GATE 2022 Chemical Engineering (CH) Question Paper with Solutions PDFs are available to download. GATE 2022 CH was conducted by IIT Kharagpur on 6th February 2022. The exam was held in the Afternoon slot (2:30 PM to 5:30 PM). The overall difficulty level of GATE 2022 CH was rated moderate to tough. As per the test-takers, the question paper was difficult as compared to last year. The General Aptitude section was rated easy, while the questions related to core subject were considered difficult.
GATE 2022 Chemical Engineering (CH) Question Paper with Solutions
| GATE 2022 Chemical Engineering (CH) Question Paper | Check Solutions |

Inhaling the smoke from a burning __________ could __________ you quickly.
A sphere of radius \(r\) cm is packed in a box of cubical shape.
What should be the minimum volume (in cm\(^3\)) of the box that can enclose the sphere?
Pipes P and Q can fill a storage tank in full with water in 10 and 6 minutes, respectively. Pipe R draws the water out from the storage tank at a rate of 34 litres per minute. P, Q and R operate at a constant rate.
If it takes one hour to completely empty a full storage tank with all the pipes operating simultaneously, what is the capacity of the storage tank (in litres)?
Six persons P, Q, R, S, T, and U are sitting around a circular table facing the center not necessarily in the same order. Consider the following statements:
P sits next to S and T.
Q sits diametrically opposite to P.
The shortest distance between S and R is equal to the shortest distance between T and U.
Based on the above statements, Q is a neighbor of
A building has several rooms and doors as shown in the top view of the building given below. The doors are closed initially.
What is the minimum number of doors that need to be opened in order to go from the point P to the point Q?

Rice, a versatile and inexpensive source of carbohydrate, is a critical component of diet worldwide. Climate change, causing extreme weather, poses a threat to sustained availability of rice. Scientists are working on developing Green Super Rice (GSR), which is resilient under extreme weather conditions yet gives higher yields sustainably.
Which one of the following is the CORRECT logical inference based on the information given in the above passage?
A game consists of spinning an arrow around a stationary disk as shown below.
When the arrow comes to rest, there are eight equally likely outcomes. It could come to rest in any one of the sectors numbered 1, 2, 3, 4, 5, 6, 7, or 8 as shown.
Two such disks are used in a game where their arrows are independently spun.
What is the probability that the sum of the numbers on the resulting sectors upon spinning the two disks is equal to 8 after the arrows come to rest?

Consider the following inequalities.
(i) \( 3p - q < 4 \)
(ii) \( 3q - p < 12 \)
Which one of the following expressions below satisfies the above two inequalities?
Given below are three statements and four conclusions drawn based on the statements.
Statement 1: Some engineers are writers.
Statement 2: No writer is an actor.
Statement 3: All actors are engineers.
Conclusion I: Some writers are engineers.
Conclusion II: All engineers are actors.
Conclusion III: No actor is a writer.
Conclusion IV: Some actors are writers.
Which one of the following options can be logically inferred?
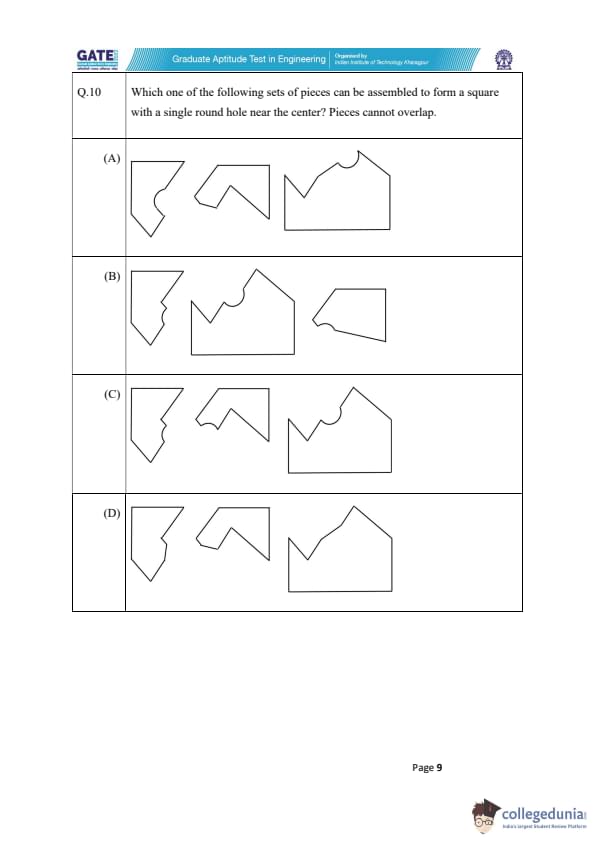
Which one of the following sets of pieces can be assembled to form a square with a single round hole near the center? Pieces cannot overlap.

