GATE 2021 Engineering Sciences (XE) Question Paper with Solutions is now available to download. IIT Bombay successfully concluded the GATE 2021 XE exam. The Question Paper of GATE 2021 XE was a mixture of General Aptitude and core XE sections. There were a total of 65 questions with 10 questions from the General Aptitude section. The overall difficulty level of the GATE 2021 XE was moderate to difficult. The total weightage of GATE 2021 XE was distributed General Aptitude and core XE sections. 15% and 85% weightage were distributed among General Aptitude and core XE sections respectively.
GATE 2021 Engineering Sciences (XE) Question Paper with Solutions
| GATE 2021 Engineering Sciences (XE) Question Paper | Check Solutions |

Western Boundary Current in the ocean is primarily due to
The relevant nondimensional number in deciding deepening of the thermocline driven by instability of ocean currents is
During July-August, the highest number of monsoon low pressure systems form over
CO\(_2\) concentration in the Earth’s atmosphere is increasing because 50% of the annual anthropogenic emissions are retained in the atmosphere. If nations agree to reduce annual CO\(_2\) emissions by one Giga ton every year starting from 2021, then in which year will the CO\(_2\) concentration in the atmosphere stop rising due to anthropogenic emissions?
Take the anthropogenic CO\(_2\) emissions in 2020 as 40 Giga tons.
The figure shows a schematic of Indian Ocean surface circulation. This pattern is representative of the circulation in which month of the year?
Over the open ocean, if the air-sea temperature difference is zero, then which of the following statements is/are always true?
The psychrometric equation, which is useful in measuring humidity, is derived assuming the following process(es).
The water vapour mixing ratio of an air parcel increases from 10 g kg\(^{-1}\) to 20 g kg\(^{-1}\) at a constant pressure of 1010 hPa and temperature of 300 K. The change in virtual temperature is ________ K (to one decimal place).
The Ekman layer thickness, if turbulent diffusivity is 0.01 m\(^2\) s\(^{-1}\), is ________ m. Take Coriolis parameter to be \( 10^{-4} \) s\(^{-1}\). Calculate to the nearest integer.
The figure shows vertical variation of two chemicals P and Q measured in the Pacific Ocean. Identify the correct combination showing (P, Q) pair from the list below.
Consider tropical high-level clouds and low-level stratus clouds with bases at 12 km and 1 km above the surface of the Earth, respectively. Which of the following statement(s) is/are correct?
Which of the following statement(s) is/are correct in the context of Sverdrup transport?
Which of the following statement(s) is/are true with regard to the Hadley circulation?
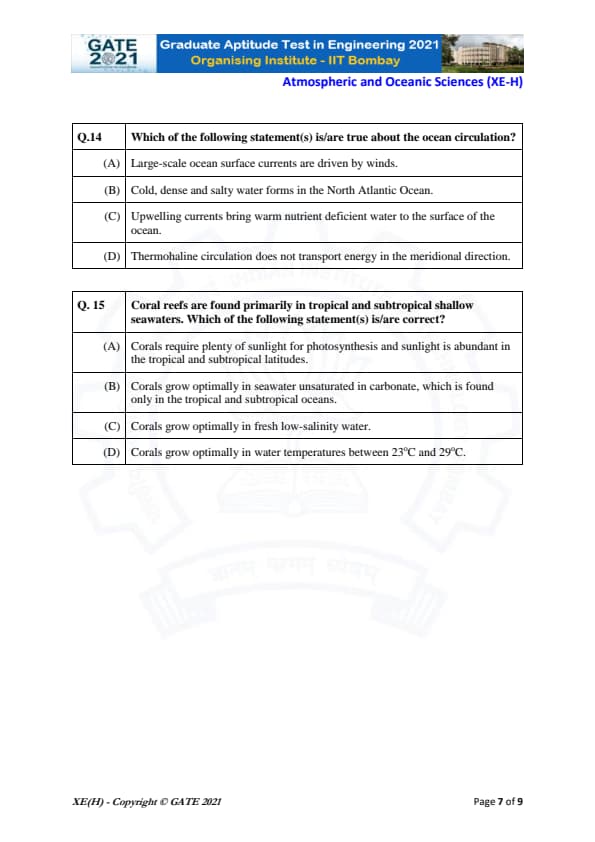
Which of the following statement(s) is/are true about the ocean circulation?
Coral reefs are found primarily in tropical and subtropical shallow seaways. Which of the following statement(s) is/are correct?
In an incompressible fluid, the horizontal divergence is -0.01 s\(^{-1}\). Then, the vertical velocity at 50 m above a flat surface is ________ m s\(^{-1}\). Round off to one decimal place.
In an atmosphere, temperature (T) decreases linearly with height above the ground (z), i.e., T(z) = T\(_0\) - \(\gamma\)z, where \(\gamma\) is a constant. Surface pressure is 900 hPa. If the atmosphere is at rest, then the value of z at which the pressure decreases to half of that at the surface is ________ m (rounded off to the nearest integer).
In a local Cartesian system, a zonal jet has a form u(y) = u\(_0\) (1 - y\(^2\)/L\(^2\)), for \(-L \le y \le L\). Here, y is the meridional distance measured from the axis of the jet and is positive northward. The vertical component of vorticity of this flow at y=L/2 is ________ s\(^{-1}\). Round off to 3 decimal places.
An eastward flow with a speed of 10 m/s goes from station M to station N, which are separated by a distance of 1 km. The temperature at station N is always higher than that at station M by 10 K. The absolute change in temperature due to advection at the mid-point between the stations in 50 s is ________ K (round off to nearest integer).
Suppose, because of the doubling of atmospheric CO\(_2\) concentration, an ocean water column receives an additional net energy input of 4 W/m\(^2\). If the entire water column of depth 1 km heats up uniformly, the water temperature will increase by 1 K in ______ years (round off to the nearest integer).
Consider a layer of atmosphere between 5 and 6 km height. The downwelling longwave radiation at 5 and 6 km is 240 and 230 Wm\(^{-2}\), respectively. The upwelling longwave radiation at these heights is 260 and 240 Wm\(^{-2}\), respectively. The longwave heating rate in this layer is ______ K per day. (Round off to one decimal place.)
A spherical asteroid, revolving around the sun in a circular orbit, is in radiative balance. Suddenly, the asteroid enters the shadow of a planet and solar radiation is cut off. Assuming that the asteroid emits as a blackbody in the longwave regime, the time taken to reduce the average temperature of the asteroid by 0.5 K is ______ seconds (rounded off to the nearest integer).
In a typical bacterial growth curve, the first order kinetics for growth rate is observed in
Which one of the following microorganisms is NOT a causative agent for food borne diseases?
Which of the following is NOT a fermented food product?
The Protein Efficiency Ratio (PER) is defined as
Which one of the following enzymes sequentially releases maltose from starch?
Which one of the following enzymes is involved in proteolysis of casein in cheese during aging?
Which one of the following compounds is present in soybean and acts as phytoestrogen?
Ultra high temperature (UHT) process of pasteurization of milk is achieved by heating at
Bittering agent in grapefruit formed after juice extraction under acidic conditions is
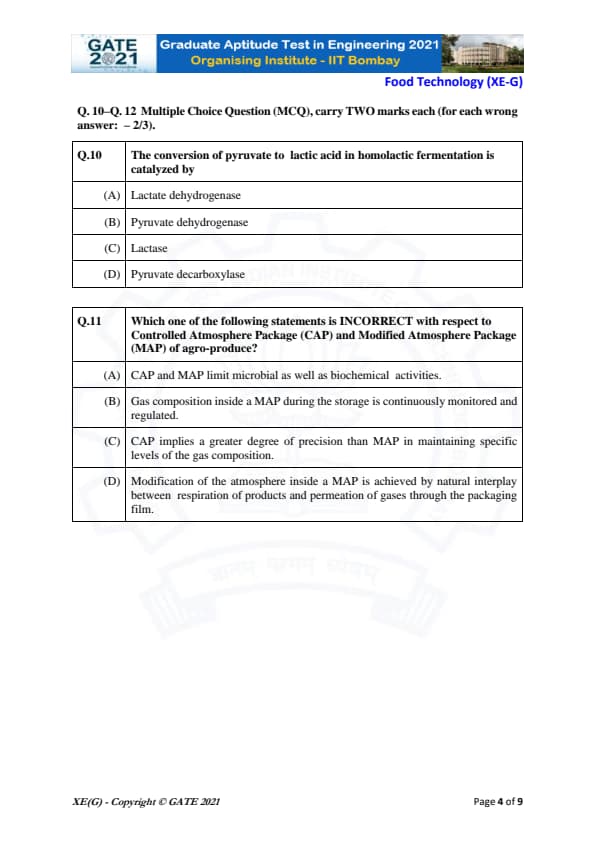
The conversion of pyruvate to lactic acid in homolactic fermentation is catalyzed by
Which one of the following statements is INCORRECT with respect to Controlled Atmosphere Package (CAP) and Modified Atmosphere Package (MAP) of agro-produce?
