?
Statement 1: All bacteria are microorganisms.
Statement 2: All pathogens are microorganisms.
Conclusion I: Some pathogens are bacteria.
Conclusion II: All pathogens are not bacteria.
Which statement summarizes the passage about anti-obesity measures (AOM)?
An ordinary differential equation (ODE), \[ \frac{dy}{dx} = 2y, \quad with an initial condition \quad y(0) = 1, has the analytical solution \quad y = e^{2x}. \]
Using Runge-Kutta second order method, numerically integrate the ODE to calculate y at \(x = 0.5\) using a step size of \(h = 0.5\).
If the relative percentage error is defined as, \[ \epsilon = \left| \frac{y_{analytical} - y_{numerical}}{y_{analytical}} \right| \times 100, \]
then the value of \(\epsilon\) at \(x = 0.5\) is _______.
The function \(\cos(x)\) is approximated using the Taylor series around \(x = 0\) as
\[ \cos(x) \approx 1 + a x + b x^2 + c x^3 + d x^4. \]
The values of \(a, b, c\) and \(d\) are:
The heat of combustion of methane, carbon monoxide and hydrogen are \(P\), \(Q\) and \(R\) respectively. For the reaction
\[ CH_4 + H_2O \longrightarrow CO + 3H_2 \]
the heat of reaction is given by
A batch settling experiment is performed with equal number of type A and type B particles in water. Stokes' law is valid. The settled bed would
A three-dimensional velocity field is given by \(V = 5x^2y\,\mathbf{i} + Cy\,\mathbf{j} - 10xyz\,\mathbf{k}\),
where \(\mathbf{i},\mathbf{j},\mathbf{k}\) are unit vectors in \(x,y,z\) directions.
If \(V\) describes an incompressible fluid flow, the value of \(C\) is
Heat transfer coefficient for a vapor condensing as a film on a vertical surface is given by
In a double-pipe heat exchanger (10 m), hot fluid flows in annulus and cold fluid in inner pipe.
Temperatures vary as: \(T_h(x) = 80 - 3x\), \(T_c(x) = 20 + 2x\),
where \(T\) in °C and \(x\) in m.
The logarithmic mean temperature difference (LMTD) is
For a shell-and-tube heat exchanger, the clean overall heat transfer coefficient is calculated as 250 W m\(^{-2}\) K\(^{-1}\) for a specific process condition. It is expected that the heat exchanger may be fouled during the operation, and a fouling resistance of 0.001 m\(^{2}\) K W\(^{-1}\) is prescribed. The dirt overall heat transfer coefficient is ________ W m\(^{-2}\) K\(^{-1}\).
In reverse osmosis, the hydraulic pressure and osmotic pressure at the feed side of the membrane are \(P_1\) and \(\pi_1\), respectively. The corresponding values are \(P_2\) and \(\pi_2\) at the permeate side. The membrane, feed, and permeate are at the same temperature. For equilibrium to prevail, the general criterion that should be satisfied is:
Ethylene adsorbs on the vacant active sites V of a transition metal catalyst according to the following mechanism.

