GATE 2021 Chemistry (CY) Question Paper with Solutions can be downloaded from here. More than 23500 students took GATE 2021 CY. This year IIT Bombay conducted GATE 2021. GATE 2021 CY exam took place on February 6, 2021 in the forenoon session (9:30 AM to 12:30 PM). As per the students, the difficulty level of the exam lies between easy to moderate. The number of General Aptitude questions remained the same in GATE 2021 CY.
Students aiming for GATE next year can download the previous year GATE question papers and answer key PDFs to check their preparation level.
GATE 2021 Chemistry (CY) Question Paper with Solutions
| GATE 2021 Chemistry (CY) Question Paper | Check Solutions |

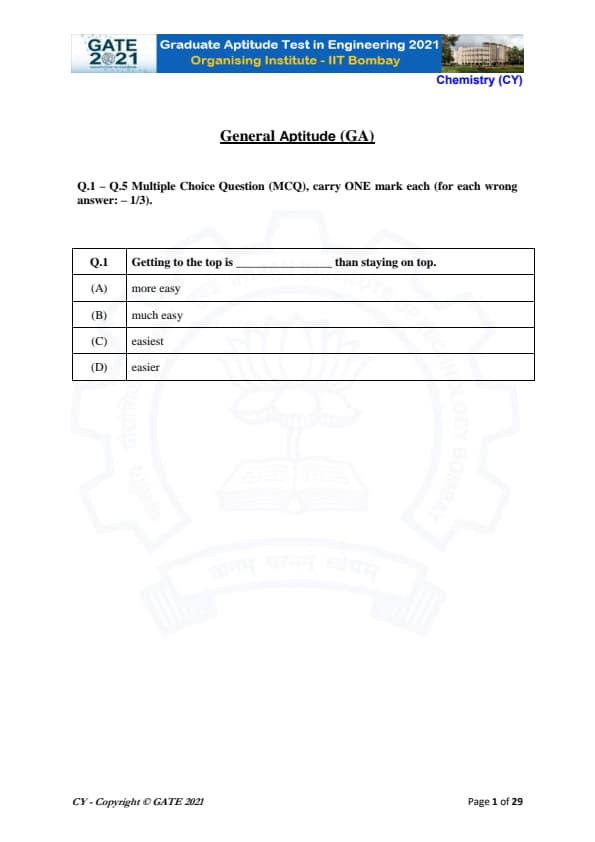
Getting to the top is _______ than staying on top.
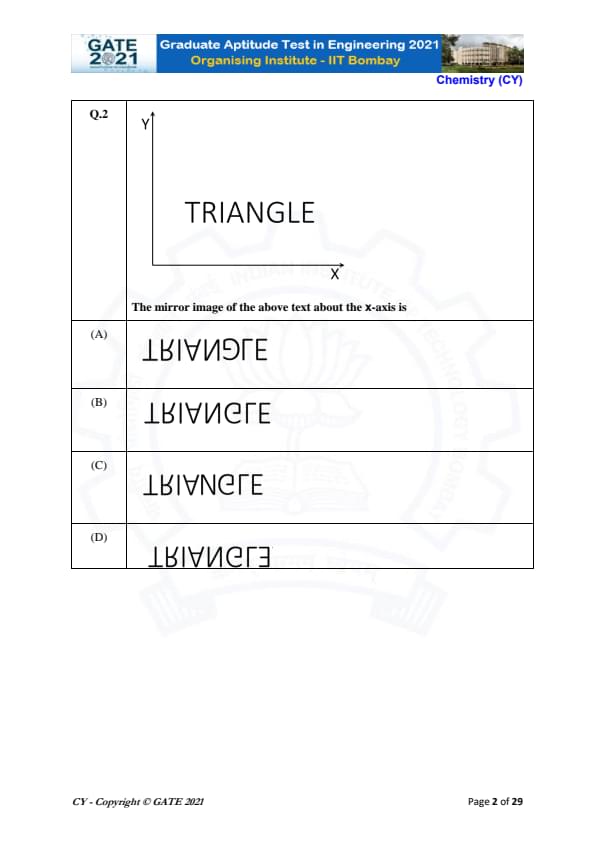
The mirror image of the above text about the x-axis is:


In a company, 35% of the employees drink coffee, 40% of the employees drink tea, and 10% of the employees drink both tea and coffee. What % of employees drink neither tea nor coffee?
