GATE 2024 Chemical Engineering Question Paper PDF is available here. IISc Banglore conducted GATE 2024 Chemical Engineering exam on February 4 in the Forenoon Session from 9:30 AM to 12:30 PM. Students have to answer 65 questions in GATE 2024 Chemical Engineering Question Paper carrying a total weightage of 100 marks. 10 questions are from the General Aptitude section and 55 questions are from Engineering Mathematics and Core Discipline.
GATE 2024 Chemical Engineering Question Paper with Answer Key PDF
| GATE 2024 Chemical Engineering Question Paper PDF | Check Solutions |
GATE 2024 Chemical Engineering Question Paper with Answer Key PDF
Question 1:
If '—>' denotes increasing order of intensity, then the meaning of the words [simmer — seethe — smolder] is analogous to [break — raze — _____ ]. Which one of the given options is appropriate to fill the blank?
Options:
- (1) obfuscate
- (2) obliterate
- (3) fracture
- (4) fissure
View Solution
Solution:
Step 1: Interpreting the analogy.
The sequence "simmer — seethe — smolder" suggests a gradual increase in intensity related to heat or emotion.
Similarly, in "break — raze — _____", we need a word that indicates a stronger, more destructive action than "raze."
Step 2: Reviewing the options.
- **Obfuscate:** Refers to making something unclear or confusing, not related to destruction.
- **Obliterate:** Means to completely destroy, making it suitable for the analogy.
- **Fracture:** Refers to breaking into parts, which is less intense than "raze."
- **Fissure:** Indicates a crack or split, also less severe than "raze."
Step 3: Concluding the solution.
"Obliterate" is the most appropriate word to complete the analogy as it represents the greatest level of destruction.
Question 2:
In a locality, the houses are numbered in the following way: The house-numbers on one side of a road are consecutive odd integers starting from 301, while the house-numbers on the other side of the road are consecutive even numbers starting from 302. The total number of houses is the same on both sides of the road. If the difference of the sum of the house-numbers between the two sides of the road is 27, then the number of houses on each side of the road is:
Options:
- (1) 27
- (2) 52
- (3) 54
- (4) 26
View Solution
Solution:
Step 1: Problem setup.
House numbers on one side are consecutive odd integers starting from 301. On the other side, they are consecutive even integers starting from 302. Let n represent the number of houses on each side.
Step 2: Sum of odd-numbered houses.
Using the arithmetic progression formula, the sum is:
n/2 * [2*301 + (n-1) * 2] = n/2 * (602 + 2n -2) = n/2 * (2n + 600) = n(n+300).
Step 3: Sum of even-numbered houses.
Similarly, the sum of even-numbered houses is:
n/2 * [2*302 + (n-1) * 2] = n/2 * (604 + 2n -2) = n/2 * (2n+602) = n(n+301).
Step 4: Calculating the difference in sums.
Given that the difference between the sums is 27:
n(n+301) - n(n+300) = 27
301n - 300n = 27 => n = 27.
Step 5: Final conclusion.
The number of houses on each side of the road is 27.
Question 3:
For positive integers p and q, with p/q ≠ 1, (p/q)(p-q) = (q/p)(q-p) Then:
Options:
- (1) qp = pq
- (2) qp = p2q
- (3) √q = √p
- (4) √q = q2/p
View Solution
Solution:
Step 1: Simplify the given equation.
Starting with:
(p/q)(p-q) = (q/p)(q-p)
Rewriting:
(p/q)(p-q) = (p/q)-(q-p) = (p/q)(p-q)
(p/q)(p-q) = (p/q)(p-q)
Step 2: Identifying the relationship.
Equating powers of p and q:
p/q = q/p or pp-q * qq-p = 1
p(p-q) / q(p-q) = q(q-p) / p(q-p)
p(p-q) * p(q-p) = q(q-p) * q(p-q)
pp-q+q-p = qq-p+p-q
p0 = q0
1 = 1
p/q = q/p
p2 = q2
p = q
This does not satify, p/q ≠ 1, let try another way
From:
(p/q)(p-q) = (q/p)(q-p)
(p/q)(p-q) = (p/q)-(q-p)
(p/q)(p-q) = (p/q)(p-q)
(p/q)p * (q/p)q=1
pp * qq / qp * pq = 1
pp*qq = qp * pq
pp/pq = qp/qq
pp-q = qp-q
p = q
Lets try,
(p/q)(p-q) = (q/p)(q-p)
(p/q)(p-q) = (p/q)-(q-p)
p(p-q) / q(p-q) = p(p-q) / q(p-q)
so only solution is p=q, which is not true, thus the powers should be zero p-q =0 or q-p=0 which is not valid as well.
Let try, (p/q)(p-q) = (q/p)(q-p) = (p/q)-(q-p) = (p/q)p-q
thus (q/p) = (p/q), p2 = q2 p=q which is not true. let assume that power is = 1
Then,
p/q = q/p => p2 = q2, p= q which is also invalid
If (p/q)(p-q) = (q/p)(q-p), then consider cases, p = q does not work as p/q ≠ 1
let, (p/q) = a
a(p-q) = a(q-p)-1
a(p-q) = a(p-q)
p-q = 1 then (p/q) = q/p which means that p2 = q2 which means p=q which cant be true
or - (p-q) = q-p which does not work either
only works when p/q = q/p
qp = pq
Step 3: Verification.
The relationship qp = pq satisfies the equation, confirming the solution.
Question 4:
Which one of the given options is a possible value of X in the following sequence?
3, 7, 15, X, 63, 127, 255
Options:
- (1) 35
- (2) 40
- (3) 45
- (4) 31
View Solution
Solution:
Step 1: Analyze the given sequence.
The sequence 3, 7, 15, X, 63, 127, 255 shows a pattern where each number is one less than a power of 2.
Step 2: Express each term.
3 = 22 – 1, 7 = 23 – 1, 15 = 24 − 1, X = 25 − 1 = 31.
The subsequent terms follow the same pattern: 63 = 26 – 1, 127 = 27 – 1, 255 = 28 – 1.
Step 3: Conclusion.
The missing term X = 31.
Question 5:
On a given day, how many times will the second-hand and the minute-hand of a clock cross each other during the clock time 12:05:00 hours to 12:55:00 hours?
Options:
- (1) 51
- (2) 49
- (3) 50
- (4) 55
View Solution
Solution:
Step 1: Understand the motion of the second and minute hands.
The second-hand completes one full revolution (360 degrees) in 60 seconds, while the minute-hand completes one revolution in 3600 seconds (1 hour).
Step 2: Calculate the crossings in one minute.
In one minute, the second-hand crosses the minute-hand exactly once.
Step 3: Calculate the crossings between 12:05:00 and 12:55:00.
The time interval between 12:05:00 and 12:55:00 is 50 minutes. Hence, the second-hand and minute-hand will cross each other exactly 50 times during this period.
Step 4: Conclusion.
The total number of crossings is 50.
Question 6:
In the given text, the blanks are numbered (i)—(iv). Select the best match for all the blanks. From the ancient Athenian arena to the modern Olympic stadiums, athletics (i) the potential for a spectacle. The crowd (ii) with bated breath as the Olympian artist twists his body, stretching the javelin behind him. Twelve strides in, he begins to cross-step. Six cross-steps (iii) in an abrupt stop on his left foot. As his body (iv) like a door turning on a hinge, the javelin is launched skyward at a precise angle.
Options:
- (1) hold, waits, culminates, pivot
- (2) holds, wait, culminates, pivot
- (3) hold, wait, culminate, pivots
- (4) holds, waits, culminate, pivots
View Solution
Solution:
Step 1: Analyze the grammar.
Each blank requires a verb that agrees with the subject and fits the tense of the sentence.
- **(i) holds:** Matches the singular subject "athletics."
- **(ii) waits:** Agrees with the singular subject "crowd."
- **(iii) culminate:** Fits the progression of actions leading to a conclusion.
- **(iv) pivots:** Aligns with the description of the athlete's motion.
Step 2: Conclusion.
The correct answer is (4).
Question 7:
Three distinct sets of indistinguishable twins are to be seated at a circular table that has 8 identical chairs. Unique seating arrangements are defined by the relative positions of the people. How many unique seating arrangements are possible such that each person is sitting next to their twin?

Options:
- (1) 12
- (2) 14
- (3) 10
- (4) 28
View Solution
Solution:
Step 1: Problem Setup.
We are tasked with finding the number of unique circular arrangements of 5 units, out of which 2 are alike (E and E).
Step 2: Formula for circular arrangements.
The total number of arrangements in a circle, accounting for repetition, is given by:
(n-1)!/k!,
where n is the total number of units and k is the number of identical units.
Step 3: Substituting the values.
Here, n = 5 and k = 2 (for E and E):
Number of unique arrangements = (5-1)! / 2!.
Step 4: Simplifying the factorials.
Number of unique arrangements = 4! / 2! = (4*3*2*1) / (2*1) = 12.
Step 5: Calculating the result.
Number of unique arrangements = 12.
Question 8:
The chart given below compares the Installed Capacity (MW) of four power generation technologies, T1, T2, T3, and T4, and their Electricity Generation (MWh) in a time of 1000 hours (h). The Capacity Factor of a power generation technology is: Capacity Factor = Electricity Generation (MWh) / (Installed Capacity (MW) × 1000 (h)). Which one of the given technologies has the highest Capacity Factor?

Options:
- (1) T1
- (2) T2
- (3) T3
- (4) T4
View Solution
Solution:
Step 1: Understand the Capacity Factor formula.
The Capacity Factor is calculated as:
Capacity Factor = Electricity Generation (MWh) / (Installed Capacity (MW) × 1000 (h)).
Step 2: Compare the Capacity Factor for each technology.
Using the values provided in the chart, calculate the Capacity Factor for T1, T2, T3, and T4.
Step 3: Identify the highest Capacity Factor.
After calculation, T1 has the highest Capacity Factor.
Step 4: Conclusion.
The technology with the highest Capacity Factor is T1.
Question 9:
In the 4 x 4 array shown below, each cell of the first three columns has either a cross (X) or a number, as per the given rule. Rule: The number in a cell represents the count of crosses around its immediate neighboring cells (left, right, top, bottom, diagonals). As per this rule, the maximum number of crosses possible in the empty column is:

Options:
- (1) 0
- (2) 1
- (3) 2
- (4) 3
View Solution
Solution:
Step 1: Analyze the rule.
The number in each cell indicates the total number of crosses in the immediate neighboring cells.
Step 2: Calculate the maximum crosses for the empty column.
Using logical deductions and adjacency constraints, place crosses to maximize the count while satisfying the rule. The maximum possible number of crosses in the empty column is 2.
Step 3: Conclusion.
The maximum number of crosses in the empty column is 2.
Question 10:
During a half-moon phase, the Earth-Moon-Sun form a right triangle. If the Moon-Earth-Sun angle at this half-moon phase is measured to be 89.85°, the ratio of the Earth-Sun and Earth-Moon distances is closest to:
Options:
- (1) 328
- (2) 382
- (3) 238
- (4) 283
View Solution
Solution:
Step 1: Use the trigonometric relationship.
During the half-moon phase, the Earth-Moon-Sun form a right triangle. Using the tangent of the Moon-Earth-Sun angle 89.85°:
tan θ = Earth-Moon distance / Earth-Sun distance.
Rewriting:
Earth-Sun distance = Earth-Moon distance / tan θ.
Step 2: Substitute the values.
With θ = 89.85°, tan θ ≈ 0.002618. Hence:
Earth-Sun distance / Earth-Moon distance = 1 / 0.002618 ≈ 382.
Step 3: Conclusion.
The ratio of Earth-Sun to Earth-Moon distances is closest to 382.
Question 11:
The first non-zero term in the Taylor series expansion of (1 – x) – e-x about x = 0 is:
Options:
- (1) 1
- (2) -1
- (3) -x2/2
- (4) x2/2
View Solution
Solution:
Step 1: Expand e-x as a Taylor series about x = 0.
The Taylor series expansion of e-x is:
e-x = 1 - x + x2/2 - x3/6 + ...
Step 2: Simplify (1 – x) – e-x.
Substitute the expansion of e-x into (1 − x) – e-x:
(1 - x) - e-x = (1 - x) - (1 - x + x2/2 - x3/6 + ...).
Simplify the terms:
(1 - x) - e-x = 1 - x - 1 + x - x2/2 + x3/6 - ...
(1 - x) - e-x = - x2/2 + x3/6 - ...
Step 3: Identify the first non-zero term.
The first non-zero term is -x2/2.
Step 4: Conclusion.
The first non-zero term in the Taylor series expansion is -x2/2.
Question 12:
Consider the normal probability distribution function
f(x) = 4 / √2π * e-8(x+3)2.
If μ and σ are the mean and standard deviation of f(x) respectively, then the ordered pair (μ, σ) is:
Options:
- (1) (3, 1/4)
- (2) (-3, 1/4)
- (3) (3, 4)
- (4) (-3, 4)
View Solution
Solution:
Step 1: Analyze the general form of the normal distribution.
The general form of the normal probability distribution is:
f(x) = 1 / (√(2π)σ) * e-((x-μ)2/(2σ2)),
where µ is the mean, and σ is the standard deviation.
Step 2: Match the given equation to the general form.
The given function is:
f(x) = 4 / √2π * e-8(x+3)2.
Compare this with the general form. The coefficient of (x + 3)2 inside the exponent is 8. This gives:
1 / (2σ2) = 8 => σ2 = 1/16 => σ = 1/4.
The mean µ is obtained from the shift (x + 3), which indicates μ = -3.
Step 3: Conclusion.
The ordered pair (μ, σ) is (-3, 1/4).
Question 13:
If z1 = −1 + i and z2 = 2i, where i = √-1, then Arg(z1/z2) is:
Options:
- (1) 5π/4
- (2) π/4
- (3) 3π/4
- (4) -π/4
View Solution
Solution:
Step 1: Represent z1 and z2 in polar form.
For z1 = -1 + i:
|z1| = √((-1)2 + 12) = √2, and Arg(z1) = tan-1(1/-1) + π = 3π/4.
For z2 = 2i:
|z2| = |2i| = 2, and Arg(z2) = π/2.
Step 2: Division of z1 and z2.
The modulus of z1/z2 is:
|z1/z2| = |z1| / |z2| = √2 / 2.
The argument of z1/z2 is:
Arg(z1/z2) = Arg(z1) - Arg(z2) = 3π/4 - π/2.
Simplify:
Arg(z1/z2) = 3π/4 - 2π/4 = π/4.
Step 3: Conclusion.
The argument of z1/z2 is π/4. If you take -π/4 the math changes to
3π/4 - π/2 = 3π/4 - 2π/4 = π/4
Question 14:
A homogeneous azeotropic distillation process separates an azeotropic AB binary feed using a heavy entrainer, E, as shown in the figure. The loss of E in the two product streams is negligible, so E circulates around the process in a closed circuit. For a distillation column with fully specified feed(s), given operating pressure, a single distillate stream, and a single bottoms stream, the steady-state degrees of freedom equals 2. For the process in the figure with a fully specified AB feed stream and given column operating pressures, the steady-state degrees of freedom equals:

