GATE 2023 Chemistry Question Paper PDF is available here for download. IIT Kanpur conducted GATE 2023 Chemistry exam on February 5, 2023 in the Afternoon Session from 02:30 PM to 05:30 PM. Students have to answer 65 questions in GATE 2023 Chemistry Question Paper carrying a total weightage of 100 marks. 10 questions are from the General Aptitude section and 55 questions are from Core Discipline.
GATE 2023 Chemistry Question Paper with Solutions PDF
| GATE 2023 Chemistry Question Paper with Solutions PDF | Download PDF | Check Solutions |
“I cannot support this proposal. My ______ will not permit it.”
Courts : _____ : : Parliament : Legislature (By word meaning)
What is the smallest number with distinct digits whose digits add up to 45?
In a class of 100 students,
How many rectangles are present in the given figure?

Forestland is a planet inhabited by different kinds of creatures. Among other creatures, it is populated by animals all of whom are ferocious. There are also creatures that have claws, and some that do not. All creatures that have claws are ferocious.
Based only on the information provided above, which one of the following options can be logically inferred with certainty?
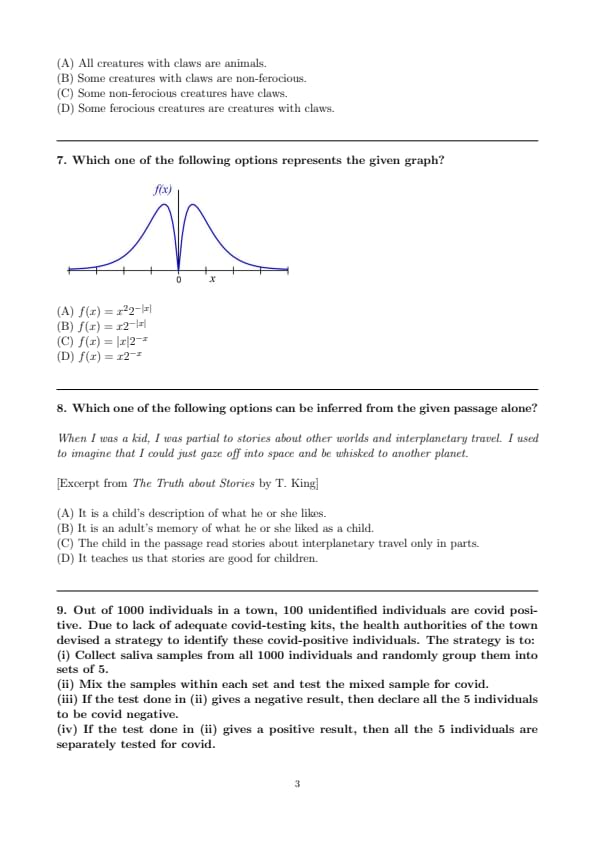
Which one of the following options represents the given graph?

