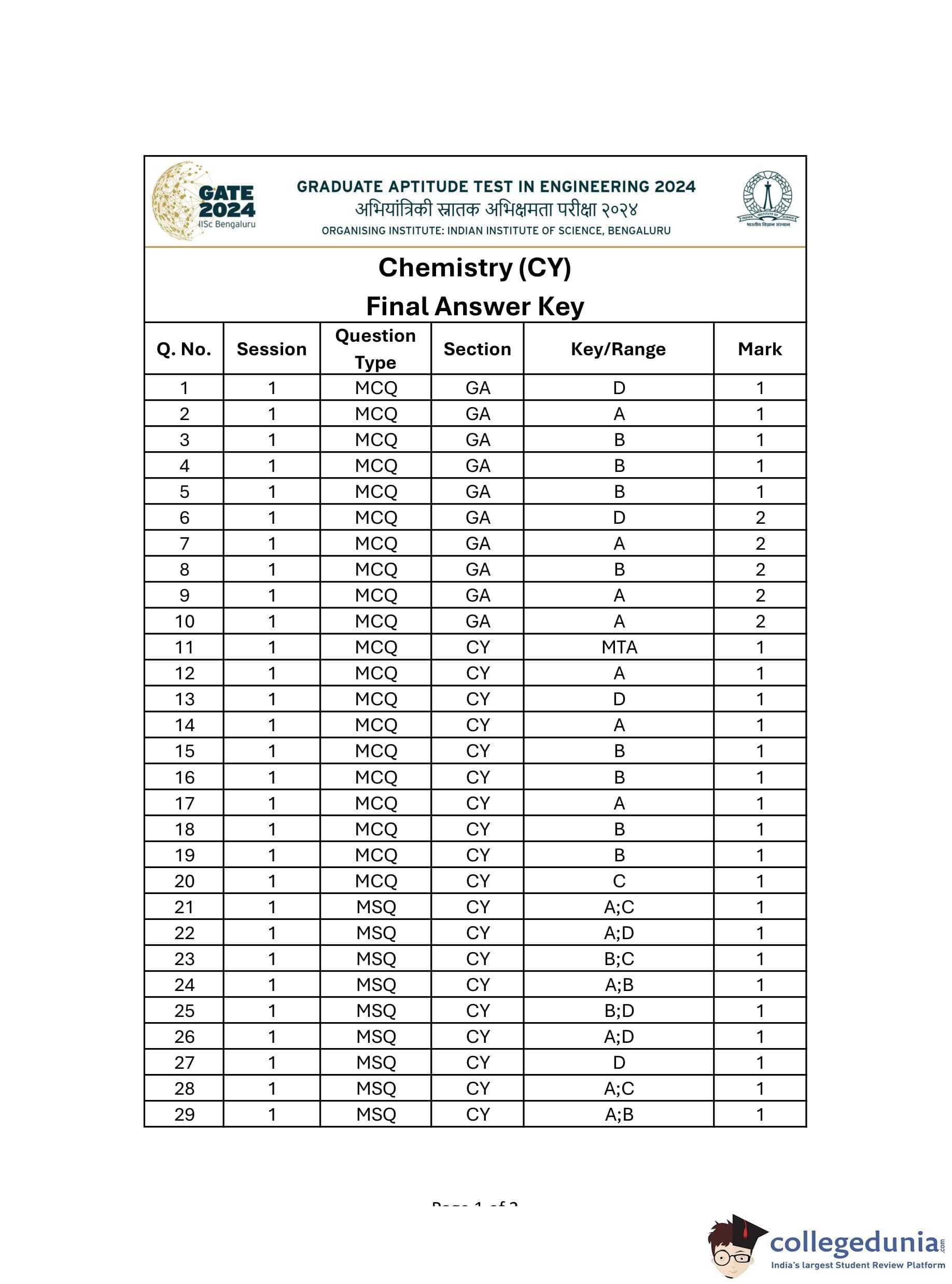
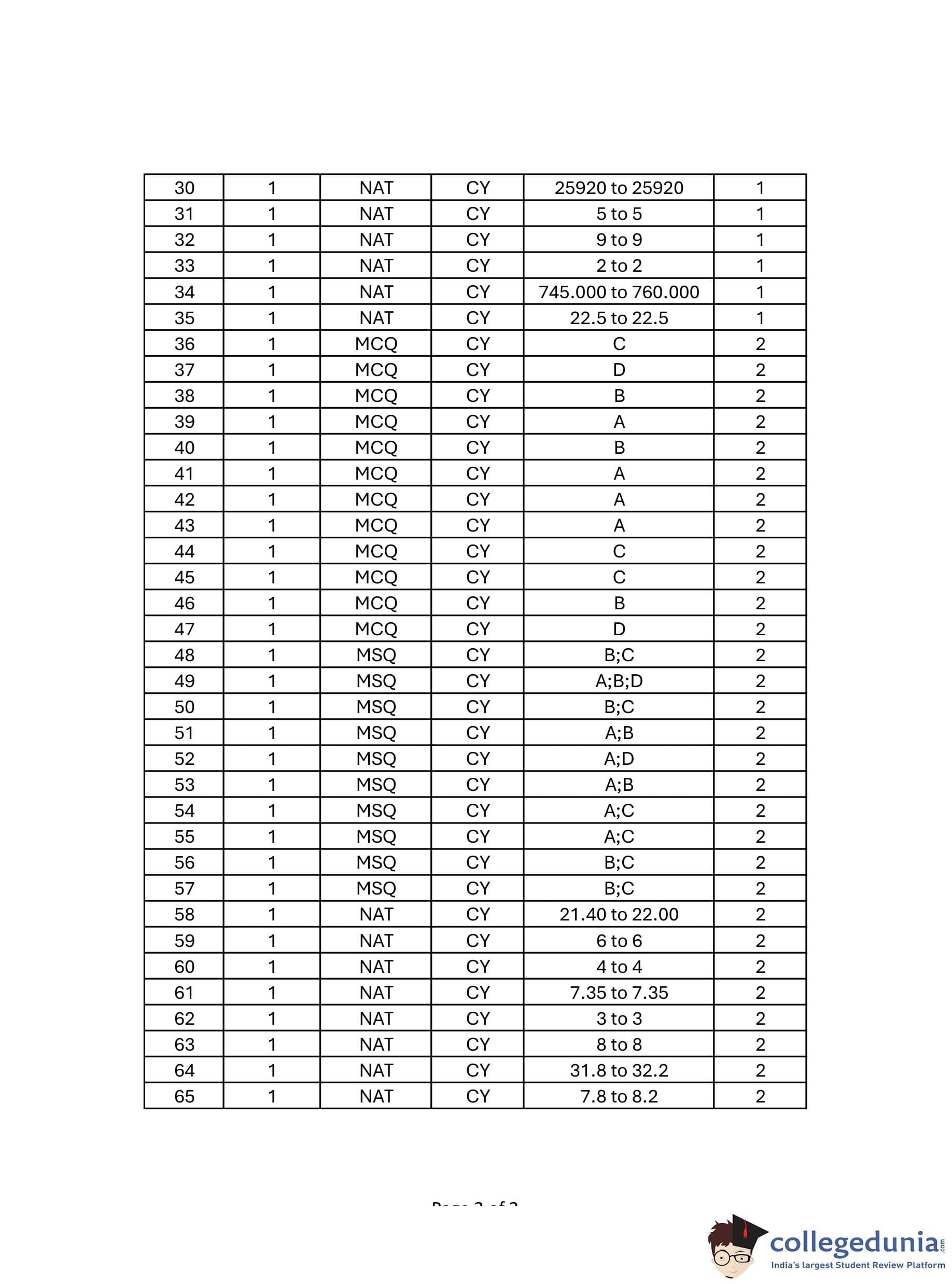
GATE 2024 Chemistry Question Paper PDF is available here. IISc Banglore conducted GATE 2024 Chemistry exam on February 3 in the Forenoon Session from 9:30 AM to 12:30 PM. Students have to answer 65 questions in GATE 2024 Chemistry Question Paper carrying a total weightage of 100 marks. 10 questions are from the General Aptitude section and 55 questions are from Core Discipline.
GATE 2024 Chemistry Question Paper with Solutions PDF
| GATE 2024 Chemistry Question Paper with Solutions PDF | Download | Check Solutions |
Also Check:
- GATE Question Paper (Available): Check Previous Year Question Paper with Solution PDF
- GATE Question Paper 2025 (Soon): Check Previous Year Question Paper with Solution PDF
- GATE Question Paper 2024 (Available): Check Previous Year Question Paper with Solution PDF
- GATE Paper Analysis, Difficulty Level, Branch-wise Question Paper Analysis, Weightage of Topics
GATE Chemistry Question Paper with Solution 2024
If `\(\rightarrow\)` denotes increasing order of intensity, then the meaning of the words \([ sick \rightarrow infirm \rightarrow moribund ]\) is analogous to \([ silly \rightarrow \_\_\_\_ \rightarrow daft ]\).
Which one of the given options is appropriate to fill the blank?
View Solution
Concept:
The arrow \( \rightarrow \) represents an increasing order of intensity in meaning.
Hence, the words must follow a progression where each subsequent word expresses a stronger degree of the same quality.
sick \(\rightarrow\) infirm \(\rightarrow\) moribund shows increasing severity of illness.
Similarly, the second set must show increasing foolishness.
Step 1: Analyze the given analogy. \[ silly \rightarrow \_\_\_\_ \rightarrow daft \]
silly: mildly foolish
daft: extremely foolish
The missing word should indicate a level of foolishness that lies between \textit{silly and \textit{daft.
Step 2: Evaluate the options.
frown: a facial expression (not related)
fawn: to flatter excessively (not related)
vein: a blood vessel (irrelevant)
vain: foolishly proud; lacking good judgment
Step 3: Form the correct sequence. \[ silly \rightarrow vain \rightarrow daft \]
Thus, the correct option is vain. Quick Tip: In analogy questions based on intensity: Identify the common trait, Arrange meanings from weak to strong, Eliminate contextually unrelated words.
The 15 parts of the given figure are to be painted such that no two adjacent parts
with shared boundaries (excluding corners) have the same color.
The minimum number of colors required is
View Solution
Concept:
This is a map coloring problem. The objective is to color different regions of a figure such that:
No two adjacent regions sharing a boundary have the same color,
Regions touching only at corners are allowed to have the same color,
The total number of colors used is minimum.
Step 1: Observe the structure of the figure.
The figure consists of:
An inner circular region divided into 3 sectors,
A middle square-like structure divided into triangular parts,
An outer oval region further divided into multiple sections.
Each region shares boundaries with multiple neighboring regions, making the coloring constraint tight.
Step 2: Start coloring the central regions.
The innermost circular region has three mutually adjacent sectors.
Hence, these three regions must be colored with three different colors.
Step 3: Extend the coloring outward.
Now consider the surrounding regions:
Each outer region is adjacent to at most two differently colored regions,
A third color is always available without causing any conflict.
Thus, all remaining regions can be colored using only the same three colors by repeating them carefully.
Step 4: Check minimality.
Using only two colors is impossible due to the triangular adjacency in the center,
Since three colors suffice for the entire figure, no additional color is required. Quick Tip: In coloring problems: Look for triangular or cyclic adjacency — they determine the minimum colors, Start coloring from the most constrained (central) region, Expand outward while reusing colors wherever possible.
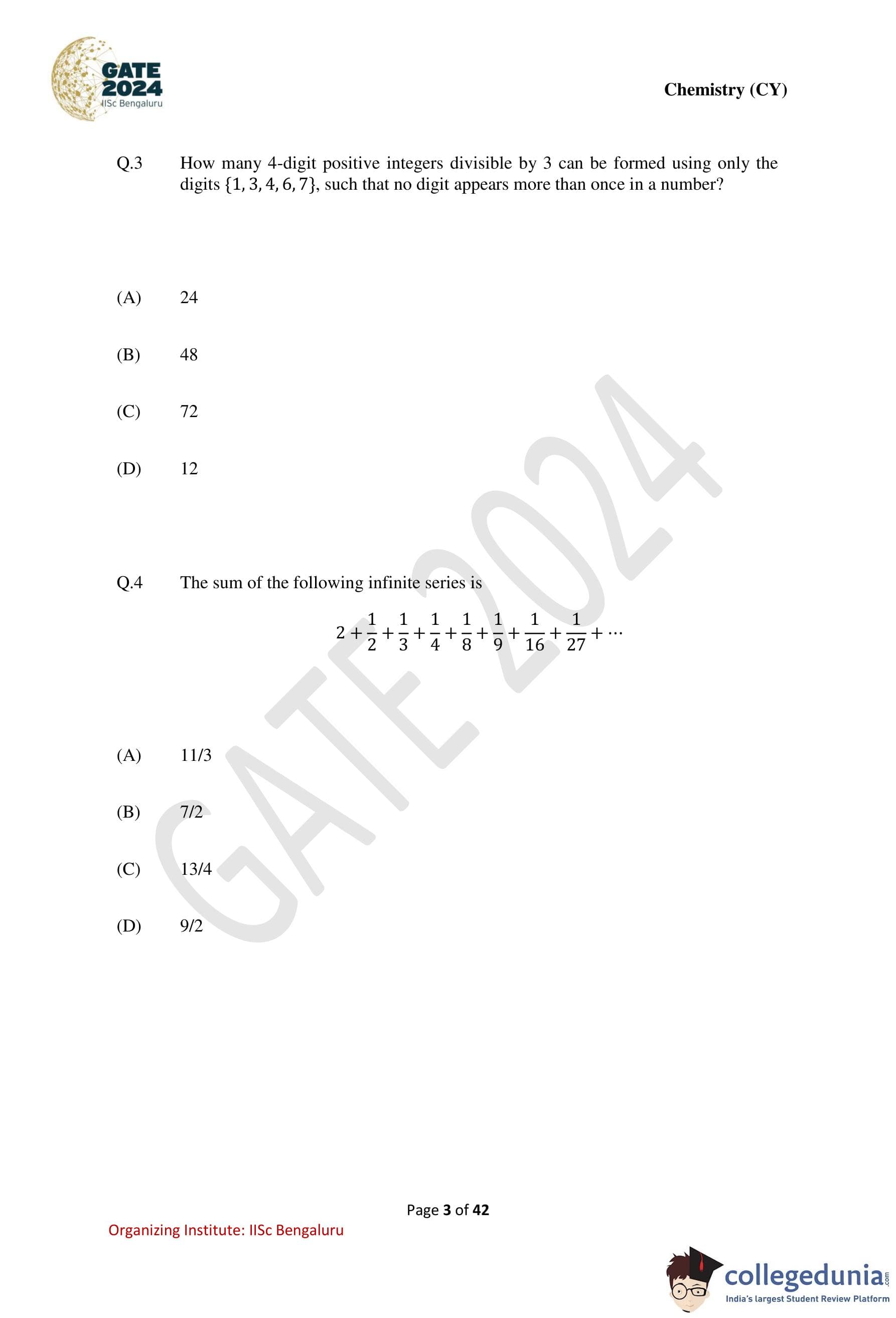
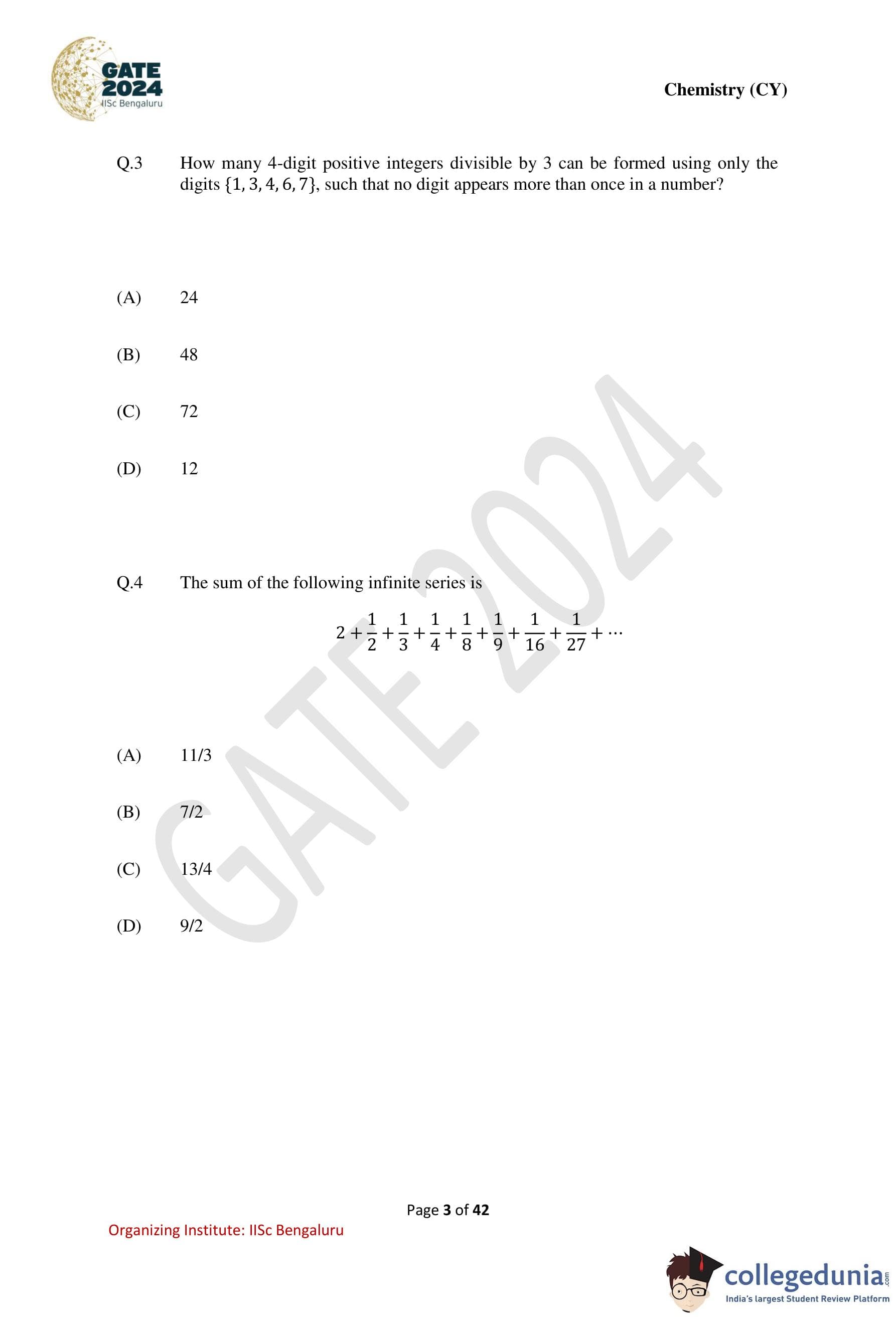
How many 4-digit positive integers divisible by 3 can be formed using only the digits \(\{1, 3, 4, 6, 7\}\), such that no digit appears more than once in a number?