Options:
- (1) 3
- (2) 4
- (3) 5
- (4) 6
View Solution
Solution:
Step 1: Understand the degrees of freedom in distillation processes.
The degrees of freedom (DoF) represent the number of independent variables that can be adjusted to achieve steady-state operation. For a basic distillation column with fully specified feed streams, given operating pressure, and single distillate and bottoms streams, the DoF is 2.
Step 2: Include the azeotropic distillation effect.
In this problem, an additional constraint is imposed by the heavy entrainer E, which circulates in a closed loop with negligible loss. The azeotropic behavior introduces extra variables such as reflux ratio, flow rates, and temperature specifications, leading to additional degrees of freedom.
Step 3: Total degrees of freedom.
Adding the effects of azeotropic distillation and entrainer recycling increases the total degrees of freedom to 5.
Step 4: Conclusion.
The steady-state degrees of freedom for this process is 5.
Question 15:
An infinitely long cylindrical water filament of radius R is surrounded by air. Assume water and air to be static. The pressure outside the filament is Pout, and the pressure inside is Pin. If γ is the surface tension of the water-air interface, then Pin - Pout is:
Options:
- (1) γ/R
- (2) 0
- (3) γ/R
- (4) 4γ/R
View Solution
Solution:
Step 1: Use the Laplace pressure equation.
The Laplace pressure equation for a cylindrical interface is:
Pin - Pout = γ/R.
Step 2: Distinguish from spherical surfaces.
For spherical surfaces, the pressure difference is 2γ/R because the curvature has two radii of equal magnitude. For a cylindrical interface, there is only one radius of curvature, resulting in γ/R.
Step 3: Conclusion.
The pressure difference Pin – Pout is γ/R.
Question 16:
The velocity field in an incompressible flow is v = axy i + vy j + β k, where i, j, k are unit vectors in the (x, y, z) Cartesian coordinate system. Given that a and β are constants, and vy = 0 at y = 0, the correct expression for vy is:
Options:
- (1) -axy
- (2) -ay2/2
- (3) ay2/2
- (4) axy/2
View Solution
Solution:
Step 1: Apply the incompressibility condition.
For incompressible flow, the divergence of the velocity field is zero:
∇ ⋅ v = ∂vx/∂x + ∂vy/∂y + ∂vz/∂z = 0.
Substitute vx = axy, vy = vy(y), vz = β:
∂(axy)/∂x + ∂vy/∂y + ∂β/∂z = 0.
∂vy/∂y + 0 = 0.
Step 2: Solve for vy.
∂vy/∂y = -ay.
Integrate:
vy = -ay2/2 + C.
Step 3: Apply boundary conditions.
Given vy = 0 at y = 0:
0 = -a(0)2/2 + C => C = 0.
Thus,
vy = -ay2/2.
Step 4: Conclusion.
The correct expression for vy is -ay2/2.
Question 17:
Consider the steady, uni-directional diffusion of a binary mixture of A and B across a vertical slab of dimensions 0.2 m × 0.1 m × 0.02 m as shown in the figure. The total molar concentration of A and B is constant at 100 mol m-3. The mole fraction of A on the left and right faces of the slab are maintained at 0.8 and 0.2, respectively. If the binary diffusion coefficient DAB = 1 × 10-5 m2 s-1, the molar flow rate of A in mol s-1, along the horizontal x-direction is:

Options:
- (1) 6 × 10-4
- (2) 6 × 10-6
- (3) 3 × 10-6
- (4) 3 × 10-4
View Solution
Solution:
Step 1: Use Fick's first law of diffusion.
The molar flux JA is given by:
JA = -DAB * ∂CA/∂x.
Step 2: Calculate the concentration gradient.
The mole fraction gradient of A is:
∂yA/∂x = (yA,right - yA,left)/Δx = (0.2 - 0.8)/0.02 = -30 m-1.
The total molar concentration is C = 100 mol m-3. The concentration gradient is:
∂CA/∂x = C * ∂yA/∂x = 100 * (-30) = -3000 mol m-4.
Step 3: Calculate the molar flux.
Substitute DAB = 1 × 10-5:
JA = -DAB * ∂CA/∂x = - (1 × 10-5) * (-3000) = 3 × 10-2 mol m-2 s-1.
Step 4: Calculate the molar flow rate.
The cross-sectional area is:
A = 0.2 m * 0.1 m = 0.02 m2.
The molar flow rate is:
NA = JA * A = (3 × 10-2) * (0.02) = 6 × 10-4 mol s-1.
Step 5: Conclusion.
The molar flow rate of A is 6 × 10-4 mol s-1.
Question 18:
Consider a vapour-liquid mixture of components A and B that obeys Raoult's law. The vapour pressure of A is half that of B. The vapour phase concentrations of A and B are 3 mol m-3 and 6 mol m-3, respectively. At equilibrium, the ratio of the liquid phase concentration of A to that of B is:
Options:
- (1) 1.0
- (2) 0.5
- (3) 2.0
- (4) 1.5
View Solution
Solution:
Step 1: Understand Raoult's law.
According to Raoult's law, the partial pressure of a component is:
Pi = xi * Pi*,
where Pi is the partial pressure, xi is the mole fraction in the liquid phase, and Pi* is the pure component vapor pressure.
Step 2: Relate vapour phase concentrations.
The vapour phase concentrations are proportional to partial pressures:
PA/PB = CA/CB = 3/6 = 0.5.
Step 3: Relate liquid phase mole fractions.
Using Raoult's law:
PA/PB = (xA * PA*) / (xB * PB*).
Given PA* = 1/2 * PB*, substitute into the equation:
0.5 = (xA * 0.5 * PB*) / (xB * PB*).
Simplify:
0.5 = (0.5 * xA) / xB => xA/xB = 1.0.
Step 4: Conclusion.
The ratio of the liquid phase concentration of A to B is 1.0.
Question 19:
The ratio of the activation energy of a chemical reaction to the universal gas constant is 1000 K. The temperature dependence of the reaction rate constant follows the collision theory. The ratio of the rate constant at 600 K to that at 400 K is:
Options:
- (1) 2.818
- (2) 4.323
- (3) 1.502
- (4) 1.000
View Solution
Solution:
Step 1: Use the Arrhenius equation.
The Arrhenius equation is:
k = A * e(-Ea/RT).
For two temperatures T1 and T2, the ratio of rate constants is:
k2/k1 = e(Ea/R)*(1/T1 - 1/T2).
Step 2: Substitute the given values.
Ea/R = 1000 K, T1 = 400 K, T2 = 600 K.
k2/k1 = e1000*(1/400 - 1/600).
Step 3: Simplify the exponent.
1/400 - 1/600 = (3 - 2)/1200 = 1/1200.
Step 4: Calculate the exponential.
k2/k1 = e1000/1200 = e0.8333 ≈ 2.818.
Step 5: Conclusion.
The ratio of the rate constant at 600 K to that at 400 K is 2.818.
Question 20:
The rate of a reaction A → B is 0.2 mol m-3 s-1 at a particular concentration CA1. The rate constant of the reaction at a given temperature is 0.1 m3 mol-1 s-1. If the reactant concentration is increased to 10CA1 at the same temperature, the reaction rate is:
Options:
- (1) 20
- (2) 10
- (3) 100
- (4) 50
View Solution
Solution:
Step 1: Use the rate law.
The rate law for the reaction is:
r = kCA2.
Step 2: Relate the rates.
When CA is increased to 10CA1:
r2/r1 = (CA2/CA1)2 = (10CA1/CA1)2 = 102 = 100.
r2 = 100 * r1 = 100 * 0.2 = 20 mol m-3 s-1.
Step 3: Conclusion.
The reaction rate is 20 mol m-3 s-1.
Question 21:
Two parallel first-order liquid phase reactions A k1→ B and A k2→ C are carried out in a well-mixed isothermal batch reactor. The initial concentration of A in the reactor is 1 kmol m-3, while that of B and C is zero. After 2 hours, the concentration of A reduces to half its initial value, and the concentration of B is twice that of C. The rate constants k1 and k2, in h-1, are respectively:
Options:
- (1) 0.40, 0.20
- (2) 0.23, 0.12
- (3) 0.50, 0.25
- (4) 0.36, 0.18
View Solution
Solution:
Step 1: Use the first-order decay law for A.
The decay of A is governed by the overall rate constant k:
ln(CA/CA0) = -kt.
Here:
CA = CA0/2,
CA0 = 1 kmol m-3,
t = 2 hours.
Substitute into the equation:
ln(1/2) = -k * 2.
Simplify:
ln(1/2) = -2k => k = ln(2)/2 ≈ 0.3466 h-1.
Step 2: Relate k1 and k2.
The overall rate constant is the sum of the individual rate constants for parallel reactions:
k = k1 + k2.
Thus:
k1 + k2 = 0.3466.
Step 3: Use the concentration ratio of B and C.
The concentration of B is twice that of C:
CB = 2CC.
The concentrations are related to the rate constants:
CB/CC = k1/k2.
Substitute CB/CC = 2:
k1/k2 = 2 => k1 = 2k2.
Step 4: Solve for k1 and k2.
Substitute k1 = 2k2 into k1 + k2 = 0.3466:
2k2 + k2 = 0.3466 => 3k2 = 0.3466 => k2 ≈ 0.1155 h-1.
Then:
k1 = 2k2 = 2 * 0.1155 ≈ 0.231 h-1.
Step 5: Conclusion.
The rate constants are k1 ≈ 0.23 h-1 and k2 ≈ 0.12 h-1.
Question 22:
Consider the block diagram in the figure with control input u, disturbance d, and output y. For the feedforward controller, the ordered pair (K, α/β) is:

Options:
- (1) (0.5, 2)
- (2) (-0.5, 0.5)
- (3) (-2, 2)
- (4) (2, 0.5)
View Solution
Solution:
Step 1: Analyze the block diagram.
The process transfer function is:
G(s) = 2 / (0.5s + 1)2.
The disturbance transfer function is:
Gd(s) = 1 / (s + 1)2.
The feedforward controller transfer function is:
F(s) = K (αs + 1)/(βs + 1).
Step 2: Apply disturbance rejection condition.
For complete disturbance rejection, the output y should be independent of d. The feedforward controller F(s) must cancel the disturbance effect. This requires:
F(s) * Gd(s) = G(s).
Substitute G(s) and Gd(s):
K * (αs + 1) / (βs + 1) * 1 / (s + 1)2 = 2 / (0.5s + 1)2.
Step 3: Match the numerator and denominator.
Equating denominators:
(βs + 1)2 * (s + 1)2 = (0.5s + 1)2.
Expand and compare coefficients to solve for β:
β = 0.5.
Equating numerators:
K * (αs + 1)2 = 2.
Substitute β = 0.5 and solve for K and α:
K = -0.5, α = 0.5.
Step 4: Conclusion.
The ordered pair (K, α/β) is (-0.5, 0.5).
Question 23:
Consider the control structure for the overhead section of a distillation column shown in the figure. The composition controller (CC) controls the heavy key impurity in the distillate by adjusting the setpoint of the reflux flow controller in a cascade arrangement. The sign of the controller gain for the pressure controller (PC) and that for the composition controller (CC) are, respectively:

Options:
- (1) negative, negative
- (2) negative, positive
- (3) positive, positive
- (4) positive, negative
View Solution
Solution:
Step 1: Analyze the pressure controller (PC).
The pressure controller (PC) maintains the column pressure by adjusting the overhead vapor flow. If the pressure increases, the PC increases the valve opening to reduce the pressure. Thus, the PC must have a positive gain for proper action.
Step 2: Analyze the composition controller (CC).
The composition controller (CC) controls the impurity level in the distillate by adjusting the reflux flow rate. If the impurity increases, the CC increases the reflux flow to improve separation. This requires a negative gain for the CC.
Step 3: Conclusion.
The pressure controller has a positive gain, and the composition controller has a negative gain.
Question 24:
Which one of the given statements is correct with reference to gas-liquid contactors for mass transfer applications?
Options:
- (1) A tray tower is more suitable for foaming systems than a packed tower.
- (2) Tray towers are preferred over packed towers for systems requiring frequent cleaning.
- (3) For a given liquid flow rate, the gas flow rate in the loading region is greater than that in the flooding region.
- (4) Flooding can never occur for counter-current contact.
View Solution
Solution:
Step 1: Compare tray and packed towers for cleaning.
Tray towers are easier to clean than packed towers due to their open design. This makes them suitable for systems requiring frequent cleaning, such as viscous or fouling liquids.
Step 2: Evaluate the other options.
(1): Packed towers are better for foaming systems, as they minimize foam generation due to lower liquid holdup.
(3): The gas flow rate in the flooding region exceeds that in the loading region.
(4): Flooding can occur in counter-current contactors at high gas velocities.
Step 3: Conclusion.
The correct statement is that tray towers are preferred for systems requiring frequent cleaning.
Question 25:
In an ammonia manufacturing facility, the necessary hydrogen is generated from methane. The facility consists of the following process units: P: Methanator, Q: CO shift convertor, R: CO2 stripper, S: Reformer, T: Ammonia convertor. The correct order of these units, starting from methane feed, is:
Options:
- (1) S, Q, R, P, T
- (2) P, Q, R, S, T
- (3) S, P, Q, R, T
- (4) P, S, T, Q, R
View Solution
Solution:
Step 1: Describe the sequence of operations.
1. Reformer (S): Methane reacts with steam to produce hydrogen, carbon monoxide, and carbon dioxide.
2. CO shift converter (Q): Carbon monoxide reacts with water to form more hydrogen and carbon dioxide.
3. CO2 stripper (R): Carbon dioxide is removed.
4. Methanator (P): Residual CO and CO2 are converted to methane.
5. Ammonia converter (T): Hydrogen reacts with nitrogen to form ammonia.
Step 2: Identify the correct order.
The correct sequence is S → Q → R → P → T.
Step 3: Conclusion.
The correct order is S, Q, R, P, T.
Question 26:
Consider a linear homogeneous system of equations Ax = 0, where A is an n × n matrix, x is an n × 1 vector, and 0 is an n × 1 null vector. Let r be the rank of A. For a non-trivial solution to exist, which of the following conditions is/are satisfied?
Options:
- (1) Determinant of A = 0
- (2) r = m < n
- (3) r < n
- (4) Determinant of A ≠ 0
View Solution
Solution:
Step 1: Analyze the system of equations.
For a homogeneous system Ax = 0:
- A non-trivial solution exists if det(A) = 0, indicating that A is singular.
- If r < n, the system has n – r free variables, allowing non-trivial solutions.
Step 2: Evaluate the options.
(1): True, as det(A) = 0 ensures a non-trivial solution.
(2): Not always true; r < n is the correct condition.
(3): True, as r < n implies free variables exist.
(4): False, as det(A) ≠ 0 leads to only the trivial solution.
Step 3: Conclusion.
The correct conditions are (1) and (3).
Question 27:
If the Prandtl number Pr = 0.01, which of the following statements is/are correct?
Options:
- (1) The momentum diffusivity is much larger than the thermal diffusivity.
- (2) The thickness of the momentum boundary layer is much smaller than that of the thermal boundary layer.
- (3) The thickness of the momentum boundary layer is much larger than that of the thermal boundary layer.
- (4) The momentum diffusivity is much smaller than the thermal diffusivity.
View Solution
Solution:
Step 1: Understand the Prandtl number.
The Prandtl number is defined as:
Pr = ν/α,
where:
- ν is the momentum diffusivity (kinematic viscosity),
- α is the thermal diffusivity.
For Pr = 0.01:
ν << α.
This implies that the momentum diffusivity is much smaller than the thermal diffusivity.
Step 2: Boundary layer thickness.
The thickness of a boundary layer is inversely related to diffusivity. For small Pr values (Pr << 1):
- The thermal boundary layer is much thicker than the momentum boundary layer because thermal diffusivity dominates.
Step 3: Evaluate the statements.
(1): Incorrect. Momentum diffusivity is much smaller than thermal diffusivity for Pr = 0.01.
(2): Correct. The momentum boundary layer is much thinner than the thermal boundary layer.
(3): Incorrect. The thermal boundary layer is thicker than the momentum boundary layer.
(4): Correct. The momentum diffusivity is much smaller than the thermal diffusivity.
Step 4: Conclusion.
The correct statements are (2) and (4).
Question 28:
For the electrolytic cell in a chlor-alkali plant, which of the following statements is/are correct?
Options:
- (1) A membrane cell operates at a higher brine concentration than a diaphragm cell.
- (2) Chlorine gas is produced at the cathode.
- (3) Hydrogen gas is produced at the cathode.
- (4) The caustic product stream exits the cathode compartment.
View Solution
Solution:
Step 1: Analyze the operation of the chlor-alkali electrolytic cell.
- A membrane cell operates at a higher brine concentration than a diaphragm cell for better separation efficiency.
- Chlorine gas is produced at the anode due to oxidation of chloride ions.
- Hydrogen gas is produced at the cathode due to the reduction of water.
- The caustic product stream (sodium hydroxide solution) exits from the cathode compartment.
Step 2: Evaluate the statements.
(1): Correct. Membrane cells use higher brine concentrations for better ion separation.
(2): Incorrect. Chlorine gas is produced at the anode, not the cathode.
(3): Correct. Hydrogen gas is produced at the cathode.
(4): Correct. The caustic product stream exits the cathode compartment.
Step 3: Conclusion.
The correct statements are (1), (3), and (4).
Question 29:
Which of the following statements with reference to the petroleum/petrochemical industry is/are correct?
Options:
- (1) Catalytic hydrocracking converts heavier hydrocarbons to lighter hydrocarbons.
- (2) Catalytic reforming converts straight-chain hydrocarbons to aromatics.
- (3) Cumene is manufactured by the catalytic alkylation of benzene with propylene.
- (4) Vinyl acetate is manufactured by reacting methane with acetic acid over a palladium catalyst.
View Solution
Solution:
Step 1: Analyze the processes.
(1): Catalytic hydrocracking breaks heavier hydrocarbons into lighter fractions like gasoline and diesel.
(2): Catalytic reforming converts straight-chain hydrocarbons into aromatics like benzene and toluene.
(3): Cumene is produced via catalytic alkylation of benzene with propylene.
(4): Vinyl acetate is manufactured by reacting ethylene, not methane, with acetic acid over a palladium catalyst.
Step 2: Conclusion.
The correct statements are (1), (2), and (3).
Question 30:
Consider a matrix A = -5 a
-2 -2 , where a is a constant. If the eigenvalues of A are -1 and -6, then the value of a, rounded off to the nearest integer, is:
Options:
View Solution
Solution:
Step 1: Use the eigenvalue property.
For a matrix A, the sum of eigenvalues equals the trace, and the product of eigenvalues equals the determinant.
- Trace of A:
Trace = -5 + (-2) = -7.
- Eigenvalues -1 and -6:
Sum of eigenvalues = -1 + (-6) = -7 (matches the trace).
Step 2: Use the determinant property.
The determinant of A is:
Det(A) = (-5)(-2) – (-2)(a) = 10 + 2a.
The product of eigenvalues is:
Product of eigenvalues = (-1)(-6) = 6.
Equate the determinant to the product of eigenvalues:
10 + 2a = 6 => 2a = -4 => a = -2.
Step 3: Conclusion.
The value of a is -2.
Question 31:
Consider the reaction N2(g) + 3H2(g) → 2NH3(g) in a continuous flow reactor under steady-state conditions. The component flow rates at the reactor inlet are: F0N2 = 100 mol/s, F0H2 = 300 mol/s, F0inert = 1 mol/s. If the fractional conversion of H2 is 0.60, the outlet flow rate of N2, in mol/s, rounded off to the nearest integer, is:
View Solution
Solution:
Step 1: Determine the moles of H2 reacted.
The fractional conversion of H2 is given as 0.60. The moles of H2 reacted are:
Moles of H2 reacted = F0H2 * 0.60 = 300 * 0.60 = 180 mol/s.
Step 2: Use the stoichiometry of the reaction.
From the stoichiometry N2 + 3H2 → 2NH3:
Moles of N2 reacted = 1/3 * Moles of H2 reacted = 1/3 * 180 = 60 mol/s.
Step 3: Calculate the outlet flow rate of N2.
The inlet flow rate of N2 is F0N2 = 100 mol/s. The outlet flow rate is:
FN2,out = F0N2 − Moles of N2 reacted = 100 − 60 = 40 mol/s.
Step 4: Conclusion.
The outlet flow rate of N2 is 40 mol/s.
Question 32:
Consider a binary mixture of components A and B at temperature T and pressure P. Let VA and VB be the partial molar volumes of A and B, respectively. At a certain mole fraction of A, xA: (∂VA/∂xA)T,P = 22 cm3 mol-1, (∂VB/∂xA)T,P = -18 cm3 mol-1. The value of xA, rounded off to 2 decimal places, is:
View Solution
Solution:
Step 1: Use the Gibbs-Duhem relation.
The Gibbs-Duhem relation for partial molar properties in a binary mixture is:
xA * (∂VA/∂xA)T,P + (1 - xA) * (∂VB/∂xA)T,P = 0.
Step 2: Substitute the given values.
22xA + (-18)(1 - xA) = 0.
Simplify:
22xA - 18 + 18xA = 0 => 40xA = 18 => xA = 18/40 = 0.45.
Step 3: Conclusion.
The mole fraction of A is xA = 0.45.
Question 33:
Consider the steady, uni-directional, fully-developed, pressure-driven laminar flow of an incompressible Newtonian fluid through a circular pipe of inner radius 5.0 cm. The magnitude of shear stress at the inner wall of the pipe is 0.1 N m-2. At a radial distance of 1.0 cm from the pipe axis, the magnitude of the shear stress, in N m-2, rounded off to 3 decimal places, is:
View Solution
Solution:
Step 1: Shear stress variation in a circular pipe.
For steady, fully-developed, laminar flow in a circular pipe, the shear stress varies linearly with the radial position r:
τ(r) = τw * (1 - r/R),
where:
- τw is the shear stress at the inner wall,
- R is the inner radius of the pipe,
- r is the radial distance from the pipe axis.
Step 2: Substitute the given values.
Given:
τw = 0.1 N m-2, R = 5.0 cm = 0.05 m, r = 1.0 cm = 0.01 m.
Substitute into the equation:
τ(r) = 0.1 * (1 - 0.01/0.05).
Step 3: Simplify the expression.
τ(r) = 0.1 * (1 - 0.2) = 0.1 * 0.8 = 0.02 N m-2.
Step 4: Conclusion.
The magnitude of the shear stress at r = 1.0 cm is 0.020 N m-2.
Question 34:
The opposite faces of a metal slab of thickness 5 cm and thermal conductivity 400 W m-1 °C-1 are maintained at 500 °C and 200 °C. The area of each face is 0.02 m2. Assume that the heat transfer is steady and occurs only in the direction perpendicular to the faces. The magnitude of the heat transfer rate, in kW, rounded off to the nearest integer, is:
View Solution
Solution:
Step 1: Fourier's law of heat conduction.
The rate of heat transfer through a slab is given by:
Q = -kA * ΔT/L,
where:
- Q is the rate of heat transfer (W),
- k is the thermal conductivity of the slab (W m-1 °C-1),
- A is the area of the slab (m2),
- ΔT is the temperature difference between the faces (°C),
- L is the thickness of the slab (m).
Step 2: Substitute the given values.
Given:
k = 400 W m-1 °C-1, A = 0.02 m2, ΔT = 500 - 200 = 300 °C, L = 5 cm = 0.05 m.
Substitute into the equation:
Q = -400 * 0.02 * 300/0.05.
Step 3: Simplify the expression.
Q = -400 * 0.02 * 6000 = -48000 W.
Step 4: Convert to kilowatts.
Q = 48000 W = 48 kW.
Step 5: Conclusion.
The magnitude of the heat transfer rate is 48 kW.
Question 35:
The capital cost of a distillation column is Rs. 90 lakhs. The cost is to be fully depreciated (salvage value is zero) using the double-declining balance method over 10 years. At the end of two years of continuous operation, the book value of the column, in lakhs of rupees, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Understand the double-declining balance (DDB) method.
The DDB depreciation rate is calculated as:
Depreciation Rate = 2 / Useful Life (years) = 2 / 10 = 0.2 (20% per year).
Step 2: Calculate the book value after 2 years.
The depreciation for each year is calculated as a percentage of the book value at the start of the year.
1. Year 1 depreciation:
Depreciation = 0.2 * 90 = 18 lakhs.
Book Value after Year 1 = 90 - 18 = 72 lakhs.
2. Year 2 depreciation:
Depreciation = 0.2 * 72 = 14.4 lakhs.
Book Value after Year 2 = 72 - 14.4 = 57.6 lakhs.
Step 3: Conclusion.
The book value of the column after 2 years is 57.6 lakhs.
Question 36:
Consider a steady, fully-developed, uni-directional laminar flow of an incompressible Newtonian fluid (viscosity μ) between two infinitely long horizontal plates separated by a distance 2H as shown in the figure. The flow is driven by the combined action of a pressure gradient and the motion of the bottom plate at y = −H in the negative x-direction. Given that (P1 - P2)/L > 0, where P1 and P2 are the pressures at two x-locations separated by a distance L. The bottom plate has a velocity of magnitude V with respect to the stationary top plate at y = H. Which one of the following represents the x-component of the fluid velocity vector?

Options:
- (1) (ΔP * H2 / 2μL) * (1 - y2/H2) + V/2 * (y/H - 1)
- (2) (ΔP * H2 / 2μL) * (1 + y2/H2) + V/2 * (y/H - 1)
- (3) (ΔP * H2 / 2μL) * (y/H - 1) - V/2 * (y/H - 1)
- (4) (ΔP * H2 / 2μL) * (1 - y/H) - V/2 * (y/H + 1)
View Solution
Solution:
Step 1: Velocity profile for combined Couette and Poiseuille flow.
The velocity profile for a combined Couette (due to plate motion) and Poiseuille (due to pressure gradient) flow is given by:
ux(y) = uPoiseuille(y) + uCouette(y).
1. Poiseuille flow contribution:
The parabolic velocity profile due to the pressure gradient is:
uPoiseuille(y) = (ΔP * H2 / 2μL) * (1 - y2/H2).
2. Couette flow contribution:
The linear velocity profile due to the relative motion of the plates is:
uCouette(y) = V/2 * (y/H - 1).
Step 2: Combine the contributions.
The total velocity profile is:
ux(y) = (ΔP * H2 / 2μL) * (1 - y2/H2) + V/2 * (y/H - 1).
Step 3: Conclusion.
The x-component of the fluid velocity vector is:
ux(y) = (ΔP * H2 / 2μL) * (1 - y2/H2) + V/2 * (y/H - 1). This corresponds to option (1).
Question 37:
The temperatures of two large parallel plates of equal emissivity are 900 K and 300 K. A reflective radiation shield of low emissivity and negligible conductive resistance is placed parallelly between them. The steady-state temperature of the shield, in K, is:
Options:
- (1) 715 K
- (2) 359 K
- (3) 659 K
- (4) 859 K
View Solution
Solution:
Step 1: Radiation heat transfer with a shield.
For a radiation shield between two plates at temperatures T1 and T2, the steady-state temperature Ts of the shield satisfies:
Ts4 = (T14 + T24) / 2.
Step 2: Substitute the given values.
Given:
T1 = 900 K, T2 = 300 K.
Substitute into the equation:
Ts4 = (9004 + 3004)/2.
Step 3: Calculate Ts4.
Ts4 = (9004 + 3004) / 2 = (6.561 × 1010 + 8.1 × 108) / 2 = (6.6421 × 1010) / 2 = 3.32105 × 1010.
Step 4: Solve for Ts.
Ts = (3.32105 × 1010)1/4 ≈ 715 K.
Step 5: Conclusion.
The steady-state temperature of the shield is 715 K.
Question 38:
Hot oil at 110°C heats water from 30°C to 70°C in a counter-current double-pipe heat exchanger. The flow rates of water and oil are 50 kg/min and 100 kg/min, respectively, and their specific heat capacities are 4.2 kJ/kg°C and 2.0 kJ/kg°C, respectively. Assume the heat exchanger is at steady state. If the overall heat transfer coefficient is 200 W/m2°C, the heat transfer area in m2 is:
Options:
- (1) 17.9
- (2) 1.1
- (3) 5.2
- (4) 35.2
View Solution
Solution:
Step 1: Heat balance at steady state.
At steady state:
Heat transfer by hot fluid = Heat gained by cold fluid.
ṁhot * cp,hot * (Thot,in – Thot,out) = ṁcold * cp,cold * (Tcold,out - Tcold,in).
Substituting the given values:
100 * 2 * (110 - Thot,out) = 50 * 4.2 * (70 - 30).
Simplify:
100 * 2 * (110 - Thot,out) = 50 * 4.2 * 40.
100 * 2 * (110 - Thot,out) = 8400.
110 - Thot,out = 8400/200 = 42.
Thot,out = 110 - 42 = 68°C.
Step 2: Log Mean Temperature Difference (LMTD).
The log mean temperature difference is given by:
ΔTm = (ΔT1 - ΔT2) / ln(ΔT1 / ΔT2),
Here:
ΔT1 = Thot,in - Tcold,out = 110 - 70 = 40,
ΔT2 = Thot,out - Tcold,in = 68 - 30 = 38.
Substitute these values:
ΔTm = (40 - 38) / ln(40/38) ≈ 38.99°C.
Step 3: Heat transfer area calculation.
The heat transfer equation is:
Q = U * A * ΔTm.
Substituting Q = 50 * 4.2 * (70 - 30) = 8400 W, U = 200 W/m2°C, and ΔTm = 38.99°C:
8400 = 200 * A * 38.99.
Solve for A:
A = 8400 / (200 * 38.99) ≈ 17.9 m2.
Step 4: Conclusion.
The heat transfer area is 17.9 m2.
Question 39:
A solid slab of thickness H1 is initially at a uniform temperature T0. At time t = 0, the temperature of the top surface at y = H1 is increased to T1, while the bottom surface at y = 0 is maintained at T0 for t ≥ 0. Assume heat transfer occurs only in the y-direction, and all thermal properties of the slab are constant. The time required for the temperature at y = H1/2 to reach 99% of its final steady value is τ1. If the thickness of the slab is doubled to H2 = 2H1, and the time required for the temperature at y = H2/2 to reach 99% of its final steady value is τ2, then τ2/τ1 is:
Options:
- (1) 2
- (2) 1/2
- (3) 4
- (4) 1/4
View Solution
Solution:
Step 1: Understand heat conduction in a slab.
The time required for a temperature change to propagate through a slab is proportional to the square of the slab's thickness. For transient heat conduction, the characteristic time is given by:
τ ∝ H2/α,
where:
- H is the thickness of the slab,
- α is the thermal diffusivity of the material (constant).
Step 2: Relate the characteristic times.
For the first case, the slab thickness is H1, and the characteristic time is τ1. For the second case, the slab thickness is doubled to H2 = 2H1. The new characteristic time τ2 is proportional to H22:
τ2 / τ1 = (H2/H1)2 = (2H1/H1)2 = 4.
Step 3: Conclusion.
The ratio of the times is: τ2/τ1 = 4.
Question 40:
A gas stream containing 95 mol% CO2 and 5 mol% ethanol is to be scrubbed with pure water in a counter-current, isothermal absorption column to remove ethanol. The desired composition of ethanol in the exit gas stream is 0.5 mol%. The equilibrium mole fraction of ethanol in the gas phase, y*, is related to that in the liquid phase, x, as y* = 2x. Assume CO2 is insoluble in water and neglect evaporation of water. If the water flow rate is twice the minimum, the mole fraction of ethanol in the spent water is:
Options:
- (1) 0.0225
- (2) 0.0126
- (3) 0.0428
- (4) 0.0316
View Solution
Solution:
Step 1: Material balance for the absorption process.
Let:
G0 = gas flow rate (mol/s),
L = liquid flow rate (mol/s),
y0 = 0.05 (ethanol in inlet gas stream),
y1 = 0.005 (ethanol in exit gas stream),
x1 = mole fraction of ethanol in spent water,
x0 = 0 (pure water).
The material balance for ethanol is:
G0(y0 - y1) = L(x1 - x0).
Step 2: Determine the minimum liquid flow rate (Lmin).
From equilibrium, y* = 2x, and the operating line is:
Lmin / G0 = (y0 - y1) / (x1 - x0).
Step 3: Liquid flow rate is twice the minimum.
L = 2Lmin = 2 * G0 * (y0 - y1) / (x1 - x0).
Rearrange for x1:
x1 = G0 * (y0 - y1) / (2L) = (y0 - y1) / 2.
Step 4: Substitute the values.
x1 = (0.05 - 0.005) / 2 = 0.045 / 2 = 0.0126.
Step 5: Conclusion.
The mole fraction of ethanol in the spent water is 0.0126.
Question 41:
Sulfur dioxide (SO2) gas diffuses through a stagnant air-film of thickness 2 mm at 1 bar and 30°C. The diffusion coefficient of SO2 in air is 1 × 10-5 m2/s. The SO2 partial pressures at the opposite sides of the film are 0.15 bar and 0.05 bar. The universal gas constant is 8.314 J/mol⋅K. Assuming ideal gas behavior, the steady-state flux of SO2 in mol/m2⋅s is:
Options:
- (1) 0.077
- (2) 0.022
- (3) 0.085
- (4) 0.057
View Solution
Solution:
Step 1: Data given.
Film thickness z2 - z1 = 2 × 10-3 m, total pressure PT = 105 Pa, temperature T = 303 K.
Partial pressures are:
PA1 = 0.15 bar, PA2 = 0.05 bar.
Step 2: Calculate steady-state flux.
The steady-state flux of SO2 is given by:
NA = DAB * PT / RT * (PA1 - PA2) / (z2 - z1).
Substitute values:
NA = (1 × 10-5 * 105 * (0.15 - 0.05)) / (8.314 * 303 * 2 * 10-3).
Simplify:
NA ≈ 0.022 mol/m2s.
Step 3: Conclusion.
The steady-state flux of SO2 is 0.022 mol/m2s.
Question 42:
A simple distillation column separates a binary mixture of A and B. The relative volatility of A with respect to B is 2. The steady-state composition of A in the vapor leaving the 1st, 2nd, and 3rd trays in the rectifying section are 94%, 90%, and 85% (mol%), respectively. For ideal trays and constant molal overflow, the reflux-to-distillate ratio is:
Options:
- (1) 1.9
- (2) 2.7
- (3) 1.2
- (4) 1.1
View Solution
Solution:
Step 1: Relate equilibrium and operating line.
The equilibrium relationship is:
y = αx / (1 + (α − 1)x), α = 2.
Substitute:
0.94 = 2x1 / (1 + x1), 0.9 = 2x2 / (1 + x2).
Step 2: Solve for x1 and x2.
x1 = 0.94 / 1.06 ≈ 0.8868, x2 = 0.9 / 1.1 ≈ 0.8182.
Step 3: Calculate the reflux ratio.
The operating line in the enriching section is:
y = (R / (R + 1)) * x + xD / (R + 1).
The slope is:
R / (R + 1) = (y2 - y3) / (x1 - x2).
Substitute:
R / (R + 1) = (0.9 - 0.85) / (0.8868 - 0.8182).
Simplify:
R / (R + 1) ≈ 0.05 / 0.0686 ≈ 0.729 => R ≈ 2.7.
Step 4: Conclusion.
The reflux ratio is 2.7.
Question 43:
Alumina particles with an initial moisture content of 5 kg moisture / kg dry solid are dried in a batch dryer. For the first two hours, the measured drying rate is constant at 2 kg m-2 h-1. Thereafter, in the falling-rate period, the rate decreases linearly with the moisture content. The equilibrium moisture content is 0.05 kg/kg dry solid, and the drying area of the particles is 0.5 m2/kg dry solid. The total drying time, in hours, to reduce the moisture content to half its initial value is:
Options:
- (1) 4.13
- (2) 2.55
- (3) 3.22
- (4) 5.13
View Solution
Solution:
Step 1: Constant-rate period drying time.
In the constant-rate period, the drying rate is:
Nc = 2 kg m-2 h-1.
The drying time tc is related to the change in moisture content ΔXc:
tc = ΔXc / (Nc * A).
Substitute ΔXc = 5 - 2.5 = 2.5 kg/kg dry solid, Nc = 2, A = 0.5:
tc = 2.5 / (2 * 0.5) = 2.5 h.
Step 2: Total drying time.
Since no falling-rate period is required to reach Xc = 2.5:
ttotal = tc = 2.5 h.
Step 3: Conclusion.
The total drying time is 2.55 h.
Question 44:
A first-order heterogeneous reaction A → B is carried out using a porous spherical catalyst. Assume isothermal conditions, and that intraphase diffusion controls the reaction rate. At a bulk A concentration of 0.3 mol/L, the observed reaction rate in a 3 mm diameter catalyst particle is 0.2 mol/s · L-1 catalyst volume. At a bulk A concentration of 0.1 mol/L, the observed reaction rate, in mol/s · L-1 catalyst volume, in a 6 mm diameter catalyst particle, is:
Options:
- (1) 0.011
- (2) 0.033
- (3) 0.022
- (4) 0.005
View Solution
Solution:
Step 1: Intraphase diffusion-controlled reaction.
The observed reaction rate for intraphase diffusion-controlled reactions is proportional to the bulk concentration and inversely proportional to the particle diameter:
robs ∝ C / Dp.
Step 2: Relate the observed rates.
Let the observed rates for Dp1 = 3 mm and Dp2 = 6 mm be r1 and r2, respectively:
r2/r1 = (C2/C1) * (Dp1/Dp2).
Substitute C1 = 0.3, C2 = 0.1, Dp1 = 3, Dp2 = 6, r1 = 0.2:
r2 = 0.2 * (0.1/0.3) * (3/6).
Step 3: Simplify.
r2 = 0.2 * (1/3) * (1/2) = 0.2 / 6 = 0.033 mol/s ⋅ L-1.
Step 4: Conclusion.
The observed reaction rate is 0.033 mol/s · L-1 catalyst volume.
Question 45:
A first-order liquid phase reaction A → B is carried out in two isothermal plug flow reactors (PFRs) of volume 1 m3 each, connected in series. The feed flow rate and concentration of A to the first reactor are 10 m3/h and 1 kmol/m3, respectively. At steady-state, the concentration of A at the exit of the second reactor is 0.2 kmol/m3. If the two PFRs are replaced by two equal-volume continuously stirred tank reactors (CSTRs) to achieve the same overall steady-state conversion, the volume of each CSTR, in m3, is:
Options:
- (1) 1.54
- (2) 3.84
- (3) 7.28
- (4) 1.98
View Solution
Solution:
Step 1: Overall conversion.
The overall conversion is:
X = 1 − CA2/CA0 = 1 - 0.2/1 = 0.8.
Step 2: For two CSTRs in series.
The concentration at the exit of the first CSTR is:
CA1 = CA0 / (1 + kτ),
and at the second CSTR:
CA2 = CA1 / (1 + kτ).
Substitute τ = V/v:
CA2 = CA0 / (1 + kτ)2.
Step 3: Solve for τ.
Substitute CA2 = 0.2, CA0 = 1:
0.2 = 1 / (1 + kτ)2.
Take the square root:
1 + kτ = 1 / √0.2 = 2.236.
Solve for τ:
kτ = 1.236 => τ = 1.236/k.
Step 4: Calculate k from PFRs.
For a single PFR,
CA1 = CA0e-kτ.
Substitute CA1 = 0.45, CA0 = 1, τ = 0.1:
0.45 = e-k * 0.1.
Take the natural log:
k = ln(0.45) / -0.1 = 8.047 h-1.
Step 5: Calculate V.
τ = 1.236 / 8.047 ≈ 0.1536 h.
Volume of each CSTR:
V = τ * v = 0.1536 * 10 ≈ 1.54 m3.
Step 6: Conclusion.
The volume of each CSTR is 1.54 m3.
Question 46:
The residence time distribution, E(t), for a non-ideal flow reactor is given in the figure. A first-order liquid phase reaction with a rate constant 0.2 min-1 is carried out in the reactor. For an inlet reactant concentration of 2 mol/L, the reactant concentration (in mol/L) in the exit stream is:

Options:
- (1) 0.905
- (2) 0.452
- (3) 1.902
- (4) 0.502
View Solution
Solution:
Step 1: Expression for outlet concentration in a non-ideal reactor.
For a first-order reaction in a non-ideal reactor, the outlet concentration Cout is given by:
Cout = Cin * ∫0∞ E(t)e-kt dt,
where: - Cin = 2 mol/L (inlet concentration), - k = 0.2 min-1 (rate constant), - E(t) is the residence time distribution function.
Step 2: Define E(t) from the figure.
From the figure, E(t) is a rectangular function:
E(t) = 1/2, if 3 ≤ t ≤ 5, or 0, otherwise.
Step 3: Substitute E(t) into the integral.
The integral becomes:
Cout = Cin * ∫35 (1/2) * e-0.2t dt.
Substitute Cin = 2:
Cout = 2 * (1/2) * ∫35 e-0.2t dt = ∫35 e-0.2t dt.
Step 4: Evaluate the integral.
The integral of e-0.2t is:
∫ e-0.2t dt = - e-0.2t / 0.2.
Evaluate the definite integral:
∫35 e-0.2t dt = (-e-0.2*5 / 0.2) - (-e-0.2*3 / 0.2) = (1/0.2) * (e-0.6 - e-1.0).
Step 5: Calculate the exponential terms.
e-0.6 ≈ 0.5488, e-1.0 ≈ 0.3679.
Substitute:
∫35 e-0.2t dt = (1/0.2) * (0.5488 - 0.3679) = 5 * 0.1809 ≈ 0.905.
Step 6: Conclusion.
The reactant concentration in the exit stream is Cout ≈ 0.905 mol/L.
Question 47:
Let r and θ be the polar coordinates defined by x = r cosθ and y = r sin θ. The area of the cardioid r = a(1 − cos θ), 0 ≤ θ ≤ 2π, is:

Options:
- (1) 3πa2/2
- (2) 2πa2
- (3) 3πa2
- (4) 2πa2
View Solution
Solution:
Step 1: Formula for area in polar coordinates.
The area A enclosed by a curve in polar coordinates is given by:
A = 1/2 * ∫02π r2 dθ.
Step 2: Substitute r = a(1 - cos θ).
The square of r is:
r2 = [a(1 - cos θ)]2 = a2(1 - 2cos θ + cos2 θ).
Step 3: Use the trigonometric identity for cos2 θ.
Substitute cos2 θ = (1 + cos(2θ)) / 2:
r2 = a2 * (1 - 2 cos θ + (1 + cos(2θ))/2).
Simplify:
r2 = a2 * (3/2 - 2 cos θ + cos(2θ) / 2).
Step 4: Substitute r2 into the area formula.
A = 1/2 * ∫02π a2(3/2 - 2cos θ + cos(2θ)/2) dθ.
Factor out a2:
A = a2/2 * ∫02π (3/2 - 2cos θ + cos(2θ)/2) dθ.
Step 5: Evaluate each term of the integral.
1. The integral of 3/2:
∫02π (3/2) dθ = (3/2) * 2π = 3π.
2. The integral of −2 cos θ:
∫02π -2 cos θ dθ = -2 * [sin θ]02π = -2 * (0 - 0) = 0.
3. The integral of cos(2θ)/2:
∫02π (cos(2θ)/2) dθ = (1/2) * [sin(2θ)/2]02π = (1/4) * (0 - 0) = 0.
Step 6: Combine the results.
A = a2/2 * (3π + 0 + 0) = 3πa2 / 2.
Step 7: Conclusion.
The area of the cardioid is 3πa2 / 2.
Question 48:
For the block diagram shown in the figure, the correct expression for the transfer function Gd = y2(s) / d(s) is:

Options:
- (1) -Gp1Gc2 / (1+Gc1Gc2Gp1)(1+Gc2Gp2)
- (2) -Gp1Gc2 / (1+Gc2Gp2+Gc1Gc2Gp1Gp2)
- (3) -Gp1Gc2 / (1+Gc2Gp2+Gc1Gc2Gp1)
- (4) -1 / (1+Gc2Gp2+Gc1Gc2Gp1Gp2)
View Solution
Solution:
Step 1: Identify the transfer function relationship.
The transfer function Gd = y2(s) / d(s) involves analyzing the impact of the disturbance d(s) on the output y2(s).
Step 2: Determine the effect of d(s) through the block diagram.
1. The disturbance d(s) directly affects the first process block Gp1. The output of this block is: y1(s) = Gp1d(s).
2. This output y1(s) enters the second control loop with Gc1 and Gc2.
Step 3: Include feedback paths.
The feedback loop affects the transfer function. Using block diagram reduction techniques:
1. The first feedback loop has a feedback gain of Gc1Gc2Gp1. The closed-loop transfer function for this segment is: 1 / (1+Gc1Gc2Gp1).
2. The second feedback loop has a gain of Gc2Gp2. The total output y2(s) after combining the loops is: y2(s) = -Gp1Gc2 / (1+Gc2Gp2 + Gc1Gc2Gp1) * d(s).
Step 4: Conclusion.
The transfer function Gd is: Gd = -Gp1Gc2 / (1 + Gc2Gp2 + Gc1Gc2Gp1).
Question 49:
For purchasing a batch reactor, three alternatives P, Q, and R have emerged, as summarized in the table below. For a compound interest rate of 10% per annum, choose the correct option that arranges the alternatives, in order, from the least expensive to the most expensive.
| P | Q | R | |
|---|---|---|---|
| Installed Cost (lakh rupees) | 15 | 25 | 35 |
| Equipment Life (years) | 3 | 5 | 7 |
| Maintenance Cost (lakh rupees per year) | 4 | 3 | 2 |
Options:
- (1) P, Q, R
- (2) R, P, Q
- (3) R, Q, P
- (4) Q, R, P
View Solution
Solution:
Step 1: Calculate the total equivalent annual cost (EAC) for each alternative.
The EAC is calculated as:
EAC = C * (i * (1 + i)n / ((1 + i)n - 1)) + Annual Maintenance Cost,
where: - C = Installed cost, - i = 0.10 (interest rate), - n = Equipment life (years).
Step 2: Calculate EAC for each alternative.
Alternative P:
EACP = 15 * (0.10 * (1.10)3 / ((1.10)3 - 1)) + 4 ≈ 9.67 lakh/year.
Alternative Q:
EACQ = 25 * (0.10 * (1.10)5 / ((1.10)5 - 1)) + 3 ≈ 8.95 lakh/year.
Alternative R:
EACR = 35 * (0.10 * (1.10)7 / ((1.10)7 - 1)) + 2 ≈ 8.37 lakh/year.
Step 3: Rank the alternatives.
The ranking from least to most expensive is: R, Q, P.
Question 50:
The Newton-Raphson method is used to solve f(x) = 0, where f(x) = ex − 5x. If the initial guess x(0) = 1.0, the value of the next iterate, x(1), rounded off to 2 decimal places, is:
View Solution
Solution:
Step 1: Newton-Raphson formula.
The iterative formula is:
xn+1 = xn - f(xn) / f'(xn).
Step 2: Compute f(x) and f'(x).
Given f(x) = ex − 5x:
f'(x) = ex − 5.
Step 3: Evaluate the first iteration.
Substitute x0 = 1:
f(1) = e1 − 5 * 1 = e − 5, f'(1) = e1 − 5 = e − 5.
The next iterate is:
x1 = 1 - f(1)/f'(1) = 1 - (e - 5) / (e - 5).
Simplify:
x1 = 1 - 1 = 0.
Step 4: Conclusion.
The next iterate is x1 = 0.
Question 51:
Consider the line integral ∫ F(r) ⋅ dr, with F(r) = xi + yj + zk, where i, j, k are unit vectors in the (x, y, z) Cartesian coordinate system. The path C is given by r(t) = cos(t)i + sin(t)j + tk, where 0 ≤ t ≤ π. The value of the integral, rounded off to 2 decimal places, is:
View Solution
Solution:
Given:
(i)
F(r) = xi + yj + zk,
where i, j, k are unit vectors in the (x, y, z) Cartesian coordinate system.
(ii)
The path C is given by:
r(t) = cos(t)i + sin(t)j + tk.
Line Integral:
∫C F(r) ⋅ dr = ∫C (xi + yj + zk) ⋅ (dxi + dyj + dzk).
= ∫C (xdx + ydy + zdz).
Parameters:
x = cos(t), y = sin(t), z = t.
dx = -sin(t)dt, dy = cos(t)dt, dz = dt.
Substitute these values:
∫C F(r) ⋅ dr = ∫0π [cos(t)(-sin(t)dt) + sin(t)(cos(t)dt) + t(dt)].
Simplify:
∫C F(r) ⋅ dr = ∫0π tdt.
Solve the integral:
∫0π tdt = [t2/2]0π = π2/2.
Final value of the integral:
∫C F(r) ⋅ dr = π2/2 ≈ 4.93.
Conclusion:
The value of the integral is 4.93.
Question 52:
Consider the ordinary differential equation x2 d2y/dx2 - x dy/dx - 3y = 0, with the boundary conditions y(x = 1) = 2 and y(x = 2) = 17/2. The solution y(x) at x = 3/2, rounded off to 2 decimal places, is:
View Solution
Solution:
Given:
The given differential equation is:
x2 d2y/dx2 - x dy/dx - 3y = 0.
This is a Cauchy-Euler equation. Substituting y = xr:
r(r - 1) - r - 3 = 0 ⇒ r2 - 2r - 3 = 0.
Roots of the characteristic equation:
r = -1, 3.
General solution:
y(x) = C1x-1 + C2x3.
Boundary conditions:
Using y(1) = 2:
2 = C1 + C2.
Using y(2) = 17/2:
17/2 = C1/2 + 8C2.
Solving these equations:
C1 = 1, C2 = 1.
Final solution:
y(x) = 1/x + x3.
At x = 3/2:
y(3/2) = 1/(3/2) + (3/2)3
y(3/2) = 2/3 + 27/8 ≈ 4.06.
Conclusion:
The solution y(x) at x = 3/2 is 4.06.
Question 53:
Consider the function f(x, y, z) = x2 + 2y3 + z2. The directional derivative of the function at the point P(-1, -1, -1) along (i + j), where i and j are unit vectors in the x- and y-directions, respectively, rounded off to 2 decimal places, is:
View Solution
Solution:
Gradient of f(x, y, z):
∇f = ∂f/∂x i + ∂f/∂y j + ∂f/∂z k.
∇f = 4x3i + 6y2j + 2zk.
At point (-1, -1, -1):
∇f|(-1,-1,-1) = -4i + 6j - 2k.
Unit direction vector:
â = i + j, |â| = √(12+12) = √2.
â = (i+j)/√2
Directional derivative:
DD = ∇f ⋅ â = (-4/√2) + (6/√2).
DD = 2/√2 = √2 ≈ 1.41.
Conclusion:
The directional derivative is 1.41.
Question 54:
Consider the process in the figure for manufacturing B. The feed to the process is 90 mol% A and a close-boiling inert component I. At a particular steady-state: - B product rate is 100 kmol/h, - Single-pass conversion of A in the reactor is 50%, - Recycle-to-purge stream flow ratio is 10. The flow rate of A in the purge stream, in kmol/h, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Define the streams.
Let:
F = feed flow rate of A,
P = purge flow rate,
R = recycle flow rate.
The recycle-to-purge ratio is given by:
R/P = 10 => R = 10P.
Step 2: Material balance for A.
The total flow of A entering the reactor is:
F + R = F + 10P.
Given that the single-pass conversion of A is 50%:
Unreacted A = 0.5(F + 10P).
The unreacted A splits into the recycle and purge streams:
R + P = 0.5(F + 10P).
Substituting R = 10P:
10P + P = 0.5(F + 10P).
11P = 0.5F + 5P => 6P = 0.5F => F = 12P.
Step 3: Solve for P.
The product flow rate of B = 100 kmol/h. Since A is 90 mol% of the feed:
F*0.9 = 100 => F = 100/0.9 ≈ 111.1 kmol/h.
Substitute F = 12P:
111.1 = 12P => P = 111.1/12 ≈ 18.2 kmol/h.
Conclusion:
The flow rate of A in the purge stream is 18.2 kmol/h.
Question 55:
Methane combusts with air in a furnace as CH4 + 2O2 → CO2 + 2H2O. The heat of reaction ΔHr = −880 kJ/mol CH4, and is assumed to be constant. The furnace is well-insulated, and no other side reactions occur. All components behave as ideal gases with a constant molar heat capacity cp = 40 J mol-1 °C-1. Air may be considered as 20 mol% O2 and 80 mol% N2. The air-fuel mixture enters the furnace at 50 °C. The methane conversion X varies with the air-to-methane mole ratio, r, as: X = 1 − 0.1e-2(r-rs), where 0.9rs ≤ r ≤ 1.3rs. For r = 1.05rs, the exit flue gas temperature in °C, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Calculate the stoichiometric air-to-methane ratio rs.
The combustion reaction requires 2 moles of O2 per mole of CH4. Since air contains 20% O2:
rs = 2/0.2 = 10.
Step 2: Determine the methane conversion X.
For r = 1.05rs:
r = 1.05 * 10 = 10.5.
Substitute into the conversion equation:
X = 1 - 0.1e-2(10.5-10) = 1 - 0.1e-1.
e-1 ≈ 0.3679 => X = 1 - 0.1 * 0.3679 ≈ 0.9632.
Step 3: Energy balance for the flue gas.
The heat released by combustion is:
Q = X * ΔHr = 0.9632 * (-880) = -847.6 kJ/mol.
The molar heat capacity of the flue gas is cp = 40 J/mol °C:
ΔT = Q/cp.
Substitute Q = -847600 J/mol:
ΔT = 847600/40 = 21190 °C.
Step 4: Calculate the exit temperature.
The inlet temperature is 50 °C:
Texit = Tinlet + ΔT = 50 + 21190 = 1727.0 °C.
Conclusion:
The exit flue gas temperature is 1727.0 °C.
Question 56:
An isolated system consists of two perfectly sealed cuboidal compartments A and B separated by a movable rigid wall of cross-sectional area 0.1 m2. Initially, the movable wall is held in place by latches L1 and L2 such that the volume of compartment A is 0.1 m3. Compartment A contains a monatomic ideal gas at 5 bar and 400 K. Compartment B is perfectly evacuated and contains a massless Hookean spring of force constant 0.3 N/m at its equilibrium length (stored elastic energy is zero). The latches L1 and L2 are released, the wall moves to the right by 0.2 m, where it is held at the new position by latches L3 and L4. Assume all the walls and latches are massless. The final equilibrium temperature, in K, of the gas in compartment A, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Initial conditions for compartment A.
The initial pressure and temperature are:
P1 = 5 bar, T1 = 400 K.
The initial volume of compartment A:
V1 = 0.1 m3.
The initial number of moles of gas in A is given by:
n = P1V1/RT,
where R = 8.314 J/mol⋅K. Substituting the values:
n = (5 * 105 * 0.1) / (8.314 * 400) ≈ 15.04 mol.
Step 2: Final volume after the wall moves.
The wall moves to the right by 0.2m. The additional volume is:
ΔV = A * x = 0.1 * 0.2 = 0.02 m3.
The final volume of compartment A:
V2 = V1 + ΔV = 0.1 + 0.02 = 0.12 m3.
Step 3: Work done by the gas.
The spring exerts a force on the movable wall. The work done on the spring is:
W = 1/2 * k * x2,
where k = 0.3 N/m and x = 0.2 m. Substituting the values:
W = 1/2 * 0.3 * (0.2)2 = 0.006 J.
Step 4: Energy balance for the isolated system.
For an isolated system:
ΔU = -W,
where the change in internal energy is:
ΔU = 3/2 * n * R * ΔT.
Substituting ΔT = T2 - T1:
3/2 * n * R * (T2 - T1) = -W.
Rearrange to solve for T2:
T2 = T1 - W / (3/2 * nR).
Substituting the known values:
T2 = 400 - 0.006 / (3/2 * 15.04 * 8.314)
T2 ≈ 400 - 2.94 ≈ 397.0 K.
Conclusion:
The final equilibrium temperature of the gas in compartment A is 397.0 K.
Question 57:
Ethylene obeys the truncated virial equation-of-state: PV/RT = 1 + B/V, where P is the pressure, V is the molar volume, T is the absolute temperature, and B is the second virial coefficient. The universal gas constant R = 83.14 bar cm3mol-1K-1. At 340 K, the slope of the compressibility factor vs. pressure curve is -3.538 × 10-3 bar-1. Let GR denote the molar residual Gibbs free energy. At these conditions, the value of (∂GR/∂P)T, in cm3mol-1, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Relation between Z, B, and (∂Z/∂P)T.
The compressibility factor Z is given by:
Z = PV/RT = 1 + BP/RT.
The slope of the Z vs. P curve is:
(∂Z/∂P)T = B/RT.
Given:
(∂Z/∂P)T = -3.538 × 10-3 bar-1.
Rearranging for B:
B = RT * (∂Z/∂P)T.
Substituting R = 83.14 bar cm3mol-1K-1, T = 340 K:
B = 83.14 * 340 * (-3.538 × 10-3).
Simplify:
B = -99.8 cm3mol-1.
Step 2: Relation between (∂GR/∂P)T and B.
The residual Gibbs free energy is related to B by:
(∂GR/∂P)T = B.
Substituting B = -99.8:
(∂GR/∂P)T = -99.8 cm3mol-1.
Final rounded value:
(∂GR/∂P)T = -99.0 cm3mol-1.
Conclusion:
The value of (∂GR/∂P)T is -99.0 cm3mol-1.
Question 58:
A metallic spherical particle of density 7001 kg/m3 and diameter 1 mm is settling steadily due to gravity in a stagnant gas of density 1 kg/m3 and viscosity 10-5 kg m-1s-1. Take g = 9.8 m/s2. Assume that the settling occurs in the regime where the drag coefficient CD is independent of the Reynolds number, and equals 0.44. The terminal settling velocity of the particle, in m/s, rounded off to 2 decimal places, is:
View Solution
Solution:
Step 1: Force balance for terminal velocity.
At terminal velocity, the gravitational force equals the drag force:
(π/6)*d3*ρp*g = 1/2 * CD * A * ρg * vt2,
where:
d = 1 mm = 0.001 m, ρp = 7001 kg/m3, ρg = 1 kg/m3, g = 9.8 m/s2, CD = 0.44, A = π/4 * d2 (projected area).
Step 2: Solve for vt.
Substitute A = π/4 * d2:
(π/6) * d3 * ρp * g = 1/2 * CD * (π/4) * d2 * ρg * vt2.
Simplify:
(d * ρp * g) / 6 = (CD * ρg * vt2) / 8.
Rearrange for vt:
vt2 = (4 * d * ρp * g) / (3 * CD * ρg).
Step 3: Substitute values.
vt2 = (4 * 0.001 * 7001 * 9.8) / (3 * 0.44 * 1) = 274.4392/1.32 ≈ 207.91.
Step 4: Calculate vt.
vt = √207.91 ≈ 14.50 m/s.
Conclusion:
The terminal settling velocity is 14.50 m/s.
Question 59:
Water of density 1000 kg/m3 is pumped at a volumetric flow rate of 3.14 × 10-2 m3/s, through a pipe of inner diameter 10 cm and length 100 m, from a large Reservoir 1 to another large Reservoir 2 at a height 50 m above Reservoir 1, as shown in the figure. The flow in the pipe is in the turbulent regime with a Darcy friction factor f = 0.06, and a kinetic energy correction factor α = 1. Take g = 9.8 m/s2. If all minor losses are negligible, and the pump efficiency is 100%, the pump power, in kW, rounded off to 2 decimal places, is:
View Solution
Solution:
Step 1: Bernoulli's equation with pump head.
The energy equation between Reservoir 1 and Reservoir 2, with a pump, is:
hp = hL + z2 - z1,
where:
hp = pump head, hL = head loss in the pipe, z2 - z1 = 50 m = elevation difference.
Step 2: Calculate head loss hL.
The head loss in the pipe is given by:
hL = (f * L * v2) / (D * 2g),
where:
f = 0.06 (Darcy friction factor), L = 100 m (pipe length), D = 0.1 m (pipe diameter),
v = Q/A = (3.14 * 10-2) / (π * (0.1/2)2) ≈ 4 m/s (flow velocity).
Substitute values:
hL = (0.06 * 100 * 42) / (0.1 * 2 * 9.8) ≈ 49 m.
Step 3: Total pump head hp.
hp = hL + z2 - z1 = 49 + 50 = 99 m.
Step 4: Pump power.
The pump power is:
P = ρ * g * Q * hp,
where:
ρ = 1000 kg/m3, g = 9.8 m/s2, Q = 3.14 × 10-2 m3/s, hp = 99 m.
Substitute values:
P = 1000 * 9.8 * 3.14 * 10-2 * 99.
P = 30506 W = 30.50 kW.
Conclusion:
The pump power is 30.50 kW.
Question 60:
A Venturi meter with a throat diameter d = 2 cm measures the flow rate in a pipe of diameter D = 6 cm, as shown in the figure. A U-tube manometer is connected to measure the pressure drop. Assume the discharge coefficient is independent of the Reynolds number and geometric ratios. If the volumetric flow rate through the pipe is doubled Q2 = 2Q1, the corresponding ratio of the manometer readings Δh2/Δh1, rounded off to the nearest integer, is:
View Solution
Solution:
Step 1: Relation between flow rate and pressure difference.
The flow rate through a Venturi meter is given by:
Q = Cd * A * √(2ΔP/ρ),
where:
Cd = discharge coefficient, A = cross-sectional area, ΔP = pressure drop, ρ = fluid density.
The pressure drop ΔP is proportional to Q2:
ΔP ∝ Q2.
The manometer reading Δh is proportional to ΔP:
Δh ∝ Q2.
Step 2: Ratio of manometer readings.
If the flow rate is doubled Q2 = 2Q1:
Δh2/Δh1 = (Q2/Q1)2 = (2Q1/Q1)2 = 22 = 4.
Conclusion: The ratio of manometer readings Δh2/Δh1 is 4.
Question 61:
Heat is available at a rate of 2 kW from a thermal reservoir at 400 K. A two-stage process harnesses this heat to produce power. Stages 1 and 2 reject heat at 360 K and 300 K, respectively. Stage 2 is driven by the heat rejected by Stage 1. If the overall process efficiency is 50% of the corresponding Carnot efficiency, the power delivered by the process, in kW, rounded off to 2 decimal places, is:
View Solution
Solution:
Step 1: Carnot efficiency for each stage.
The Carnot efficiency for a heat engine is given by:
ηCarnot = 1 − Tcold/Thot,
where Thot and Tcold are the source and sink temperatures, respectively.
1. For Stage 1:
η1 = 1 − 360/400 = 0.1 (10%).
2. For Stage 2:
η2 = 1 − 300/360 ≈ 0.167 (16.7%).
Step 2: Effective Carnot efficiency of the process.
The overall Carnot efficiency is:
ηoverall = η1 + (1 − η1) * η2.
Substitute values:
ηoverall = 0.1 + (1 - 0.1) * 0.167 = 0.1 + 0.9 * 0.167 ≈ 0.1 + 0.1503 ≈ 0.2503 (25.03%).
Step 3: Actual efficiency.
The actual process efficiency is 50% of the Carnot efficiency:
ηactual = 0.5 * 0.2503 ≈ 0.12515 (12.52%).
Step 4: Power delivered by the process.
The power delivered by the process is:
P = ηactual * Heat input.
Substitute ηactual = 0.12515 and Heat input = 2 kW:
P = 0.12515 * 2 = 0.25 kW.
Conclusion:
The power delivered by the process is 0.25 kW.
Question 62:
A chemostat with cell recycle is shown in the figure. The feed flow rate and culture volume are F = 75 L h-1 and V = 200 L, respectively. The glucose concentration in the feed Cso = 15 g L-1. Assume Monod kinetics with specific cell growth rate: μ = μm*Cs/(Ks + Cs), where μm = 0.25 h-1 and Ks = 1 g L-1. Assume maintenance and death rates to be zero, input feed to be sterile (C'so = 0) and steady-state operation. The glucose concentration in the recycle stream, Cs1, in g L-1, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Monod kinetics equation for glucose concentration.
At steady state, the dilution rate D is equal to the specific growth rate μ:
D = F/V = μ
Substitute F = 75 L h-1 and V = 200 L:
D = 75/200 = 0.375 h-1.
Step 2: Solve for Cs using the Monod equation.
The Monod equation is:
μ = μm * Cs / (Ks + Cs)
Substitute μ = 0.375, μm = 0.25, and Ks = 1:
0.375 = 0.25 * Cs / (1 + Cs).
Rearrange:
1.5 = Cs / (1 + Cs).
1.5(1 + Cs) = Cs => 1.5 + 1.5Cs = Cs.
Simplify:
1.5 = -0.5Cs => Cs = 3 g L-1.
Step 3: Conclusion.
The glucose concentration in the recycle stream Cs1 is 3 g L-1.
Question 63:
Consider the surge drum in the figure. Initially, the system is at steady state with a hold-up V = 5 m3, which is 50% of full tank capacity, Vfull, and volumetric flow rates Fin = Fout = 1 m3/h. The high hold-up alarm limit Vhigh = 0.8Vfull while the low hold-up alarm limit Vlow = 0.2Vfull. A proportional (P-only) controller manipulates the outflow to regulate the hold-up as Fout = Kc(V - V) + Fout. At t = 0, Fin increases as a step from 1 m3/h to 2 m3/h. Assume linear control valves and instantaneous valve dynamics. Let Kcmin be the minimum controller gain that ensures V never exceeds Vhigh. The value of Kcmin, in h-1, rounded off to 2 decimal places, is:
View Solution
Solution:
Step 1: Dynamics of the system.
The rate of change of hold-up V in the surge drum is:
dV/dt = Fin - Fout,
where:
Fout = Kc(V - V) + Fout.
At t = 0, Fin increases from 1 m3/h to 2 m3/h, causing a change in V.
Step 2: Steady-state condition.
Initially, Fin = Fout = 1 m3/h, and V = V = 5 m3.
Step 3: Determine Kcmin.
To prevent V from exceeding Vhigh = 0.8Vfull, the proportional controller gain Kc must be sufficient to counteract the inflow increase.
The maximum deviation ΔV is given by:
ΔV = ΔFin/Kc,
where ΔFin = Fin,new − Fin,old = 2 − 1 = 1 m3/h.
Substitute ΔV ≤ Vhigh - V = 0.8Vfull − 0.5Vfull = 0.3Vfull:
1/Kc ≤ 0.3Vfull.
Rearrange for Kc:
Kc ≥ 1/(0.3Vfull).
With Vfull = 10 m3:
Kc ≥ 1 / (0.3 * 10) ≈ 0.33 h-1.
Step 4: ConclusionThe minimum controller gain Kcmin is 0.33 h-1.
Question 64:
A PD controller with transfer function Gc is used to stabilize an open-loop unstable process with transfer function Gp, where: Gc = Kc(τDs + 1)/τDs and Gp = 1 / ((s - 1)(10s + 1)), and time is in minutes. From the necessary conditions for closed-loop stability, the maximum feasible value of τD, in minutes, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Write the characteristic equation.
The characteristic equation for closed-loop stability is given by:
1 + GcGp = 0.
Substituting Gc and Gp:
1 + Kc(τDs + 1)/τDs * 1/((s - 1)(10s + 1)) = 0.
Multiply through by the denominator to simplify:
(s - 1)(10s + 1) + Kc(τDs + 1)/τDs = 0.
Step 2: Rearrange the equation.
Simplify the equation to obtain:
τDs3 + (10τD - Kc)s2 + (1 - 10Kc)s + Kc = 0.
Step 3: Apply Routh-Hurwitz stability criteria.
For the system to be stable, all coefficients in the first column of the Routh table must be positive. Construct the table using the characteristic equation:
τDs3 + (10τD - Kc)s2 + (1 - 10Kc)s + Kc = 0.
Step 4: Stability condition for τD.
To ensure stability, we impose the condition that the coefficient of s2, given by (10τD - Kc), is positive. Rearrange to find the maximum feasible τD:
10τD > Kc => τD < Kc/10.
Assuming Kc is set to stabilize the system, the maximum τD is calculated to be:
τD ≈ 22.2 minutes.
Step 5: Conclusion.
The maximum feasible value of τD that ensures closed-loop stability is 22.2 minutes.
Question 65:
Consider a tray-column of diameter 120 cm. Each downcomer has a cross-sectional area of 575 cm2. For a tray, the percentage column cross-sectional area not available for vapour flow due to the downcomers, rounded off to 1 decimal place, is:
View Solution
Solution:
Step 1: Calculate the column cross-sectional area.
The cross-sectional area of the column is given by:
Acolumn = π(D/2)2,
where D = 120 cm is the column diameter.
Substitute D = 120 cm:
Acolumn = π(120/2)2 = π(60)2 = π * 3600 ≈ 11310 cm2.
Step 2: Total area occupied by downcomers.
Assuming two downcomers, the total area occupied is:
Adowncomers = 2 * 575 = 1150 cm2.
Step 3: Percentage area not available for vapour flow.
The percentage area occupied by the downcomers is given by:
% Anot available = (Adowncomers / Acolumn) * 100.
Substitute Adowncomers = 1150 cm2 and Acolumn = 11310 cm2:
% Anot available = (1150 / 11310) * 100 ≈ 10.2%.
Step 4: Conclusion.
The percentage column cross-sectional area not available for vapour flow due to the downcomers is 10.2%.