The value of \( (1 + i)^{12} \), where \( i = \sqrt{-1} \), is
Given matrix \( A = \left[\begin{matrix} x & 1 & 3
y & 2 & 6
3 & 5 & 7 \end{matrix}\right] \), the ordered pair \( (x, y) \) for which \( det(A) = 0 \) is
Let \( f(x) = e^{-|x|} \), where \( x \) is real. The value of \[ \frac{df}{dx} at x = -1 is \]
The value of the real variable \( x \geq 0 \), which maximizes the function \[ f(x) = x e^x e^{-x} \]
is
For a single component system at vapor-liquid equilibrium, the extensive variables
A, V, S and N denote the Helmholtz free energy, volume, entropy, and number of moles, respectively, in a given phase. If superscripts \( (\nu) \) and \( (\ell) \) denote the vapor and liquid phase, respectively, the relation that is NOT CORRECT is
Consider turbulent flow in a pipe under isothermal conditions. Let r denote the radial coordinate and z denote the axial flow direction. On moving away from the wall towards the center of the pipe, the rz-component of the Reynolds stress
Consider two stationary spherical pure water droplets of diameters \( d_1 \) and \( 2d_1 \). CO\(_2\) diffuses into the droplets from the surroundings. If the rate of diffusion of CO\(_2\) into the smaller droplet is \( W_1 \) mol s\(^{-1}\), the rate of diffusion of CO\(_2\) into the larger droplet is
In soap manufacturing, the triglycerides present in oils and fats are hydrolyzed to mainly produce
The chemical formula of Glauber’s salt, used in the Kraft process, is
Catalytic reforming is commonly used in the petroleum industry to improve fuel quality. The undesirable reaction in the catalytic reforming of naphtha is
A control system on the jacket side of a reactor is shown in the figure. Pressurized water flows through the jacket to cool the reactor. The heated water flashes in the boiler. The exothermic reaction heat thus generates steam. Fresh boiler feed water (BFW) is added to make-up for the loss of water as steam. Assume that all control valves are air-to-open. The controller action, ‘direct’ or ‘reverse’, is defined with respect to the controller. Select the option that correctly specifies the action of the controllers.

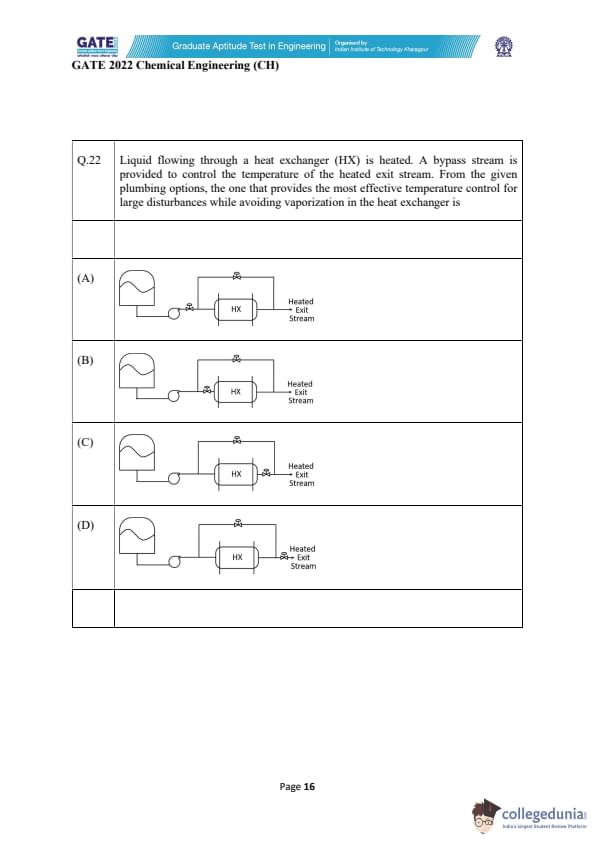
Liquid flowing through a heat exchanger (HX) is heated. A bypass stream is provided to control the temperature of the heated exit stream. From the given plumbing options, the one that provides the most effective temperature control for large disturbances while avoiding vaporization in the heat exchanger is

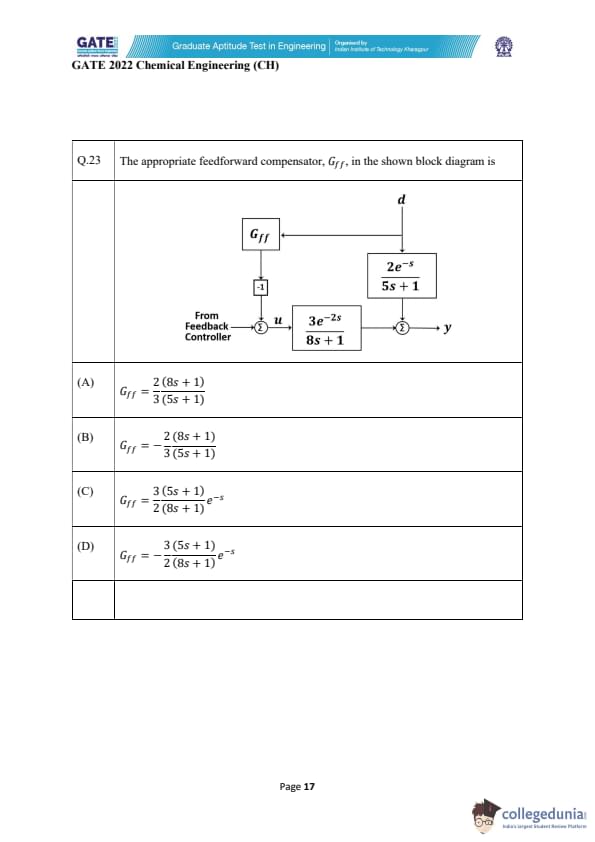
The appropriate feedforward compensator, \(G_{ff}\), in the shown block diagram is