If \(N_T\), \(N_V\) and \(N_{C_2H_4}\) denote the total number of active sites, number of vacant active sites and number of adsorbed C\(_2\)H\(_4\) molecules, respectively, the balance on the total number of active sites is given by:
Which of the following is NOT a standard to transmit measurement and control signals?
A feedforward controller can be used only if:
Turnover ratio is defined as:
A principal amount is charged a nominal annual interest rate of 10%. If interest is compounded continuously, what happens to the final amount after one year?
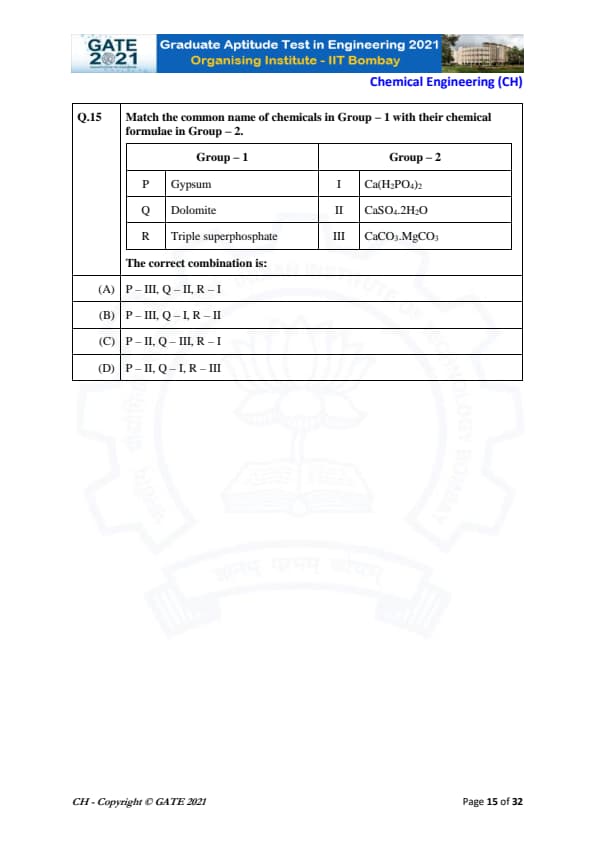
Match the common name of chemicals in Group – 1 with their chemical formulae in Group – 2.

The correct combination is:
For the function \(f(x)=\begin{cases} -x, & x<0
x^{2}, & x\ge 0 \end{cases}\)
the CORRECT statement(s) is/are
In the ternary extraction diagram shown, the CORRECT option(s) is/are

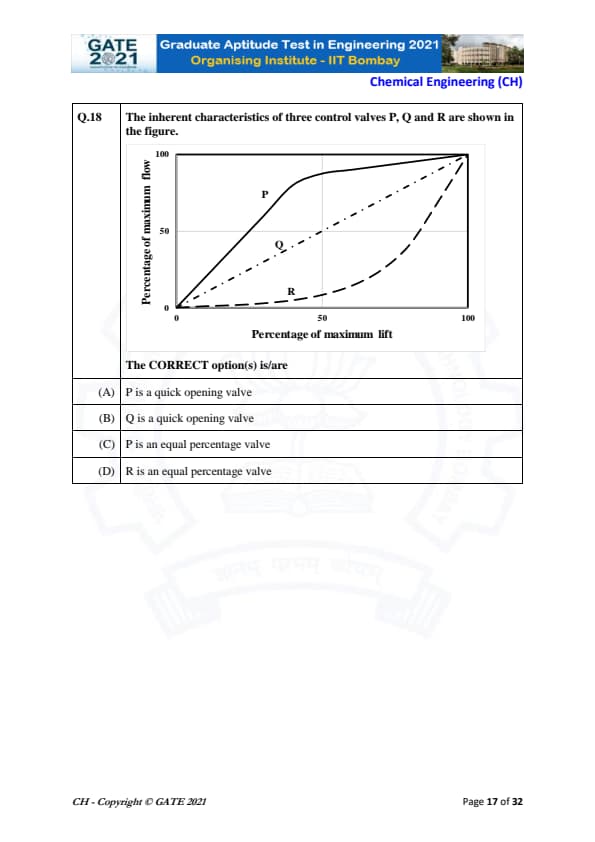
The inherent characteristics of three control valves P, Q and R are shown in the figure.

The CORRECT option(s) is/are
A source placed at the origin of a circular sample holder (radius \(r = 1\) m) emits particles uniformly in all directions. A detector of length \(l = 1\) cm is placed on the perimeter. During the experiment the detector registers 14 particles. The total number of particles emitted is _______.
A, B, C and D are vectors of length 4. The rank of the matrix