Which one of the following options can be inferred from the given passage alone?
When I was a kid, I was partial to stories about other worlds and interplanetary travel. I used to imagine that I could just gaze off into space and be whisked to another planet.
[Excerpt from The Truth about Stories by T. King]
Out of 1000 individuals in a town, 100 unidentified individuals are covid positive. Due to lack of adequate covid-testing kits, the health authorities of the town devised a strategy to identify these covid-positive individuals. The strategy is to:
(i) Collect saliva samples from all 1000 individuals and randomly group them into sets of 5.(ii) Mix the samples within each set and test the mixed sample for covid.
(iii) If the test done in (ii) gives a negative result, then declare all the 5 individuals to be covid negative.
(iv) If the test done in (ii) gives a positive result, then all the 5 individuals are separately tested for covid.
Given this strategy, no more than _____ testing kits will be required to identify all the 100 covid positive individuals irrespective of how they are grouped.
A \(100 cm \times 32 cm\) rectangular sheet is folded 5 times. Each time the sheet is folded, the long edge aligns with its opposite side. Eventually, the folded sheet is a rectangle of dimensions \(100 cm \times 1 cm\).
The total number of creases visible when the sheet is unfolded is _____.
The major product formed in the given reaction is: \[ TiCl_3(DME)_2 \quad Zn-Cu \quad DME, r.t. \quad \longrightarrow \quad 1,2-Dimethoxyethane. \]
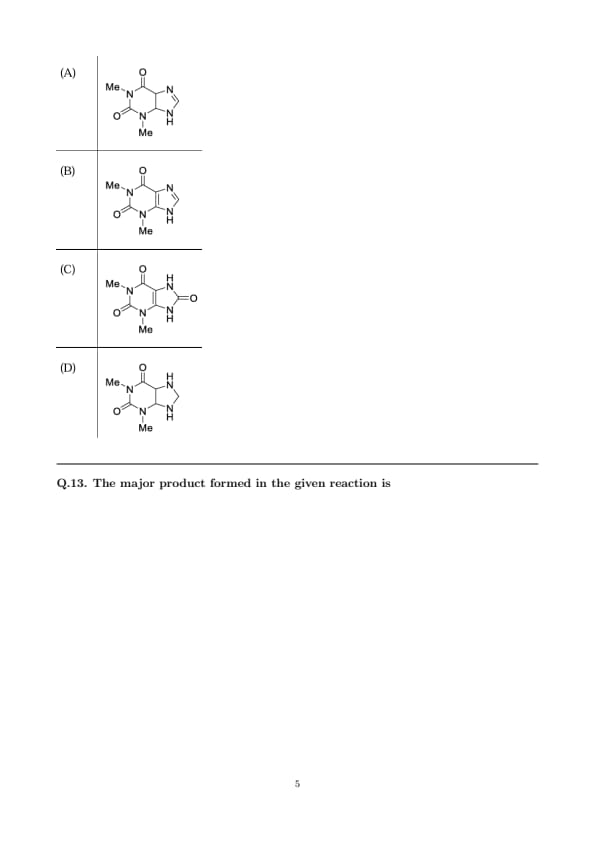
The major product formed in the given reaction is

The major product formed when the \(\,o\)-(\(\mathrm{Me_3Si}\))-aryl triflate (a Kobayashi benzyne precursor) is treated with Ph–substituted cyclohexene in the presence of \(\mathrm{CsF}\) in \(\mathrm{CH_3CN}\) at r.t. is (A)–(D) as shown in the figure.

On irradiation using UV light (\(>300\) nm), compounds X and Y predominantly undergo which photochemical pathway? (Structures as shown in the figure)

The topicity relationship of \(H_a\) and \(H_b\) in X, Y and Z (as drawn in the figure) are, respectively,

Compound \(\mathbf{P}\) was prepared based on a four-component reaction at room temperature in methanol. The required starting materials for the synthesis are (structure of \(\mathbf{P}\) shown).

The major product formed in the following reaction is \(\; \mathrm{nBu_3SnH,\ AIBN,\ C_6H_6,\ heat}\)

The reaction of Ph\(_3\)PCl\(_2\) with PhNH\(_2\) primarily produces
Formation of [M(en)\(_3\)]\(^{2+}\) from [M(H\(_2\)O)\(_6\)]\(^{2+}\) and three equivalents of ethylenediamine (en) is LEAST favored when M is
Wacker oxidation of alkenes is catalyzed by a combination of
For the conversion of [Pt(L)Cl\(_3\)]\(^-\) to trans-[Pt(L)Cl\(_2\)(H\(_2\)O)], the trans-effect is LEAST when the ligand L is
The tetracoordinated copper center in the oxidized and reduced forms of plastocyanin exhibits longest bond with
The packing efficiency (in %) of spheres for a body-centered cubic (bcc) lattice is approximately
The magnitudes of CFSE in \([\mathrm{M}(\mathrm{H}_2\mathrm{O})_6]^{n+}\) for Mn and Fe ions satisfy the relations
The organometallic catalyst for the following transformation is (intramolecular olefin closure giving a cyclohexene and ethylene, i.e., ring-closing metathesis):
Point group of naphthalene \(C_{10}H_8\) is
The \(\textbf{INCORRECT}\) statement is
For an ideal gas, the molecular partition function in the canonical ensemble, that is proportional to the system volume (V), is the
Assertion (S): The total angular momentum for light atoms (low atomic number) is obtained by Russell–Saunders coupling, whereas \(jj\)-coupling is used for heavy atoms (high atomic number).
Reasoning (R): The spin–orbit interactions are weak in light atoms (low atomic number) and strong in heavy atoms (high atomic number).
The correct option is
The acetolysis product(s) of the given reaction (solvolysis of the benzylic tosylate in AcOH) is(are)