View Solution
Concept:
A number is divisible by 3 if the sum of its digits is divisible by 3.
Also, since digits are not repeated, we deal with permutations of distinct digits.
Step 1: Find the remainders of the given digits modulo 3. \[ \begin{aligned} 1 &\equiv 1 \pmod{3}
3 &\equiv 0 \pmod{3}
4 &\equiv 1 \pmod{3}
6 &\equiv 0 \pmod{3}
7 &\equiv 1 \pmod{3} \end{aligned} \]
Thus:
Digits with remainder 0: \(\{3, 6\}\)
Digits with remainder 1: \(\{1, 4, 7\}\)
Step 2: Choose 4 digits whose sum is divisible by 3.
Let the number of digits with remainder 0 be \(k\).
Then the sum condition gives: \[ (4 - k) \equiv 0 \pmod{3} \Rightarrow 4 - k = 3 \Rightarrow k = 1 \]
Step 3: Count the valid selections.
Choose 1 digit from \(\{3,6\}\): \(2\) ways
Choose 3 digits from \(\{1,4,7\}\): \(1\) way
Total valid digit sets \(= 2\).
Step 4: Arrange each set into 4-digit numbers.
Each set has \(4! = 24\) permutations.
\[ Total numbers = 2 \times 24 = 48 \] Quick Tip: For divisibility-by-3 problems with permutations: First select digits satisfying the divisibility rule, Then count the permutations of those digits.
The sum of the following infinite series is \[ 2 + \frac{1}{2} + \frac{1}{3} + \frac{1}{4} + \frac{1}{8} + \frac{1}{9} + \frac{1}{16} + \frac{1}{27} + \cdots \]
View Solution
Concept:
The given series can be split into separate geometric series:
A constant term,
A geometric series with ratio \( \frac{1}{2} \),
A geometric series with ratio \( \frac{1}{3} \).
Step 1: Rearrange the series. \[ 2 + \left(\frac{1}{2} + \frac{1}{4} + \frac{1}{8} + \frac{1}{16} + \cdots \right) + \left(\frac{1}{3} + \frac{1}{9} + \frac{1}{27} + \cdots \right) \]
Step 2: Evaluate each part.
First term: \(2\)
Second series:
\[ \frac{\frac{1}{2}}{1 - \frac{1}{2}} = 1 \]
Third series:
\[ \frac{\frac{1}{3}}{1 - \frac{1}{3}} = \frac{1}{2} \]
Step 3: Add the results. \[ 2 + 1 + \frac{1}{2} = \frac{7}{2} \] Quick Tip: When dealing with infinite series: Look for hidden geometric patterns, Split mixed sequences into simpler known series.
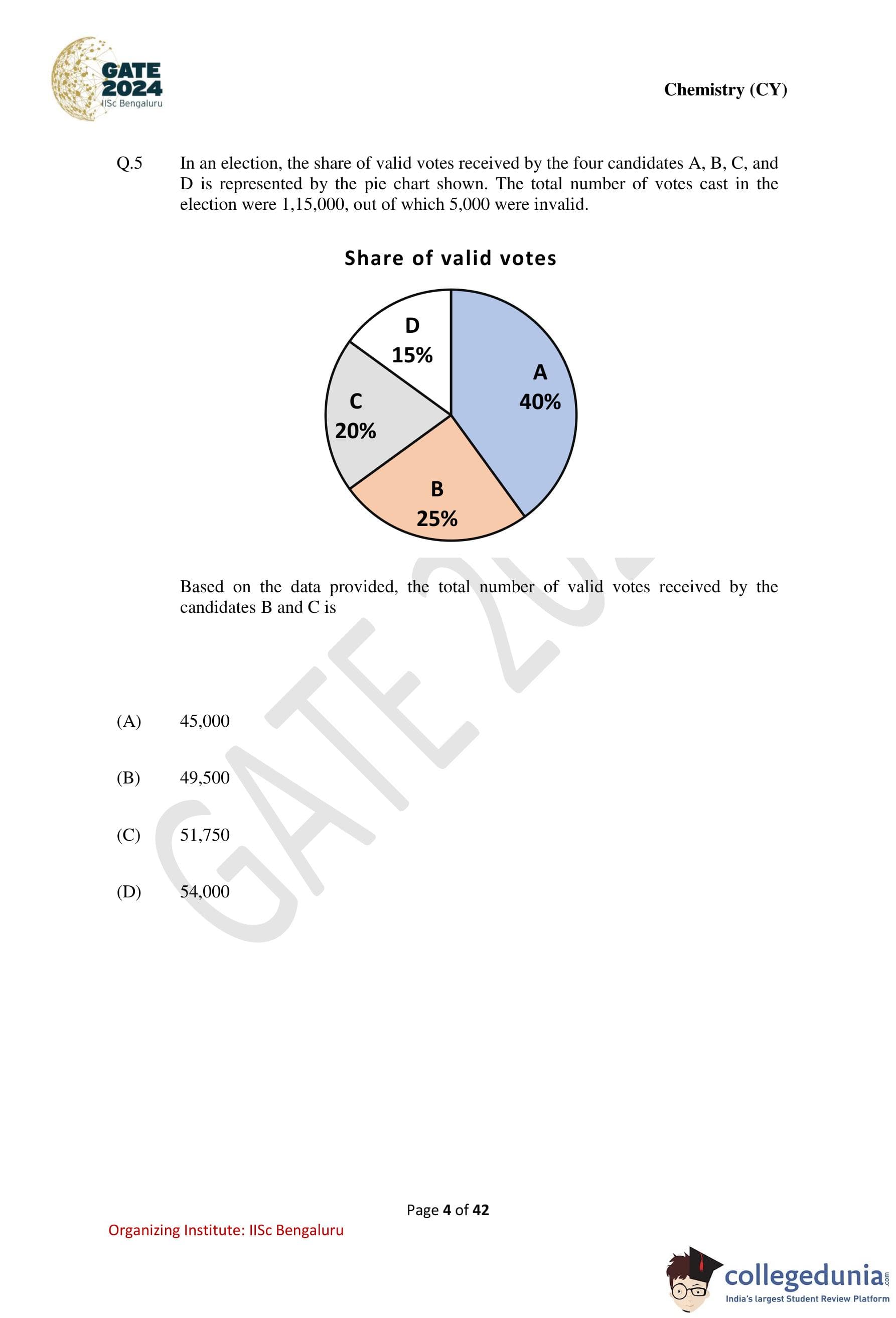
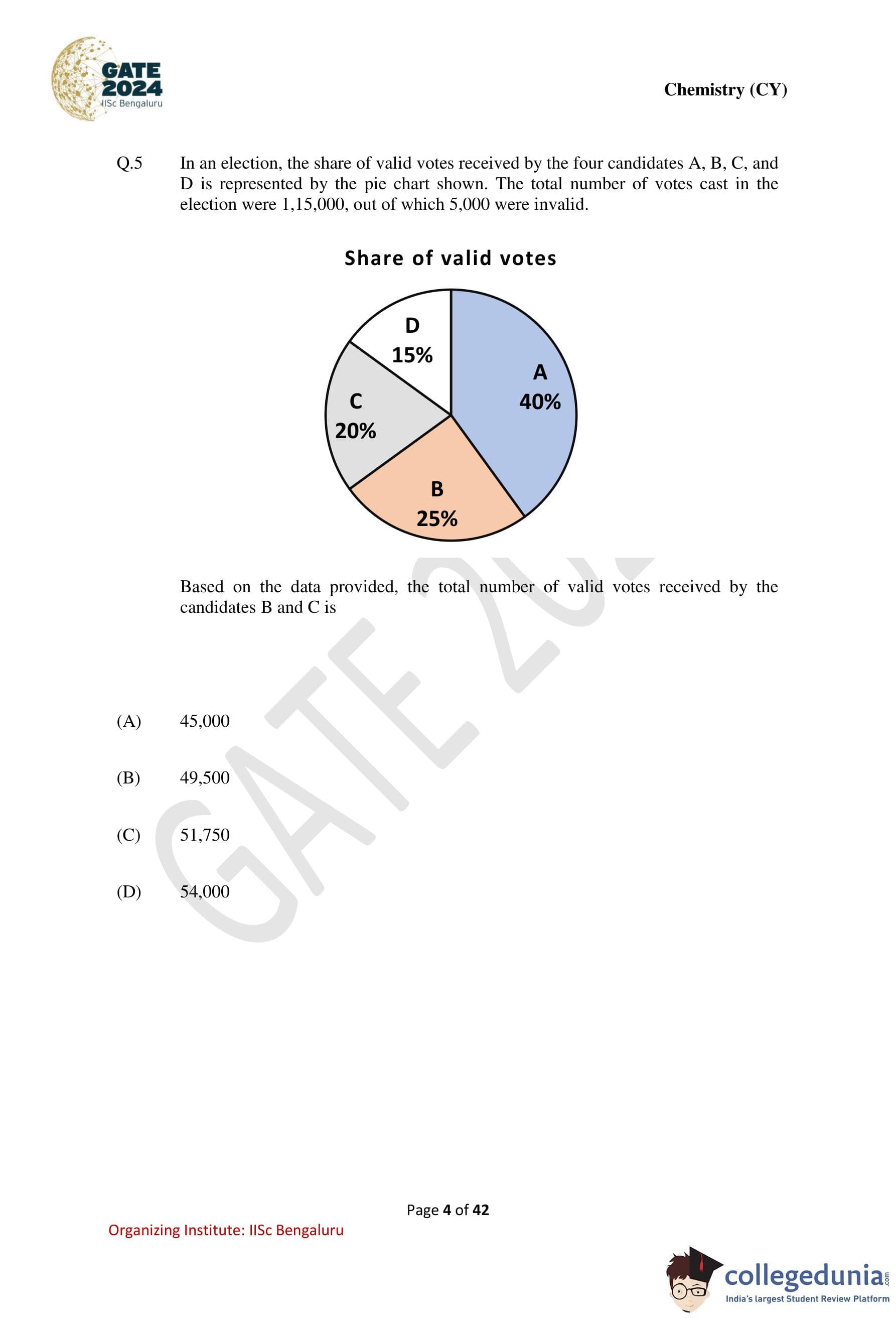
In an election, the share of valid votes received by the four candidates A, B, C, and D
is represented by the pie chart shown. The total number of votes cast in the election were
1,15,000, out of which 5,000 were invalid. Based on the data provided, the total number of valid
votes received by the candidates B and C is
View Solution
Concept:
A pie chart represents the percentage distribution of valid votes.
To find the actual number of votes:
First calculate the total number of valid votes,
Then apply the given percentages to this total.
Step 1: Calculate the total number of valid votes. \[ Total votes cast = 1,15,000 \] \[ Invalid votes = 5,000 \] \[ Valid votes = 1,15,000 - 5,000 = 1,10,000 \]
Step 2: Determine the combined percentage of votes for candidates B and C.
From the pie chart: \[ Votes for B = 25%, \quad Votes for C = 20% \] \[ Total for B and C = 25% + 20% = 45% \]
Step 3: Calculate the number of valid votes received by B and C. \[ 45% of 1,10,000 = \frac{45}{100} \times 1,10,000 = 49,500 \] Quick Tip: In pie-chart problems: Always confirm whether percentages are of total votes or valid votes, Subtract invalid data before applying percentages.
Thousands of years ago, some people began dairy farming. This coincided with a number
of mutations in a particular gene that resulted in these people developing the ability to
digest dairy milk. Based on the given passage, which of the following can be inferred?
View Solution
Concept:
Inference-based questions require conclusions that are:
Directly supported by the passage,
Logically necessary,
Free from exaggeration or assumptions beyond the text.
Step 1: Analyze the key information in the passage.
Dairy farming began thousands of years ago,
This coincided with mutations in a particular gene,
These mutations enabled people to digest dairy milk.
Step 2: Evaluate the options.
Option (A): Incorrect — the passage refers only to \emph{some people, not all humans.
Option (B): Incorrect — directly contradicts the passage.
Option (C): Incorrect — no statement about essentiality is made.
Option (D): Correct — it restates the logical inference drawn from the passage.
Thus, option (D) is the only valid inference. Quick Tip: For inference questions: Stick strictly to what is implied, Avoid options with words like \emph{all}, \emph{none}, or \emph{essential} unless clearly stated.
The probability of a boy or a girl being born is \( \frac{1}{2} \).
For a family having only three children, what is the probability of having two girls and one boy?
View Solution
Concept:
This is a basic probability and combination problem where:
Each child is equally likely to be a boy or a girl,
Births are independent events.
Step 1: Find the total number of possible outcomes.
Each child has 2 possible outcomes (boy or girl), so: \[ Total outcomes = 2^3 = 8 \]
Step 2: Count the favorable outcomes (exactly two girls and one boy).