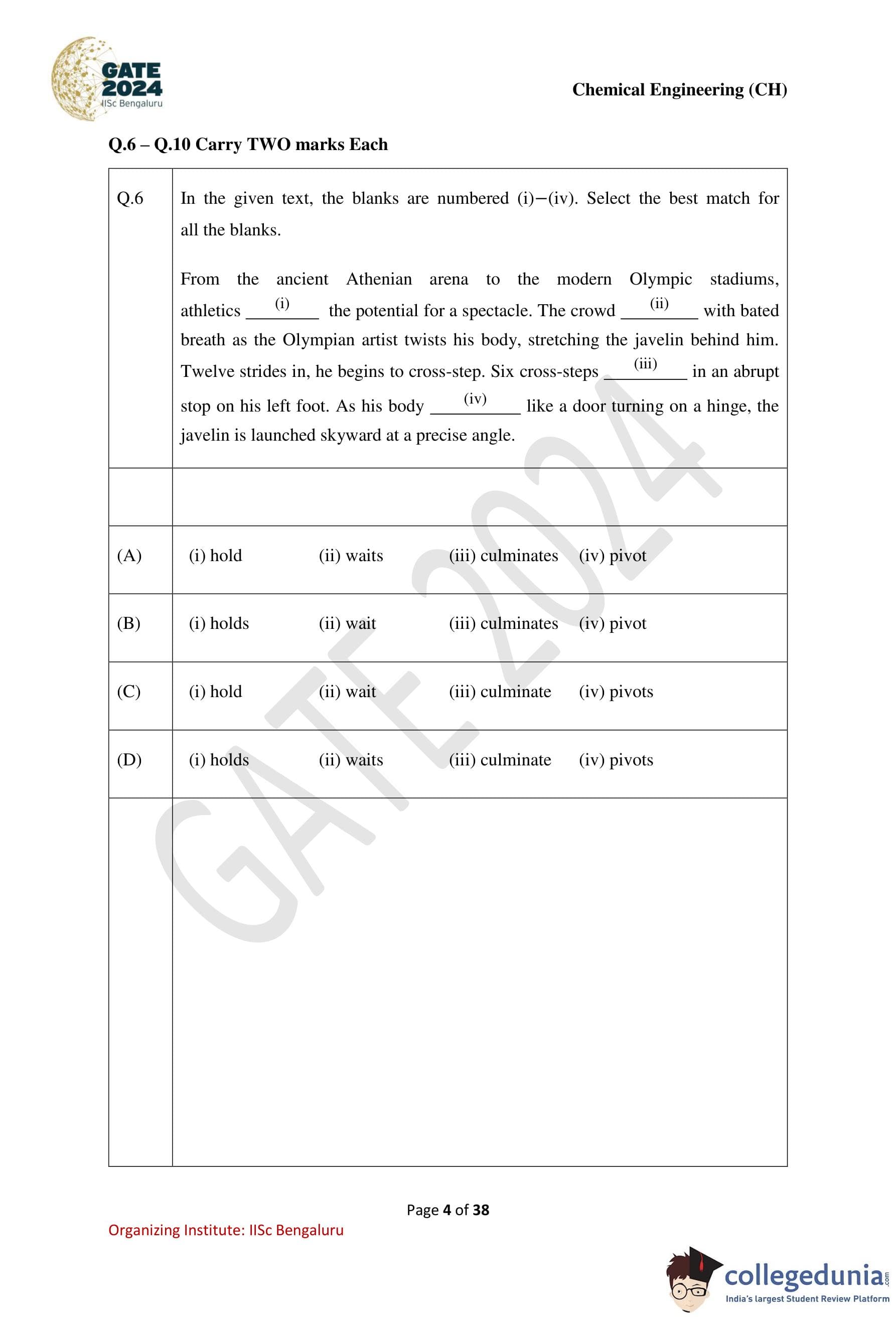

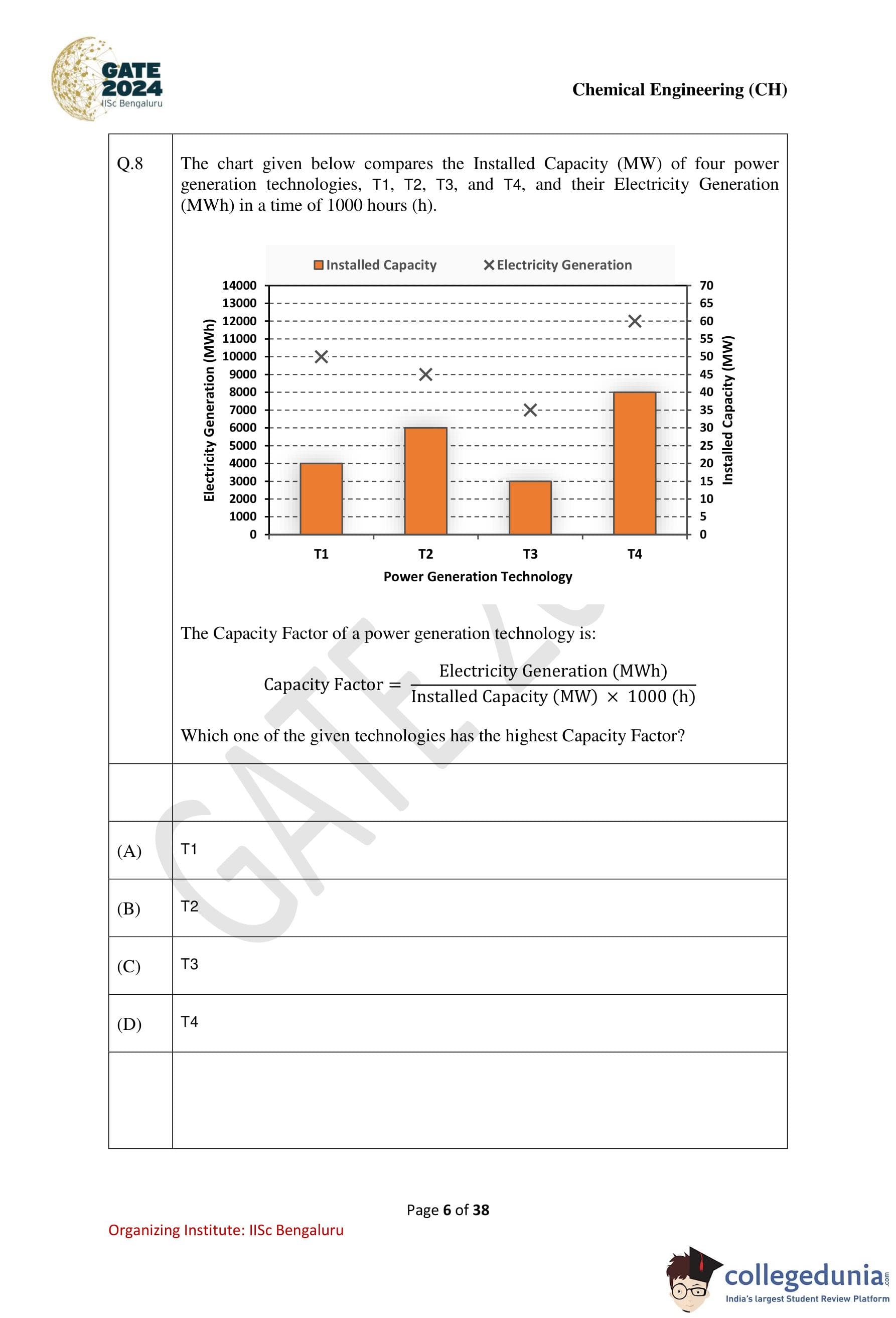
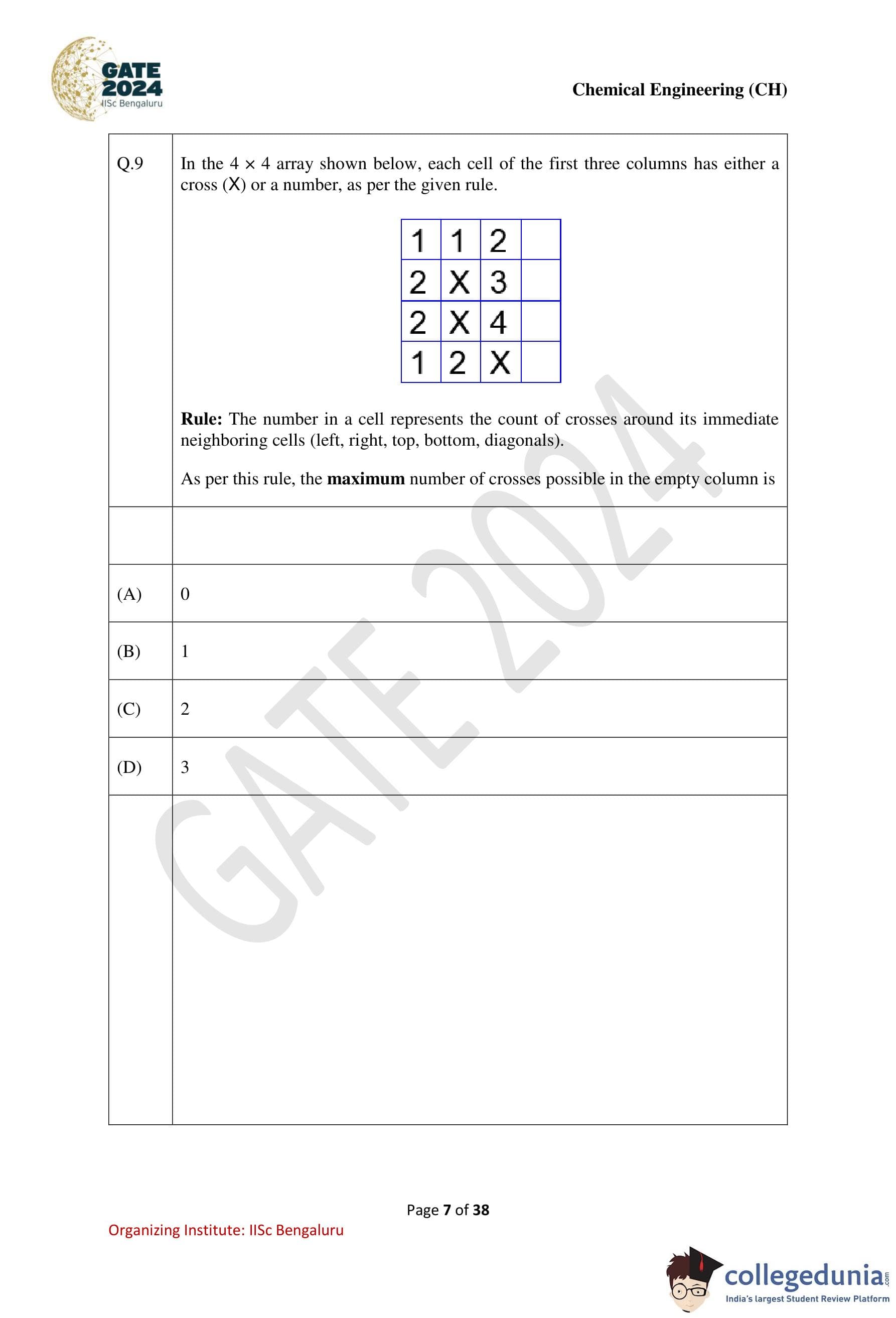