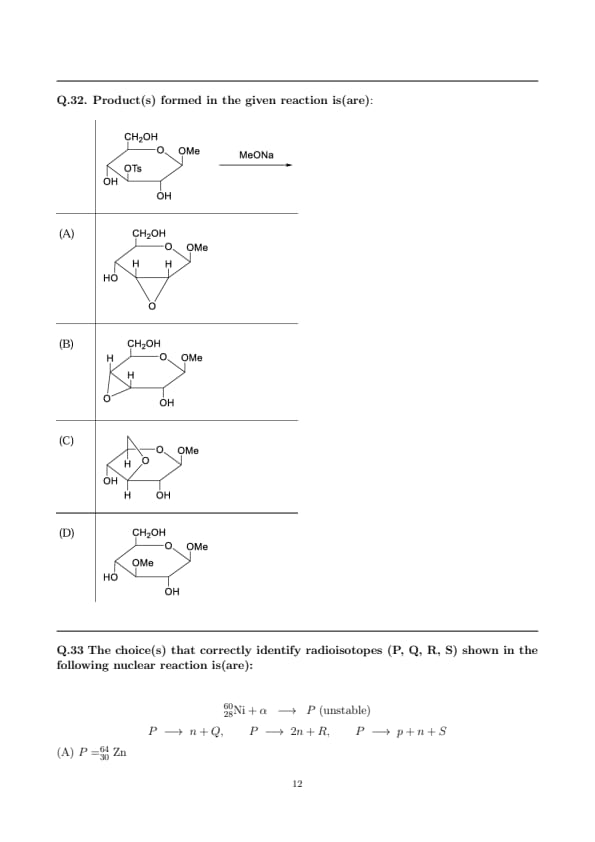
Product(s) formed in the given reaction is(are):

The choice(s) that correctly identify radioisotopes (P, Q, R, S) shown in the following nuclear reaction is(are):
\[ ^{60}_{28}Ni + \alpha \;\; \longrightarrow \;\; P \; (unstable) \] \[ P \;\longrightarrow\; n + Q, \qquad P \;\longrightarrow\; 2n + R, \qquad P \;\longrightarrow\; p + n + S \]
For the Lindemann–Hinshelwood mechanism of gas phase unimolecular reactions, the true statement(s) is(are)
The calculated magnetic moment of \([Ce(NO_3)_5]^{2-}\) is _____ BM (rounded off to two decimal places).
(Given: atomic number of Ce is 58)
A compound, \(\mathrm{C_{15}H_{16}O_2}\), has the following spectral data;
\(^1\)H NMR (ppm): 9.16 (s), 6.89 (d, \(J = 8\) Hz), 6.64 (d, \(J = 8\) Hz), 1.53 (s)
\(^ {13}\)C NMR (ppm): 154.7, 140.9, 127.1, 114.4, 40.7, 30.7
The structure of the compound is

The major product formed in the given reaction sequence is

E and F in the given reaction scheme are determined for the photolysis (\(h\nu\)) of the fused N–O heterocycle bearing an N-phenyl group, followed by thermolysis (\(\Delta\)).