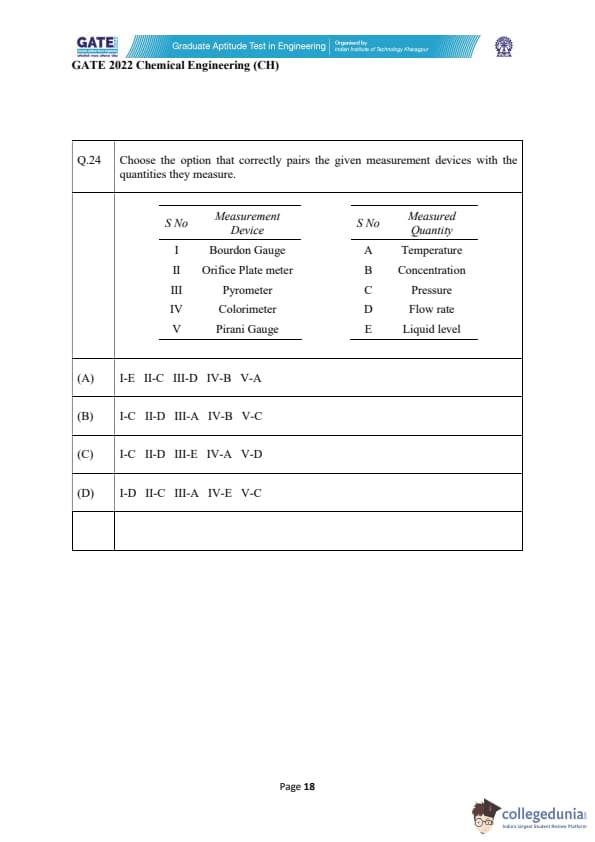
Choose the option that correctly pairs the given measurement devices with the quantities they measure.

A simple distillation column is designed to separate an ideal binary mixture to specified distillate and bottoms purities at a given column pressure. If RR\(_{\min}\) is the minimum reflux ratio for this separation, select the statement that is NOT CORRECT with regard to the variation in the total annualized cost (TAC) of the column with reflux ratio (RR).
The reaction A → B is carried out isothermally on a porous catalyst. The intrinsic reaction rate is \(k C_A^2\), where \(k\) is the rate constant and \(C_A\) is the concentration of A. If the reaction is strongly pore-diffusion controlled, the observed order of the reaction is
In an enzymatic reaction, an inhibitor (I) competes with the substrate (S) to bind with the enzyme (E), thereby reducing the rate of product (P) formation. The competitive inhibition follows the reaction mechanism shown below. Let [S] and [I] be the concentration of S and I, respectively, and \(r_s\) be the rate of consumption of S. Assuming pseudo-steady state, the correct plot of \(\frac{1}{-r_s}\) vs \(\frac{1}{[S]}\) is