The number of ways to choose positions for two girls out of three children is: \[ \binom{3}{2} = 3 \]
Step 3: Calculate the probability. \[ Probability = \frac{Favorable outcomes}{Total outcomes} = \frac{3}{8} \] Quick Tip: For such probability problems: Use combinations to count arrangements, Multiply probabilities only after fixing the number of favorable cases.
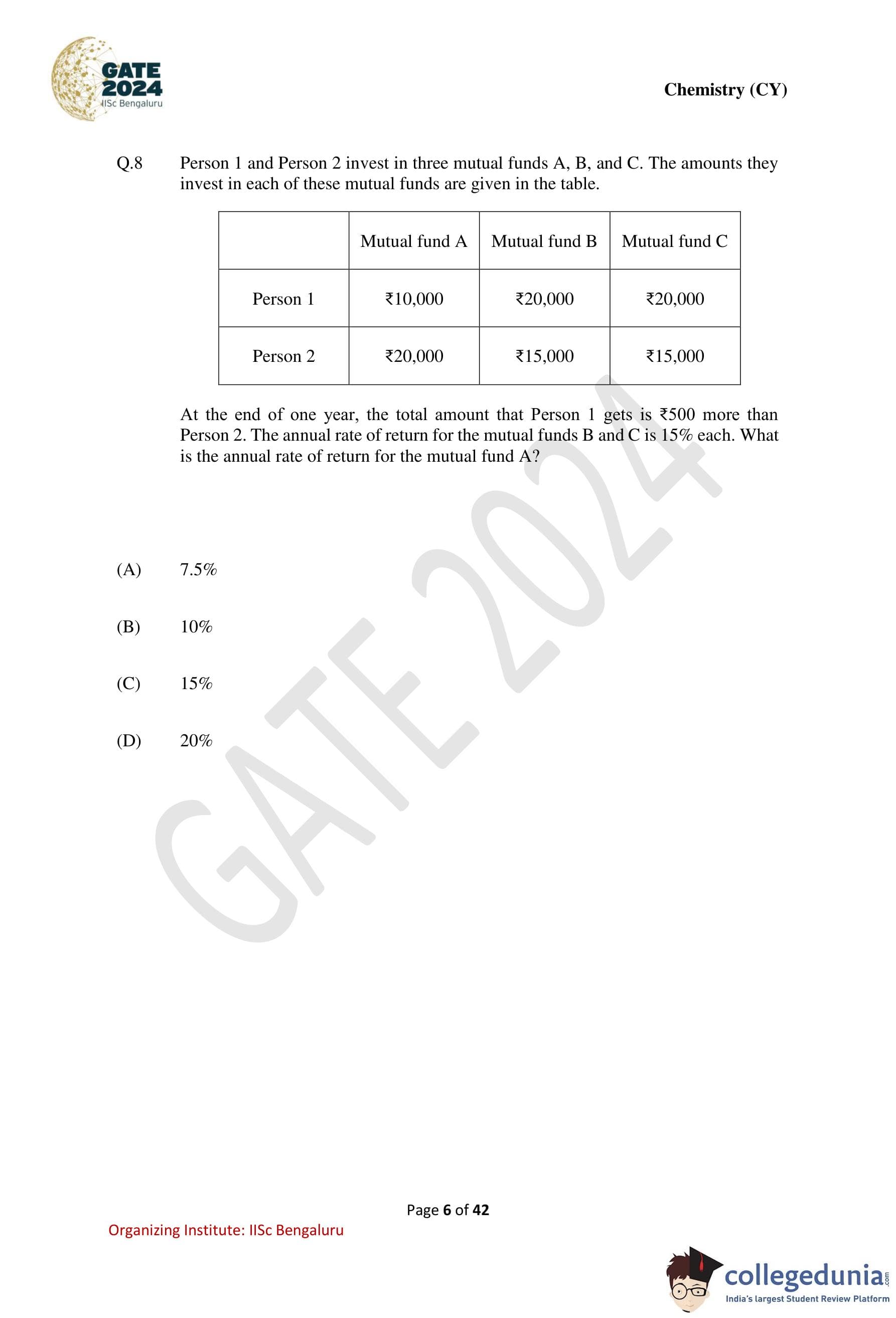
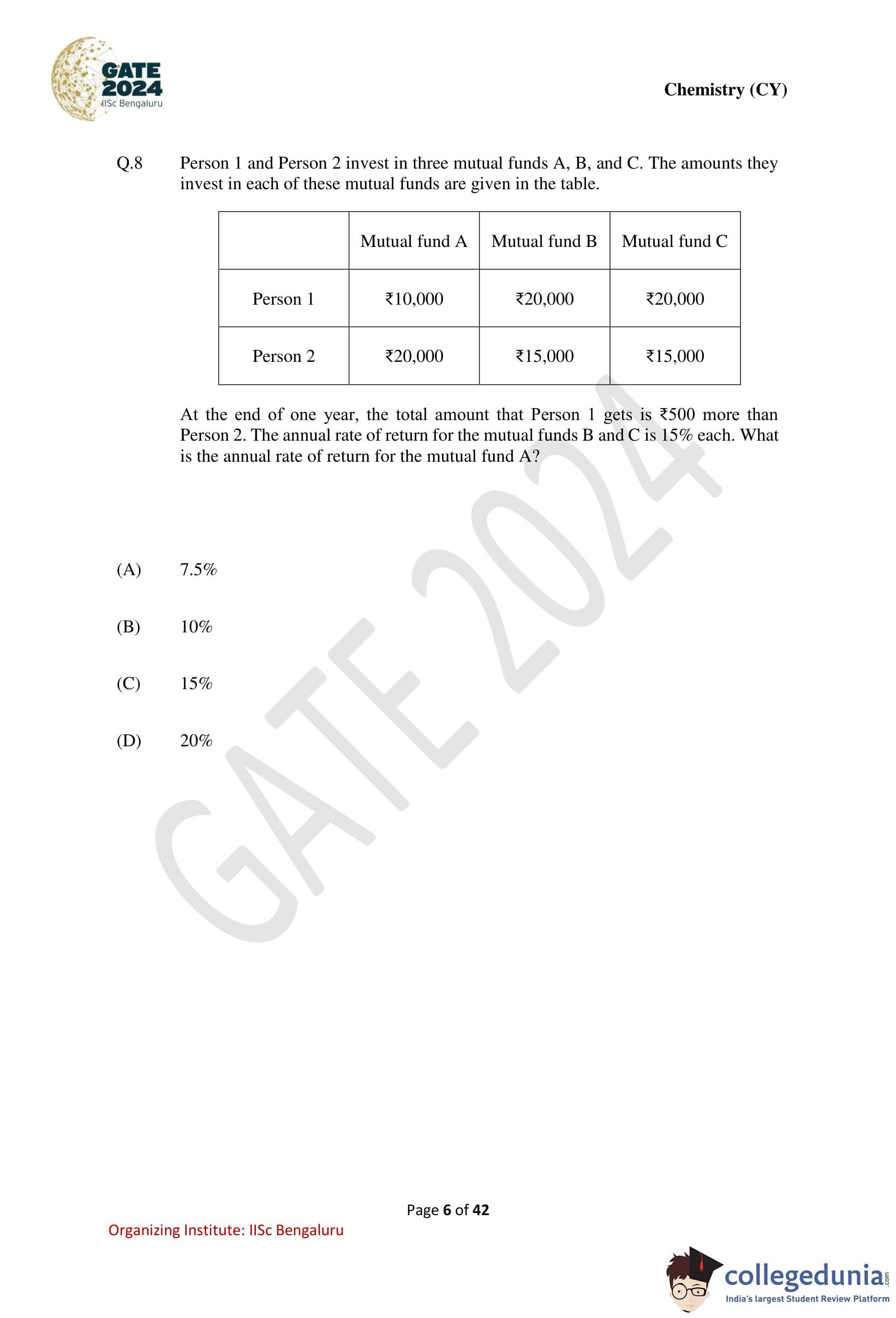
Person 1 and Person 2 invest in three mutual funds A, B, and C. The amounts they invest
in each of these mutual funds are given in the table. At the end of one year, the total amount
that Person 1 gets is \₹500 more than Person 2. The annual rate of return for the mutual funds
B and C is 15% each. What is the annual rate of return for the mutual fund A?
View Solution
Concept:
The total amount received after one year is: \[ Principal + Return \]
If the principal investments of both persons are equal, then any difference in the final amount
is solely due to the difference in returns.
Step 1: Calculate total investments of both persons.
\[ Person 1 = 10{,}000 + 20{,}000 + 20{,}000 = 50{,}000 \] \[ Person 2 = 20{,}000 + 15{,}000 + 15{,}000 = 50{,}000 \]
Since both invested the same total amount, the difference of \₹500 is due only to returns.
Step 2: Let the annual rate of return of mutual fund A be \( r \).
Returns of Person 1: \[ 10{,}000r + 20{,}000(0.15) + 20{,}000(0.15) = 10{,}000r + 6{,}000 \]
Returns of Person 2: \[ 20{,}000r + 15{,}000(0.15) + 15{,}000(0.15) = 20{,}000r + 4{,}500 \]
Step 3: Use the given condition.
\[ Return of Person 1 = Return of Person 2 + 500 \]
\[ 10{,}000r + 6{,}000 = 20{,}000r + 4{,}500 + 500 \] \[ 10{,}000r + 6{,}000 = 20{,}000r + 5{,}000 \]
Step 4: Solve for \( r \).
\[ 1{,}000 = 10{,}000r \Rightarrow r = 0.10 = 10% \] Quick Tip: In investment comparison problems: First check whether total investments are equal, If yes, compare only the returns, Assign variables only to unknown rates.
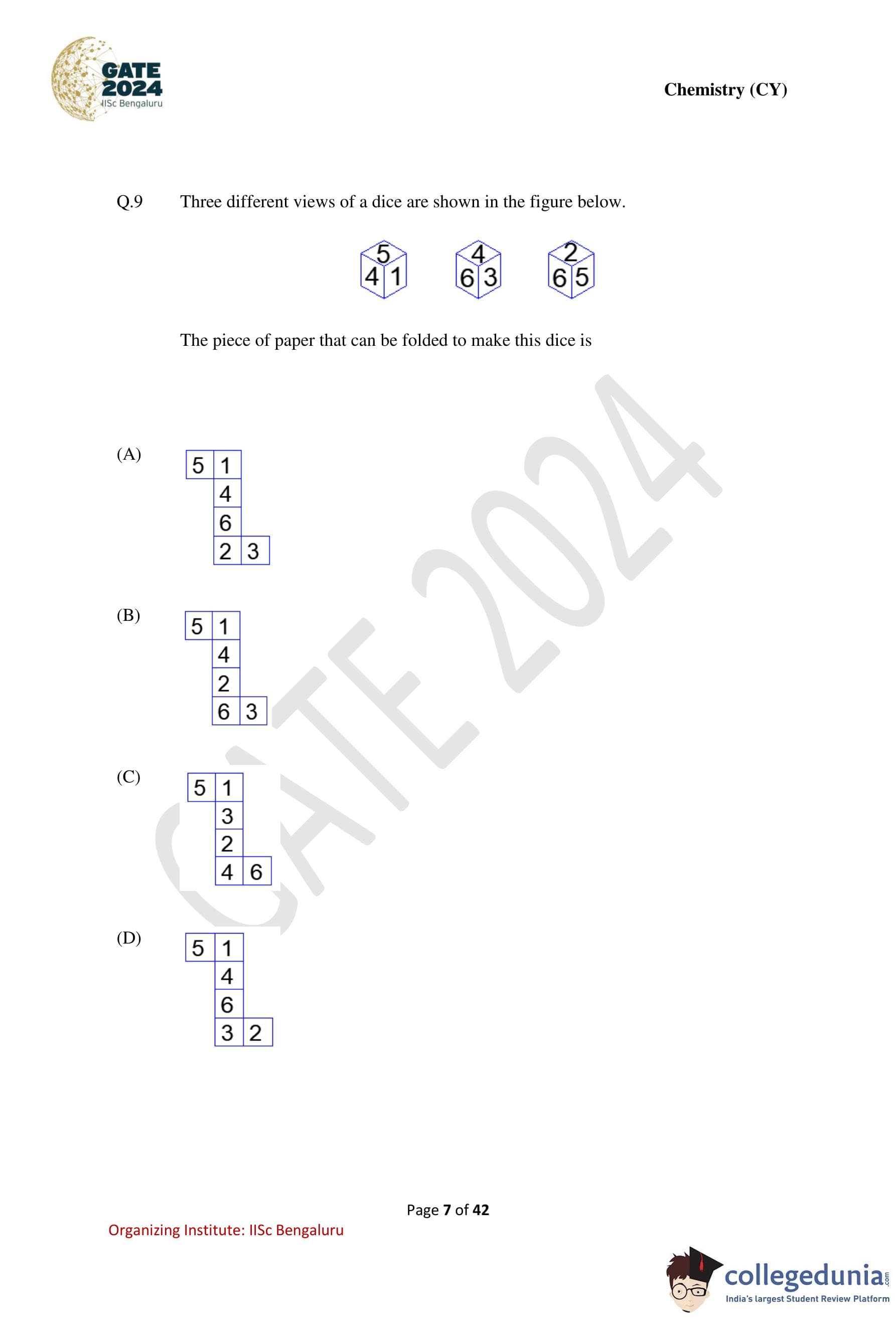
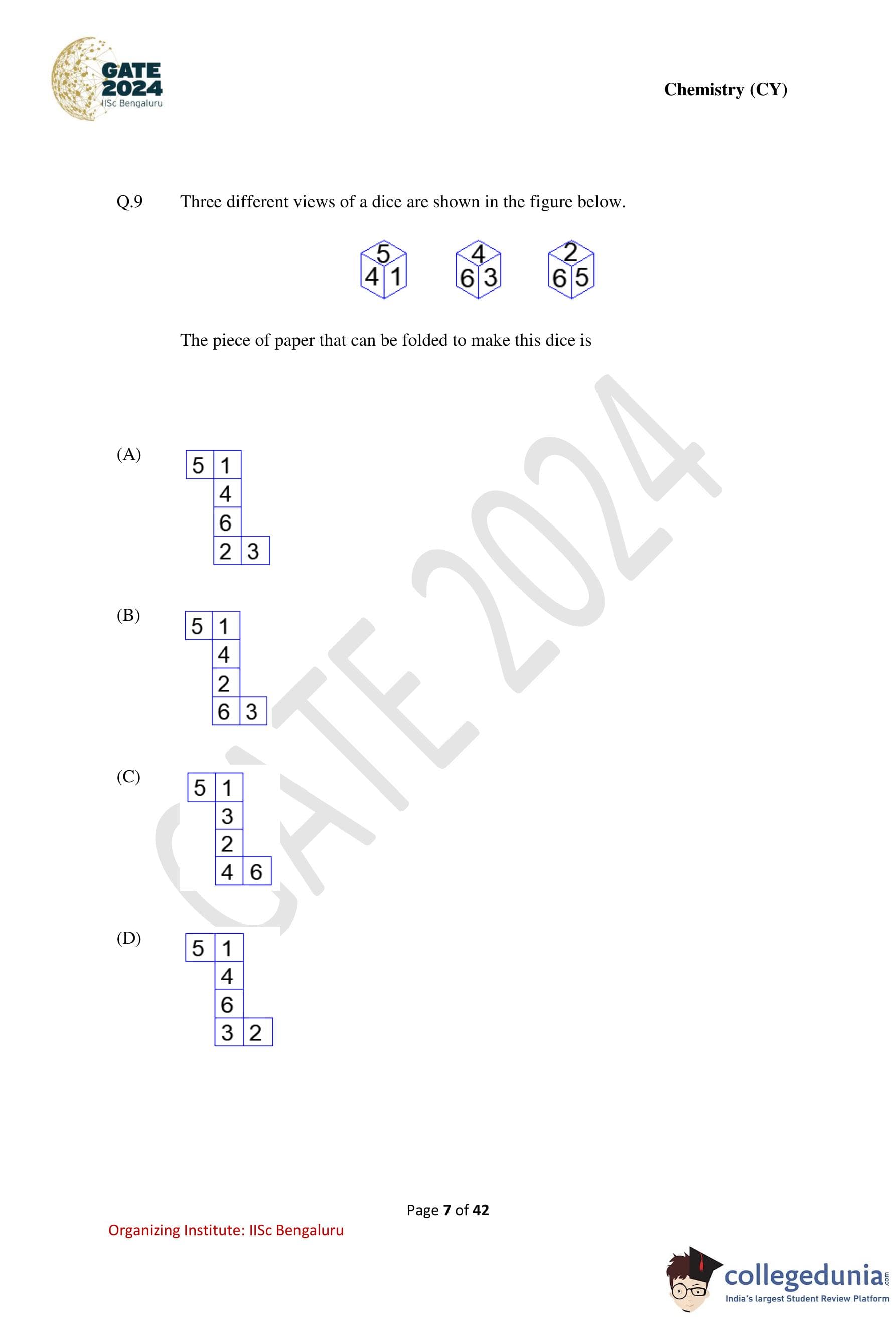
Three different views of a dice are shown in the figure below.