Let \( \oplus \) and \( \odot \) be two operators on numbers \( p \) and \( q \) such that \[ p \oplus q = \frac{p^2 + q^2}{pq} \quad and \quad p \odot q = \frac{p^2}{q}. \]
If \( x \oplus y = 2 \odot 2 \), then \( x = \: \_ \)?
Four persons P, Q, R, and S are to be seated in a row, all facing the same direction, but not necessarily in the same order. P and R cannot sit adjacent to each other. S should be seated to the right of Q. The number of distinct seating arrangements possible is:
Statement: Either P marries Q or X marries Y
Among the options below, the logical NEGATION of the above statement is:
Consider two rectangular sheets, Sheet M and Sheet N of dimensions 6 cm x 4 cm each.
Folding operation 1: The sheet is folded into half by joining the short edges of the current shape.
Folding operation 2: The sheet is folded into half by joining the long edges of the current shape.
Folding operation 1 is carried out on Sheet M three times.
Folding operation 2 is carried out on Sheet N three times.
The ratio of perimeters of the final folded shape of Sheet N to the final folded shape of Sheet M is _________.
Five line segments of equal lengths, PR, PS, QS, QT and RT are used to form a star as shown in the figure above. The value of \( \theta \), in degrees, is


A function, \( \lambda \), is defined by: \[ \lambda(p,q) = \begin{cases} (p - q)^2, & if p \geq q,
p + q, & if p < q. \end{cases} \]
The value of the expression \( \lambda(-( -3 + 2), ( -2 + 3)) \) is:
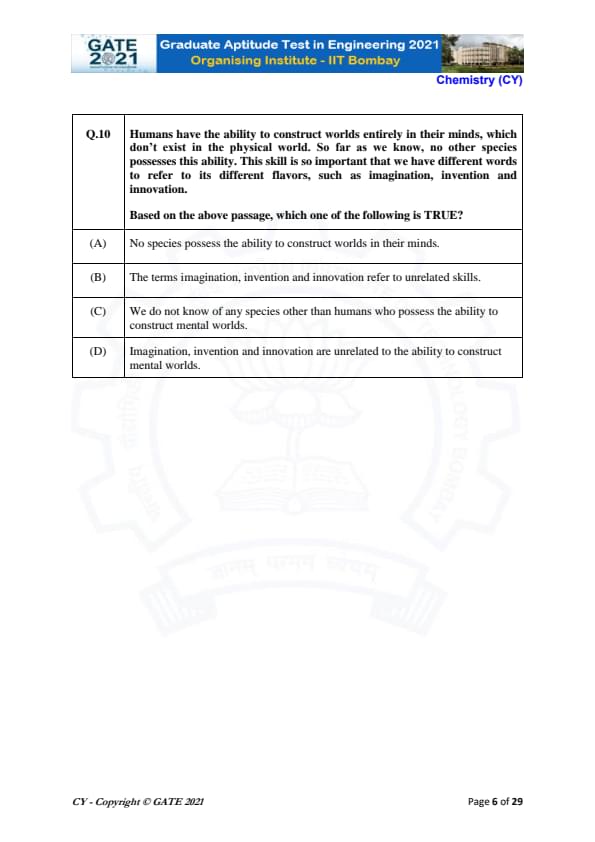
Humans have the ability to construct worlds entirely in their minds, which don't exist in the physical world. So far as we know, no other species possesses this ability. This skill is so important that we have different words to refer to its different flavors, such as imagination, invention and innovation.
Based on the above passage, which one of the following is TRUE?