It is known that B is not a scalar multiple of A. Also, C is linearly independent of A and B. Further, \[ D = 3A + 2B + C \]
The rank of the matrix \[ \begin{bmatrix} a_1 & a_2 & a_3 & a_4 \\
b_1 & b_2 & b_3 & b_4 \\
c_1 & c_2 & c_3 & c_4 \\
d_1 & d_2 & d_3 & d_4 \end{bmatrix} \] is ____
The van der Waals equation in reduced form is \[ P_r = \frac{8T_r}{3v_r - 1} - \frac{3}{v_r^2}. \]
If \(v_r = 3\) and \(T_r = \frac{4}{3}\), compute the compressibility factor \(Z\) (rounded to 2 decimals).
Consider a steady flow of an incompressible Newtonian fluid through a smooth circular pipe. For turbulent flow, the kinetic energy correction factor is \[ \alpha_{turbulent} = \left(\frac{V_0}{\overline{V}}\right)^3 \frac{2n^2}{(3+n)(3+2n)} \]
and the ratio of average velocity to centerline velocity is \[ \frac{\overline{V}}{V_0} = \frac{2n^2}{(n+1)(2n+1)}. \]
For n = 7, the value of \(\frac{\alpha_{turbulent}}{\alpha_{laminar}}\) is _____ (round to 2 decimals).
The molar heat capacity of n-pentane is \[ \frac{C_p}{R} = 2.46 + 45.4 \times 10^{-3}T - 14.1 \times 10^{-6}T^2. \]
At 1000 K, the rate of change of molar entropy at constant pressure is _____ J mol\(^{-1}\) K\(^{-2}\).
A ⇌ B ⇌ C reactions are at equilibrium. Given \(k_1 = 0.1\), \(k_{-1} = 0.2\), \(k_2 = 1\), \(k_{-2} = 10\), \(k_3 = 10\).
Determine \(k_{-3}\) (round to 1 decimal).

A company invests in a recovery unit to separate valuable metals from effluent streams. The capital investment is Rs. 10 lakhs. The recovered metals generate Rs. 4 lakhs per year. If the annual return on this investment is 15%, the annual operating costs should be ___________ lakhs of rupees (correct to 1 decimal place).
Let \(A\) be an \(n \times n\) square matrix (\(n>1\)) with elements \[ a_{ij} = \begin{cases} i \times j, & if i \ge j
0, & if i < j \end{cases} \]
The determinant of \(A\) is:
A fluid is sheared between parallel plates in two experiments, and the required shear stresses are given. Determine the rheological behavior of the fluid.
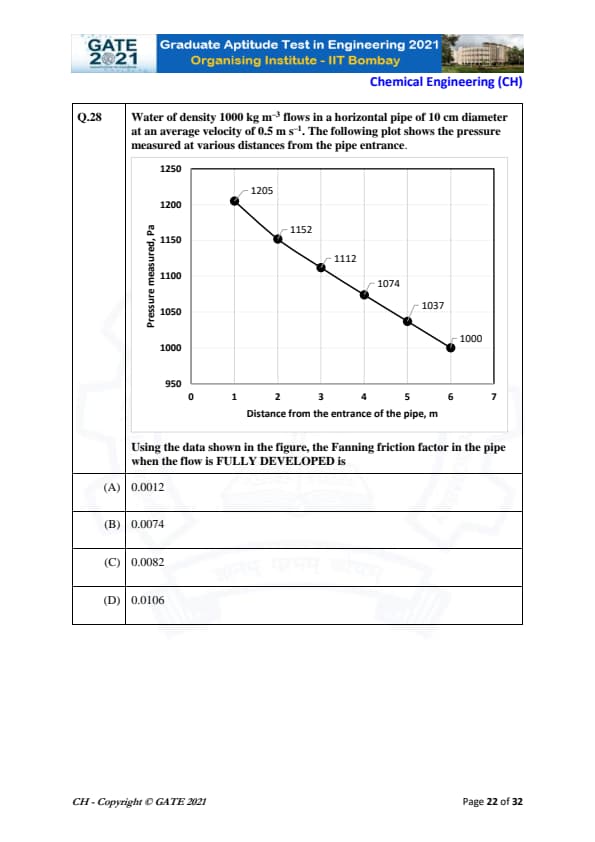
Water of density 1000 kg m\(^{-3}\) flows in a horizontal pipe of 10 cm diameter at an average velocity of 0.5 m s\(^{-1}\). The following plot shows the pressure measured at various distances from the pipe entrance.