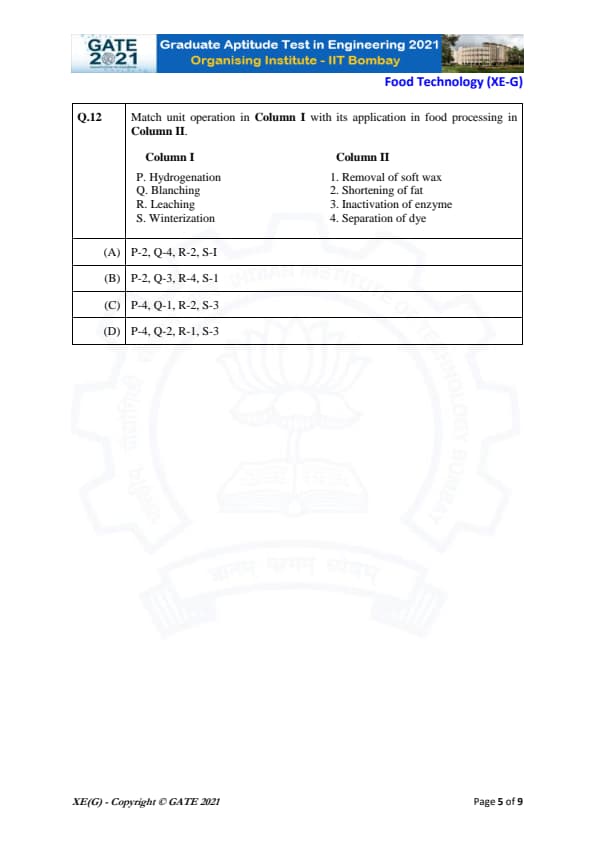
Match unit operation in Column I with its application in food processing in Column II.
Which of the following is the correct pair of GRAS chemical food preservative, affected organism, and given food matrix?
Choose the correct pair of pigment and their corresponding color in given plant product.
Which of the following compounds act as anti-nutritional factors?
Which of the followings is/are commonly used medium/media in the supercritical fluid extraction of spices and tea?
Which of the following expressions represent the Reynolds number of a fluid flowing through a uniform circular cross-section pipe?
Which of the following combinations of analytical equipment, property measured and food property are correct?
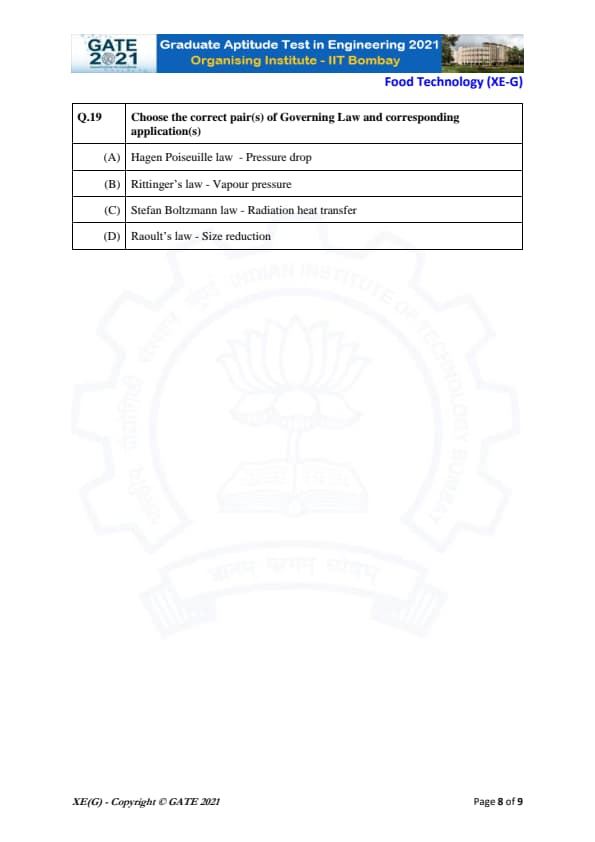
Choose the correct pair(s) of Governing Law and corresponding application(s)
An orange juice sample is concentrated from 10% to 40% (by weight) total soluble solids in a single effect evaporator with a feed rate of 3600 kg hr\(^{-1}\) at 25°C. The evaporator operates at sufficient vacuum to allow the product moisture to evaporate at 55°C. The specific heat of both feed and concentrated juice is 4.0 kJ kg\(^{-1}\) °C\(^{-1}\). If enthalpy of water vapour at 55°C is 2600 kJ kg\(^{-1}\), heat transfer rate through the heating surface area of the evaporator in kilowatt (in integer) will be ________.
Dry air is fed into a tray dryer. The percentage relative humidity of the air leaving the dryer is 60% at 70°C and 101.35 kPa. If saturated vapour pressure of water at 70°C is 31.2 kPa, the humidity of the air leaving the dryer in kg water per kg dry air (round off to 3 decimal places) will be ________.
In a cold storage plant, 5000 kg potato having a constant specific heat capacity of 3.65 kJ kg\(^{-1}\) °C\(^{-1}\) are cooled from 28°C to 2°C in 24 hours. The heat of respiration of potato per 24 hour is 3.12 kJ kg\(^{-1}\) during the storage. Assuming the efficiency of the storage plant to be 70%, the capacity of the plant in ton of refrigeration (round off to 2 decimal places) is ________.
Linear low density polyethylene (LLDPE) is a copolymer of ethylene and a small fraction of _________.
Binary polymer blends of polypropylene and polyamide 6 are immiscible. From a thermodynamic viewpoint this is due to \underline{\hspace{2cm.
Which one of the following is an elastomer?
Compression moulded isotropic polypropylene film exhibits
Which one of the following is an example of a biodegradable polymer?
Polymer crystals show a range of melting points in contrast to single melting point of crystals of small molecules, because _________.
When the rate of cooling is increased during the solidification process, the glass transition temperature of a polymer _________.
Equal and opposite forces of a constant magnitude \( F \) are applied at the two ends of a thin elastomeric rod, which is held at a temperature \( T_1 \) (where \( T_g < T_1 < T_m \), and \( T_g \) and \( T_m \) are the glass transition temperature and melting temperature respectively). If the temperature is increased to \( T_2 \) (where \( T_g < T_2 < T_m \) and \( T_2 > T_1 \)), the rod will \underline{\hspace{2cm.
The size of a coiled polymer chain in a dilute solution is \( R_G \) in a good solvent, \( R_I \) in an ideal solvent and \( R_P \) in a poor solvent. Select the correct ordering of sizes.
Match the Additive to its Function.
Match the polymer processing operation with respect to its typical range of shear rate.
Shear stress (\( \sigma \)) and shear viscosity (\( \eta \)) are plotted as functions of the shear rate (\( \dot{\gamma} \)) for idealized "solid-like with yielding (1)" and "liquid-like (2)" materials.
Associate the shear stress and viscosity plots with the appropriate material responses.
The plateau modulus of polystyrene has a value of 0.2 × 10\textsuperscript{6} Pa at 150 °C. Given, the density of polystyrene is 1.05 g/cm\textsuperscript{3}, the universal gas constant, \( R = 8.3 \, J K^{-1} mol^{-1} \), and the monomer molecular weight is 104 g/mol. The molecular weight between entanglements (rounded off to the nearest integer) of polystyrene chains is ________ g/mol.
A unidirectional composite of epoxy and carbon fiber of 50% by volume is made. The elastic modulus of epoxy and carbon fiber are 3.5 GPa and 350 GPa, respectively. The ratio (rounded off to one decimal place) of the modulus of the composite to the matrix modulus is ________.
A single screw extruder is operating at a rotational speed of 2 revolutions per second for the extrusion of a Newtonian polymer under open-discharge conditions (in absence of a die, the pressure drop, \( \Delta p = 0 \)). The extruder has a screw diameter, \( D = 5 \, cm \), a channel depth, \( H = 0.4 \, cm \), distance between flights, \( W = 1 \, cm \), and a helix angle, \( \theta = 20^\circ \). Assume the value of \( \pi = 3.14 \). The volumetric flow rate (rounded off to 2 decimal places) is _______ cm\textsuperscript{3/s.
At 215 °C, the viscosity of a polystyrene of molecular weight 250 × 10\textsuperscript{3} g/mol is 8.0 × 10\textsuperscript{3} Pa·s. The critical molecular weight of polystyrene, \( M_c = 35 \times 10^3 \) g/mol. For a similar polystyrene of molecular weight 500 × 10\textsuperscript{3 g/mol, the viscosity (rounded off to nearest integer) will be _______ × 10\textsuperscript{3 Pa·s.
There are two different PTFE polymer specimens of the following density \( \rho \) and % crystallinity. For PTFE-specimen-1, \( \rho = 2.144 \, g/cm^3 \) and % crystallinity is 50. For PTFE- specimen-2, \( \rho = 2.215 \, g/cm^3 \) and % crystallinity is 75. Assuming the polymer is pure and defect free, the density (rounded off to 3 decimal places) of 100% amorphous PTFE specimen will be _______ g/cm\textsuperscript{3.