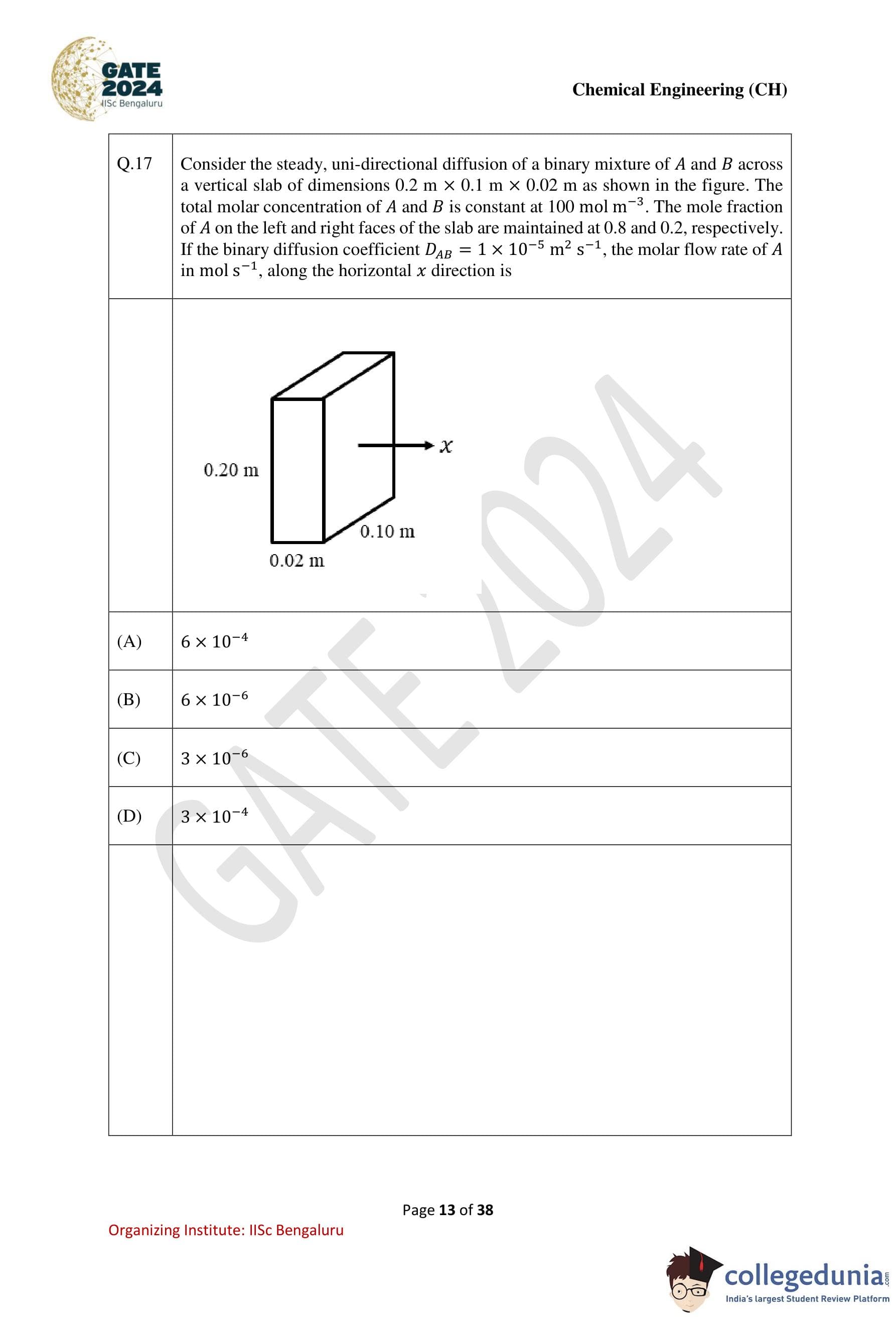


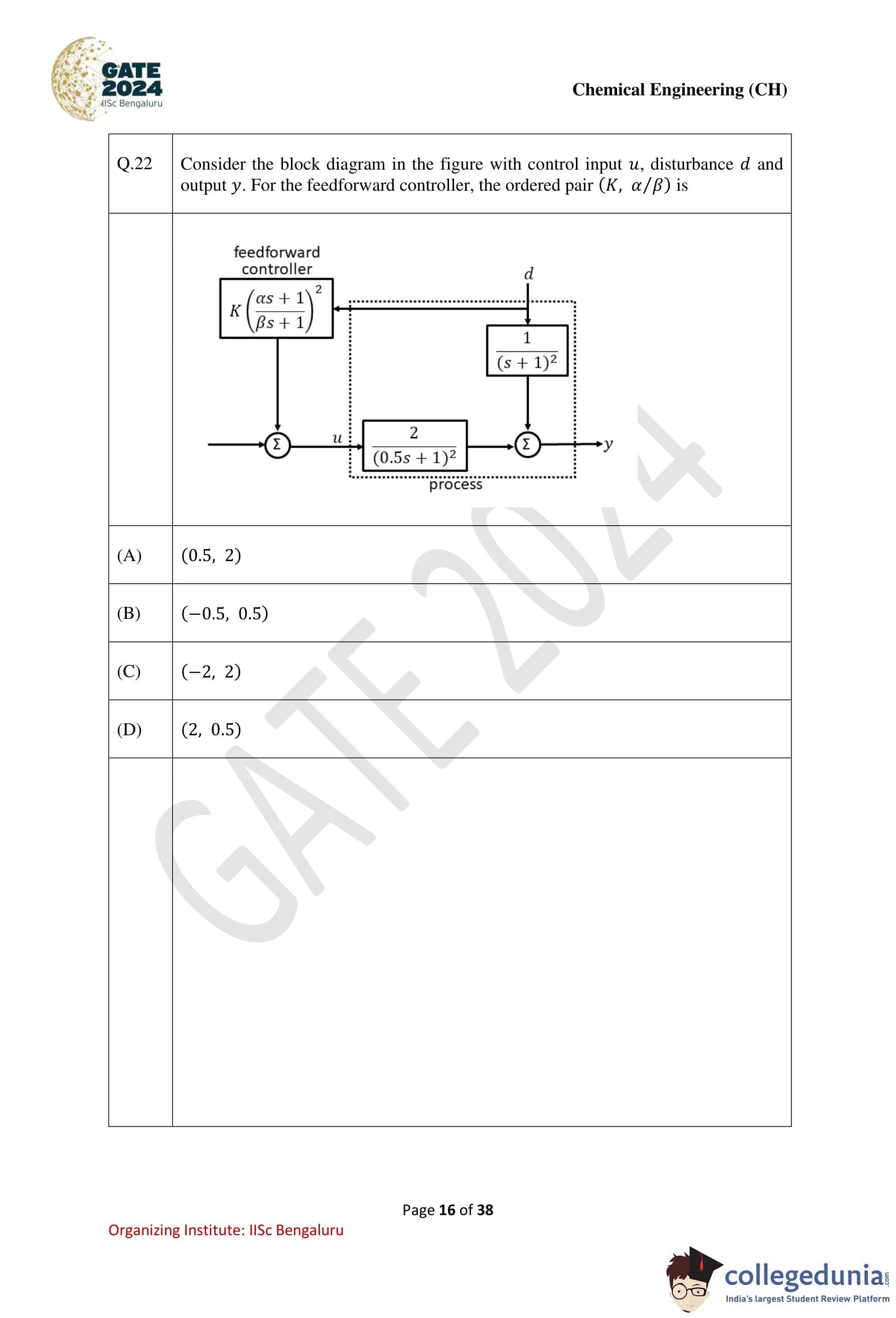


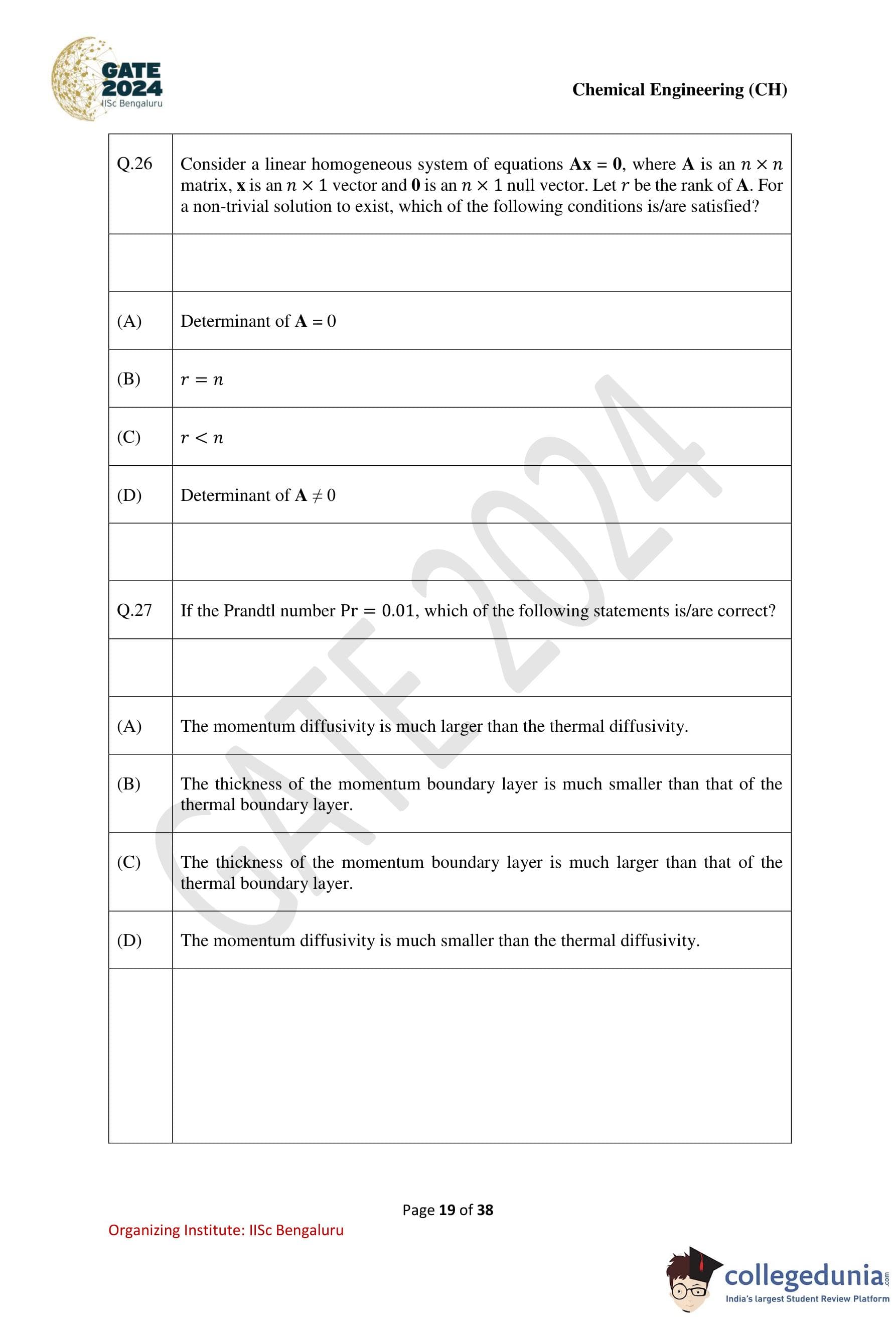





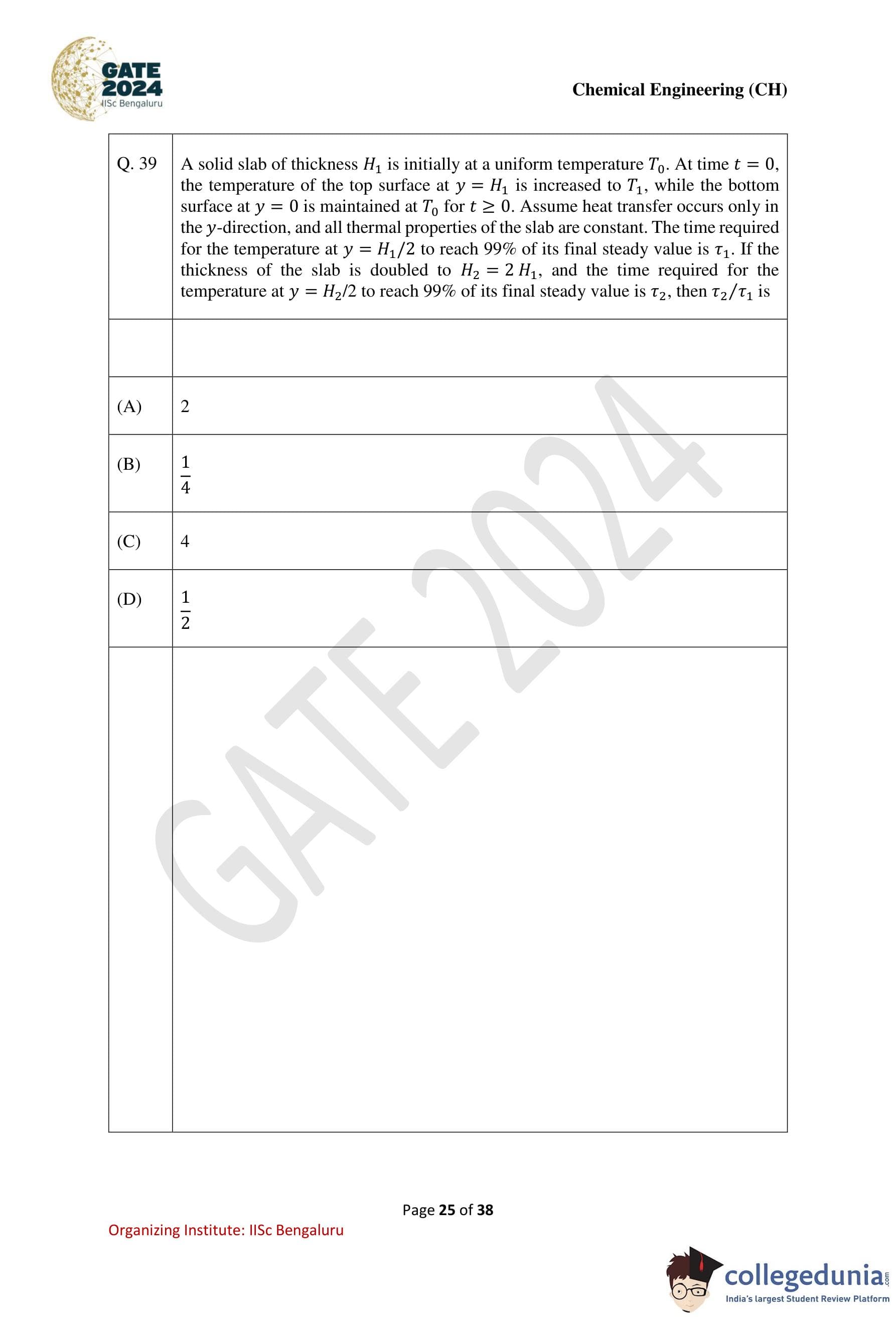
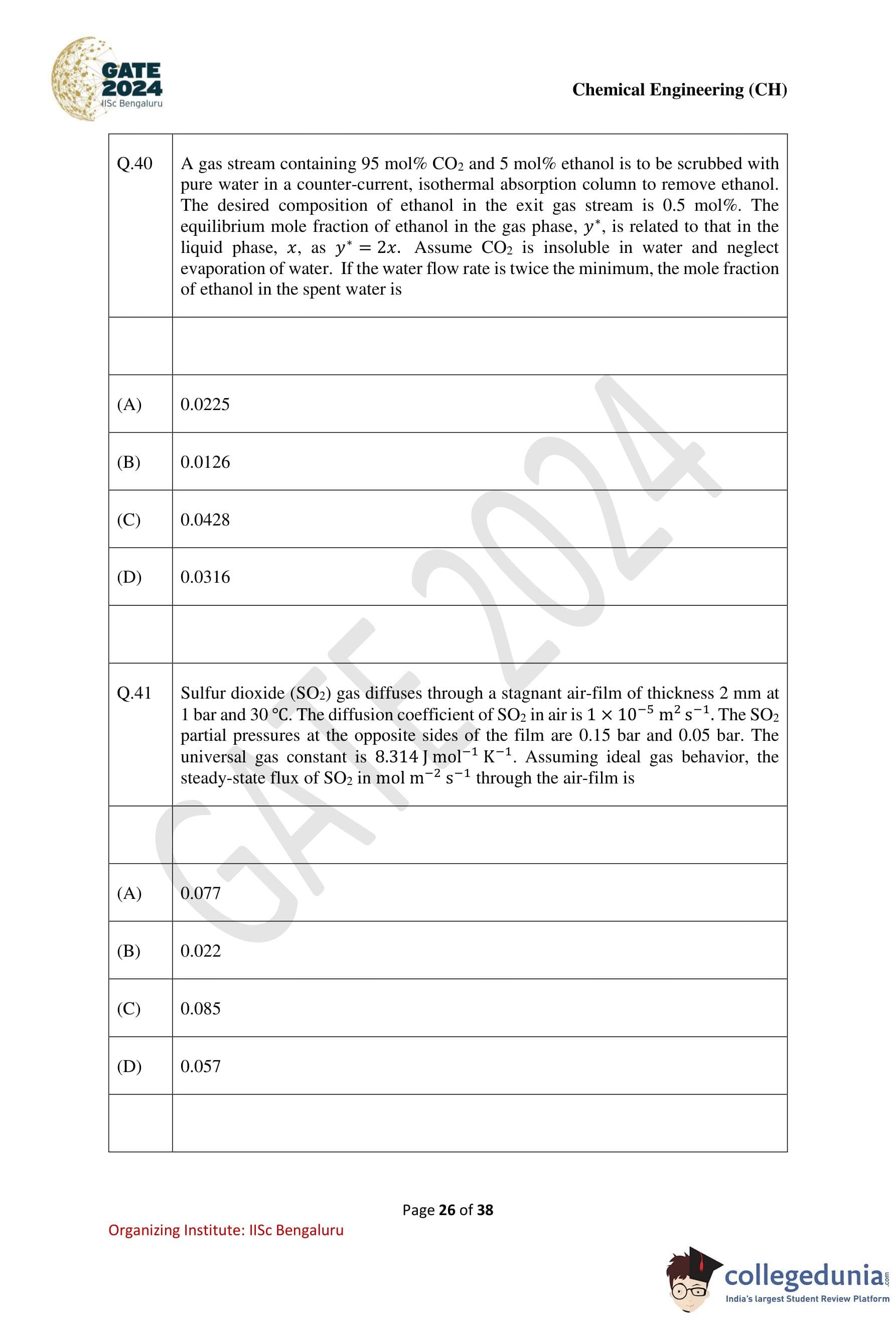