In a solvent regeneration process, a gas is used to strip a solute from a liquid in a countercurrent packed tower operating under isothermal condition. Pure gas is used in this stripping operation. All solutions are dilute and Henry's law, \(y^ = mx\), is applicable. Here, \(y^\) is the mole fraction of the solute in the gas phase in equilibrium with the liquid phase of solute mole fraction \(x\), and \(m\) is the Henry’s law constant. Let \(x_1\) be the mole fraction of the solute in the leaving liquid, and \(x_2\) be the mole fraction of solute in the entering liquid. When the value of the ratio of the liquid-to-gas molar flow rates is equal to \(m\), the overall liquid phase Number of Transfer Units, NTU\(_{OL}\), is given by
Which of these symbols can be found in piping and instrumentation diagrams?

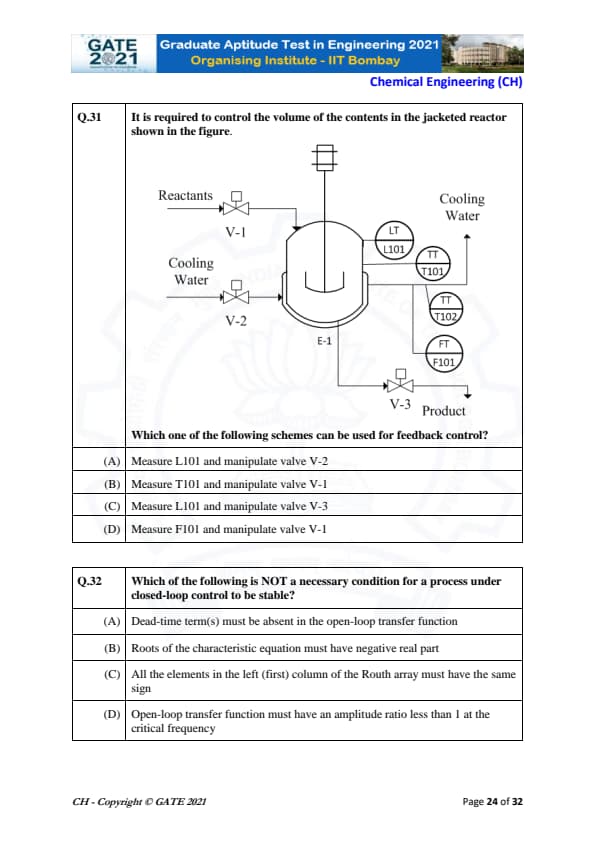
It is required to control the volume of the contents in the jacketed reactor shown in the figure.

Which one of the following schemes can be used for feedback control?
Which of the following is NOT a necessary condition for a process under closed-loop control to be stable?
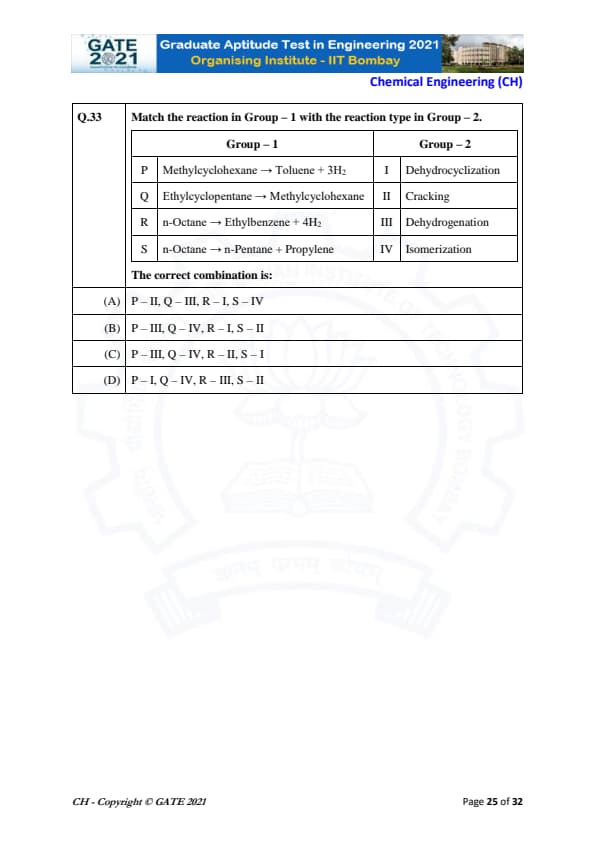
Match the reaction in Group – 1 with the reaction type in Group – 2.