The behavior of a polymer is described by a Maxwell model consisting of a spring element of modulus \( 10^{10} \) Pa in series with a dashpot of viscosity \( 10^{12} \) Pa.s. In the solid, 50 s after the sudden application of a fixed strain of 1%, the stress (rounded off to 2 decimal places) will be _______ \( \times 10^7 \) Pa.
A particular free radical polymerization process yields a polymer with a number averaged degree of polymerization, \( \overline{x_n} = 100 \). The monomer concentration is doubled and the initiator concentration is increased by four times. Assuming that all rate coefficients and other parameters remain unchanged, the value of \( \overline{x_n} \) (rounded off to the nearest integer) is _______.
A polymer is synthesized from 2 moles of terephthalic acid (molecular weight of the repeat unit, (-OCC6H4CO-), is 132 g/mol), 1 mol of ethylene glycol (molecular weight of the repeat unit, (-OCH2CH2O-), is 60 g/mol), and 1 mol of butylene glycol (molecular weight of the repeat unit, (-O(CH2)4O-), is 88 g/mol). The reaction is terminated at 99% conversion of the acid. The number averaged molecular weight, \( \overline{M_n} \) (rounded off to the nearest integer) is _______ g/mol.
A sample of natural rubber (cis-1,4-polyisoprene) is vulcanized such that one of every 240 chain carbon atoms is cross-linked. The formula unit of the isoprene monomer is \( C_5H_8 \) (molecular weight = 68 g/mol). The average molecular weight (rounded off to nearest integer) between cross-links is _______ g/mol.
A sample of an oriented semi-crystalline polymer is subjected to uniaxial tensile stress, \( \sigma \), in an X-ray diffractometer. The wavelength of X-ray radiation (Cu K\( \alpha \)) is \( \lambda = 1.542 \, Å \). The position of the (002) peak, which was found initially at a Bragg angle of 37.50° at \( \sigma = 0 \, MPa \), shifted to 37.45° at \( \sigma = 160 \, MPa \). Assuming elastic deformation, the strain (rounded off to three decimal places) in the sample along the direction of applied stress is _______ \( \times 10^{-3} \).
A refrigerator working on a reversed Carnot cycle has a Coefficient of Performance (COP) of 4. If it works as a heat pump and consumes work input of 1 kW, the heating effect will be:
The liquid phase of a pure substance is termed as _________, if its temperature is lower than the saturation temperature corresponding to its pressure \(P\).
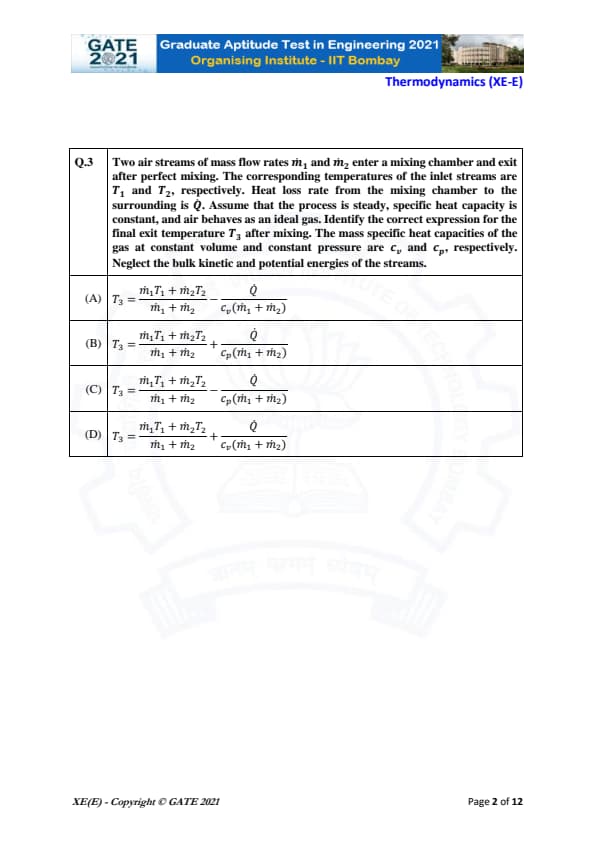
Two air streams of mass flow rates \( \dot{m_1} \) and \( \dot{m_2} \) enter a mixing chamber and exit after perfect mixing. The corresponding temperatures of the inlet streams are \( T_1 \) and \( T_2 \), respectively. Heat loss rate from the mixing chamber to the surrounding is \( \dot{Q} \). Assume that the process is steady, specific heat capacity is constant, and air behaves as an ideal gas. Identify the correct expression for the final exit temperature \( T_3 \) after mixing. The mass specific heat capacities of the gas at constant volume and constant pressure are \( c_v \) and \( c_p \), respectively. Neglect the bulk kinetic and potential energies of the streams.
If
h \text{ is the mass specific enthalpy,
s \text{ is the mass specific entropy,
P is the pressure,
T is the temperature,
C_V \text{ is the mass specific heat at constant volume,
C_P \text{ is the mass specific heat at constant pressure,
\beta \text{ is the coefficient of thermal expansion,
v \text{ is the mass specific volume,
\kappa \text{ is the isothermal compressibility,
\text{then the partial derivative \left( \frac{\partial h{\partial s \right)_P \text{ is
If
v is the mass specific volume,
s \text{ is the mass specific entropy,
P \text{ is the pressure,
T \text{ is the temperature,
then using Maxwell relations, \[ \left( \frac{\partial s{\partial P} \right)_T = \]
A closed system consists of a solution of liquid water and ethanol in equilibrium with its vapours. Using the Gibbs phase rule, the degree of freedom of the system is:
For a real gas passing through an insulated throttling valve, the outlet temperature of the gas \underline{\hspace{2cm with respect to the inlet temperature.
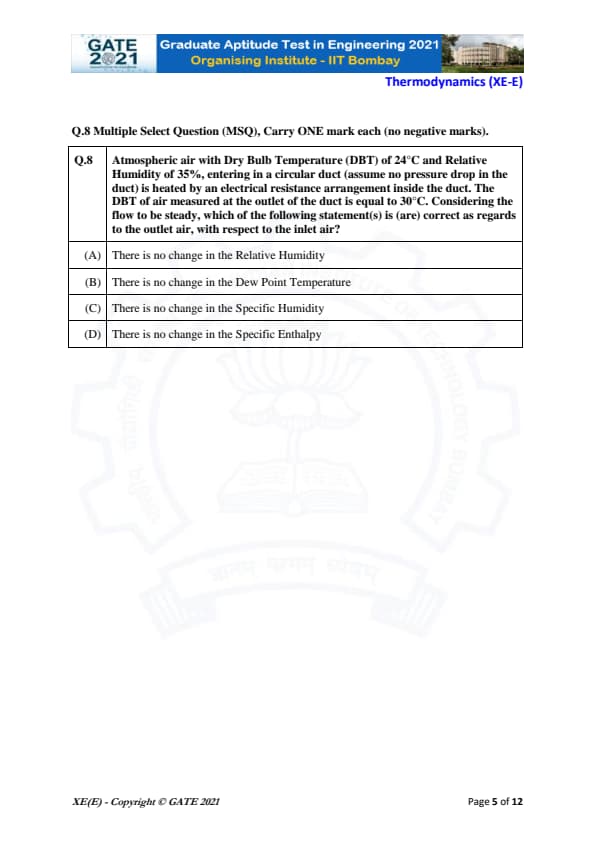
Atmospheric air with Dry Bulb Temperature (DBT) of 24°C and Relative Humidity of 35%, entering in a circular duct (assume no pressure drop in the duct) is heated by an electrical resistance arrangement inside the duct. The DBT of air measured at the outlet of the duct is equal to 30°C. Considering the flow to be steady, which of the following statement(s) is (are) correct as regards to the outlet air, with respect to the inlet air?
A cylinder of volume 1 m\(^3\) contains a mixture of CO\(_2\) (20% by mol) and O\(_2\) (80% by mol) at 100 kPa and 300 K. This cylinder is connected to a 1 MPa pressure line carrying N\(_2\) at 300 K. The cylinder is filled isothermally till the pressure of gas mixture inside it becomes 500 kPa, and then the filling is stopped. The amount of N\(_2\) gas that has entered the cylinder is _________ (in mole, 2 decimal places).
The universal gas constant is 8.3145 J/(mol K).
The saturation pressure \(P_{sat}\) of a pure liquid is represented by an equation of the form: \[ \ln P_{sat} = A - \frac{B}{T}, \]
where \(A\) and \(B\) are constants, and \(T\) is the absolute temperature. For this substance, which of the following expression for the specific entropy difference between the saturated vapour and the saturated liquid phase \((s_{fg})\) is correct?