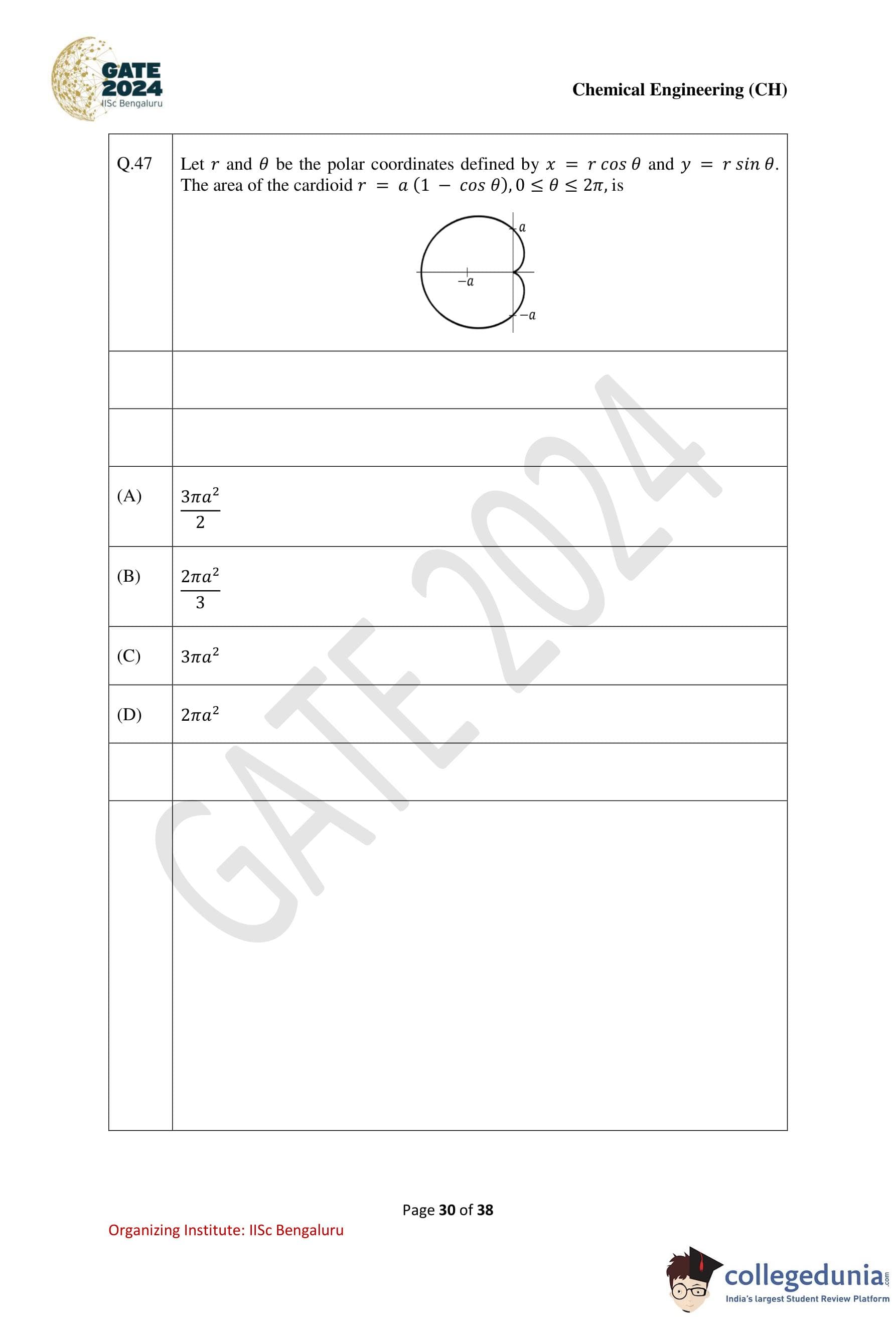
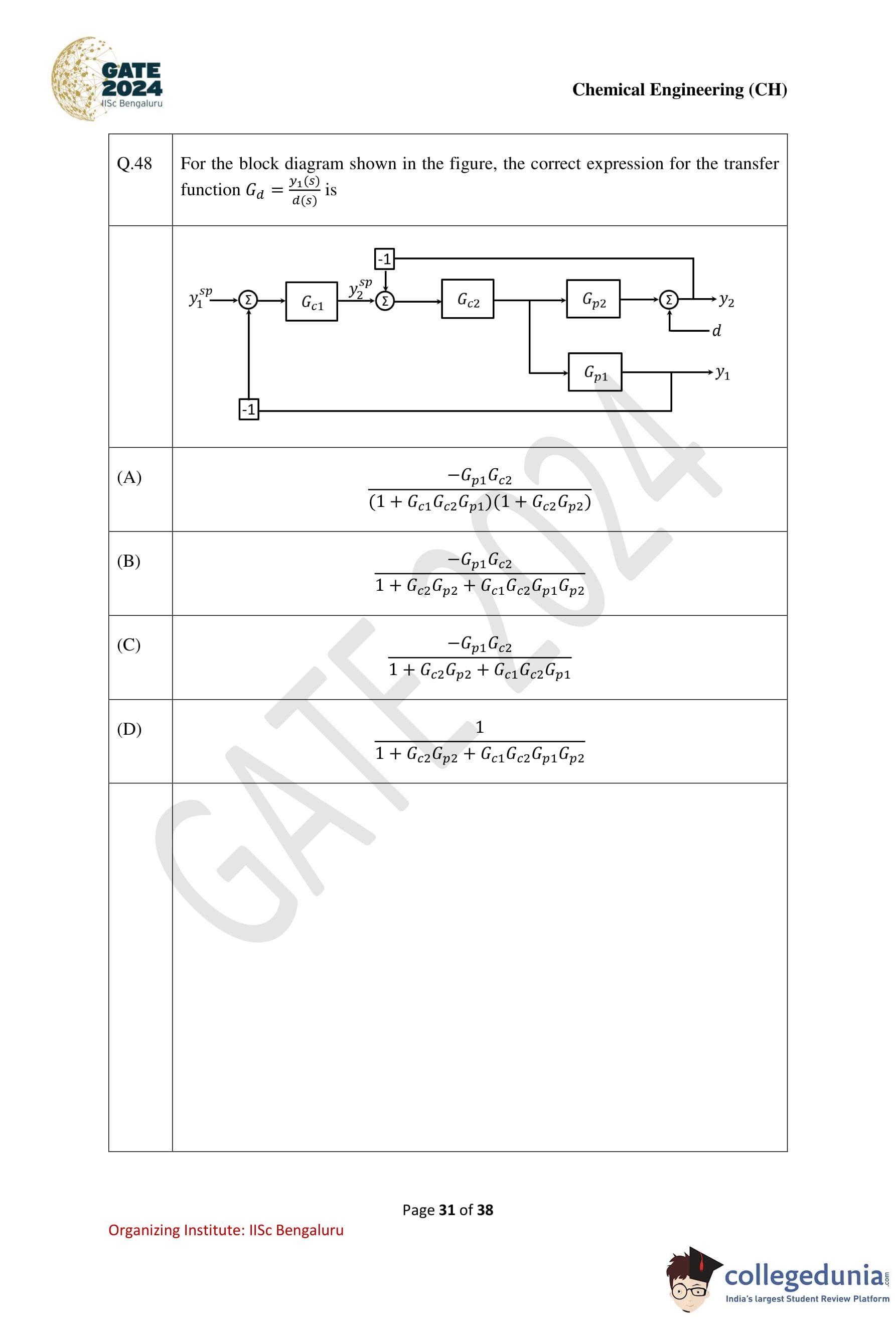
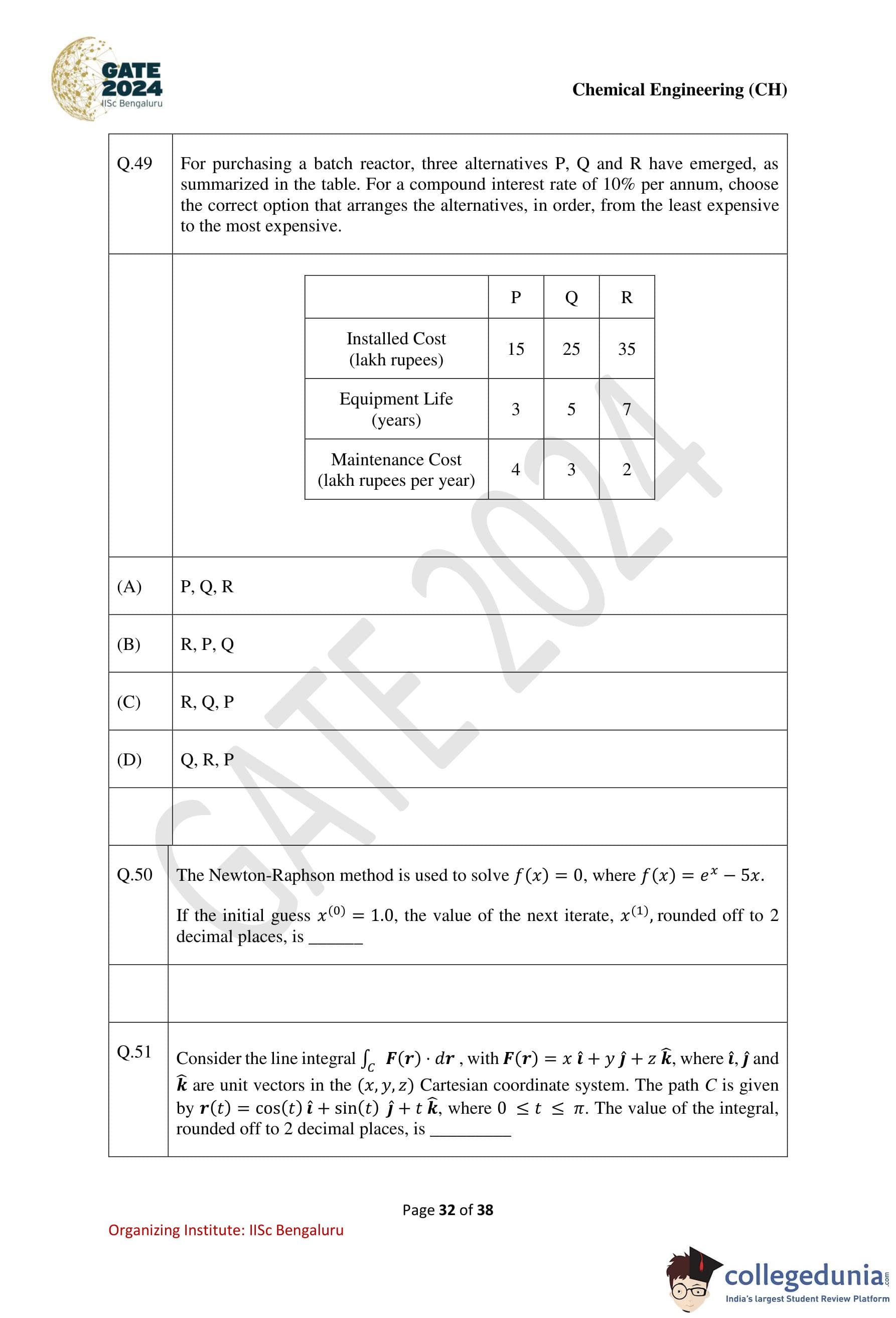






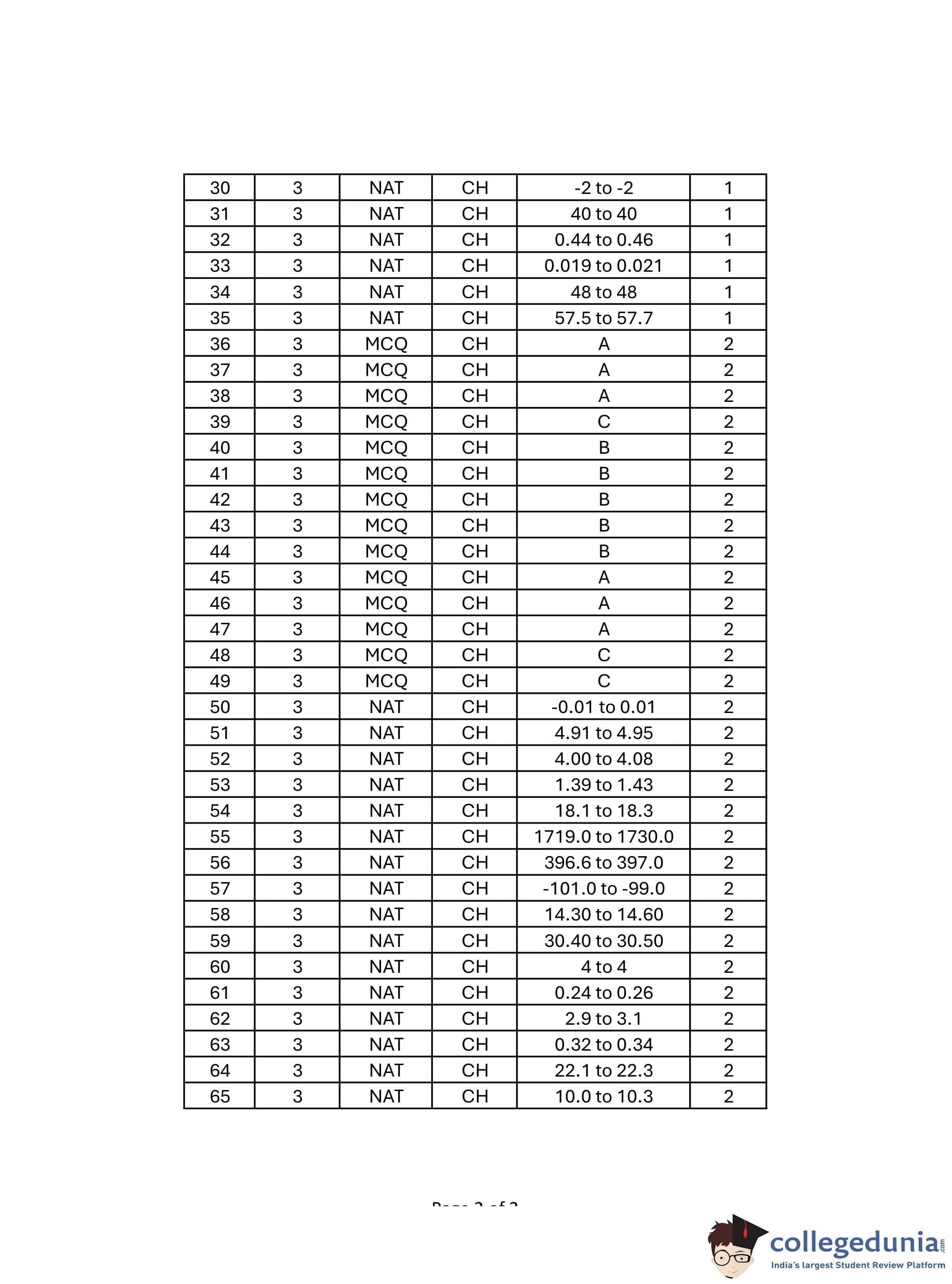









Comments