The piece of paper that can be folded to make this dice is
View Solution
Concept:
In dice problems:
Faces that appear together in a view are adjacent,
A face can have only four adjacent faces,
The remaining face (not adjacent) is the opposite face.
Step 1: Identify adjacency from the given views.
From the first view: \[ 5 is adjacent to 4 and 1 \]
From the third view: \[ 5 is adjacent to 6 and 2 \]
Thus, face 5 is adjacent to: \[ \{1, 2, 4, 6\} \]
Hence, the only number not adjacent to 5 is: \[ \boxed{3} \]
So, \(5\) and \(3\) are opposite faces.
Step 2: Determine another opposite pair.
From the first and second views: \[ 4 is adjacent to 5, 1, 6, 3 \]
Thus, the only number not adjacent to 4 is: \[ \boxed{2} \]
So, \(4\) and \(2\) are opposite faces.
Step 3: Identify the final opposite pair.
The remaining numbers are: \[ 1 and 6 \]
Hence, they must be opposite faces.
Opposite face pairs are: \[ (5,3), \quad (4,2), \quad (1,6) \]
Step 4: Check the given nets.
Only Option (D) produces exactly these opposite face pairs when folded into a cube.
All other options place at least one opposite pair on adjacent faces, which is impossible. Quick Tip: For dice problems: First determine opposite face pairs using adjacency, Then eliminate nets where opposite faces touch or share an edge.
Visualize two identical right circular cones such that one is inverted over the other
and they share a common circular base. If a cutting plane passes through the vertices of the
assembled cones, what shape does the outer boundary of the resulting cross-section make?
View Solution
Concept:
When solids are cut by a plane, the shape of the cross-section depends on:
The geometry of the solid,
The orientation of the cutting plane,
The points through which the plane passes.
Here, we are dealing with two identical right circular cones placed apex-to-apex,
sharing a common base.
Step 1: Understand the arrangement of the cones.
The cones are identical and aligned along the same axis,
One cone is inverted over the other,
Their vertices lie on the same straight line (axis),
They share a common circular base in the middle.
Step 2: Analyze the cutting plane.
The cutting plane:
Passes through the vertices of both cones,
Is symmetric about the axis of the cones.
Such a plane intersects:
Each cone in an isosceles triangle,
With equal slant heights due to identical cones.
Step 3: Determine the shape of the outer boundary.
The two triangular cross-sections join base-to-base at the common circular base.
The outer boundary therefore consists of:
Four equal slant edges,
Opposite sides equal and parallel.
This forms a rhombus. Quick Tip: For solid cross-section problems: Always sketch a rough diagram, Look for symmetry in the solid and the cutting plane, Combine individual cross-sections carefully.
Among the following, the compound with the lowest CO stretching frequency is:
The ground state of \(\left[\mathrm{Cr(H_2O)}_6\right]^{2+}\) is:
The reaction of XeF\(_2\) with HN(SO\(_2\)F)\(_2\) at 273 K in CF\(_2\)Cl\(_2\) solvent yields:
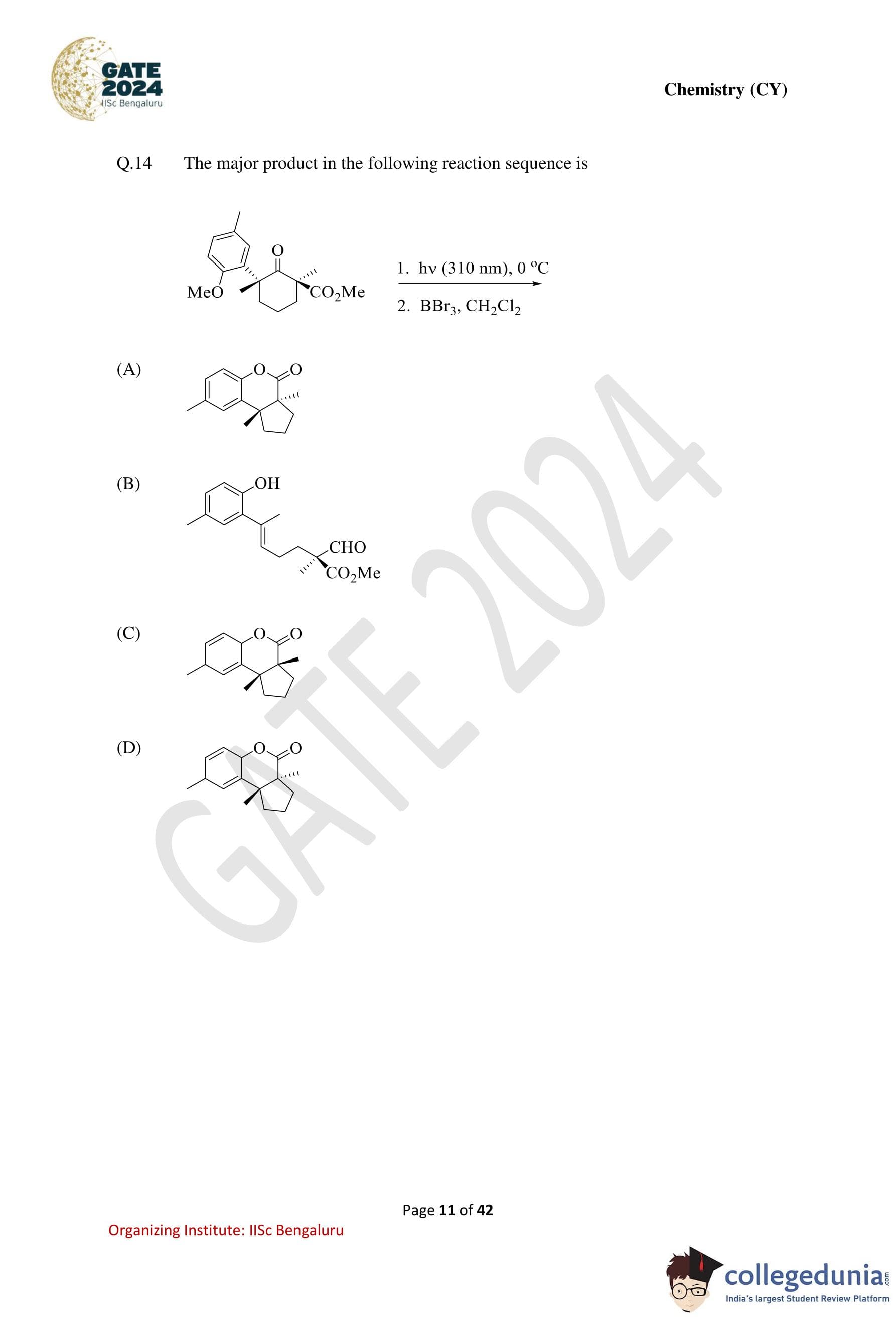
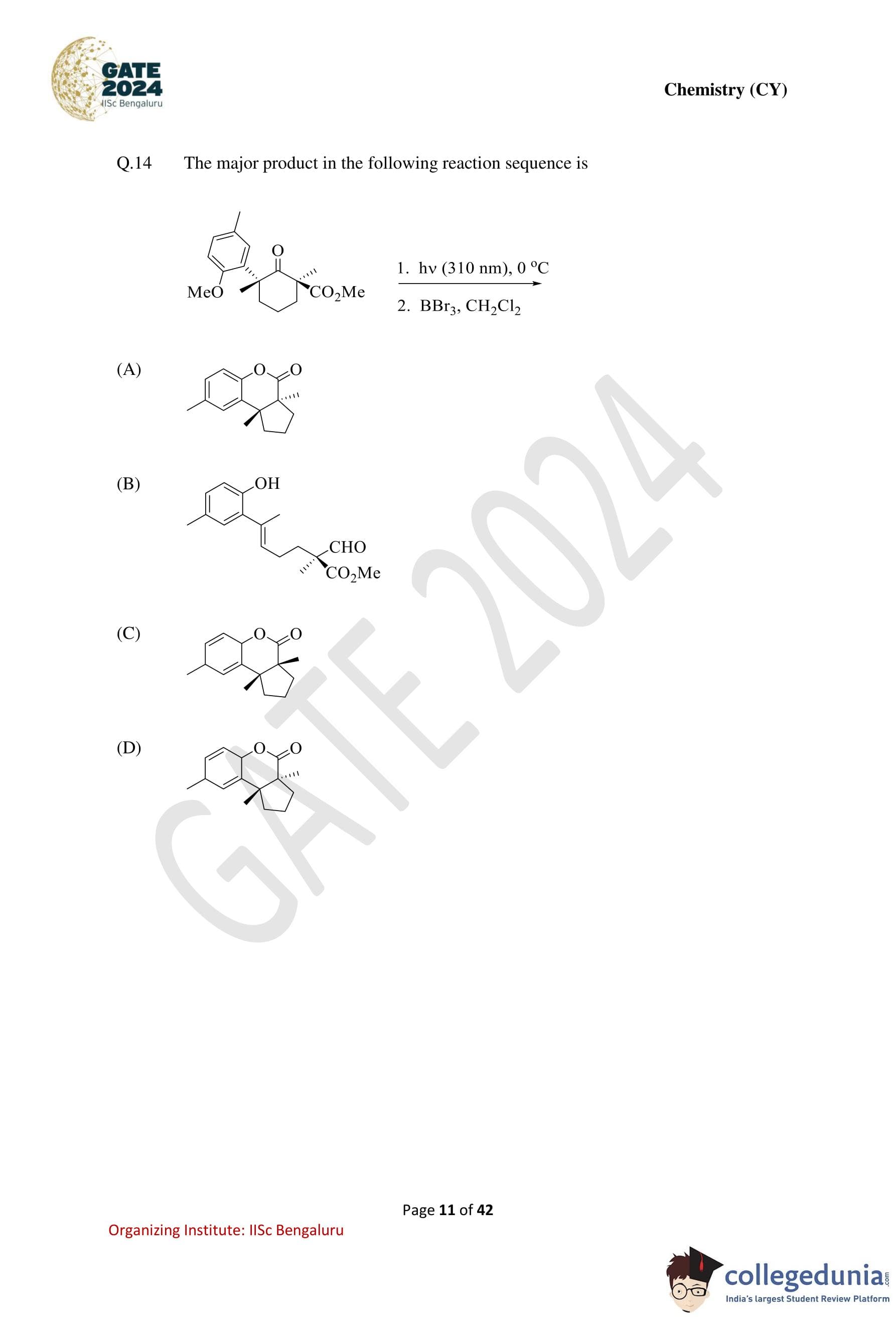
The major product in the following reaction sequence is:
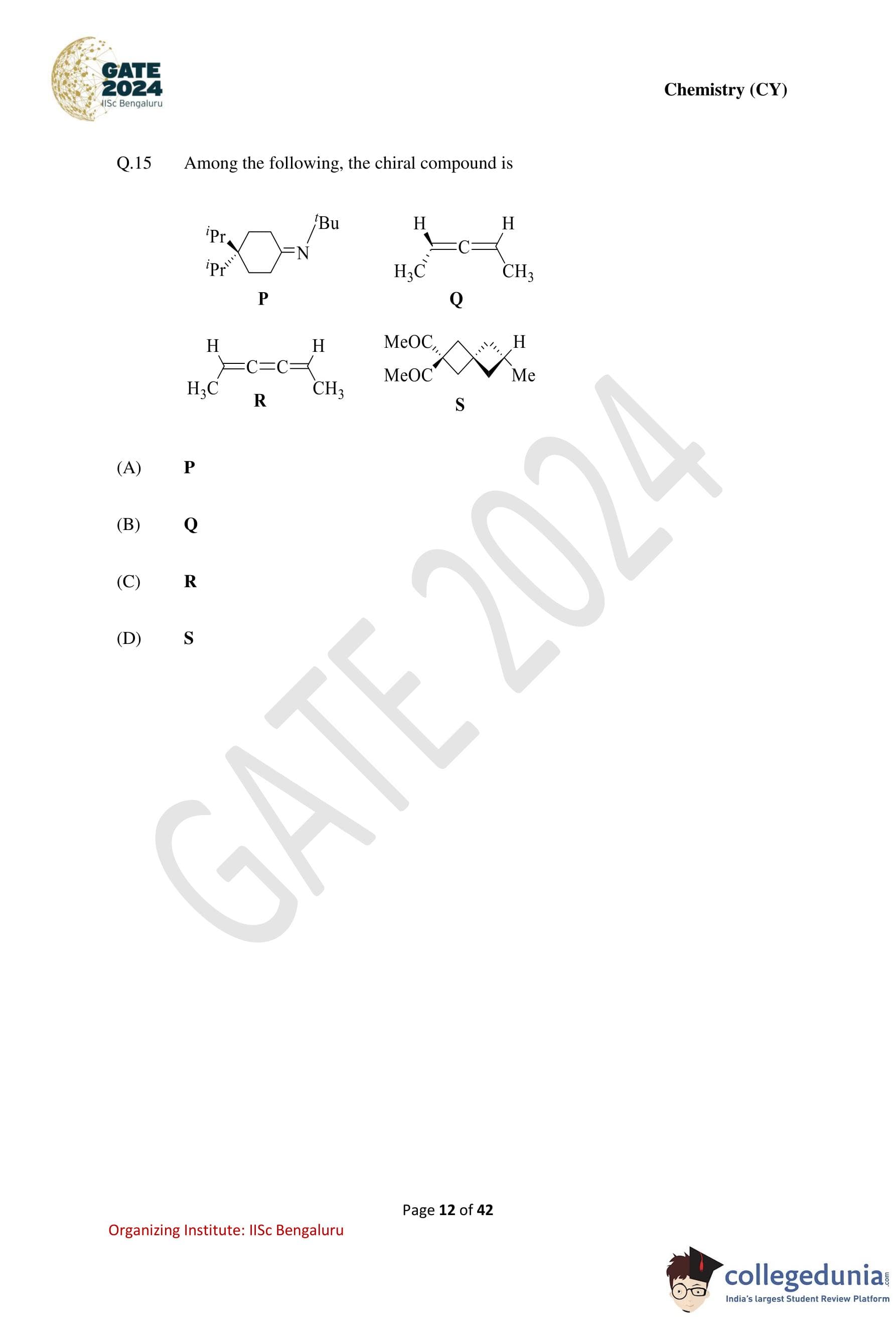
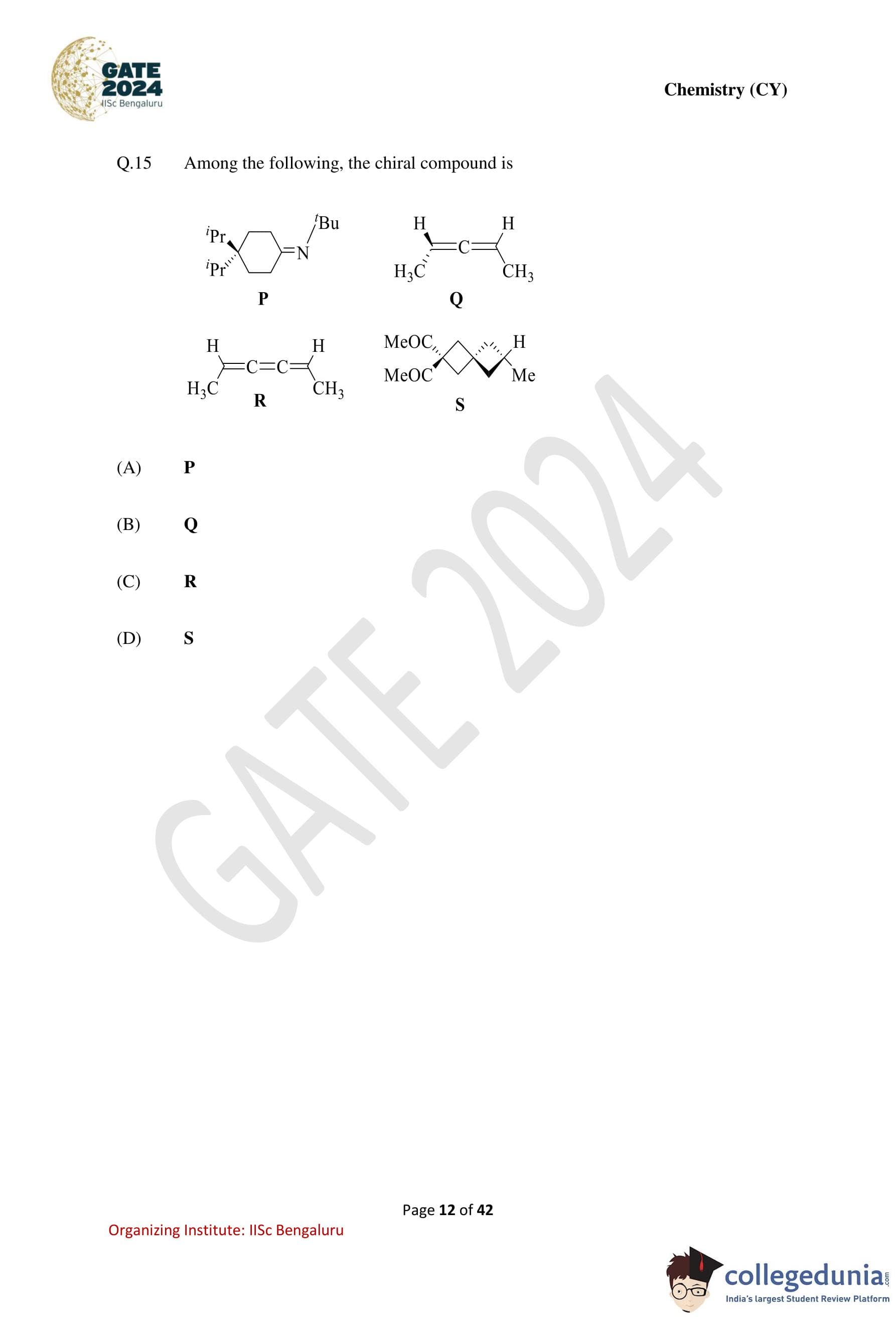
Among the following, the chiral compound is:
The major product in the given reaction sequence is Q. The mass spectrum of Q shows
([M] = molecular ion peak)
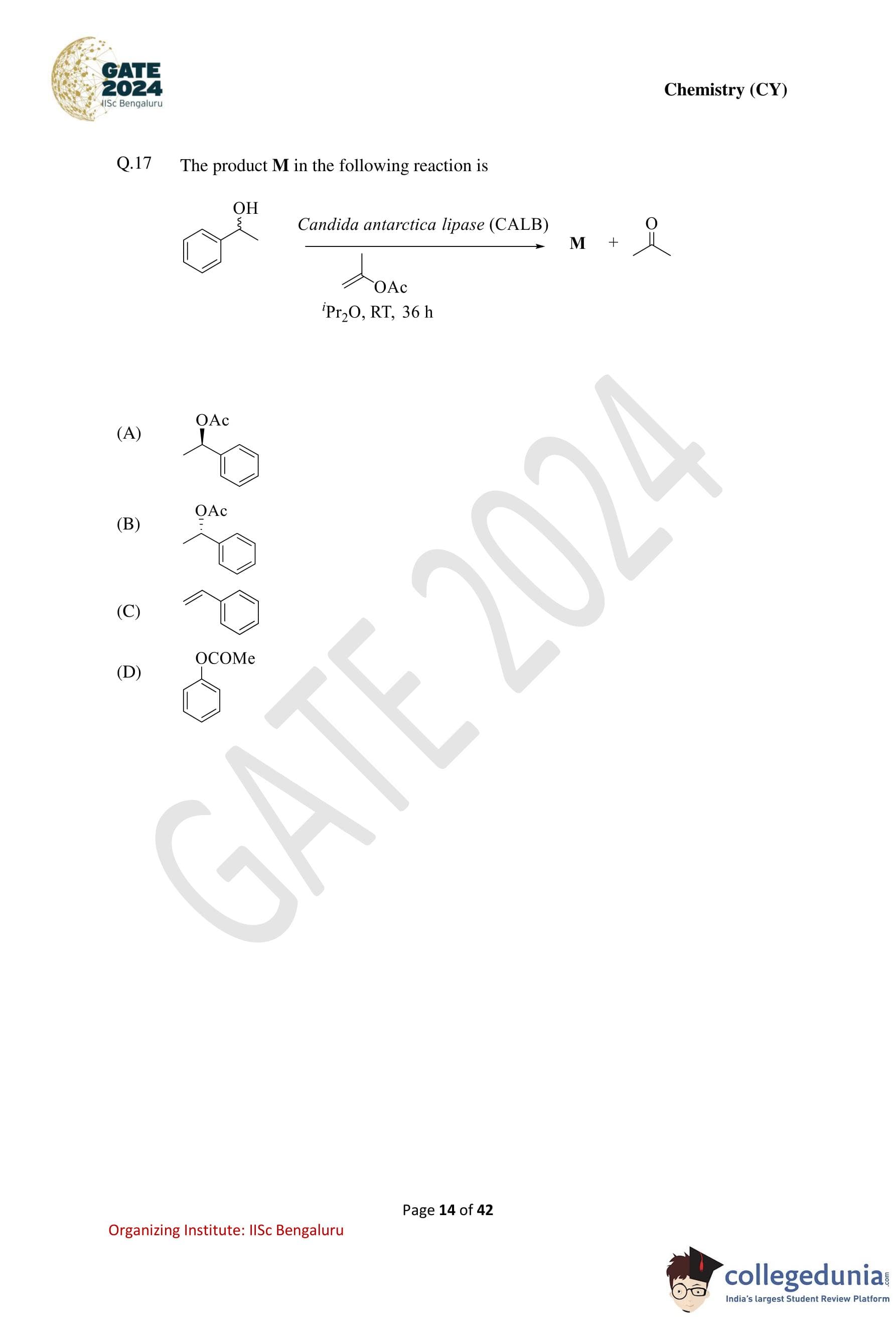
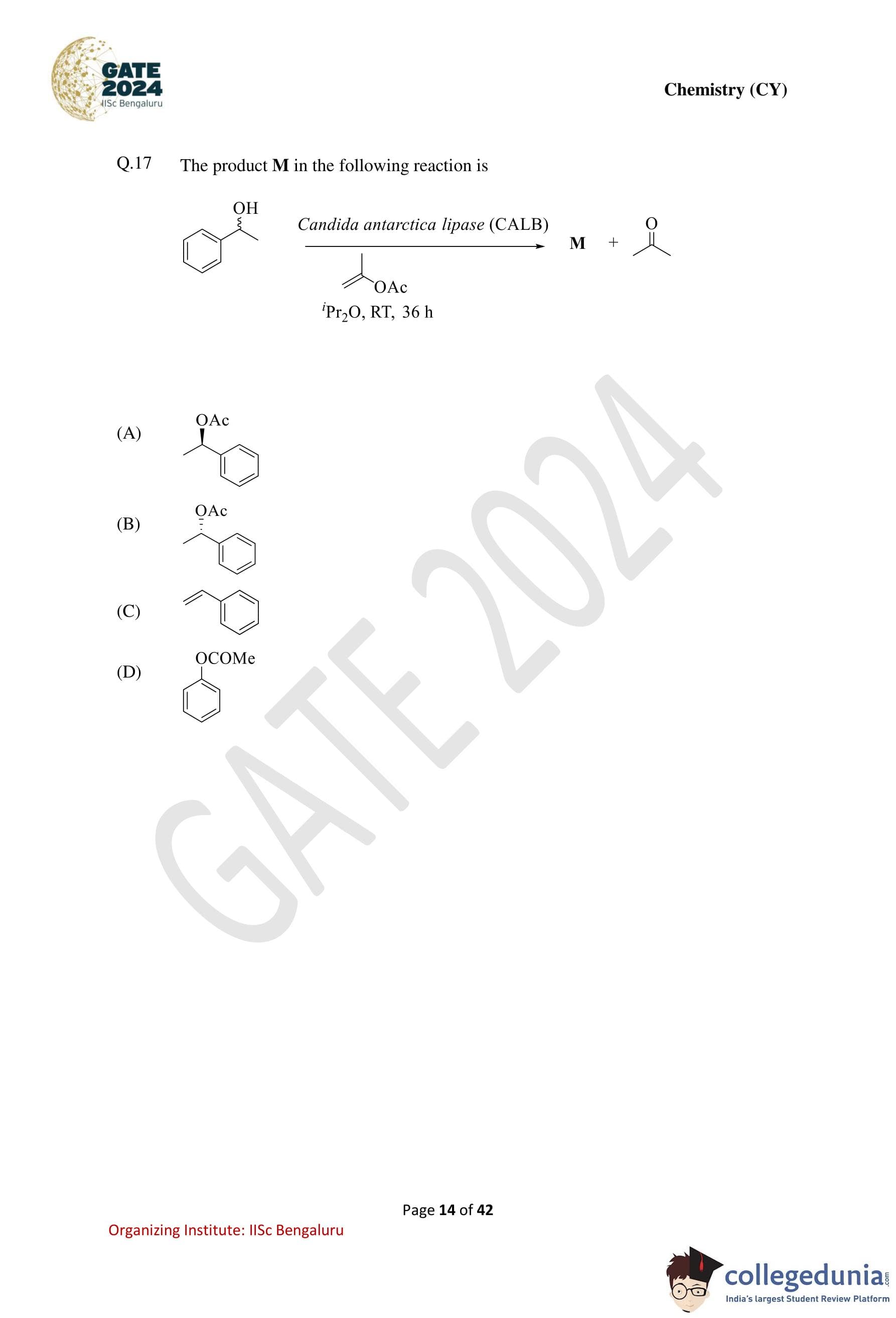
The product M in the following reaction is:
Critical micellar concentration of a surfactant is 0.008 M in water at 25 \(^\circ\)C. If the aggregation number of the micelles is 80, the concentration of the micelles (in M) present in 0.088 M aqueous solution of the surfactant at 25 \(^\circ\)C is:
The order and the number of classes present in a group with the irreducible representations \( A_1, A_2, B_1, B_2, E_1, \) and \( E_2 \), are respectively:
The molecule XY\(_2\) is microwave active and its vibration-rotation spectrum shows only P and R transitions. In the correct structure,
The complex(es) with distorted octahedral structure is (are):
The compound(s) which show(s) the perovskite structure in solid state is (are):
Among the following metalloproteins, the pair(s) of non-heme proteins is (are):
The reaction(s) that yield(s) X as the major product is (are):
\[ X = \mathrm{Ph{-}CH_2{-}CH_2{-}CO_2Me} \]
The reaction(s) that yield(s) 2-methylquinoline as the major product is (are):
The correct statement(s) for decalin is (are):
The correct statement(s) about \(^{4}D_{5/2}\) state of an atom is (are):
The correct statement(s) related to an ensemble is (are):
The non-dissociative adsorption of a gas on a given surface at a fixed temperature follows Langmuir isotherm. The plot(s) which give(s) a straight line is (are)
[Given: \( V \) = volume of the adsorbed gas, \( P \) = pressure of the gas]
The crystal field stabilization energy of \(\left[\mathrm{Cr(NH_3)}_6\right]^{3+}\) with \(\Delta_o\) value of 21600 cm\(^{-1}\) is \(y\) cm\(^{-1}\). The value of \(|y|\) is .......................
(rounded off to the nearest integer)
The number of metal–metal bond(s) in the complex \([(\eta^5-Cp)\mathrm{Mo(CO)}_2]_2\) is \(x\) and in \([(\eta^5-Cp)_2\mathrm{Fe}_2(\mathrm{CO})_3]\) is \(y\). The value of \(x+y\) is .......................
(Assume 18 electron rule is followed)
\(^1\)H NMR spectrum of a mixture containing CH\(_3\)Br (\(x\) mol) and (CH\(_3\))\(_3\)CBr (\(y\) mol) shows two singlets at 2.7 ppm and 1.8 ppm, with relative ratio of 3:1 (integration value), respectively. The value of \(x/y\) is .......................
(rounded off to the nearest integer)
The value of \(\dfrac{e^2}{2\pi \varepsilon_0 a_0}\) in atomic unit of energy is .......................
(rounded off to the nearest integer)
The partial vapor pressure of 0.1 molal solution of B in liquid A is 60 kPa at 300 K. The partial vapor pressure (in kPa) of a solution containing B with mole fraction of 0.1 in liquid A at 300 K is .......................
(Assume the solute obeys Henry’s law. The molar mass of A is 80 g mol\(^{-1}\).)
(rounded off to three decimal places)
Consider the following two parallel irreversible first-order reactions, where \(k_1 = 2k_2\) at 300 K. After complete conversion of R at 300 K, the concentration of P1 in the reaction mixture was 15 mol L\(^{-1}\). The initial concentration of R (in mol L\(^{-1}\)) was .......................
(rounded off to one decimal place)
Borax on treatment with NaOH and \( \mathrm{H_2O_2} \) forms X. The compound X on reaction with PhCN at \(60^\circ\mathrm{C}\) in methanol–water mixture gives Y as the major product. X and Y, respectively, are
In the EPR spectrum of an aqueous solution of \( \mathrm{VOSO_4} \) at room temperature, the total number of hyperfine splitting signals is
The hapticity of allyl and Cp and the ligation mode of NO in the thermodynamically stable complexes \([(\eta^x-allyl)\mathrm{Ru(CO)_2(NO)}]\) and \([(\eta^y-Cp)\mathrm{Ru(CO)_2(NO)}]\), respectively, are
(The hapticity of allyl and Cp are denoted by \(\eta^x\) and \(\eta^y\), respectively.)