For a refrigeration cycle, the ratio of actual COP to the COP of a reversible refrigerator operating between the same temperature limits is 0.8. The condenser and evaporator temperatures are \(51^\circ C\) and \(-30^\circ C\), respectively. If the cooling capacity of the plant is 2.4 kW, then the power input to the refrigerator is:
Two identical pressure cookers, Cooker A and Cooker B, each having a total internal capacity of 6 litres are available. Cooker A is filled with 2 litres of liquid water at 110°C and Cooker B is filled with 4 litres of liquid water at 110°C. The remaining space in both the cookers is filled with saturated water vapour in equilibrium with the liquid water. If \( g \) represents the specific Gibbs free energy, and subscripts \( v \) and \( l \) represent the saturated vapour and the saturated liquid phases, respectively, which of the following expressions is correct?
Four different Entropy (S) - Temperature (T) diagrams, representing liquid to vapour phase transition process of a pure substance in a closed system under constant pressure are shown. The diagram, which correctly represents the process, is:
Air having a mass flow rate of 2 kg/s enters a diffuser at 100 kPa and 30°C, with a velocity of 200 m/s. Exit area of the diffuser is 400 cm\textsuperscript{2} while the exit temperature of the air is 45°C. The rate of heat loss from the diffuser to the surrounding is 8 kJ/s. The pressure at the diffuser exit is _______ kPa (2 decimal places).
For the Refrigerant R-134 (at 1 MPa and 50°C), the difference between the specific volume computed by assuming it to be an ideal gas and its actual specific volume is: \( v_{ideal} - v_{actual} = 4.529 \times 10^{-3} \, m^3/kg \). If the compressibility factor associated with this state is \( Z = 0.84 \), then \( v_{com} - v_{actual} = \_\_\_\_\_\_\_ \times 10^{-3} \, m^3/kg \) (3 decimal places).
A mixture of air and water vapour enters a steady-flow adiabatic saturator at 50°C and 100 kPa. It leaves the saturator in a completely saturated state at temperature 25°C and pressure of 100 kPa. Liquid water enters the saturator at 25°C. If air is considered to be an ideal gas, with constant specific heat capacity, the relative humidity of the air entering the saturator is _______ % (1 decimal place).
Air at a pressure of 1 MPa and 300 K is flowing in a pipe. An insulated evacuated rigid tank is connected to this pipe through an insulated valve. The volume of the tank is 1 m\textsuperscript{3}. The valve is opened and the tank is filled with air until the pressure in the tank is 1 MPa. Subsequently, the valve is closed. Consider air to be an ideal gas and neglect bulk kinetic and potential energy. The final temperature of air in the tank is _______ K (1 decimal place).
A cylinder of volume 0.1 m\textsuperscript{3} is filled with 100 mol of propane (C\textsubscript{3}H\textsubscript{8}) at 2 MPa. If propane is assumed to obey the van der Waals equation of state, then its temperature is _______ K (1 decimal place).
A frictionless piston cylinder device contains 1 kg of an ideal gas. The gas is compressed according to \( P v^{1.3} = constant \) (P is pressure and v is mass specific volume), from 100 kPa, 250 K, till it reaches a temperature of 500 K. The heat transfer from the piston cylinder device to its surroundings is _______ kJ (2 decimal places).
A 0.8 m\textsuperscript{3} insulated rigid tank contains 1.5 kg of an ideal gas at 100 kPa. Electric work is done on the system until the pressure in the tank rises to 135 kPa. The loss in availability (exergy) associated with the process is _______ kJ (2 decimal places).
A rigid tank contains 1.0 kg of pure water consisting of liquid and vapour phases in equilibrium at 10 bar. If the liquid and vapour phase each occupies one half of the volume of the tank, then the net enthalpy of the contents of the tank is _______ kJ (1 decimal place).
An air-standard Diesel cycle with a compression ratio of 16 takes air at 1 bar and 300 K. If the maximum temperature in the cycle is 2100 K, then the thermal efficiency of the cycle is _________ % (1 decimal place).
The ratio of the specific heat capacities of air is 1.4.
A force \( F = 40 \, kN \) is applied on the hook as shown. The equivalent force-couple system at B is
A rigid rod OA rotates clockwise at an angular velocity of 10 rad/s. A bead B (OB = 1 m) translates outward on the rod at a speed of 5 m/s and acceleration 2.5 m/s² (both quantities with respect to the rod). The Coriolis component of acceleration is
A two force member in equilibrium is one in which
If the yield point shear stress obtained from the torsion test of a cylindrical specimen is \( \tau_y \), then what is the maximum value of principal strain at yielding? (\( \mu \) is Poisson’s ratio and \( E \) is Young’s modulus)
If the ratio of Young’s modulus to bulk modulus of a material is \( \frac{3}{2} \), then the ratio of shear modulus to the Young’s modulus of the material is
With respect to the plane of maximum shear stress, which of the following statements is INCORRECT?
A simply supported beam of length L is loaded by two symmetrically applied point loads P at \( L/3 \) from each support. Both the loads are then shifted to new points which are at a distance \( L/4 \) from each support. The bending moments at the mid-section of the beam in both the cases are same. The magnitude of \( P_1 \) in terms of P is:
A beam having rectangular cross section is subjected to transverse loading. The ratio of maximum shear stress developed in the beam to the average shear stress is:
During an earthquake, a structure vibrates and the vibration can be assumed to be in simple harmonic motion at 5 Hz. At a measurement point, the RMS value of acceleration is 10 m/s\(^2\). The approximate amplitude of motion (in mm) at this point (rounded off to two decimal places) is _________.
For the state of plane stress shown, the components of normal and shear stresses are given in terms of stress \( \sigma \) and unknown constants \( m \) and \( n \). If the normal and shear components of stress on a 45° plane are \( 2\sigma \) and zero, the values of \( m \) and \( n \) would be:
For a state of plane strain, the normal strains are given by
\epsilon_{xx = 1000 \times 10^{-6, \quad \epsilon_{yy = 200 \times 10^{-6, \quad \text{and the maximum shear strain is \, \gamma_{\text{max = 1000 \times 10^{-6. \quad \text{The value of shear strain \, \gamma_{xy \, \text{for this strain state is
A thin cylinder (closed at its ends) of radius \( r \) and thickness \( t \) (\( r \gg t \)) is subjected to internal pressure \( p \). The maximum shear stress in the wall of the cylinder is
The truss shown is subjected to a force \( P \). All members of the truss have the same length \( L \). The reaction at A and force in member AB are
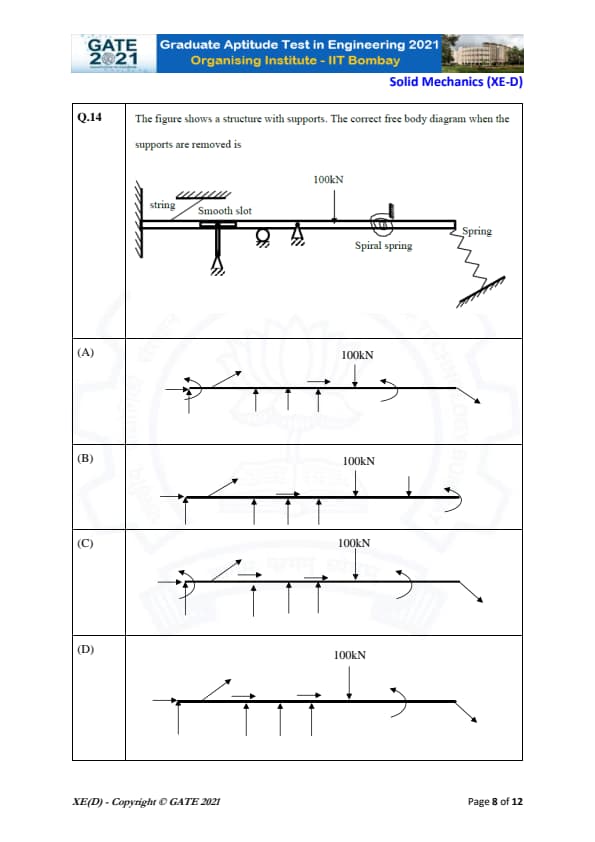
The figure shows a structure with supports. The correct free body diagram when the supports are removed is
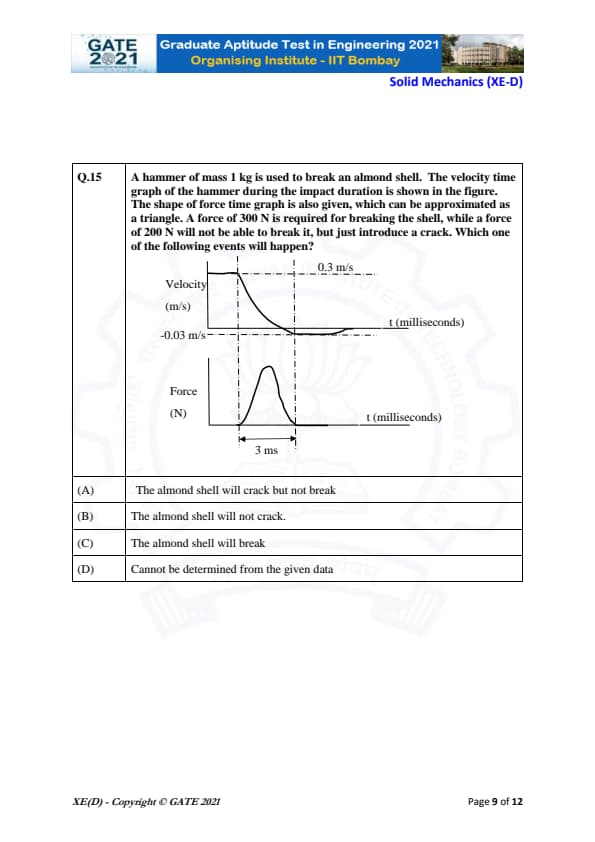
A hammer of mass 1 kg is used to break an almond shell. The velocity time graph of the hammer during the impact duration is shown in the figure. The shape of force time graph is also given, which can be approximated as a triangle. A force of 300 N is required for breaking the shell, while a force of 200 N will not be able to break it, but just introduce a crack. Which one of the following events will happen?