The area of a circular field is 25 m\(^2\). The radius, r, is to be determined using the Newton-Raphson iterative method. For an initial guess of r = 2.500 m, the revised estimate of r after one iteration is _________ m (rounded off to three decimal places).
5 moles of liquid benzene, 8 moles of liquid toluene and 7 moles of liquid xylene are mixed at 25\(^\circ\)C and 1 bar. Assuming the formation of an ideal solution and using the universal gas constant \(R = 8.314\) J mol\(^{-1}\) K\(^{-1}\), the total entropy change is _________ J K\(^{-1}\) (rounded off to one decimal place).
A perfectly insulated double pipe heat exchanger is operating at steady state. Saturated steam enters the inner pipe at 100 °C and leaves as saturated water at 100 °C. Cooling water enters the outer pipe at 75 °C and exits at 95 °C. The overall heat transfer coefficient is 1 kW m\(^{-2}\) K\(^{-1}\) and the heat transfer area is 1 m\(^{2}\). The average specific heat capacity of water is 4.2 kJ kg\(^{-1}\) K\(^{-1}\). The required cooling water flow rate is _________ kg s\(^{-1}\) (rounded off to two decimal places).
Consider steady-state diffusion in a binary A-B liquid at constant temperature and pressure. The mole-fraction of A at two different locations is 0.8 and 0.1. Let \(N_{A1}\) be the diffusive flux of A calculated assuming B to be non-diffusing, and \(N_{A2}\) be the diffusive flux of A calculated assuming equimolar counter-diffusion. The quantity \(\frac{(N_{A1} - N_{A2})}{N_{A1}} \times 100\) is _________ (rounded off to one decimal place).
Consider interphase mass transfer of a species S between two immiscible liquids A and B. The interfacial mass transfer coefficient of S in liquid A is twice of that in liquid B. The equilibrium distribution of S between the liquids is given by \(y_S^A = 0.5 y_S^B\), where \(y_S^A\) and \(y_S^B\) are the mole-fractions of S in A and B, respectively. The bulk phase mole-fraction of S in A and B is 0.10 and 0.02, respectively. If the steady-state flux of S is estimated to be 10 kmol h\(^{-1}\) m\(^{-2}\), the mass transfer coefficient of S in A is ________ kmol h\(^{-1}\) m\(^{-2}\) (rounded off to one decimal place).
A wet solid containing 20% (w/w) moisture (based on mass of bone-dry solid) is dried in a tray-dryer. The critical moisture content of the solid is 10% (w/w). The drying rate (kg m\(^{-2}\) s\(^{-1}\)) is constant for the first 4 hours, and then decreases linearly to half the initial value in the next 1 hour. At the end of 5 hours of drying, the percentage moisture content of the solid is ________ % (w/w) (rounded off to one decimal place).
A process described by the transfer function \[ G_p(s) = \frac{10s + 1}{5s + 1} \]
is forced by a unit step input at time \(t = 0\). The output value immediately after the step input (at \(t = 0^+\)) is _________ (rounded off to the nearest integer).
A compressor with a life of 10 years costs Rs 10 lakhs. Its yearly operating cost is Rs 0.5 lakh. If the annual compound interest rate is 8%, the amount needed at present to fund perpetual operation of the compressor is Rs _________ lakhs (rounded to first decimal place).
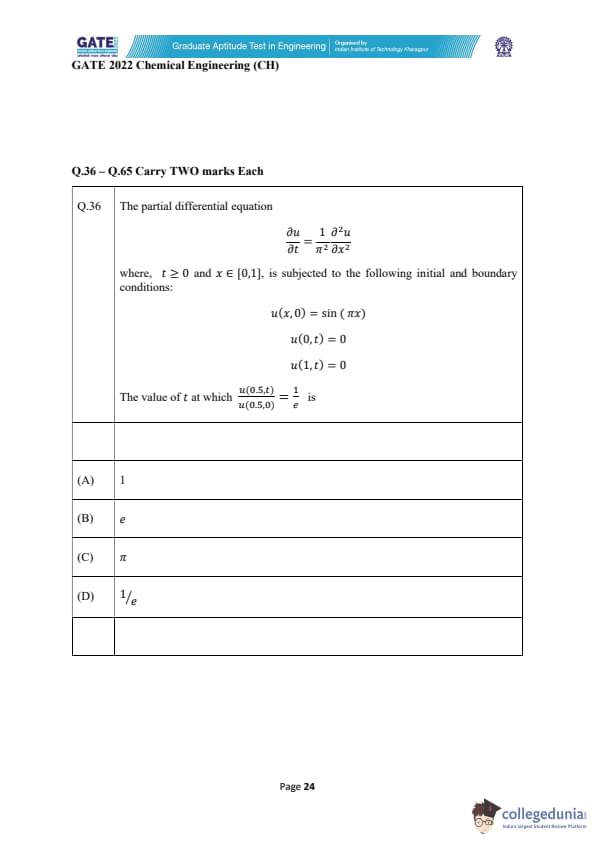
The partial differential equation
\[ \frac{\partial u}{\partial t} = \frac{1}{\pi^2} \frac{\partial^2 u}{\partial x^2} \]
where \(t \ge 0\) and \(x \in [0,1]\), is subjected to the following initial and boundary conditions:
\[ u(x,0) = \sin(\pi x) \]
\[ u(0,t) = 0 \]
\[ u(1,t) = 0 \]
The value of \(t\) at which \[ \frac{u(0.5,t)}{u(0.5,0)} = \frac{1}{e} \]
is
N moles of an ideal gas undergo a two-step process as shown in the figure. Let \( P, V, T \) denote the pressure, volume and temperature of the gas. The gas, initially at state-1 \( (P_1, V_1, T_1) \), undergoes an isochoric (constant volume) process to reach state-A, and then undergoes an isobaric (constant pressure) expansion to reach state-2 \( (P_2, V_2, T_2) \). For an ideal gas, \( C_P - C_V = NR \), where \( C_P \) and \( C_V \) are heat capacities at constant pressure and volume. The heat gained by the gas in the two-step process is given by

A horizontal cylindrical water jet of diameter \(D_1 = 2 cm\) strikes a vertical solid plate with a hole of diameter \(D_2 = 1 cm\), as shown in the figure. A part of the jet passes through the hole and the rest is deflected along the plate. The density of water is \(1000\ kg m^{-3}\). If the jet speed is \(20\ m s^{-1}\), the magnitude of the horizontal force required to hold the plate stationary is

Consider a horizontal rod of radius \(aR\) (\(a < 1\)) in a stationary pipe of radius \(R\). The rod is pulled coaxially at a constant velocity \(V\) as shown in the figure. The annular region is filled with a Newtonian incompressible fluid of viscosity \(\mu\). The steady–state fully–developed axial velocity profile in the fluid is \[ u(r) = V\frac{\ln(r/R)}{\ln(a)}, \]
where \(r\) is the radial coordinate. Ignoring end effects, the magnitude of the pulling force per unit rod length is