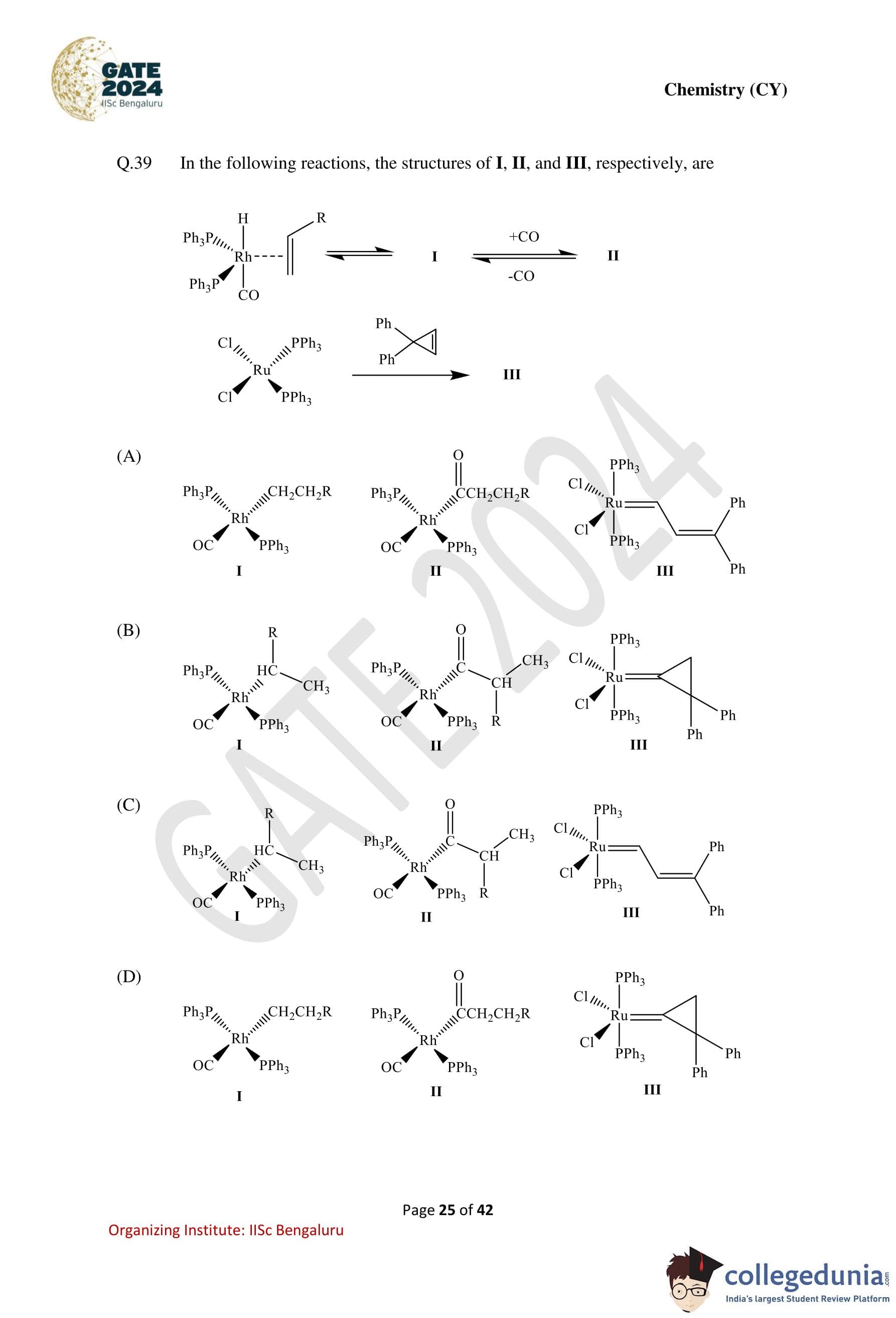
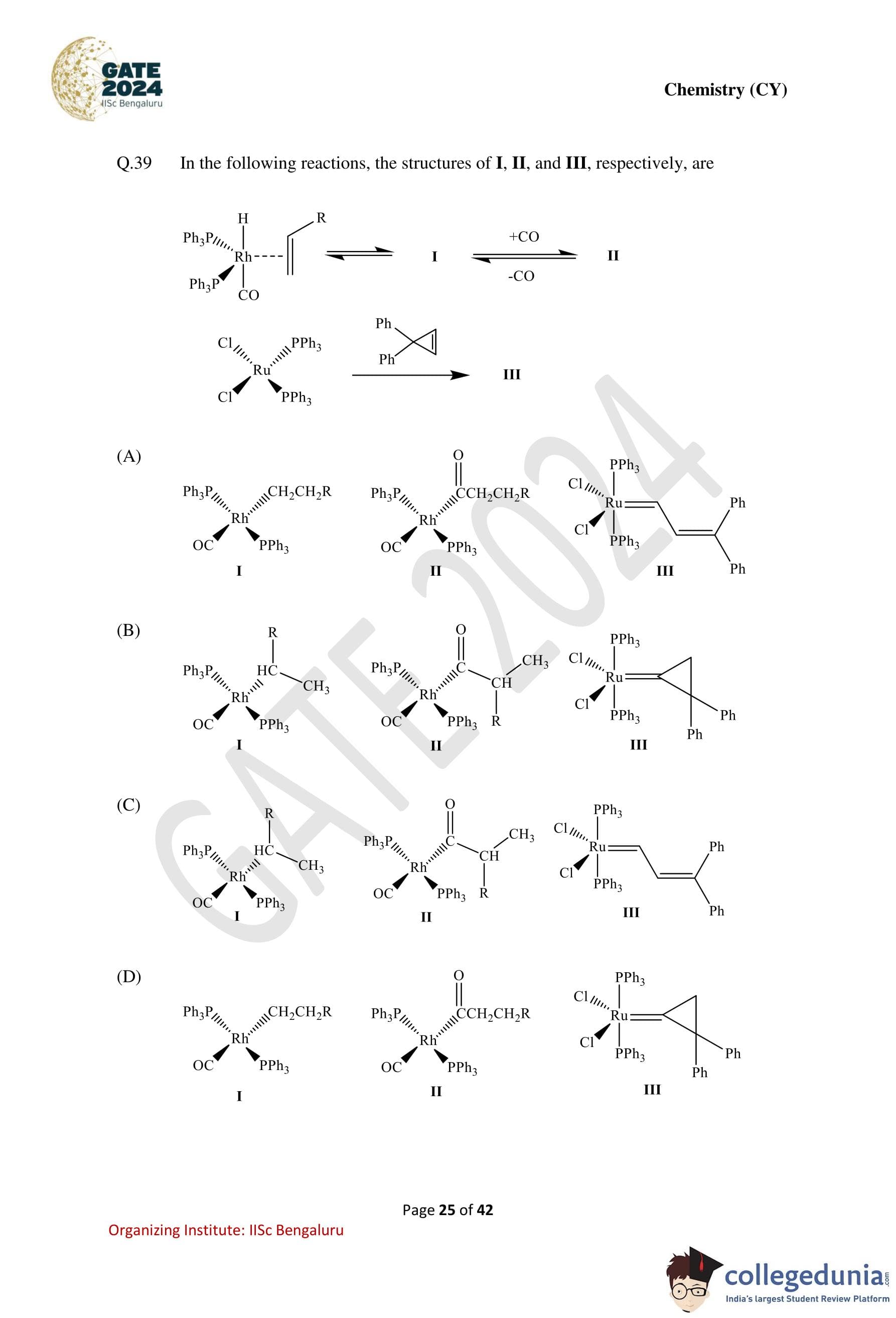
In the following reactions, the structures of I, II, and III, respectively, are
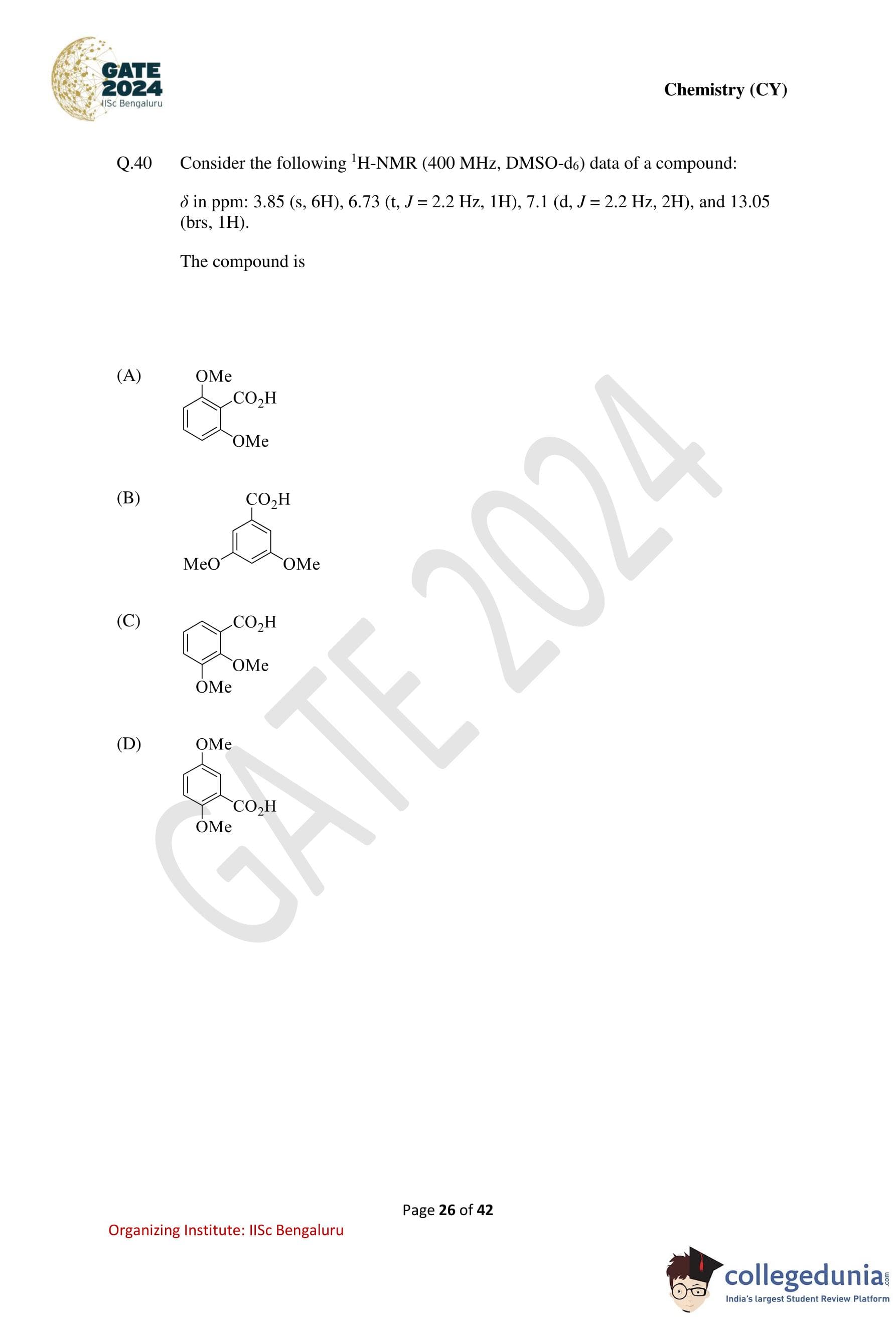
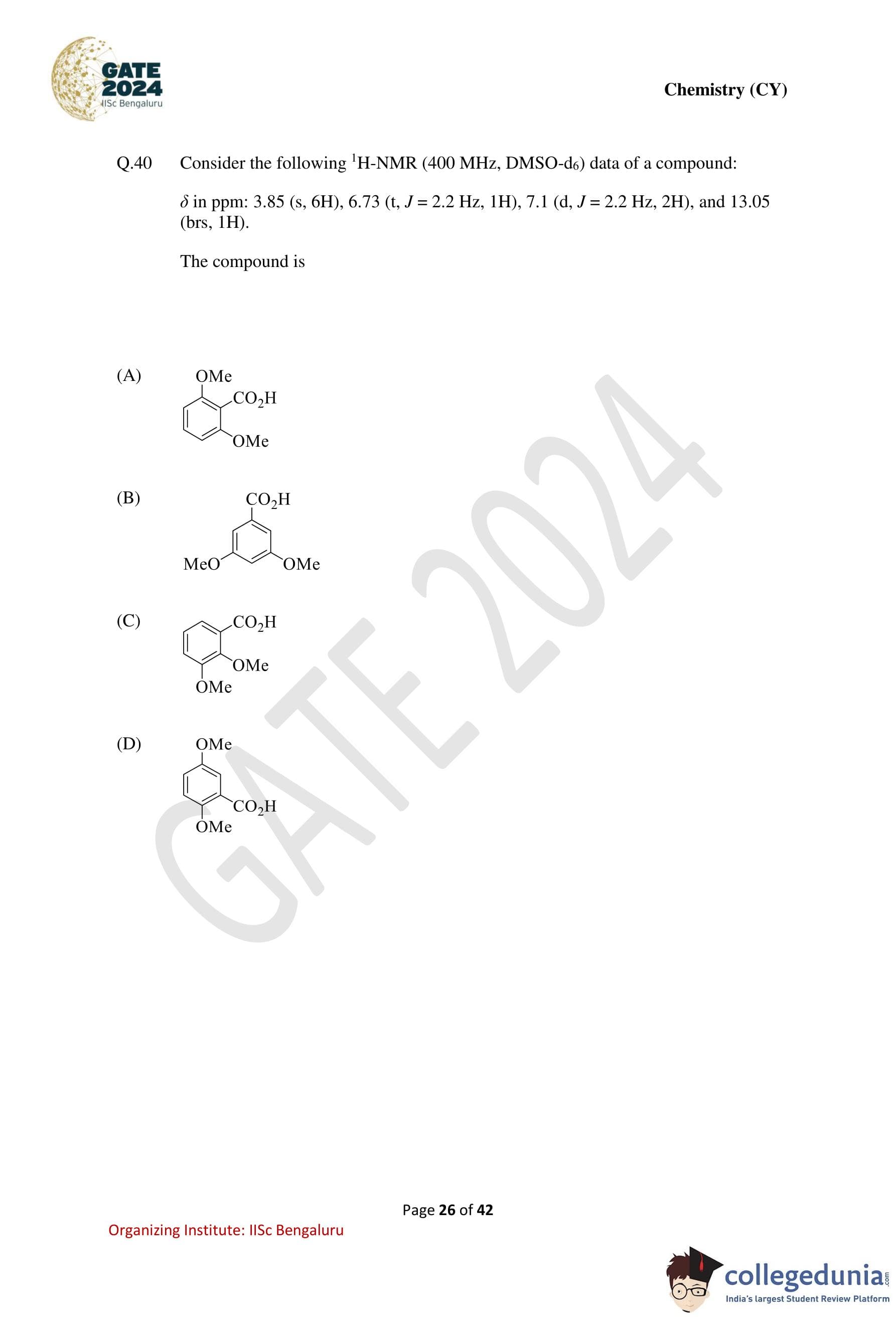
Consider the following \(^1\)H-NMR (400 MHz, DMSO-\(d_6\)) data of a compound:
\[ \delta (ppm): 3.85\ (s, 6H),\ 6.73\ (t,\ J = 2.2\ Hz,\ 1H),\ 7.1\ (d,\ J = 2.2\ Hz,\ 2H),\ 13.05\ (br\,s,\ 1H) \]
The compound is
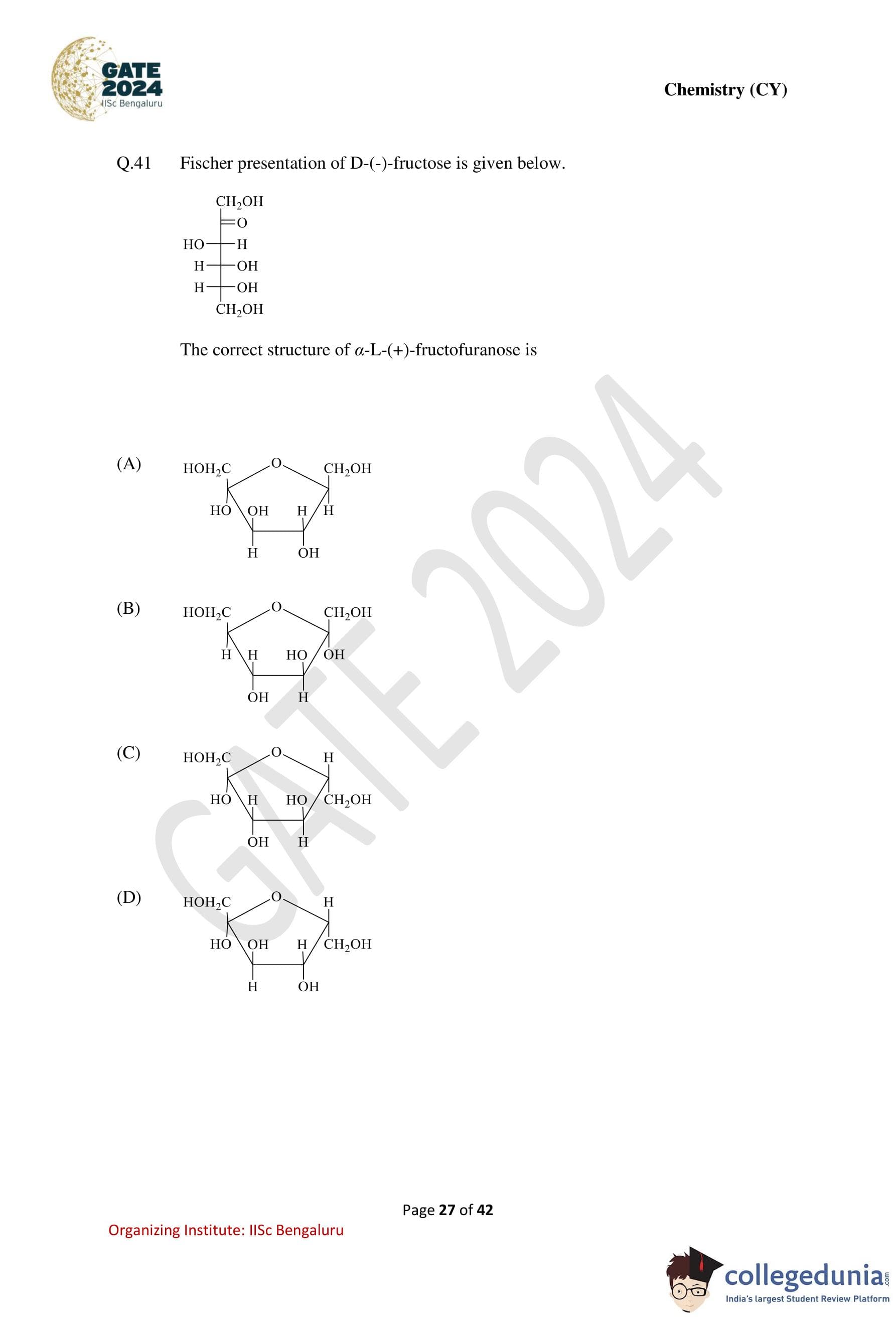
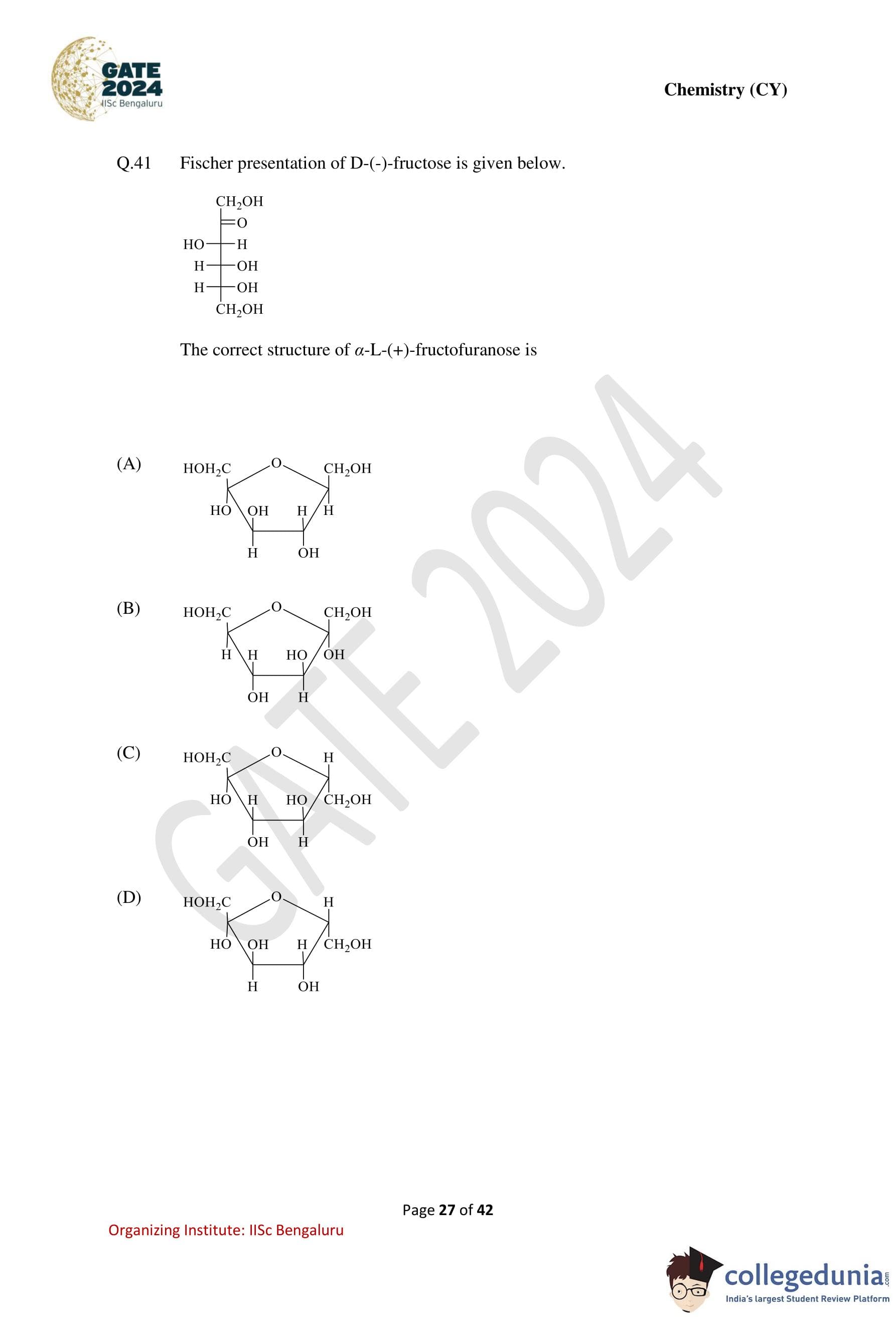
Fischer presentation of D-(-)-fructose is given below.
The correct structure of \(\alpha\)-L-(+)-fructofuranose is
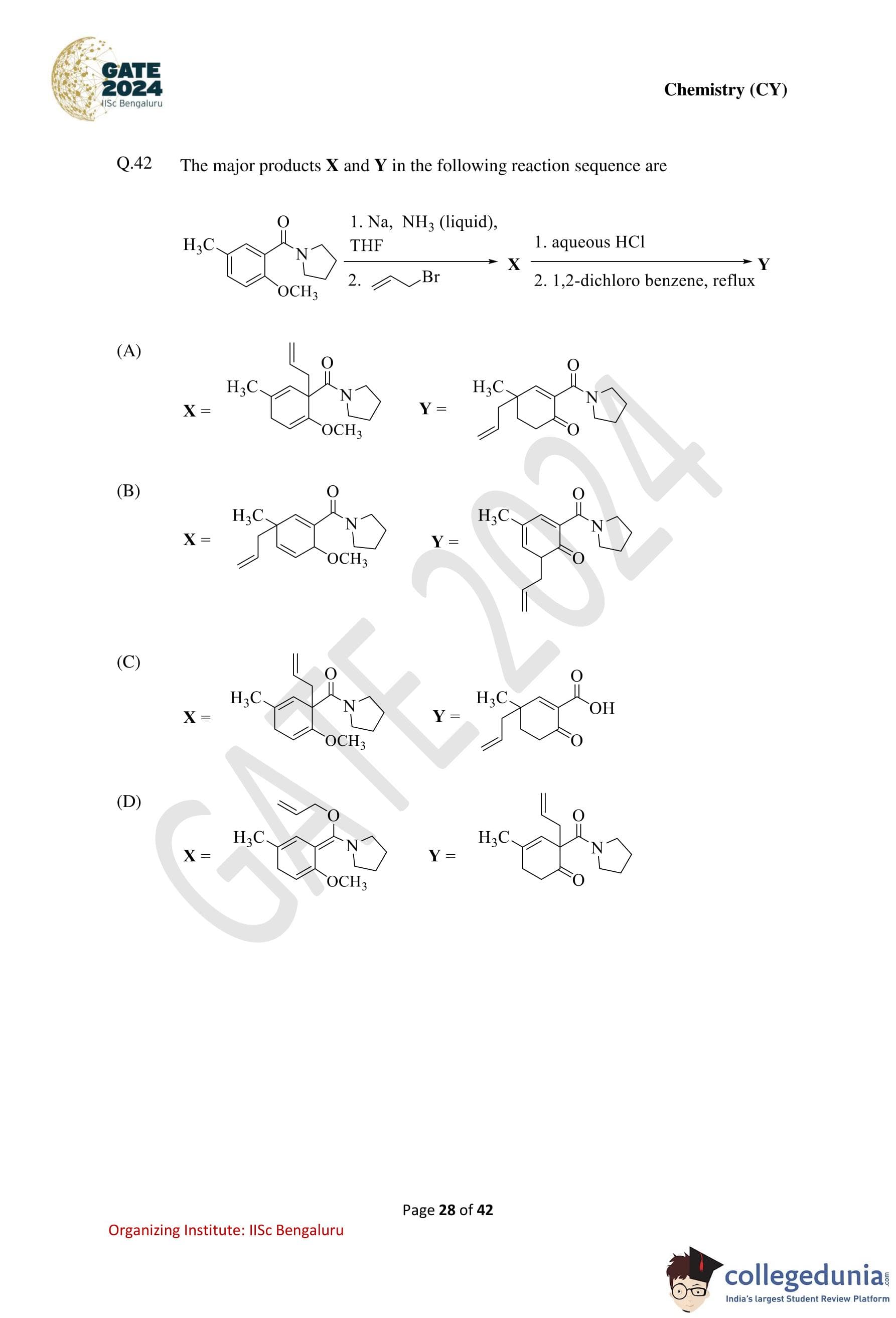
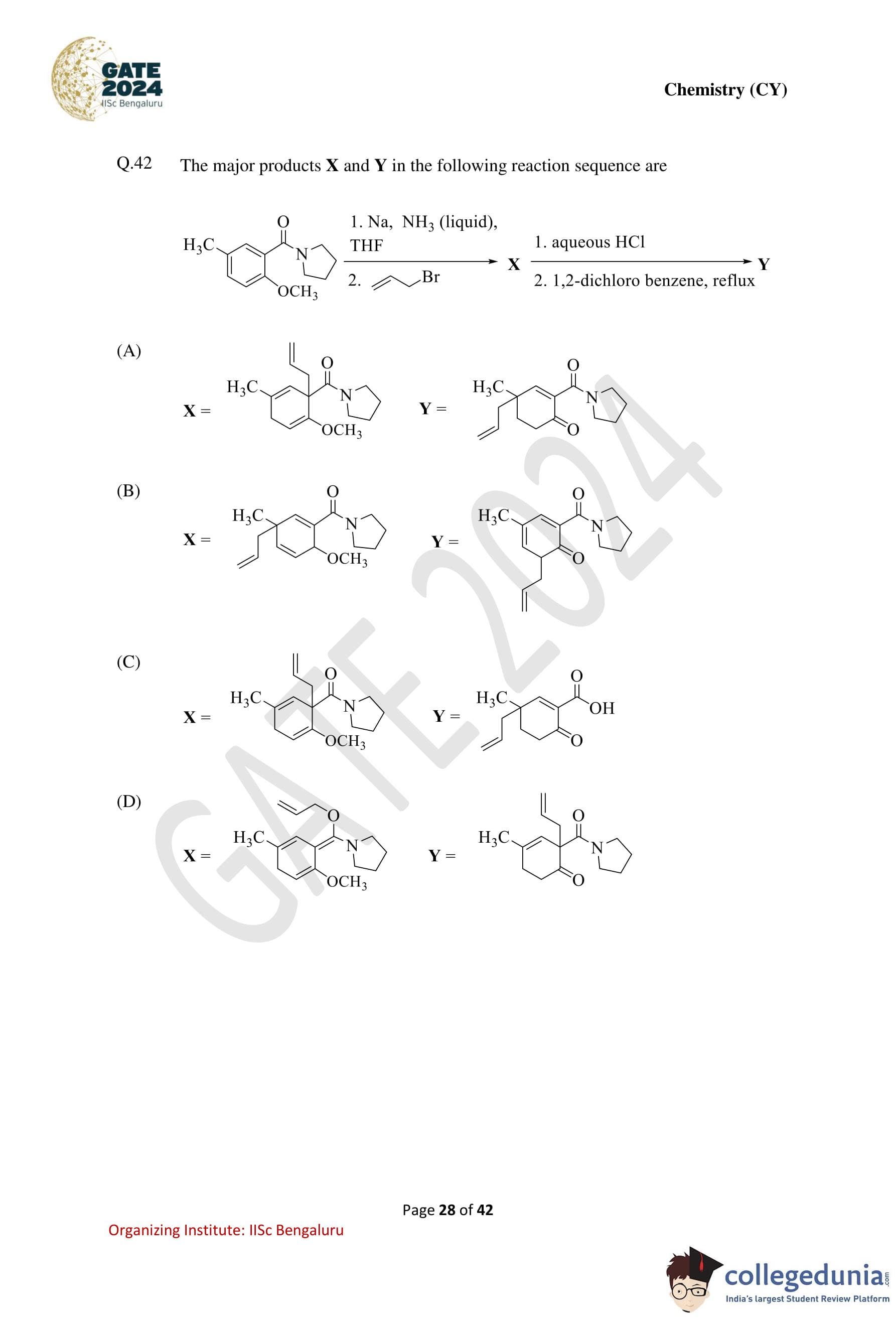
The major products X and Y in the following reaction sequence are
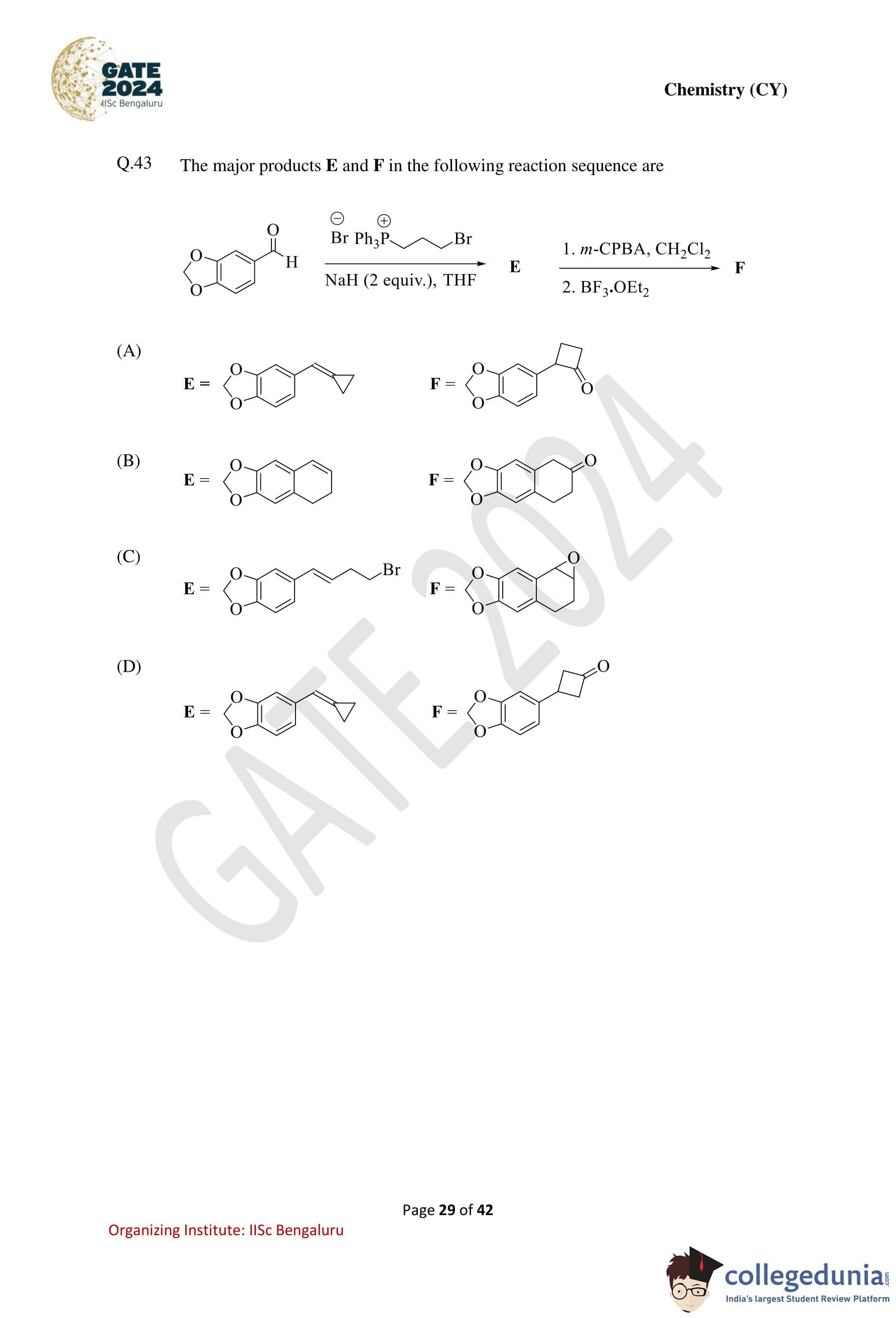
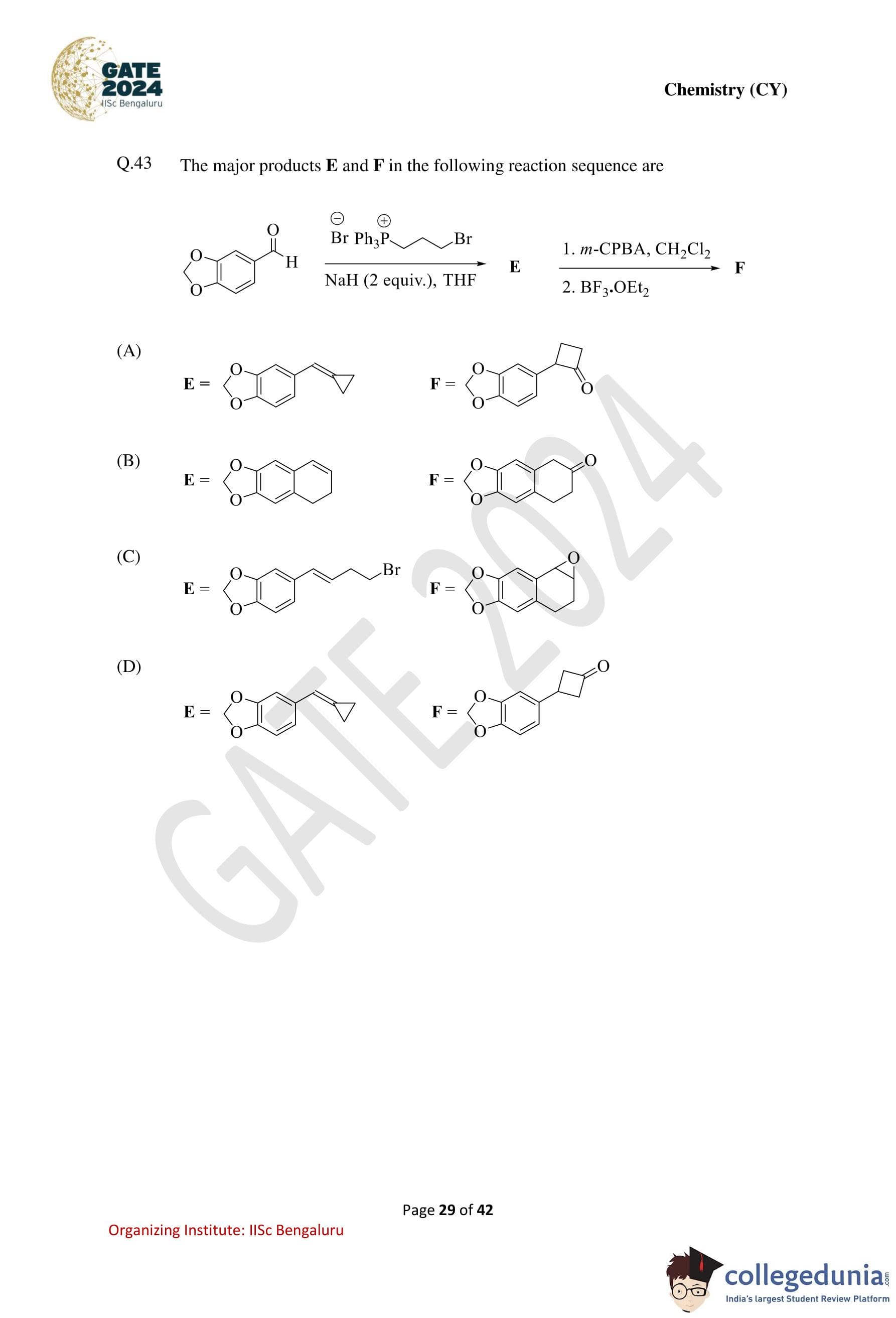
The major products E and F in the following reaction sequence are