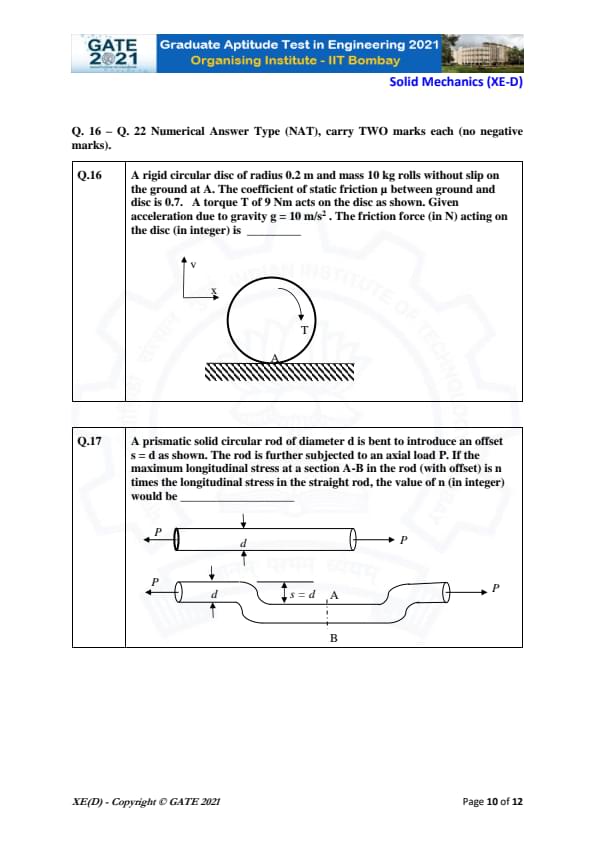
A rigid circular disc of radius 0.2 m and mass 10 kg rolls without slip on the ground at A. The coefficient of static friction \( \mu \) between ground and disc is 0.7. A torque \( T \) of 9 Nm acts on the disc as shown. Given acceleration due to gravity \( g = 10 \, m/s^2 \). The friction force (in N) acting on the disc (in integer) is _______.
A prismatic solid circular rod of diameter \( d \) is bent to introduce an offset \( s = d \) as shown. The rod is further subjected to an axial load \( P \). If the maximum longitudinal stress at a section A-B in the rod (with offset) is \( n \) times the longitudinal stress in the straight rod, the value of \( n \) (in integer) would be _______.
A naturally curved steel beam AB having Young’s modulus 208 GPa, area moment of inertia \( I = 26.7 \, cm^4 \) and radius \( R = 2 \, m \) is subjected to a vertical load \( P = 1000 \, N \) at B. The end A at \( \theta = 90^\circ \) is rigidly fixed. The bending strain energy of the beam (in Nm, rounded off to two decimal places) is _________.
At room temperature of 25°C, a gap of 1 mm exists between the ends of the rods 1 and 2 as shown. Given the cross section area \( A \) of the rods is \( 1500 \, mm^2 \), Young’s modulus \( E = 75 \, GPa \), and the coefficient of thermal expansion \( \alpha = 23 \times 10^{-6} \, °C^{-1} \). When the temperature has reached 150°C, the magnitude of normal stress in each of the rods (in MPa, rounded off to two decimal places) is _________.
A tube of inner radius 4 cm and outer radius 5 cm can carry a maximum torque of T. This tube is now replaced by a solid circular shaft of the same material. The minimum radius of the solid circular shaft (in cm, rounded off to two decimal places) to carry the same amount of torque \( T \) is _________.
In System A, a rectangular block of mass M is centrally supported on a spring of stiffness K as shown. In System B, the mass is hinged at one of its ends and is supported centrally by the spring. The ratio of natural frequency of System B to that of System A (rounded off to two decimal places) is ________.
A coronavirus droplet of mass 1 microgram ejects from the mouth of a patient with a velocity of 0.7 m/s and travels through air. The gravitational force experienced by it can be neglected due to the buoyancy effect. However, the droplet experiences air drag force proportional to its velocity and the drag coefficient is given as 1.0 μN-s/m. The distance travelled by the droplet before its velocity drops to 10% of its initial velocity (in m, rounded off to two decimal places) is _______.
Condition to be satisfied for \( \alpha \) and \( \beta \) phases to be in equilibrium in a two-component (A and B) system at constant temperature and pressure is
Amino acids react to form peptides and proteins. This process is known as
The most favoured slip system in face centered cubic metal is
The dielectric constant of a material at ultraviolet frequencies is mainly due to
Match the different transformations/reactions in Column I with the most suitable information in Column II.
In scanning electron microscopy, the resolution of backscattered electron (BSE) image is poorer compared to that of secondary electron (SE) image, because
Which of the following deposition conditions favour the formation of larger grains in thin film?
Atmospheric air with Dry Bulb Temperature (DBT) of 24°C and Relative Humidity of 35%, entering in a circular duct (assume no pressure drop in the duct) is heated by an electrical resistance arrangement inside the duct. The DBT of air measured at the outlet of the duct is equal to 30°C. Considering the flow to be steady, which of the following statement(s) is (are) correct as regards to the outlet air, with respect to the inlet air?
A magnet of mass 50 g has a magnetic moment of \( 4.2 \times 10^{-7} \, A m^2 \). The density of the magnet is 7.2 g/cm\textsuperscript{3. The intensity of magnetization in A/m is _______ (round off to 3 decimal places).
In the context of scanning electron microscopy, match the information in Column I with the most appropriate information in Column II.
Match the heat treatment processes given in Column I with the most suitable outcomes in Column II.
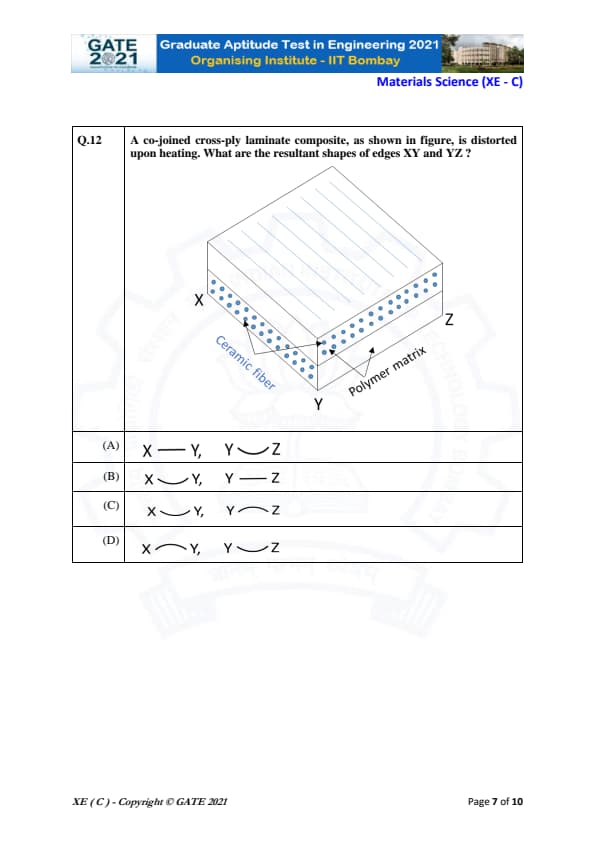
A co-joined cross-ply laminate composite, as shown in the figure, is distorted upon heating. What are the resultant shapes of edges XY and YZ?
X-ray diffraction peak broadening enables the estimation of
Fe - 10 atom % C austenite (fcc), having no Fe vacancies, has a lattice parameter of 4 Å. The density of austenite in g/cm\textsuperscript{3} is _______ (round off to 2 decimal places).