M and N in the given reaction scheme are (Simmons–Smith on the alkene, then OsO\(_4\)/NMO):

In the \(^{1}\)H NMR spectrum, multiplicity of the signal (bold and underlined H atom) in the following species is
(I) \([\mathbf{H}\mathrm{Ni(OPEt_3)_4}]^{+}\)
(II) \(\mathrm{Ph_2Si(Me)\mathbf{H}}\)
(III) \(\mathrm{PH_3}\)
(IV) \((\mathrm{Cp^*})_2\mathrm{Zr}\mathbf{H}_2\) (Cp\(^*\)=pentamethylcyclopentadienyl)
The major product obtained by the treatment of \((\eta^{5}-\mathrm{C_5H_5})_2\mathrm{Ni}\) with Na/Hg in ethanol is
The number of shared corners of the constituent \(SiO_4\) units in orthosilicate, pyrosilicate, cyclic silicate and sheet silicate, respectively, are
Concentration of Q in a consecutive reaction \(P \xrightarrow{k_1} Q \xrightarrow{k_2} R\) is given by \([Q]=\dfrac{k_1[P]_0}{k_2-k_1}\left(e^{-k_1 t}-e^{-k_2 t}\right)\). If \(k_2=25~\mathrm{s^{-1}}\), the value of \(k_1\) that leads to the longest waiting time for Q to reach its maximum is
The wavefunction for Be\(^{3+}\) in a certain state is given by \(\psi = N e^{-\left(\tfrac{4}{a_0}\right)r}\), where \(N\) is the normalization constant, \(r\) is the distance of electron from the nucleus and \(a_0\) is the Bohr radius. The most probable distance of the electron from the nucleus in this state is
Match the following

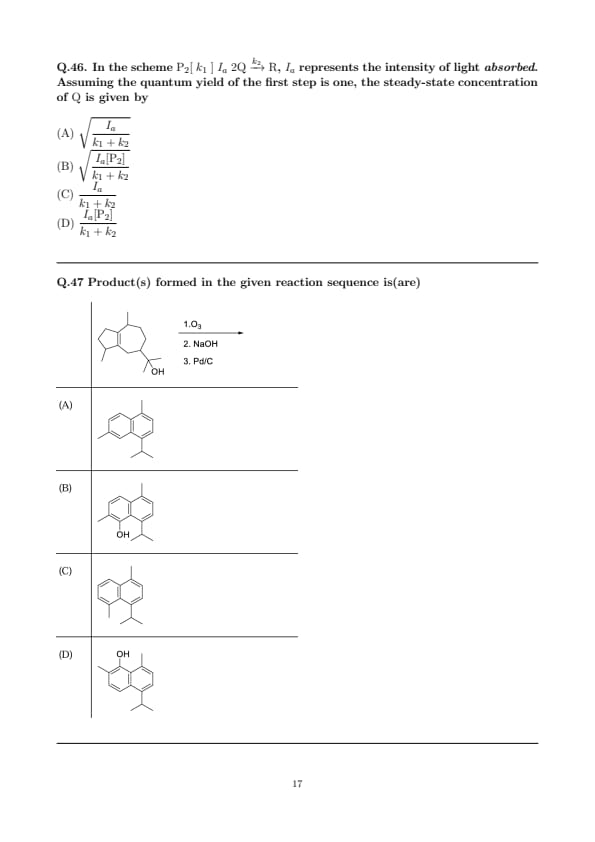
In the scheme \( \mathrm{P_2} [\;k_1\;]{\;I_a\;} 2\mathrm{Q} \xrightarrow{k_2} \mathrm{R} \), \(I_a\) represents the intensity of light absorbed. Assuming the quantum yield of the first step is one, the steady-state concentration of \(\mathrm{Q}\) is given by
Product(s) formed in the given reaction sequence is(are)