To solve an algebraic equation \( f(x) = 0 \), an iterative scheme of the type \( x_{n+1} = g(x_n) \) is proposed, where \( g(x) = x - \frac{f(x)}{f'(x)} \).
At the solution \( x = s \), \( g'(s) = 0 \) and \( g''(s) \neq 0 \).
The order of convergence for this iterative scheme near the solution is _____.
The probability distribution function of a random variable \( X \) is shown in the figure. From this distribution, random samples with sample size \( n = 68 \) are taken. If \( \bar{X} \) is the sample mean, the standard deviation of the probability distribution of \( \bar{X} \), i.e. \( \sigma_{\bar{X}} \), is ______ (rounded off to 3 decimal places).

For the ordinary differential equation \[ \frac{d^3 y}{dt^3} + 6 \frac{d^2 y}{dt^2} + 11 \frac{dy}{dt} + 6y = 1 \]
with initial conditions \( y(0) = y'(0) = y''(0) = y'''(0) = 0 \), the value of \( \lim_{t \to \infty} y(t) \) is _____ (rounded off to 3 decimal places).
Formaldehyde is produced by the oxidation of methane in a reactor. The following two parallel reactions occur: \[ CH_4 + O_2 \rightarrow HCHO + H_2O \] \[ CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O \]
Methane and oxygen are fed to the reactor. The product gases leaving the reactor include methane, oxygen, formaldehyde, carbon dioxide and water vapor.
60 mol/s of methane enters the reactor. The molar flowrate (in mol/s) of CH₄, O₂ and CO₂ leaving the reactor are 26, 2, and 4, respectively. The molar flowrate of oxygen entering the reactor is _____ mol/s.
The combustion of carbon monoxide is carried out in a closed, rigid and insulated vessel. 1 mol of CO, 0.5 mol of O₂ and 2 mol of N₂ are taken initially at 1 bar and 298 K, and the combustion is carried out to completion. The standard molar internal energy change of reaction (\(\Delta u^\circ_R\)) for the combustion of carbon monoxide at 298 K is -282 kJ mol\(^{-1}\). At constant pressure, the molar heat capacities of N₂ and CO₂ are 33.314 J mol\(^{-1}\) K\(^{-1}\) and 58.314 J mol\(^{-1}\) K\(^{-1}\), respectively. Assume the heat capacities are independent of temperature, and the gases are ideal. Take R = 8.314 J mol\(^{-1}\) K\(^{-1}\). The final pressure in the vessel at the completion of the reaction is ____ bar (round off to 1 decimal place).
A gaseous mixture at 1 bar and 300 K consists of 20 mol% CO₂ and 80 mol% inert gas. Assume the gases to be ideal. Take R = 8.314 J mol\(^{-1}\) K\(^{-1}\). The magnitude of minimum work required to separate 100 mol of this mixture at 1 bar and 300 K into pure CO₂ and inert gas at the same temperature and pressure is ____ kJ (round off to nearest integer).
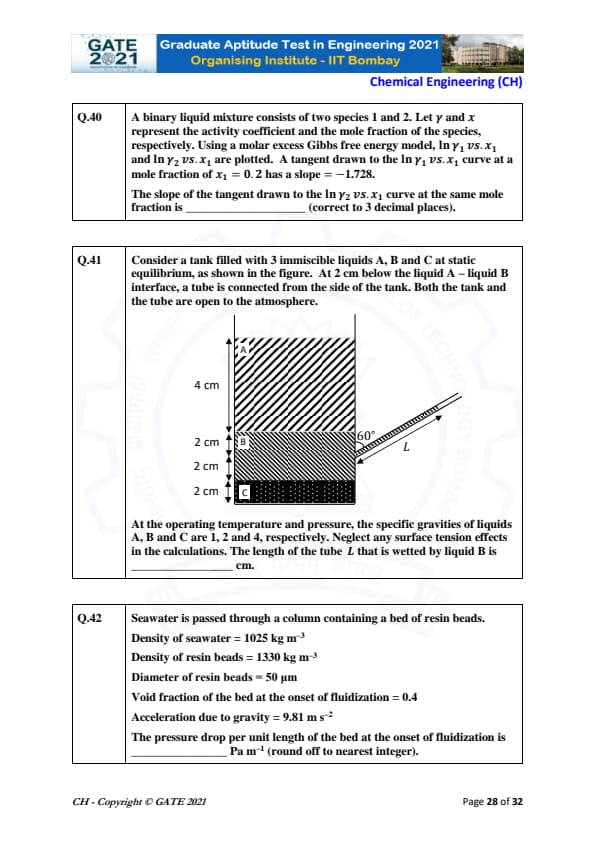
A binary liquid mixture consists of two species 1 and 2. Let \( \gamma \) and \( x \) represent the activity coefficient and the mole fraction of the species, respectively. Using a molar excess Gibbs free energy model, \( \ln \gamma_1 \) vs. \( x_1 \) and \( \ln \gamma_2 \) vs. \( x_1 \) are plotted. A tangent drawn to the \( \ln \gamma_1 \) vs. \( x_1 \) curve at a mole fraction of \( x_1 = 0.2 \) has a slope = -1.728. The slope of the tangent drawn to the \( \ln \gamma_2 \) vs. \( x_1 \) curve at the same mole fraction is _______ (correct to 3 decimal places).
Consider a tank filled with 3 immiscible liquids A, B, and C at static equilibrium. At 2 cm below the liquid A-liquid B interface, a tube is connected from the side of the tank. Both the tank and the tube are open to the atmosphere. At the operating temperature and pressure, the specific gravities of liquids A, B, and C are 1, 2, and 4, respectively. Neglect any surface tension effects in the calculations. The length of the tube \( L \) that is wetted by liquid B is _____ cm.