An element transforms from \( \alpha \) to \( \beta \) at 773 K and 1 atm pressure with 912 J mol\(^{-1}\) as enthalpy of transformation. The molar volumes of \( \alpha \) and \( \beta \) phases are 7.377 cm\textsuperscript{3 and 7.317 cm\textsuperscript{3, respectively. Assume that the difference in molar volumes of \( \alpha \) and \( \beta \) is independent of pressure. The pressure (in atm) required for \( \alpha \) to \( \beta \) transformation to occur at 723 K is _______ (round off to nearest integer).
A binary A-B alloy has \( \alpha \) and \( \beta \) phases at equilibrium. The ratio of weight percentages (wt.%) of \( \alpha \) to \( \beta \) is 4. The wt.% of A in \( \alpha \) and \( \beta \) phases is 70 and 20, respectively. The wt.% of B in the alloy is _______ (round off to nearest integer).
During heating, Ti undergoes allotropic transformation from hcp to bcc at 882 °C. The percent volume change accompanying this transformation is _______ (round off to 1 decimal place).
Vickers hardness test is performed with an indenter of square-base diamond pyramid having an included angle of 136° between the opposite faces of the pyramid. If the applied load is 10 kg and the average length of diagonals of square indentation is 0.5 mm, the Vickers hardness in kg/mm\textsuperscript{2} is _______ (round off to nearest integer).
The drift mobility of electron in an n-type Si crystal doped with \( 10^{16} \, cm^{-3} \) phosphorous atoms is ______ (round off to nearest integer).
At 1000 K, the linear thermal expansion coefficients of graphite, parallel and perpendicular to the graphite layers, are \( 0.8 \times 10^{-6} \, K^{-1} \) and \( 29 \times 10^{-6} \, K^{-1} \), respectively. The percentage increase in the volume of graphite when heated from 900 K to 1100 K is _______ (round off to 2 decimal places).
A certain ceramic has a theoretical density and sintered density of \( 6.76 \, g/cm^3 \) and \( 6.60 \, g/cm^3 \), respectively. The green compact has 18 volume percent porosity. For a sintered cube of side 2 cm, the required side of the cubic green compact in cm is _______ (round off to 2 decimal places).
When a metal (M) is immersed in de-aerated acid electrolyte, it polarizes anodically by 0.4 V. The M/M\(^{n+}\) exchange current density is \( 10^{-5} \, A m^{-2} \) and Tafel slope is 0.1 V/decade for the anodic reaction. Assume that corrosion is uniform and anodic and cathodic reactions are under activation control. The rate of metal dissolution in A m\textsuperscript{-2 is _______ (round off to 1 decimal place).
The general relationship between shear stress, \( \tau \), and the velocity gradient \[ \tau = k \left( \frac{du}{dy} \right)^n, \]
where \( k \) is a constant with appropriate units. The fluid is Newtonian if
Which one of the following options is TRUE?
If \( P_{in} = 1.2 \, Pa \) and \( P_{out} = 1.0 \, Pa \) are the average pressures at inlet and outlet respectively for a fully-developed flow inside a channel having a height of 50 cm, then the absolute value of average shear stress (in Pa) acting on the walls of the channel of length 5 m is
Consider the fully-developed flow of a Newtonian fluid (density \( \rho \); viscosity \( \mu \)) through a smooth pipe of diameter \( D \) and length \( L \). The average velocity of the flow is \( V \). If the length of the pipe is doubled, keeping \( V \), \( D \), \( \rho \), \( \mu \) constant, the friction factor
The absolute value of pressure difference between the inside and outside of a spherical soap bubble of radius \( R \), and surface tension \( \gamma \), is:
Which one of the following statements is TRUE about the continuity equation \( \frac{\partial u}{\partial x} + \frac{\partial v}{\partial y} + \frac{\partial w}{\partial z} = 0 \) (where \( u \), \( v \), \( w \) are the velocity components along the \( x \), \( y \), and \( z \) coordinates respectively):
The head loss (\( K_L \)) associated with the flow entry of water to an internal passage depends on the shape of the entry. The following figure shows three different types of flow entry into a pipe. Which one of the following relationships correctly represents the head loss associated with the three different flow entries?
The form and friction drags together contribute to the total drag when flow of air occurs past any object. Two orientations of a finite flat plate are shown in the figure. In Orientation-1, the plate is placed perpendicular to the flow while in Orientation-2, the plate is placed parallel to the flow. If the velocity (\( V \)) of air in both orientations is the same, which one of the following options is TRUE?
A spherical ball is steadily supported against gravity by an upward air jet as shown in the figure. Take acceleration due to gravity to be \( g = 10 \, m/s^2 \). The mass flow rate of air, reaching the ball, is 0.01 kg/s and the air reaches the ball at an upward velocity of 3 m/s. Neglecting the buoyancy force, and using the principle of integral momentum balance, the mass (in grams, up to one decimal place) of the ball is ______.
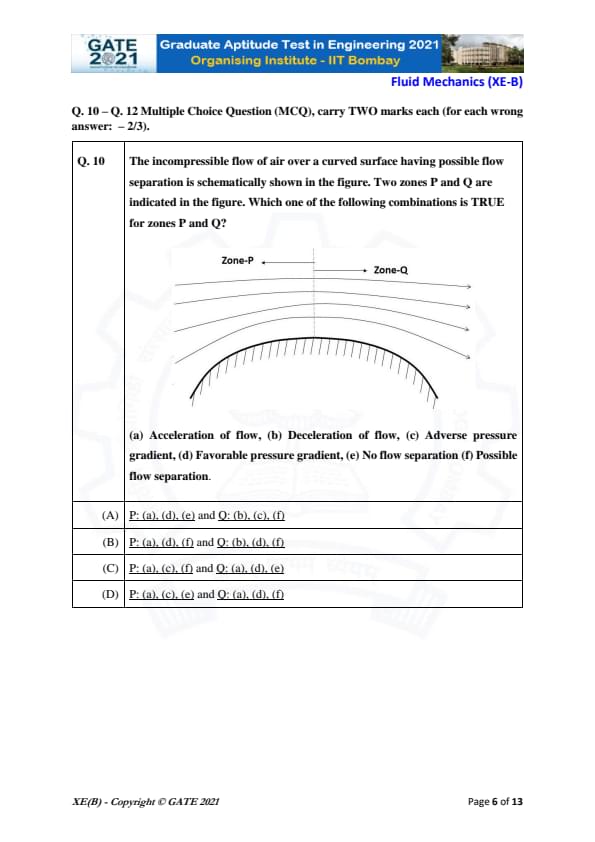
The incompressible flow of air over a curved surface having possible flow separation is schematically shown in the figure. Two zones P and Q are indicated in the figure. Which one of the following combinations is TRUE for zones P and Q?
A spherical metal ball (of density \( \rho_s \) and diameter \( D \)), attached to a string, is exposed to a crossflow (of velocity \( U_\infty \)) of a viscous fluid (of viscosity \( \mu \) and density \( \rho_f \)). Due to the crossflow, the string makes an angle of inclination \( \theta \) with the top surface as shown in the figure. The acceleration due to gravity is denoted by \( g \). For this flow, Reynolds number, \( Re = \frac{\rho_f U_\infty D}{\mu} \ll 1 \) and buoyancy force in the fluid is negligible compared to viscous force. Assuming the string to be weightless and offering negligible drag, the expression for \( \theta \) is
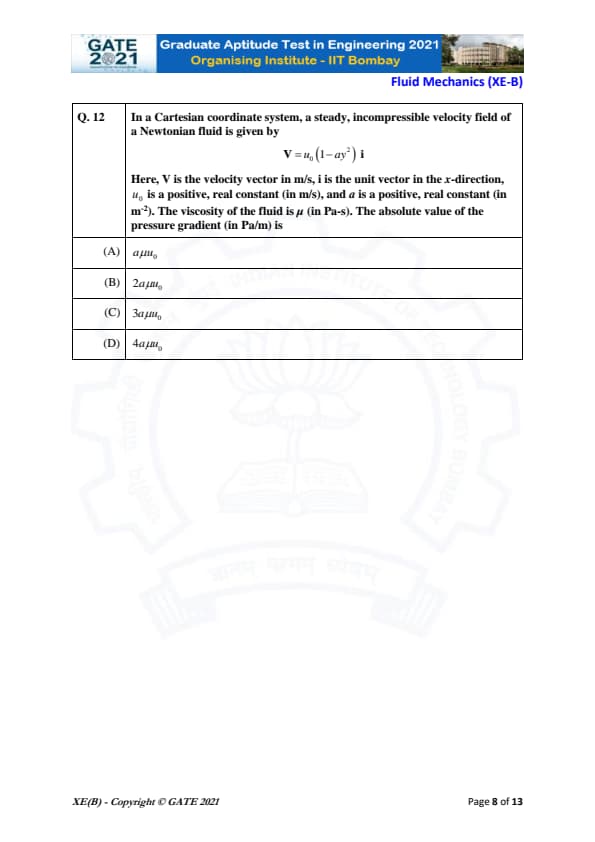
In a Cartesian coordinate system, a steady, incompressible velocity field of a Newtonian fluid is given by
\[ V = u_0 \left( 1 - a y^2 \right) \hat{i} \]
Here, \( V \) is the velocity vector in m/s, \( \hat{i} \) is the unit vector in the x-direction, \( u_0 \) is a positive, real constant in m/s, and \( a \) is a positive, real constant in m\(^{-2}\). The viscosity of the fluid is \( \mu \) in Pa-s. The absolute value of the pressure gradient (in Pa/m) is
In a laminar, incompressible, fully-developed pipe flow of a Newtonian fluid, as shown in the figure, the velocity profile over a cross-section is given by \( u = U \left( 1 - \frac{r^2}{R^2} \right) \), where \( U \) is a constant. The pipe length is \( L \) and the fluid viscosity is \( \mu \). The power \( P \) required to sustain the flow is expressed as \( P = c \mu L U^2 \), where \( c \) is a dimensionless constant. The value of the constant \( c \) (up to one decimal place) is ______.