\(\psi_1, \psi_2, \psi_3,\) and \(\psi_4\) are four Hückel molecular orbitals of benzene with orbital energies \(E_1, E_2, E_3,\) and \(E_4\), respectively,
\[ \psi_1 = \frac{1}{2}(\phi_B + \phi_C - \phi_E - \phi_F) \]
\[ \psi_2 = \frac{1}{6^{1/2}}(\phi_A - \phi_B + \phi_C - \phi_D + \phi_E - \phi_F) \]
\[ \psi_3 = \frac{1}{6^{1/2}}(\phi_A + \phi_B + \phi_C + \phi_D + \phi_E + \phi_F) \]
\[ \psi_4 = \frac{1}{12^{1/2}}(2\phi_A + \phi_B - \phi_C - 2\phi_D - \phi_E + \phi_F) \]
The correct order of the orbital energies is
(The six carbon atoms of benzene are denoted by A to F and \(\phi_j\) is the 2\(p_z\) orbital of the \(j^{th}\) carbon of benzene.)
Consider the following six vibrational modes:
symmetric stretching of CO\(_2\)
O–H symmetric stretching of H\(_2\)O
stretching of HCl
stretching of H\(_2\)
N–H symmetric stretching of NH\(_3\)
bending of CO\(_2\)
Among these modes, if \(k\) number of modes are IR active but Raman inactive, \(l\) number of modes are IR inactive but Raman active, and \(m\) number of modes are both IR and Raman active, then \(k\), \(l\), and \(m\), respectively, are