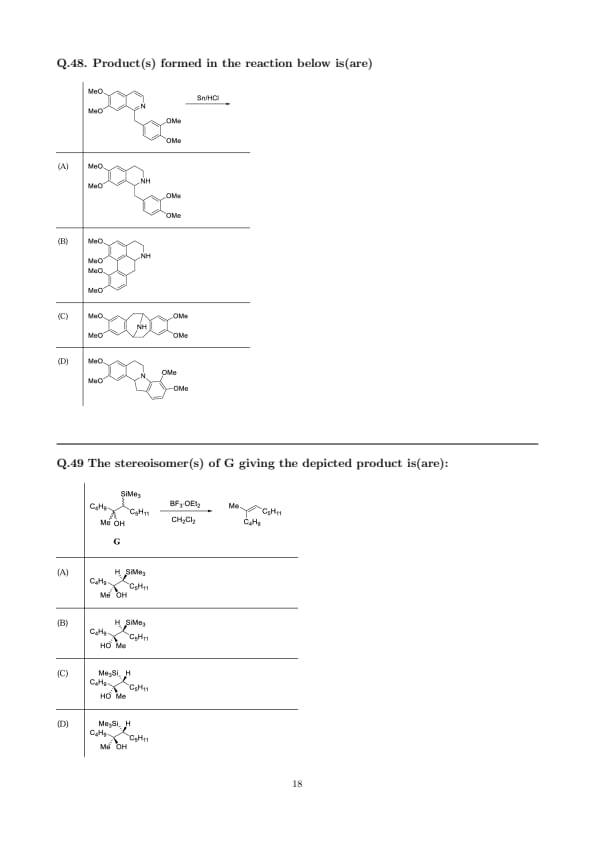
Product(s) formed in the reaction below is(are)

The stereoisomer(s) of G giving the depicted product is(are):

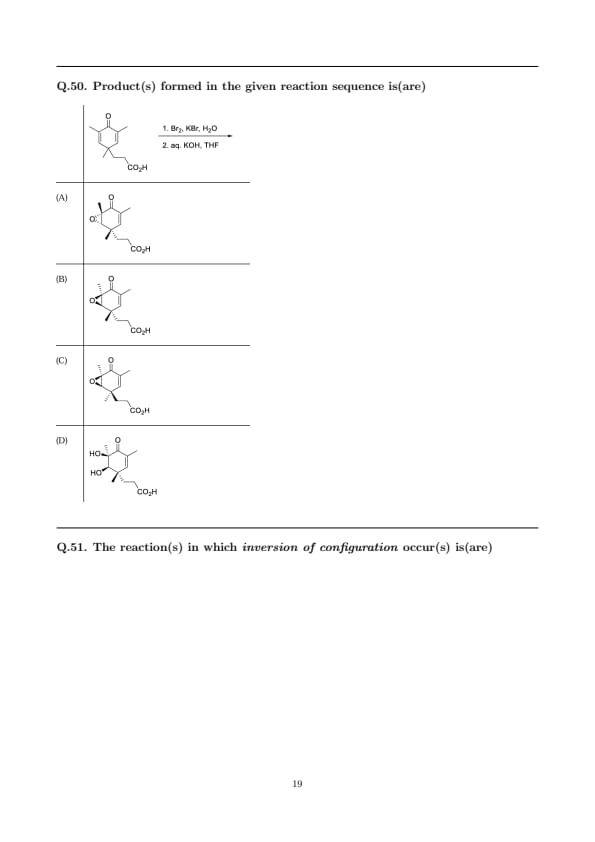
Product(s) formed in the given reaction sequence is(are)