The two-dimensional velocity field \( \mathbf{V} \) of a flow in a Cartesian coordinate system is given in dimensionless form by \( \mathbf{V} = (x^2 - axy) \hat{i} + \left( bxy - \frac{y^2}{2} \right) \hat{j} \). Here, \( \hat{i} \) and \( \hat{j} \) are the unit vectors along the \( x \) and \( y \) directions respectively, \( a \) and \( b \) are independent of \( x \), \( y \) and time. If the flow is incompressible, then the value of \( (a - b) \), up to one decimal place, is ______.
For the configuration shown in the figure, oil of density 800 kg/m\(^3\) lies above water of density 1000 kg/m\(^3\). Assuming hydrostatic conditions and acceleration due to gravity \( g = 10 \, m/s^2 \), the length \( L \) (in meters, up to one decimal place) of water in the inclined tube is _________.
A two-dimensional Eulerian velocity field is given (in m/s) by \( \mathbf{V} = \left[ (\sqrt{5})x \right] \hat{i} - \left[ (\sqrt{12})y \right] \hat{j} \), where \( x \) and \( y \) are the coordinates (in meters) in a Cartesian coordinate system. The magnitude of the acceleration (in m/s\(^2\), up to one decimal place) of a fluid particle at \( x = 1 \, m \) and \( y = -1 \, m \) is _________.
A large pump is to deliver oil at an average velocity \( V = 1.5 \, m/s \). The pump has an impeller diameter \( D \) of 40 cm and the pressure rise across the pump is 400 kPa. To design this pump, a lab-scale model pump with an impeller diameter of 4 cm is to be used with water as the fluid. The viscosity \( \mu \) of the oil is 100 times that of water, and the densities \( \rho \) of oil and water are identical. A complete geometric similarity is maintained between the model and prototype. If the pressure rise is a function only of \( V, D, \rho \) and \( \mu \), the pressure rise (in kPa, up to one decimal place) across the model pump is _________.
Water (density = \( 10^3 \, kg/m^3 \)) enters steadily into a horizontal pipe bend, which is part of a larger piping system, as shown in the figure. The volumetric flow rate of water is 0.1 m\(^3\)/s. The gage pressure at the inlet is 500 kPa, while the exit is open to atmosphere. The \( x \)-component of the force on the support is \( F_x \). The absolute value of \( F_x \) (in kN, up to one decimal place) is ______.
Air (of density 0.5 kg/m\(^3\)) enters horizontally into a jet engine at a steady speed of 200 m/s through an inlet area of 1.0 m\(^2\). Upon entering the engine, the air passes through the combustion chamber and the exhaust gas exits the jet engine horizontally at a constant speed of 700 m/s. The fuel mass flow rate added in the combustion chamber is negligible compared to the air mass flow rate. Also neglect the pressure difference between the inlet air and the exhaust gas. The absolute value of the horizontal force (in kN, up to one decimal place) on the jet engine is ______.
Water discharges from a cylindrical tank through an orifice, as shown in the figure. The flow is considered frictionless. Initially, the water level in the tank was \( h_1 = 2 \, m \). The diameter of the tank is \( D = 1 \, m \), while the diameter of the jet is \( d = 10 \, cm \), and the acceleration due to gravity is \( g = 10 \, m/s^2 \). The time taken (in seconds, up to one decimal place) for the water level in the tank to come down to \( h_2 = 1 \, m \) is _________.
Water discharges steadily from a large reservoir through a long pipeline, as shown in the figure. The Darcy friction factor in the pipe is 0.02. The pipe diameter is 20 cm and the discharge of water is 360 m\(^3\)/h. Water level in the reservoir is 10 m and acceleration due to gravity is \( g = 10 \, m/s^2 \). If minor losses are negligible, the length \( L \) (in meters, up to one decimal place) of the pipeline is _________.
Water is flowing with a flow rate \( Q \) in a horizontal circular pipe. Due to the low pressure created at the venturi section (Section-1 in the figure), water from a reservoir is drawn upward using a connecting pipe as shown in the figure. Take acceleration due to gravity \( g = 10 \, m/s^2 \). The flow rate \( Q = 0.1 \, m^3/s \), \( D_1 = 8 \, cm \), and \( D_2 = 20 \, cm \). The maximum height \( h \) (in meters, up to one decimal place) of the venturi from the reservoir just sufficient to raise the liquid up to Section-1 is ______.
Gauri said that she can play the keyboard _________ her sister.
A transparent square sheet shown above is folded along the dotted line. The folded sheet will look like _________.
If \( \theta \) is the angle, in degrees, between the longest diagonal of the cube and any one of the edges of the cube, then \( \cos \theta = \)
If \[ \left( x - \frac{1}{2} \right)^2 - \left( x - \frac{3}{2} \right)^2 = x + 2, then the value of x is: \]
Pen : Write :: Knife : _________
Which one of the following options maintains a similar logical relation in the above?
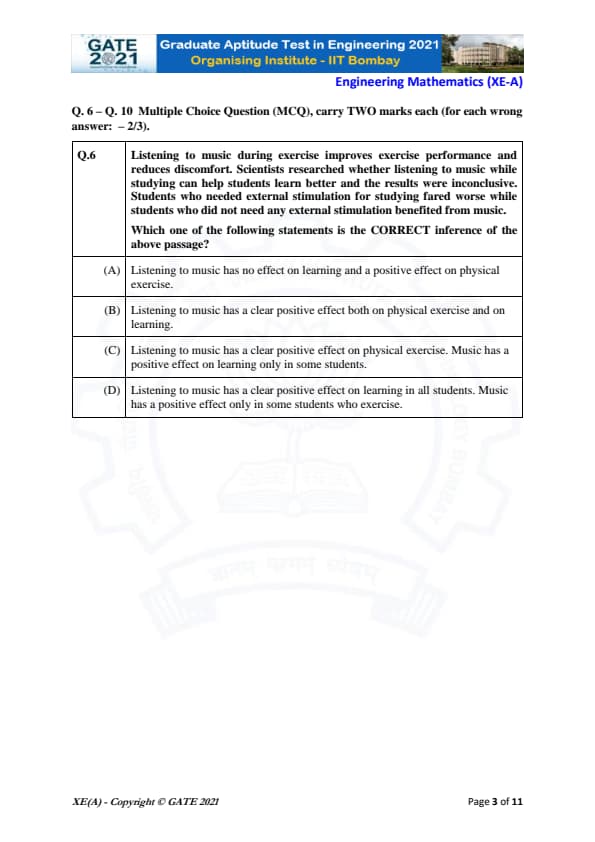
Listening to music during exercise improves exercise performance and reduces discomfort. Scientists researched whether listening to music while studying can help students learn better and the results were inconclusive. Students who needed external stimulation for studying fared worse while students who did not need any external stimulation benefited from music.
Which one of the following statements is the CORRECT inference of the above passage?
A jigsaw puzzle has 2 pieces. One of the pieces is shown above. Which one of the given options for the missing piece, when assembled, will form a rectangle? The piece can be moved, rotated, or flipped to assemble with the above piece.