Consider a bare long copper wire of 1 mm diameter. Its surface temperature is \(T_s\) and the ambient temperature is \(T_a\) (\(T_s > T_a\)). The wire is to be coated with a 2 mm thick insulation. The convective heat transfer coefficient is 20 W m\(^{-2}\) K\(^{-1}\). Assume that \(T_s\) and \(T_a\) remain unchanged. To reduce heat loss from the wire, the maximum allowed thermal conductivity of the insulating material, in W m\(^{-1}\) K\(^{-1}\), rounded off to two decimal places, is
Two large parallel planar walls are maintained at 1000 K and 500 K. Parallel radiation shields are to be installed between the two walls. Assume emissivities of walls and shields are equal. If the melting temperature of the shields is 900 K, the maximum number of shield(s) that can be installed between the walls is (are)
Saturated steam condenses on a vertical plate maintained at a constant wall temperature. If \(x\) is the vertical distance from the top edge of the plate, then the local heat transfer coefficient \(h(x) \propto \Gamma(x)^{-1/3}\), where \(\Gamma(x)\) is the local mass flow rate of the condensate per unit plate width. The ratio of the average heat transfer coefficient over the entire plate to the heat transfer coefficient at the bottom of the plate is
Match the product in Group-1 with the manufacturing process in Group-2. The correct combination is

The directional derivative of \(f(x,y,z)=4x^{2}+2y^{2}+z^{2}\) at the point (1,1,1) in the direction of the vector \(\vec{v}=\hat{i}-\hat{k}\) is ________ (rounded off to two decimal places).
Consider a sphere of radius 4, centered at the origin, with outward unit normal \(\hat{n}\) on its surface \(S\). The value of the surface integral \(\iint_{S}\dfrac{2x\hat{i}+3y\hat{j}+4z\hat{k}}{4\pi}\cdot \hat{n}\, dA\) is ________ (rounded off to one decimal place).
The equation \(\dfrac{dy}{dx} = xy^{2} + 2y + x - 4.5\) with the initial condition \(y(x=0)=1\) is to be solved using a predictor-corrector approach. Use a predictor based on the implicit Euler’s method and a corrector based on the trapezoidal rule of integration, each with a full-step size of 0.5. Considering only positive values of \(y\), the value of \(y\) at \(x=0.5\) is _________ (rounded off to three decimal places).
A substance at 4\(^\circ\)C has a thermal expansion coefficient \(\beta = \dfrac{1}{v}\left(\dfrac{\partial v}{\partial T}\right)_P = 0\) K\(^{-1}\), an isothermal compressibility, \(\kappa_T = -\dfrac{1}{v}\left(\dfrac{\partial v}{\partial P}\right)_T = 5\times10^{-4}\) Pa\(^{-1}\) and a molar volume \(v = 18\times10^{-6}\) m\(^3\) mol\(^{-1}\). If \(s\) is the molar entropy, then at 4\(^\circ\)C, the quantity \(\left[v\left(\dfrac{\partial s}{\partial v}\right)_T\right]\) evaluated for the substance is ________ J mol\(^{-1}\) K\(^{-1}\) (rounded off to the nearest integer).
The molar excess Gibbs free energy (\(g^E\)) of a liquid mixture of \(A\) and \(B\) is given by \[ \frac{g^E}{RT} = x_A x_B [C_1 + C_2(x_A - x_B)] \]
where \(x_A\) and \(x_B\) are the mole fraction of \(A\) and \(B\), respectively, the universal gas constant, \(R = 8.314\) J K\(^{-1}\) mol\(^{-1}\), \(T\) is the temperature in K, and \(C_1, C_2\) are temperature-dependent parameters. At 300 K, \(C_1 = 0.45\) and \(C_2 = -0.018\). If \(\gamma_A\) and \(\gamma_B\) are the activity coefficients of \(A\) and \(B\), respectively, the value of \[ \int_{0}^{1} \ln\left(\frac{\gamma_A}{\gamma_B}\right)\,dx_A \]
at 300 K and 1 bar is ________ (rounded off to the nearest integer).
For a pure substance, the following data at saturated conditions are given:
\[ \begin{array}{c c} \ln P^{sat} \, (bar) & T \,(K)
0.693 & 350
1.386 & 370
\end{array} \]
Assume the vapor behaves ideally, liquid molar volume is negligible, and latent heat of vaporization is constant over this range. The universal gas constant is \(R=8.314\) J mol\(^{-1}\) K\(^{-1}\). From the above data, the estimated latent heat of vaporization at 360 K is ________ kJ/mol (rounded to one decimal place).
A liquid-phase irreversible reaction \(A \rightarrow B\) occurs in a CSTR of volume 1 m\(^{3}\) with rate \(-r_{A}=164\, x_{A}\) kmol m\(^{-3}\) h\(^{-1}\). The reactor output is split ideally to obtain pure B in bottoms, while the distillate (free of B) is partly purged (10 kmol h\(^{-1}\)) and the rest recycled (200 kmol h\(^{-1}\)). At steady state, pure B product rate is 100 kmol h\(^{-1}\). A small amount of inert \(I\) is added with the fresh feed. Compute the inert feed rate into the process (kmol h\(^{-1}\)), rounded to two decimals.