The correct statement(s) regarding myoglobin (Mb) and haemoglobin (Hb) is(are)
The correct statement(s) regarding Co\(_2\)(CO)\(_8\) is(are)
The compound(s) having [Xe]4f\(^1\) configuration is(are)
(Given the atomic numbers Ce:58, Lu:71, Pr:59 and Nd:60)
The correct statement(s) for XeF\(_2\) is(are)
For the non-dissociative adsorption of a gas on solid,
Two different enzyme catalysis reactions I and II have identical Y-intercepts for the Lineweaver–Burke plots. The slope for reaction I is twice that of reaction II. If the initial concentrations of enzymes in I and II are same, the correct statement(s) is(are)
\[ \frac{1}{v}=\frac{1}{v_{\max}}+\frac{K_M}{v_{\max}}\frac{1}{[S]} \]
The enthalpy change for the exothermic reaction between BeI\(_2\) and HgF\(_2\) is _____ kJ mol\(^{-1}\) (rounded off to the nearest integer).
(Given bond dissociation energies in kJ mol\(^{-1}\): Be–F = 632, Be–I = 289, Hg–F = 268, Hg–I = 145)
Number of carbon atoms connected to the metal center in [W(C\(_{60}\))(CO)\(_5\)] is ____ (rounded off to the nearest integer).
(Given: atomic number of W = 74)
Two-component solid–liquid system of naphthalene–benzene forms a simple eutectic mixture. Assuming an ideal solution, the mole fraction of naphthalene in benzene at \(300~K\) and \(1~bar\) is _____ (rounded off to two decimal places).
(Given: freezing point \(T_{\mathrm{fp}}\) and enthalpy of fusion \(\Delta H_{\mathrm{fus}}\) of naphthalene are \(353~K\) and \(19.28~kJ mol^{-1}\), respectively; \(R=8.31~J K^{-1}mol^{-1}\))
The intrinsic viscosity of a sample of polystyrene in toluene is \(84\ cm^3\ g^{-1}\) at \(30\,^{\circ}C\). It follows the Mark–Houwink equation with empirical constants \(K = 1.05\times 10^{-2}\ cm^3\ g^{-1}\) and \(a = 0.75\). The molecular weight of the polymer is \(\_\_\_\_\_\_\times10^{3}\ g mol^{-1}\) (rounded off to the nearest integer).
According to Debye–Hückel limiting law, the mean molal activity coefficient for 0.87 g K\(_2\)SO\(_4\) (molar mass = 174 g mol\(^{-1}\)) in 1 kg of water at 25 °C is ____ (rounded off to two decimal places).
A solution is prepared by dissolving 128 g of naphthalene \(C_{10}H_8\) in 780 g of benzene \(C_6H_6\). The vapor pressure of pure benzene is 12.6 kPa at 25\(^\circ\)C. Assuming that naphthalene in benzene is an ideal solution, the partial vapor pressure of benzene is _____ kPa (rounded off to two decimal places).
For the galvanic cell: H\(_2\)(g) | HCl(aq) | Cl\(_2\)(g), the standard electromotive force is \[ E^0 = 1.73 - (1.25\times10^{-3})T + (1.00\times10^{-6})T^2 \quad (V, with T in K). \]
Find the standard enthalpy change \(\Delta_r H^0\) at \(300\ K\) (kJ mol\(^{-1}\)). Assume the cell reaction is written per mole of HCl, i.e., \(\tfrac12\mathrm{H_2} + \tfrac12\mathrm{Cl_2} \rightarrow \mathrm{HCl(aq)}\) so that \(n=1\). (Given \(F=96500\ C mol^{-1}\)).
A solution of three non-interacting compounds P, Q, and R is taken in a cuvette of 1 cm path length. Their concentrations are [P] = \(1\times10^{-6}\) M, [Q] = \(2\times10^{-6}\) M, [R] = \(3\times10^{-6}\) M and the molar extinction coefficients at 300 nm are \(\varepsilon_P = 1\times10^{5}\ \mathrm{M^{-1}\,cm^{-1}}\), \(\varepsilon_Q = 2\times10^{5}\ \mathrm{M^{-1}\,cm^{-1}}\) and \(\varepsilon_R = 3\times10^{5}\ \mathrm{M^{-1}\,cm^{-1}}\). The % transmittance at 300 nm is ___________ (rounded off to two decimal places)




























Comments