Seawater is passed through a column containing a bed of resin beads.
Density of seawater = 1025 kg m\(^{-3}\)
Density of resin beads = 1330 kg m\(^{-3}\)
Diameter of resin beads = 50 \(\mu\)m
Void fraction of the bed at the onset of fluidization = 0.4
Acceleration due to gravity = 9.81 m s\(^{-2}\)
The pressure drop per unit length of the bed at the onset of fluidization is _____ Pa m\(^{-1}\) (round off to nearest integer).
A straight fin of uniform circular cross section and adiabatic tip has an aspect ratio (length/diameter) of 4. If the Biot number (based on radius of the fin as the characteristic length) is 0.04, the fin efficiency is _____ % (round off to nearest integer).
A double-effect evaporator is used to concentrate a solution. Steam is sent to the first effect at 110 °C and the boiling point of the solution in the second effect is 63.3 °C. The overall heat transfer coefficient in the first effect and second effect are 2000 W m\(^{-2}\) K\(^{-1}\) and 1500 W m\(^{-2}\) K\(^{-1}\), respectively. The heat required to raise the temperature of the feed to the boiling point can be neglected. The heat flux in the two evaporators can be assumed to be equal. The temperature at which the solution boils in the first effect is _____ °C (round off to nearest integer).
Consider a solid slab of thickness 2L and uniform cross section A. The volumetric rate of heat generation within the slab is \(\dot{q}\) (W m\(^{-3}\)). The slab loses heat by convection at both the ends to air with heat transfer coefficient \(h\). Assuming steady state, one-dimensional heat transfer, the temperature profile within the slab along the thickness is given by: \[ T(x) = \frac{\dot{q}L^2}{2k} \left[1 - \left(\frac{x}{L}\right)^2 \right] + T_s \quad for \quad -L \leq x \leq L \]
where \(k\) is the thermal conductivity of the slab and \(T_s\) is the surface temperature. If \(T_s = 350\) K, ambient air temperature \(T_\infty = 300\) K, and Biot number (based on L as the characteristic length) is 0.5, the maximum temperature in the slab is ____ K (round off to nearest integer).
A distillation column handling a binary mixture of A and B is operating at total reflux. It has two ideal stages including the reboiler. The mole fraction of the more volatile component in the residue (\(x_w\)) is 0.1. The average relative volatility \(\alpha_{AB}\) is 4. The mole fraction of A in the distillate (\(x_D\)) is _____ (round off to 2 decimal places).
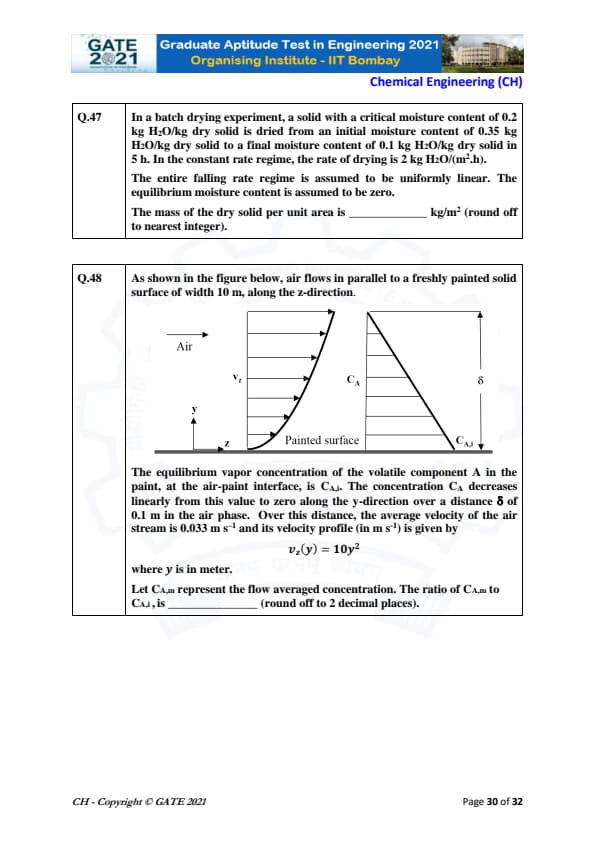
In a batch drying experiment, a solid with a critical moisture content of 0.2 kg H\(_2\)O/kg dry solid is dried from an initial moisture content of 0.35 kg H\(_2\)O/kg dry solid to a final moisture content of 0.1 kg H\(_2\)O/kg dry solid in 5 hours. In the constant rate regime, the rate of drying is 2 kg H\(_2\)O/(m\(^2\)·h). The entire falling rate regime is assumed to be uniformly linear. The equilibrium moisture content is assumed to be zero. The mass of the dry solid per unit area is _____ kg/m\(^2\) (round off to nearest integer).
As shown in the figure below, air flows in parallel to a freshly painted solid surface of width 10 m, along the z-direction.