Two reservoirs located at the same altitude are connected by a straight horizontal pipe of length 120 m and inner diameter 0.5 m. A pump transfers a liquid of density 800 kg m\(^{-3}\) at a flow rate of 1 m\(^{3}\) s\(^{-1}\) from Reservoir-1 to Reservoir-2. The liquid levels in Reservoir-1 and Reservoir-2 are 2 m and 10 m, respectively. The friction factor is 0.01. The acceleration due to gravity is 9.8 m s\(^{-2}\). The required power of the pump is ________ kW (rounded off to one decimal place).

A venturi meter (venturi coefficient \(C_v = 0.98\)) is installed in a pipe of inner diameter 50 mm. Water (density 1000 kg m\(^{-3}\)) flows through the pipe. The pressure drop across the venturi meter is 50 kPa. If the venturi throat diameter is 20 mm, the estimated flow rate is _______ \(\times 10^{-3}\) m\(^{3}\) s\(^{-1}\) (rounded off to two decimal places).
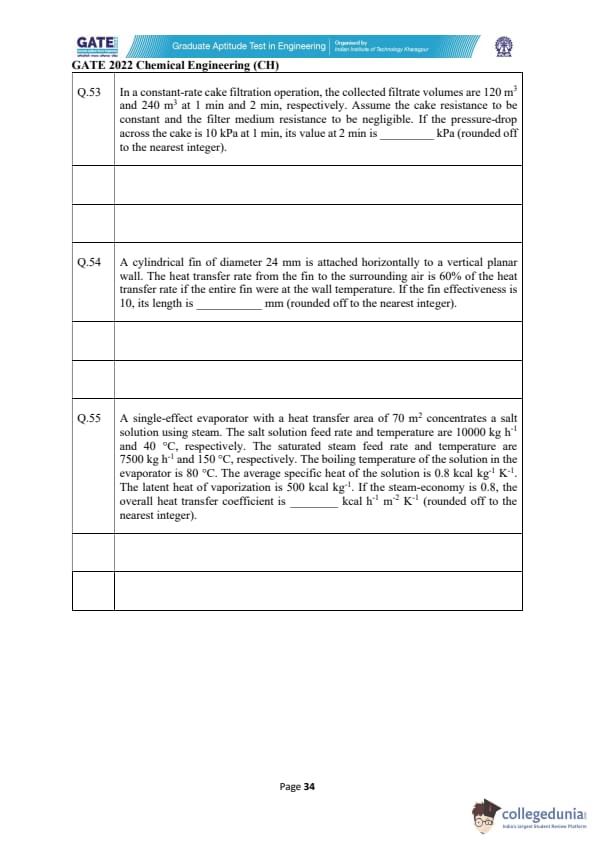
In a constant-rate cake filtration operation, the collected filtrate volumes are 120 m\(^{3}\) and 240 m\(^{3}\) at 1 min and 2 min, respectively. Assume the cake resistance to be constant and the filter medium resistance to be negligible. If the pressure-drop across the cake is 10 kPa at 1 min, its value at 2 min is ________ kPa (rounded off to the nearest integer).
A cylindrical fin of diameter 24 mm is attached horizontally to a vertical planar wall. The heat transfer rate from the fin to the surrounding air is 60% of the heat transfer rate if the entire fin were at the wall temperature. If the fin effectiveness is 10, its length is ________ mm (rounded off to the nearest integer).
A single-effect evaporator with a heat transfer area of 70 m\(^{2}\) concentrates a salt solution using steam. The salt solution feed rate and temperature are 10000 kg h\(^{-1}\) and 40\(^\circ\)C, respectively. The saturated steam feed rate and temperature are 7500 kg h\(^{-1}\) and 150\(^\circ\)C, respectively. The boiling temperature of the solution in the evaporator is 80\(^\circ\)C. The average specific heat is 0.8 kcal kg\(^{-1}\) K\(^{-1}\). The latent heat of vaporization is 500 kcal kg\(^{-1}\). If the steam-economy is 0.8, the overall heat transfer coefficient is ________ kcal h\(^{-1}\) m\(^{-2}\) K\(^{-1}\) (rounded off to the nearest integer).
An equimolar binary mixture is to be separated in a simple tray-distillation column. The feed rate is 50 kmol min\(^{-1}\). The mole fractions of the more volatile component in the top and bottom products are 0.90 and 0.01, respectively. The feed and the reflux stream are saturated liquids. On application of the McCabe-Thiele method, the operating line for the stripping section is obtained as \[ y = 1.5x - 0.005 \]
where \(y\) and \(x\) are the mole fractions of the more volatile component in the vapor and liquid phases, respectively. The reflux ratio is _______ (rounded off to two decimal places).
The dry-bulb temperature of air in a room is 30 °C. The Antoine equation for water is \[ \ln P^{sat} = 12.00 - \frac{4000}{T - 40} \]
where \(T\) is in K and \(P^{sat}\) is in bar. The latent heat of vaporization is 2000 kJ kg\(^{-1}\), humid heat is 1.0 kJ kg\(^{-1}\) K\(^{-1}\), and the molecular weights of air and water are 28 and 18 kg kmol\(^{-1}\). If absolute humidity is \(Y'\) kg moisture per kg dry air, then for a wet-bulb depression of 9 °C, \(1000\,Y' =\) ________ (rounded to one decimal place).
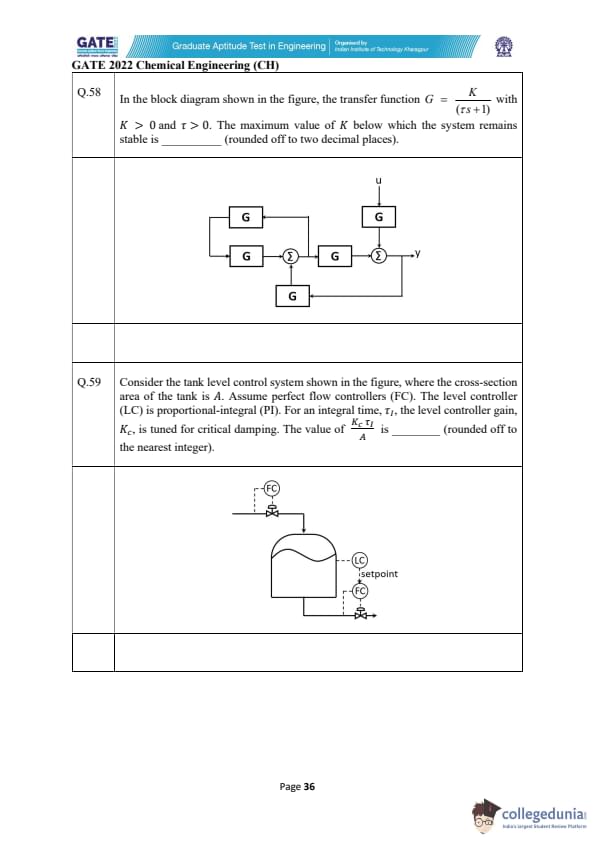
In the block diagram shown in the figure, the transfer function \(G=\dfrac{K}{(\tau s+1)}\) with \(K>0\) and \(\tau>0\). The maximum value of \(K\) below which the system remains stable is ________ (rounded off to two decimal places).