The rates of alkaline hydrolysis of the compounds shown below follow the order:
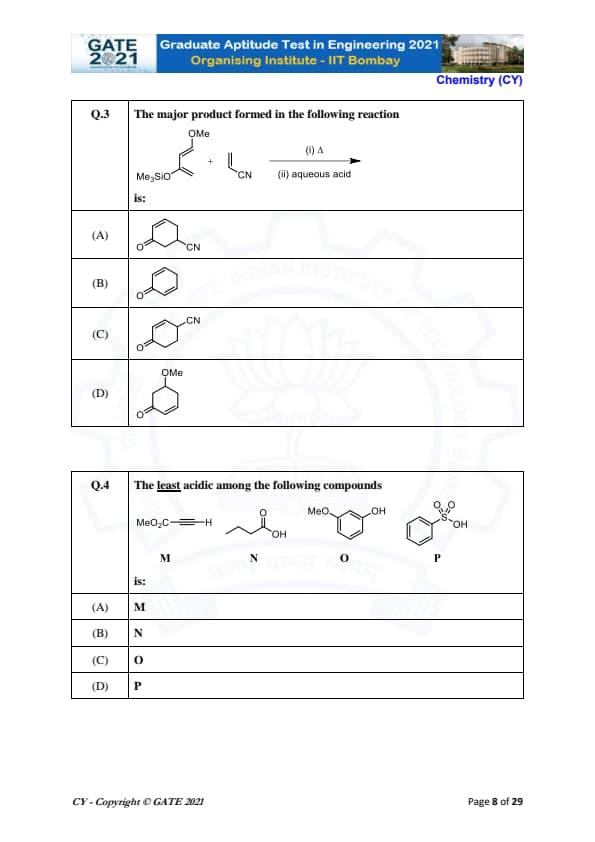
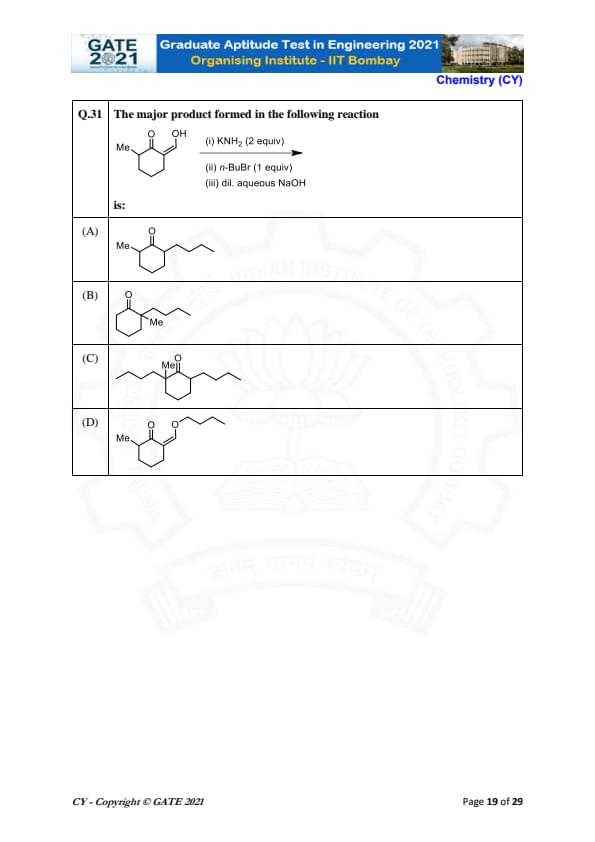
The major product formed in the following reaction is:
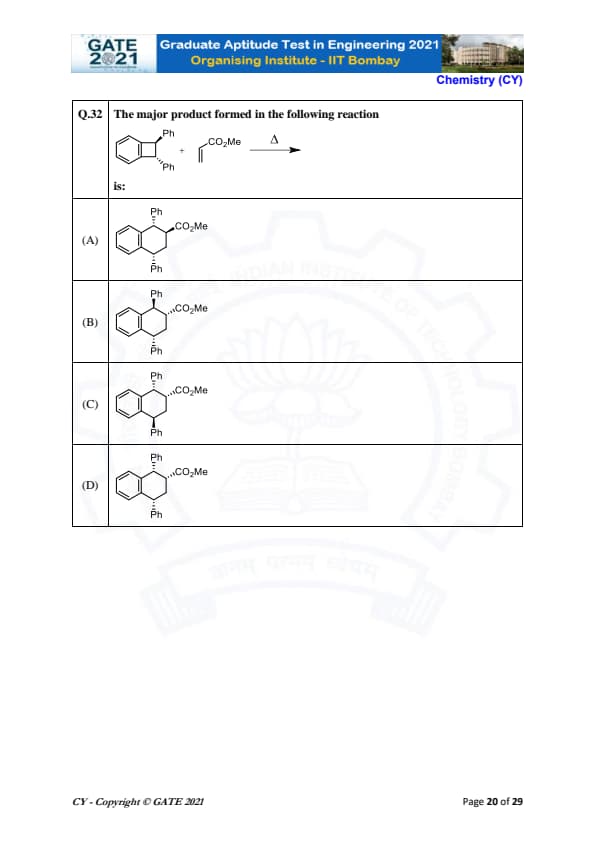
The major product formed in the following reaction:
The least acidic among the following compounds:
is:
The major product formed in the following reaction:
is:
The reagent(s) required for the conversion of hex-3-yne to (E)-hex-3-ene is/are:
An organic compound exhibits the [M]+, [M+2]+, and [M+4]+ peaks in the intensity ratio 1:2:1 in the mass spectrum, and shows a singlet at \( \delta \) 7.49 in the \( ^1H \) NMR spectrum in CDCl\(_3\). The compound is:
Reaction of LiAlH\(_4\) with one equivalent of Me\(_3\)N·HCl gives a tetrahedral compound, which reacts with another equivalent of Me\(_3\)N·HCl to give compound N. The compound N and its geometry, respectively, are:
Which one of the following is a non-heme protein?
A correct example of a nucleotide is:
The equilibrium constant for the reaction \[ 3 \, NO (g) \rightleftharpoons N_2O (g) + NO_2 (g) \]
at 25°C is closest to: \[ [\Delta G^\circ = -104.18 \, kJ; R = 8.314 \, J mol^{-1} K^{-1}] \]
The reaction of NiBr_2 with two equivalents of PPh_3 in CS_2 at -78°C gives a red-colored diamagnetic complex, [NiBr_2 \text{(PPh_3\text{)_2\text{]. This transforms to a green-colored paramagnetic complex with the same molecular formula at 25°C. The geometry and the number of unpaired electrons in the green-colored complex, respectively, are:
The rate of the substitution reaction of [Co(CN)\(_5\)Cl]\(^3^-\) with OH\(^-\) to give [Co(CN)\(_5\)(OH)]\(^3^-\) follows:
The \(\Delta_o\) of [Cr(H\(_2\)O)\(_6\)]\(^3+\), [CrF\(_6\)]\(^3-\), and [Cr(CN)\(_6\)]\(^3-\) follows the order:
The phase diagram of CO\(_2\) is shown below:
Phase diagram of CO\(_2\) with solid, liquid, and gas phases labeled along with critical temperature \(T_c\) and triple point temperature \(T_1\)
The correct statement(s) about CO\(_2\) is/are:
Acceptable wavefunctions for a quantum particle must be:
The characters of \( E, C_2, \sigma_v, \) and \( \sigma' \) symmetry operations, in this order, for valid irreducible representation(s) of the \( C_2v \) point group is/are:
The normal mode(s) of vibration of H\(_2\)O is/are:
A reversible heat engine absorbs 20 kJ of heat from a source at 500 K and dissipates it to the reservoir at 400 K. The efficiency of the heat engine is ________ %.
Among the following eight compounds, the number of compound(s) which can exhibit stereoisomerism is ________.
The Mo–Mo bond order in [(η5-C5H5)Mo(CO)2]2 which obeys the 18-electron rule is ________.
The change in enthalpy (\( \Delta H \)) for the reaction
\[ 2 \, P (s) + 3 \, Br_2 (l) \rightarrow 2 \, PBr_3 (l) \]
is -243 kJ. In this reaction, if the amount of phosphorus consumed is 3.1 g, the change in enthalpy (rounded off to two decimal places) is ________ kJ.
The number of signal(s) in the \( ^1H \) NMR spectrum of the following compound
recorded at 25°C in CDCl_3 is ________.
A 5 V battery delivers a steady current of 1.5 A for a period of 2 h. The total charge that has passed through the circuit is _________ Coulombs.