The equilibrium vapor concentration of the volatile component A in the paint, at the air-paint interface, is \( C_{A,i} \). The concentration \( C_A \) decreases linearly from this value to zero along the y-direction over a distance \( \delta \) of 0.1 m in the air phase. Over this distance, the average velocity of the air stream is 0.033 m s\(^{-1}\) and its velocity profile \( v_z(y) \) is given by \[ v_z(y) = 10 y^2 \]
where \( y \) is in meter. Let \( C_{A,m} \) represent the flow averaged concentration. The ratio of \( C_{A,m} \) to \( C_{A,i} \) is _____ (round off to 2 decimal places).
The following isothermal autocatalytic reaction:
\[ A + B \longrightarrow 2B \quad \left( -r_A = 0.1 C_A C_B \, (mol L^{-1} s^{-1}) \right) \]
is carried out in an ideal continuous stirred tank reactor (CSTR) operating at steady state. Pure A at 1 mol L\(^{-1}\) is fed, and 90% of A is converted in the CSTR. The space time of the CSTR is _____ seconds.
Reactant A decomposes to products B and C in the presence of an enzyme in a well-stirred batch reactor. The kinetic rate expression is given by
\[ -r_A = \frac{0.01 C_A}{0.05 + C_A} \, (mol L^{-1} min^{-1}) \]
If the initial concentration of A is 0.02 mol L\(^{-1}\), the time taken to achieve 50% conversion of A is _____ min (rounded to 2 decimal places).
The following homogeneous, irreversible reaction involving ideal gases:
\[ A \longrightarrow B + C \quad \left( -r_A = 0.5 C_A \, (mol L^{-1} s^{-1}) \right) \]
is carried out in a steady state ideal plug flow reactor (PFR) operating at isothermal and isobaric conditions. The feed stream consists of pure A, entering at 2 m s\(^{-1}\). In order to achieve 50% conversion of A, the required length of the PFR is _____ meter (rounded off to 2 decimal places).
A system has a transfer function
\[ G(s) = \frac{3e^{-4s}}{12s^2 + 1} \]
When a step change of magnitude M is given to the system input, the final value of the system output is measured to be 120. The value of M is _____ .
A process has a transfer function
\[ G(s) = \frac{Y(s)}{X(s)} = \frac{20}{90000s^2 + 240s + 1} \]
Initially the process is at steady state with \( x(t = 0) = 0.4 \) and \( y(t = 0) = 100 \). If a step change in \( x \) is given from 0.4 to 0.5, the maximum value of \( y \) that will be observed before it reaches the new steady state is ____ (round off to 1 decimal place).
Operating labor requirements L in the chemical process industry is described in terms of the plant capacity C (kg day\(^{-1}\)) over a wide range (10\(^3\) − 10\(^6\)) by a power law relationship: \[ L = \alpha C^\beta \]
where \(\alpha\) and \(\beta\) are constants. It is known that \[ L = 60 when C = 2 \times 10^4 \quad and \quad L = 70 when C = 6 \times 10^4 \]
The value of L when C = \(10^5\) kg day\(^{-1}\) is _____ (round off to nearest integer).
A viscous liquid is pumped through a pipe network in a chemical plant. The annual pumping cost per unit length of pipe is given by \[ C_{pump} = \frac{48.13 q^2 \mu}{D^4} \]
The annual cost of the installed piping system per unit length of pipe is given by \[ C_{piping} = 45.92 D \]
Here, D is the inner diameter of the pipe in meter, q is the volumetric flowrate of the liquid in m\(^3\)s\(^{-1}\) and \(\mu\) is the viscosity of the liquid in Pa.s. If the viscosity of the liquid is \(20 \times 10^{-3}\) Pa.s and the volumetric flow rate of the liquid is \(10^{-4}\) m\(^3\)s\(^{-1}\), the economic inner diameter of the pipe is _____ meter (round off to 3 decimal places).




