In the tank-level control system shown, the tank cross-sectional area is \(A\). The flow controllers are perfect. The level controller (LC) is PI. For an integral time \(\tau_I\), the controller gain \(K_c\) is tuned for critical damping. The value of \(\dfrac{K_c\tau_I}{A}\) is ________ (rounded off to nearest integer).

Consider a single-input-single-output (SISO) system with the transfer function \[ G_p(s)=\frac{2(s+1)}{\left(\frac{1}{2}s+1\right)\left(\frac{1}{4}s+1\right)} \]
where the time constants are in minutes. The system is forced by a unit step input at time \(t=0\). The time at which the output response reaches its maximum is ________ minutes (rounded off to two decimal places).
Information for a proposed greenfield project is provided in the table. The discounted cash flow for the fourth year is Rs ______ crores (rounded off to one decimal place).

Consider the process in the figure. The liquid-phase elementary reactions \[ A + B \rightarrow P \quad -r_{B1} = k_1 x_A x_B \] \[ P + B \rightarrow S \quad -r_{B2} = k_2 x_P x_B \] \[ S + A \rightarrow 2P \quad -r_{S3} = k_3 x_S x_A \]
occur in the continuous stirred tank reactor (CSTR). All fresh feeds, exit streams and recycle streams are pure. At steady state, the net generation of the undesired product \(S\) in the CSTR is zero. Let \(q = x_A/x_B\) in the reactor. For 90% single-pass conversion of \(B\) and fixed product rate, determine the value of \(q\) that minimizes the sum of the molar flow rates of the A and S recycle streams (rounded to one decimal place).

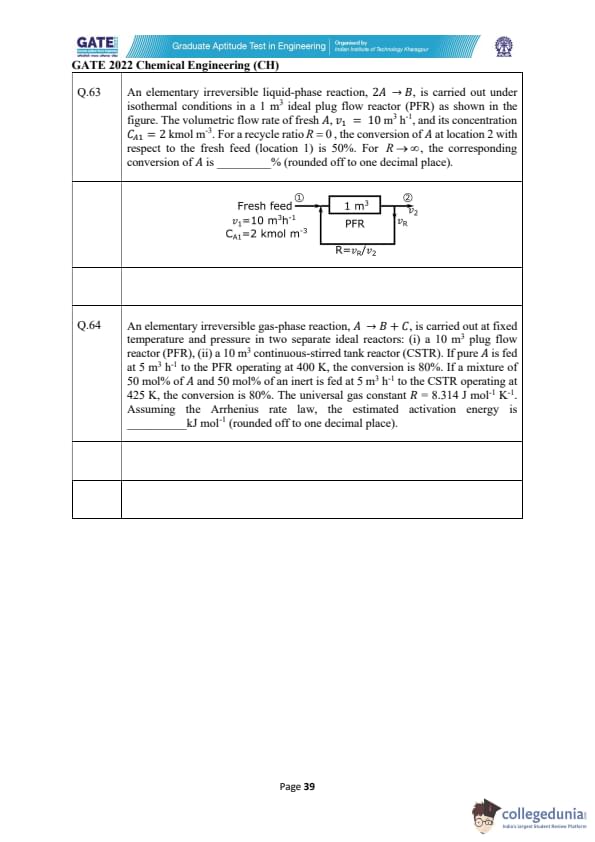
An elementary irreversible liquid-phase reaction, \(2A \rightarrow B\), is carried out under isothermal conditions in a 1 m\(^{3}\) ideal plug flow reactor (PFR) as shown. The volumetric flow rate of fresh A is \(v_{1}=10\) m\(^{3}\) h\(^{-1}\) and its concentration is \(C_{A1}=2\) kmol m\(^{-3}\). For a recycle ratio \(R=0\), the conversion of A at location 2 with respect to fresh feed (location 1) is 50%. For \(R\rightarrow\infty\), the corresponding conversion of A is ______% (rounded off to one decimal place).