The spin-only magnetic moment of [Co(H_2\text{O)_6^{2+ (rounded off to one decimal place) is _________ BM.
The geometry and the number of unpaired electrons in tetrakis(1-norbomyl)Co, respectively, are:
The yellow color of an aqueous solution of K\(_2\)CrO\(_4\) changes to red-orange upon the addition of a few drops of HCl. The red-orange complex, the oxidation state of its central element(s), and the origin of its color, respectively, are:
The shapes of the compounds ClF\(_3\), XeOF\(_2\), N\(_3^-\) and XeO\(_3\)F\(_2\) respectively, are:
The metal borides that contain isolated boron atoms are:
The major product formed in the following reaction
is:
The major product formed in the following reaction is:
The major product formed in the following reaction:
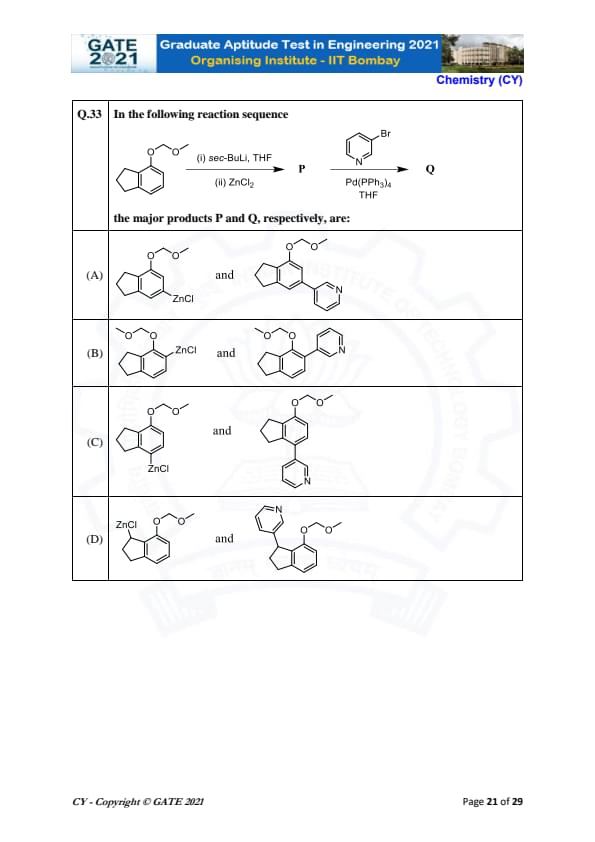
In the following reaction sequence:
The major products P and Q, respectively, are:
In an electrochemical cell, Ag\(^+\) ions in AgNO\(_3\) are reduced to Ag metal at the cathode and Cu is oxidized to Cu\(^{2+}\) at the anode. A current of 0.7 A is passed through the cell for 10 min. The mass (in grams) of silver deposited and copper dissolved, respectively, are:
Among the following, the compounds which can be prepared by nucleophilic substitution reaction are:
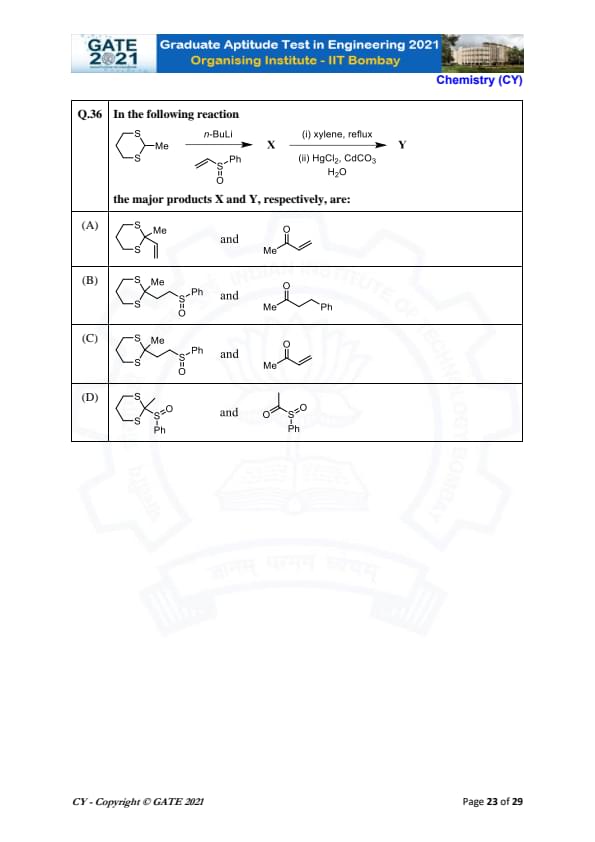
In the following reaction
the major products X and Y, respectively, are:
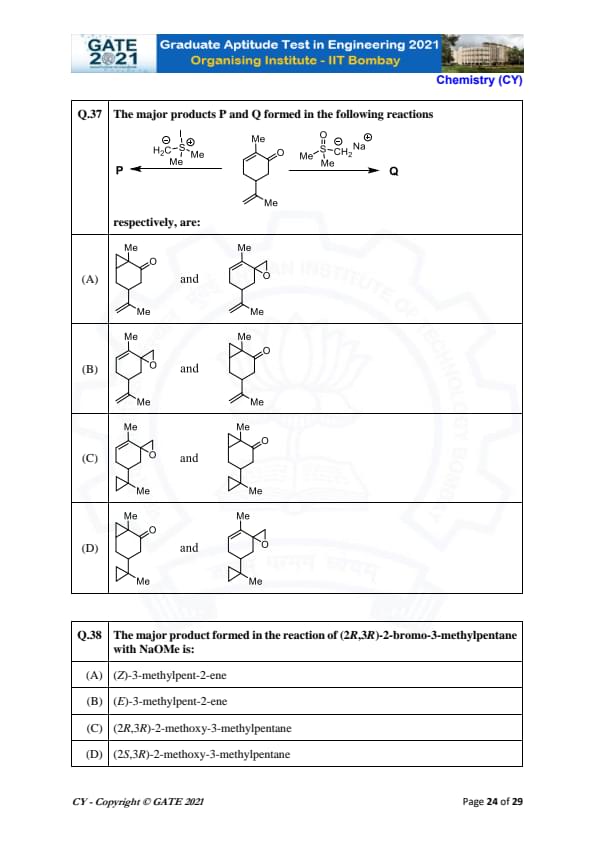
The major products P and Q formed in the following reactions respectively, are:
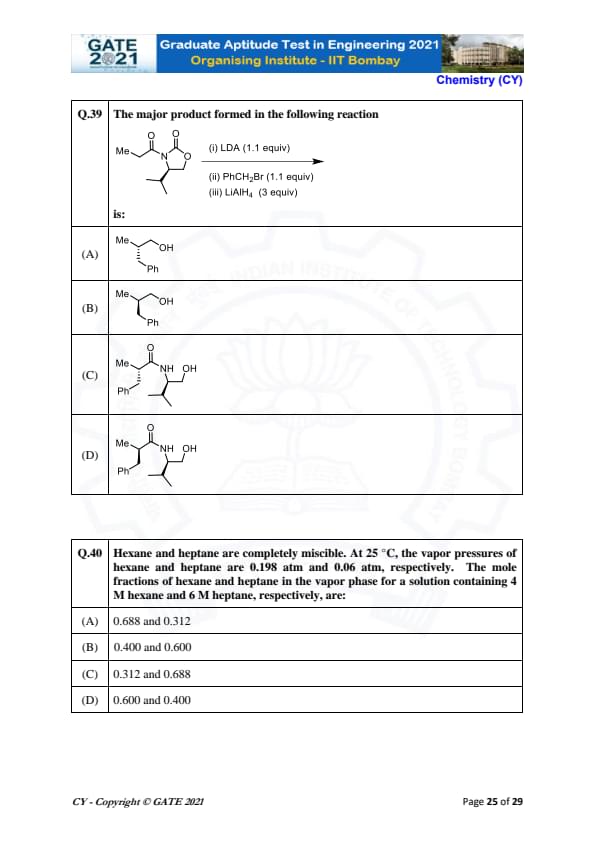
The major product formed in the reaction of (2R,3R)-2-bromo-3-methylpentane with NaOMe is:
The major product formed in the following reaction:
is:
Hexane and heptane are completely miscible. At 25°C, the vapor pressures of hexane and heptane are 0.198 atm and 0.06 atm, respectively. The mole fractions of hexane and heptane in the vapor phase for a solution containing 4 M hexane and 6 M heptane, respectively, are:
The correct order of Lewis acid strengths of BF\(_2\)Cl, BFClBr, BF\(_2\)Br and BFBr\(_2\) is:
The correct order of increasing intensity (molar absorptivity) of the UV-visible absorption bands for the ions [Ti(H\(_2\)O)\(_6\)]\(^{3+}\), [Mn(H\(_2\)O)\(_6\)]\(^{2+}\), [CrO\(_4\)]\(^{2-}\), and [NiCl\(_4\)]\(^{2-}\) is:
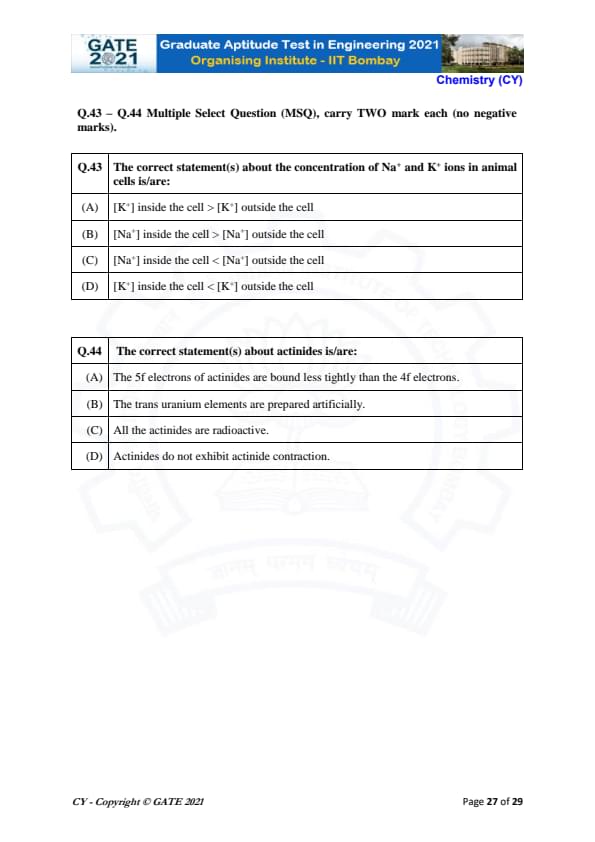
The correct statement(s) about the concentration of Na\(^+\) and K\(^+\) ions in animal cells is/are:
The correct statement(s) about actinides is/are:
The number of photons emitted per nanosecond by a deuterium lamp (400 nm) having a power of 1 microwatt (rounded off to the nearest integer) is ________.
Given: \( h = 6.626 \times 10^{-34} \, kg m^2 s^{-1}, c = 3.0 \times 10^8 \, m s^{-1} \).
Given the initial weight of 1 mg of radioactive \( ^{60} \)Co (half-life = 5.27 years), the amount disintegrated in 1 year (rounded off to two decimal places) is ________ mg.
The de Broglie wavelength of an argon atom (mass = 40 amu) traveling at a speed of 250 m/s (rounded off to one decimal place) is ________ picometers.
Given: \( N = 6.022 \times 10^{23}, h = 6.626 \times 10^{-34} \, kg m^2 s^{-1} \).
The molar absorption coefficient of a substance dissolved in cyclohexane is 1710 L mol\(^{-1}\) cm\(^{-1}\) at 500 nm. The reduction in intensity of light of the same wavelength that passes through a cell of 1 mm path length containing a 2 mmol L\(^{-1}\) solution (rounded off to one decimal place) is ________ %.
The fundamental vibrational frequency of \( ^1H^{127}I \) is 2309 cm\(^{-1}\). The force constant for this molecule (rounded off to the nearest integer) is ________ N m\(^{-1}\).
Given: \( N = 6.022 \times 10^{23}, c = 3.0 \times 10^8 \, m s^{-1} \).