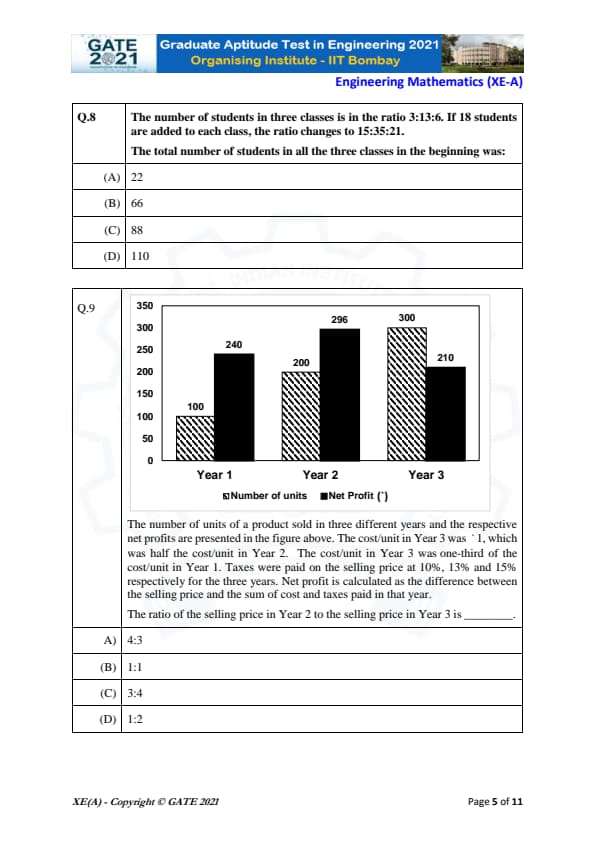
The number of students in three classes is in the ratio 3:13:6. If 18 students are added to each class, the ratio changes to 15:35:21. The total number of students in all the three classes in the beginning was:
The number of units of a product sold in three different years and the respective net profits are presented in the figure above. The cost/unit in Year 3 was `1, which was half the cost/unit in Year 2. The cost/unit in Year 3 was one-third of the cost/unit in Year 1. Taxes were paid on the selling price at 10%, 13% and 15% respectively for the three years. Net profit is calculated as the difference between the selling price and the sum of cost and taxes paid in that year. The ratio of the selling price in Year 2 to the selling price in Year 3 is _________.
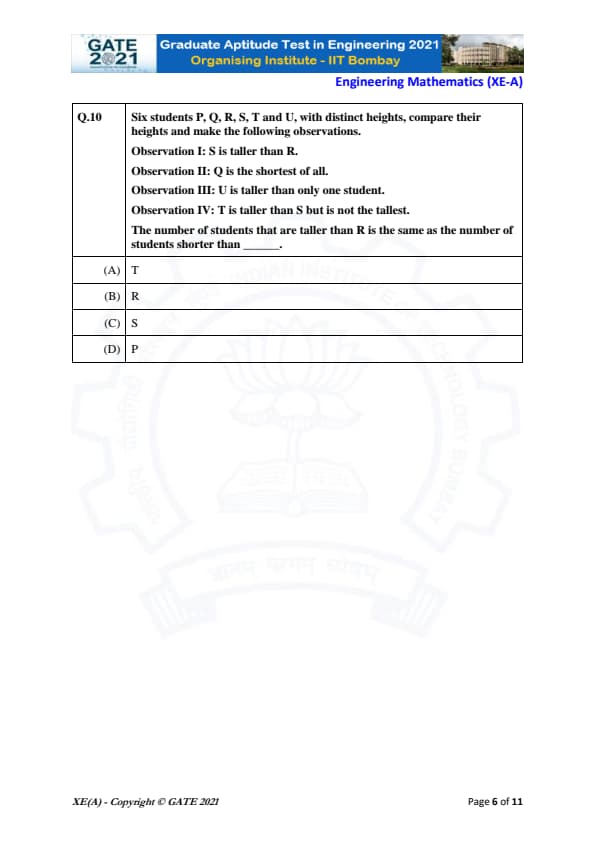
Six students P, Q, R, S, T and U, with distinct heights, compare their heights and make the following observations.
% Observation I
Observation I: S is taller than R.
Observation II: Q is the shortest of all.
Observation III: U is taller than only one student.
Observation IV: T is taller than S but is not the tallest.
The number of students that are taller than R is the same as the number of students shorter than _________.
Let \[ S = \left\{ AX : A = \begin{bmatrix} 2 & -4
1 & 1
1 & -1 \end{bmatrix}, \, X = \begin{bmatrix} x_1
x_2 \end{bmatrix} \right\}. \]
If \[ \begin{bmatrix} -1
\alpha
1 \end{bmatrix} \in S, then the value of \alpha is \]
Let \( C \) be the boundary of the region \( R : 0 \leq x \leq \pi, 0 \leq y \leq \sin x \) in the xy-plane and \( \alpha \) be the area of the region \( R \). If \( C \) traverses once in the counterclockwise direction, then the value of the line integral \[ \int_C (2y \, dx + 5x \, dy) \]
is equal to
Given that \( i = \sqrt{-1} \). The value of \[ \lim_{z \to e^{i \pi/3}} \frac{z^3 + 1}{z^4 + z^2 + 1} \]
is
Let \( f(x) \) be a non-negative continuous function of real variable \( x \). If the area under the curve \( y = f(x) \) from \( x = 0 \) to \( x = a \) is \( \frac{a^2}{2} + \frac{a}{2} \sin a + \frac{\pi}{2} \cos a - \frac{\pi}{2} \), then the value of \( f \left( \frac{\pi}{2} \right) \) is _______ (round off to one decimal place).
If the numerical approximation of the value of the integral \( \int_0^4 2^\alpha x \, dx \) using the Trapezoidal rule with two subintervals is 9, then the value of the real constant \( \alpha \) is _______ (round off to one decimal place).
Let the transformation \( y(x) = e^x \nu(x) \) reduce the ordinary differential equation \( x \frac{d^2y}{dx^2} + 2(1 - x) \frac{dy}{dx} + (x - 2) y = 0 \), where \( \alpha, \beta, \gamma \) are real constants. Then, the arithmetic mean of \( \alpha, \beta, \gamma \) is _______ (round off to three decimal places).
A person, who speaks the truth 3 out of 4 times, throws a fair die with six faces and informs that the outcome is 5. The probability that the outcome is really 5 is ________ (round off to three decimal places).
Let \[ f(x, y) = x^4 + y^4 - 2x^2 + 4xy - 2y^2 + \alpha \]
be a real valued function. Then, which one of the following statements is TRUE for all \( \alpha \)?
Let \[ u(x, y) = (x^2 - y^2)v(x, y) \]
be such that both \( u(x, y) \) and \( v(x, y) \) satisfy the Laplace equation in a domain \( \Omega \) of the xy-plane. Then, which one of the following is TRUE in \( \Omega \)?
Let \( I \) denote the identity matrix of order 7, and \( A \) be a \( 7 \times 7 \) real matrix having characteristic polynomial \( C_A(\lambda) = \lambda^2 (\lambda - 1)^\alpha (\lambda + 2)^\beta \), where \( \alpha \) and \( \beta \) are positive integers. If \( A \) is diagonalizable and \( rank(A) = rank(A + 2I) \), then \( rank(A - I) \) is _______ (in integer).
Let \( C_1 \) be the line segment from \( (0, 1) \) to \( \left( \frac{4}{5}, \frac{3}{5} \right) \), and let \( C_2 \) be the arc of the circle \( x^2 + y^2 = 1 \) from \( (0, 1) \) to \( \left( \frac{4}{5}, \frac{3}{5} \right) \). If
\[ \alpha = \int_{C_1} \left( \frac{2x}{y} \hat{i} + \frac{1 - x^2}{y^2} \hat{j} \right) \cdot d\vec{r} \]
and \[ \beta = \int_{C_2} \left( \frac{2x}{y} \hat{i} + \frac{1 - x^2}{y^2} \hat{j} \right) \cdot d\vec{r}, \]
where \( \vec{r} = x \hat{i} + y \hat{j} \), then the value of \( \alpha^2 + \beta^2 \) is _______ (round off to two decimal places).


































































Quick Links:
GATE 2021 XE Exam Pattern
There were three sections in GATE 2021 XE exam-
1.General Aptitude
2.Mathematics (Compulsory Section)
3.Any two optional papers from sections such as Fluid Mechanics (XE-B), Materials Science (XE-C), Solid Mechanics (XE-D),Thermodynamics (XE-E), Polymer Science and Engineering (XE-F), Food Technology (XE-G), and Atmospheric and Oceanic Sciences (XE-H)
The General Aptitude section carried 10 MCQs, holding 15 marks (each 5 questions 1 mark each, the remaining 5 questions carried 2 marks each. The Mathematics section comprises 11 questions, contributing 15 marks. The Mathematics section carried 3 and 2 MCQs carrying 1 and 2 respectively. Coming to NAT (Numerical Answer Types) questions, 4 NATs carrying 1 mark each, and 2 NATs carrying 2 marks each, listed in GATE 2021 XE-A. The remaining 70 marks belong to the Optional Sections. Each optional section comprises 22 questions, carrying 35 marks each section.
GATE 2021 XE Marking Scheme
Each question carried either 1 or 2 marks. ⅓ marks would be reduced for wrong MCQs carrying 1 mark, and ⅔ marks would be deducted for every wrong MCQs carrying 2 marks. For every correct NAT, 1 or 2 marks would be rewarded, depending upon the marks of the question
Also Check:
GATE Previous Year Question Papers:
| GATE 2022 Question Papers | GATE 2021 Question Papers | GATE 2020 Question Papers |
| GATE 2019 Question Papers | GATE 2018 Question Papers | GATE 2017 Question Papers |










Comments