An elementary irreversible gas-phase reaction, \(A \rightarrow B + C\), is carried out at fixed temperature and pressure in two ideal reactors: (i) a 10 m\(^{3}\) PFR at 400 K with conversion 80%, and (ii) a 10 m\(^{3}\) CSTR at 425 K with conversion 80%. Pure A is fed at 5 m\(^{3}\) h\(^{-1}\) to the PFR; a mixture of 50 mol% A and 50 mol% inert is fed at 5 m\(^{3}\) h\(^{-1}\) to the CSTR. Assume Arrhenius kinetics. Estimate activation energy (rounded to 1 decimal place).
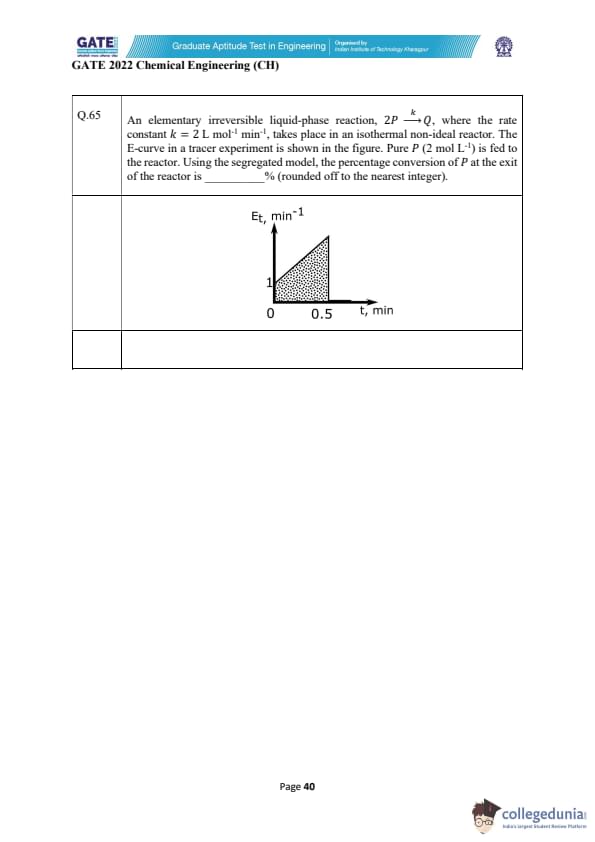
An elementary irreversible liquid-phase reaction, \(2P \xrightarrow{k} Q\), with rate constant \(k = 2\ L mol^{-1}min^{-1}\), occurs in an isothermal non-ideal reactor. Pure \(P\) (2 mol L\(^{-1}\)) is fed. The \(E\)-curve from a tracer test is triangular from \(t=0\) to \(t=0.5\) min, rising linearly from 1 to 2 min\(^{-1}\). Using the segregated model, the percentage conversion of \(P\) at the reactor exit is ________% (rounded to the nearest integer).
































Quick Links:
GATE 2022 CH Detailed Paper Analysis
IIT Kharagpur listed 35 MCQs (Multiple Choice Questions) and 30 NATs (Numerical Answer Type) questions in GATE 2022 CH. Please refer to the below-mentioned table in order to get the details of listed MCQs and NATs as per the carried marks-
| Question Types | Question Frequency | Carried Marks |
|---|---|---|
| No. Of 1 Mark MCQs | 22 | 22 |
| No. Of 2 Mark MCQs | 13 | 26 |
| No. Of 1 Mark NATs | 8 | 8 |
| No. Of 2 Marks NATs | 22 | 44 |
| Total | 65 | 100 |
- No MSQs (Multiple Select Questions) were asked in GATE 2022 CH
- This year, maximum weightage was carried by NATs in the question paper of GATE 2022 CH
- Sections such as Heat Transfer, IPC, Mass Transfer, CRE, and FM carried the maximum weightage in the question paper
- The Engineering Mathematics section carried 13 out of 100 marks
- NAT questions were more complex as compared to the other questions
- A candidate must have attempted more than 45 questions in order to qualify the cutoff
Also Check:
GATE Previous Year Question Papers:
| GATE 2022 Question Papers | GATE 2021 Question Papers | GATE 2020 Question Papers |
| GATE 2019 Question Papers | GATE 2018 Question Papers | GATE 2017 Question Papers |









Comments