A laser Raman spectrometer operating at 532 nm is used to record the vibrational spectrum of \( Cl_2 \) having its fundamental vibration at 560 cm\(^{-1}\). The Stokes line corresponding to this vibration will be observed at ________ cm\(^{-1}\).
The vapor pressure of toluene (Mol. Wt. = 92) is 0.13 atm at 25 °C. If 6 g of a hydrocarbon is dissolved in 92 g of toluene, the vapor pressure drops to 0.12 atm. The molar mass of the hydrocarbon (rounded off to the nearest integer) is ________.
The reaction
\[ CO (g) + Cl_2 (g) \rightleftharpoons COCl_2 (g) \]
at 500 °C, with initial pressures of 0.7 bar of CO and 1.0 bar of Cl_2, is allowed to reach equilibrium. The partial pressure of COCl_2 (g) at equilibrium is 0.15 bar. The equilibrium constant for this reaction at 500 °C (rounded off to two decimal places) is ________.
The rate constants for the decomposition of a molecule in the presence of oxygen are 0.237 \times 10^{-4} L mol\(^{-1}\) s\(^{-1}\) at 0 °C and 2.64 \times 10^{-4 L mol\(^{-1}\) s\(^{-1}\) at 25 °C. The activation energy for this reaction (rounded off to one decimal place) is ________ kJ mol\(^{-1}\).
Given: \( R = 8.314 \, J mol^{-1} K^{-1} \).
2 L of a gas at 1 atm pressure is reversibly heated to reach a final volume of 3.5 L. The absolute value of the work done on the gas (rounded off to the nearest integer) is ________ Joules.
The quantity of the cobalt ore [Co\(_3\)(AsO\(_4\))\(_2\)\( \cdot \) H\(_2\)O] required to obtain 1 kg of cobalt (rounded off to two decimal places) is ________ kg.
Given: Atomic Wt. of Co = 59, As = 75, O = 16, H = 1.


















Quick Links:
GATE 2021 CY: Paper Analysis
The question paper was full of easy questions. General Aptitude and Inorganic Chemistry questions were quite easy to attempt.
The below-mentioned table shows No. of questions asked as per the topics-
| Sections | No. Of Question Asked | Asked Topics |
|---|---|---|
| Inorganic Chemistry | 3-4 | Base Hydrolysis, A question on Chemical Bonding - CLF3 XeoF2 |
| Physical Spectra | 2-3 | Pericycle |
| Thermodynamics | 2-3 | - |
| Electrochemistry | 2-3 | Faraday First Law |
| Solutions | 2 (Numerical Questions) | - |
| Quantum | 2 | Acceptable Wave Function, De Broglie Wavelength |
| Chemical Equilibrium | 2 | Coordination, Reagent, Chemical Bonding, Geometry |
| F-Block | 1 | - |
- 40-50 attempted questions would be considered a good attempting rate
- Reagent holds the maximum weightage in the paper
- Questions from Physical Chemistry, Organic Chemistry, and Inorganic Chemistry considered the easiest questions to attempt
- Questions appeared from Quantum section were the most difficult questions to attempt
- Not even a single question asked from solid-state section
Also Check:
GATE 2021 CY: Exam Pattern and Marking Scheme
The conducting authorities had MSQs from the year 2021. MSQs comes with one or more than one correct answers. A student had to choose all the correct answers to solve MSQs correctly. Likewise, NATs no marks would be deducted if students attempt wrong or incorrect MSQs. MSQs, MCQs and NATs constituted the GATE 2021 CY. Each question holds either 1 or 2 marks. There were a total of 65 questions holding 100 marks.
- Candidates will have to pay penalty of ⅓ and ⅔ marks for every wrong question carried 1 and 2 marks
- Marks would be deducted only for the incorrect MCQs
- The cutoff for 2021 goes up to 36.2, 32.5, and 24.1 for General, OBC, and SC/ST candidates
GATE Previous Year Question Papers
| GATE 2022 Question Papers | GATE 2021 Question Papers | GATE 2020 Question Papers |
| GATE 2019 Question Papers | GATE 2018 Question Papers | GATE 2017 Question Papers |










Comments