Quick Links:
GATE 2021 CH: Marks Weightage as per the Sections
Engineering Mathematics carried weightage somewhere between 11-22%. Questions related to Engineering Mathematics were of moderate level.
The below-mentioned table shows the weightage of marks based on topics-
| Sections | Marks Weightage (in %) |
|---|---|
| Engineering Mathematics | 11-22% |
| General Aptitude | 15% |
| Chemical Reaction Engineering | 12% |
| IRC | 12% |
| Fluid Mechanics | 10% |
| Mass Transfer | 7% |
| CT | 4% |
| Rest of the sections | Remaining weightage as per marks |
- 10 questions appeared from General Aptitude section, carried 15% of the total weightage in GATE 2021 CH
- Other 85% weightage were distributed across the different sections of the core chemical engineering syllabus
- CT section stood with the least number of questions which were of easy to moderate level
- Out of 65 questions, 33 questions were MCQs carried 46 marks
- The weightage of NATs in terms of appearance were 29, holds 51 marks
- Only 3 MSQs appeared in GATE 2021 CH, carried 1 mark each
Also Check:
GATE 2021 Exam Pattern and Marking Scheme
In 2021, MSQs appeared in GATE 2021 CH, making it a little more complex to attempt. MSQs comes with one or more than one correct option. Only after choosing all the correct options, a candidate will be rewarded with the marks.
- The question paper comprised of 65 questions holding 100 marks
- There would be no negative marking if wrong NATs or MSQs would be attempted
- For every incorrect attempted MCQ, ⅓ or ⅔ marks would be reduced for the questions holding 1 and 2 marks
The cutoff for General candidates was 36.2, 32.5 for OBC students, and 24.1 for SC/ST students. The cutoff for 2021 was slightly higher as compared to 2020. There were a total of 16705 candidates, competing to clear the cutoff. Although 20.35% of the total candidates qualified.
GATE Previous Year Question Papers
| GATE 2022 Question Papers | GATE 2021 Question Papers | GATE 2020 Question Papers |
| GATE 2019 Question Papers | GATE 2018 Question Papers | GATE 2017 Question Papers |










Comments