The correct statement for a thermally initiated radical polymerization in a solution is
(Assume: steady-state and equal reactivity of the propagating radicals, termination reactions are only by combination, and no chain transfer reaction.)
[Given: \(R_p\) = rate of polymerization, DP = degree of polymerization, [I] = initiator concentration, and [M] = monomer concentration.]
If \(q_t\) and \(Q_{t,m}\) are the molecular and molar translational partition functions of X\(_2\), respectively, then \[ \ln(Q_{t,m}) = \]
(N is the Avogadro number)
Among the following, the NMR active nucleus(nuclei) is (are):
The complex(es) that exhibit(s) optical isomerism is (are):
In aqueous solution of K\(_4\)[Fe(CN)\(_6\)], the allowed transition(s) is (are):
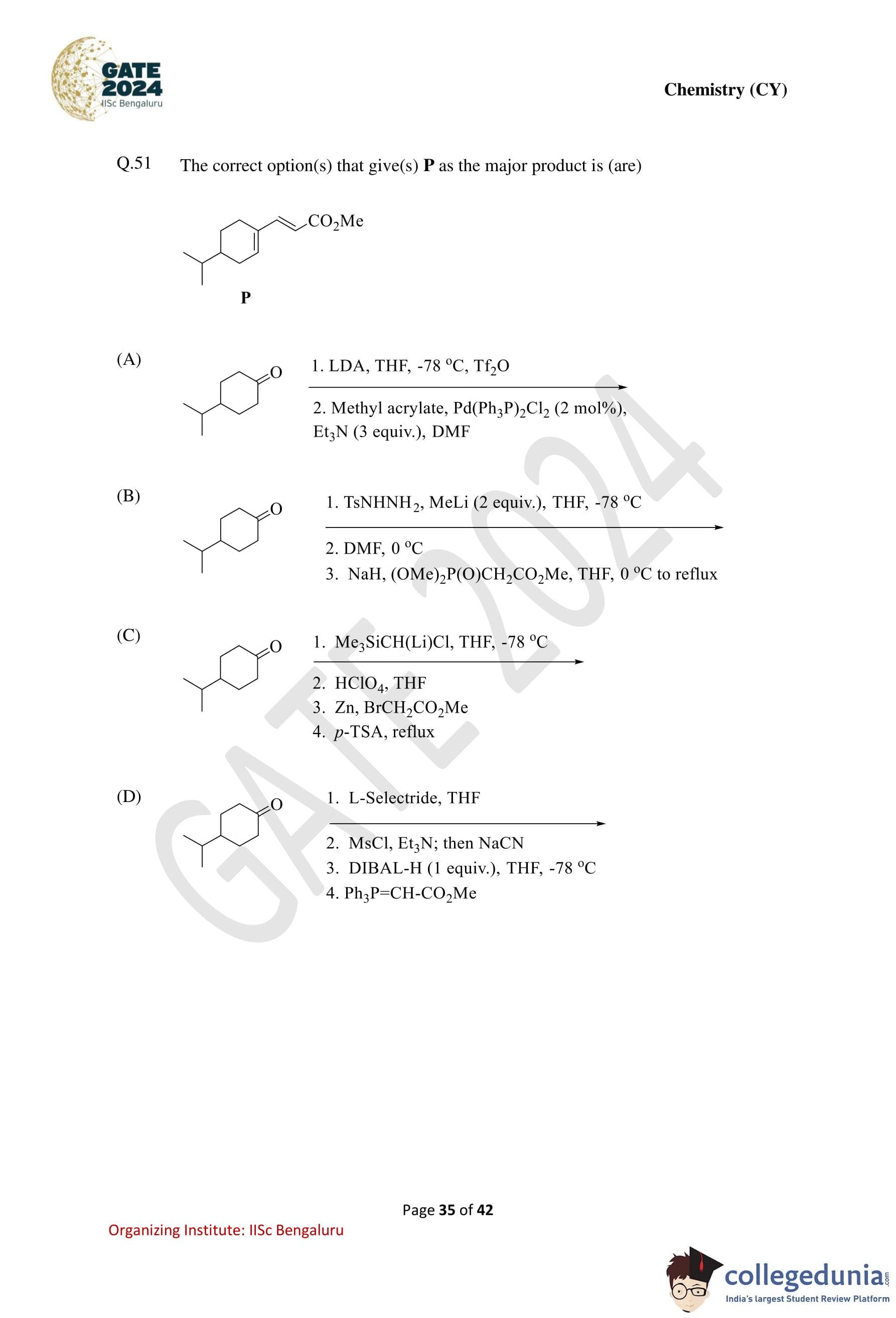
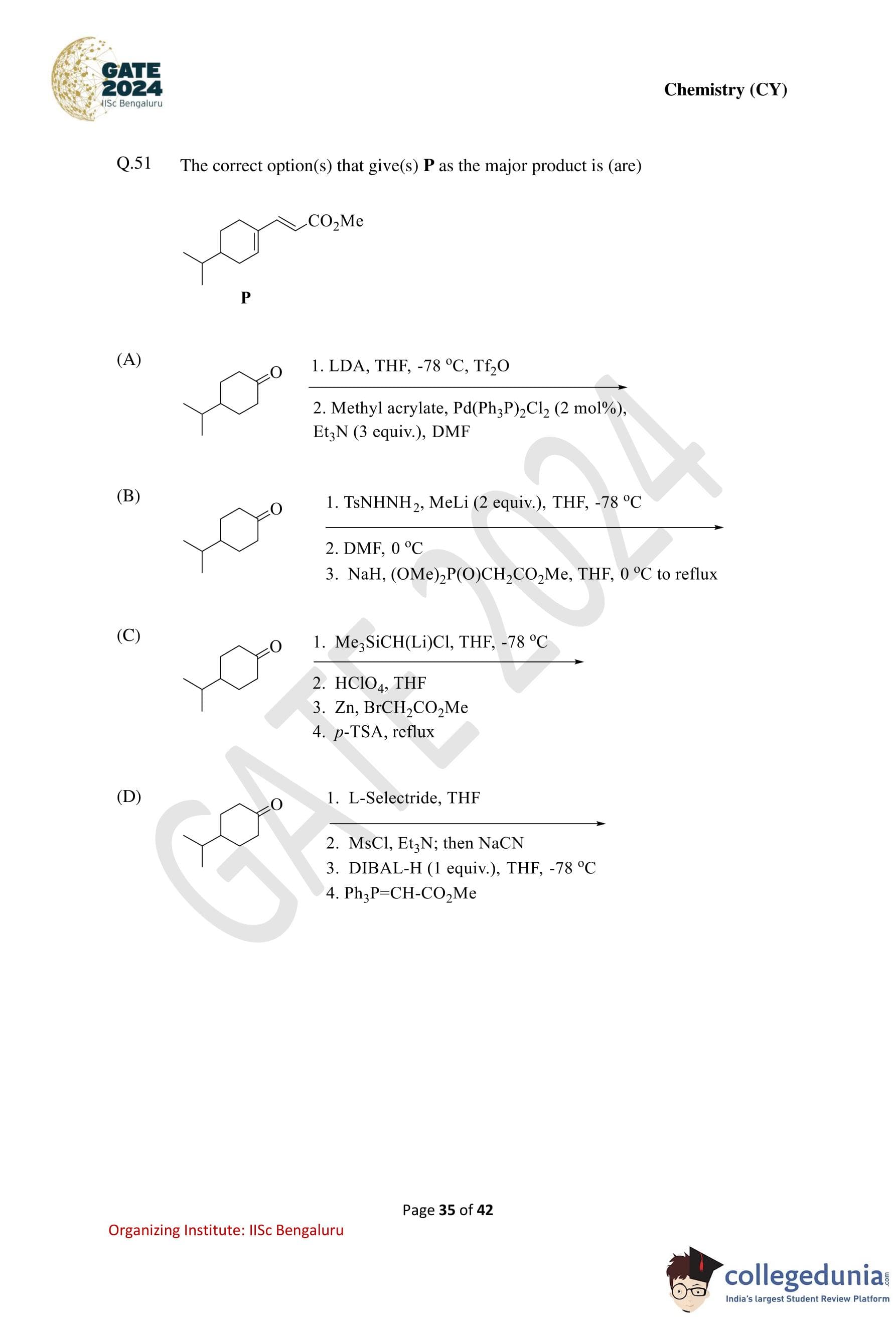
The correct option(s) that give(s) P as the major product is (are):
The correct statement(s) regarding P, Q, R, and S is (are):
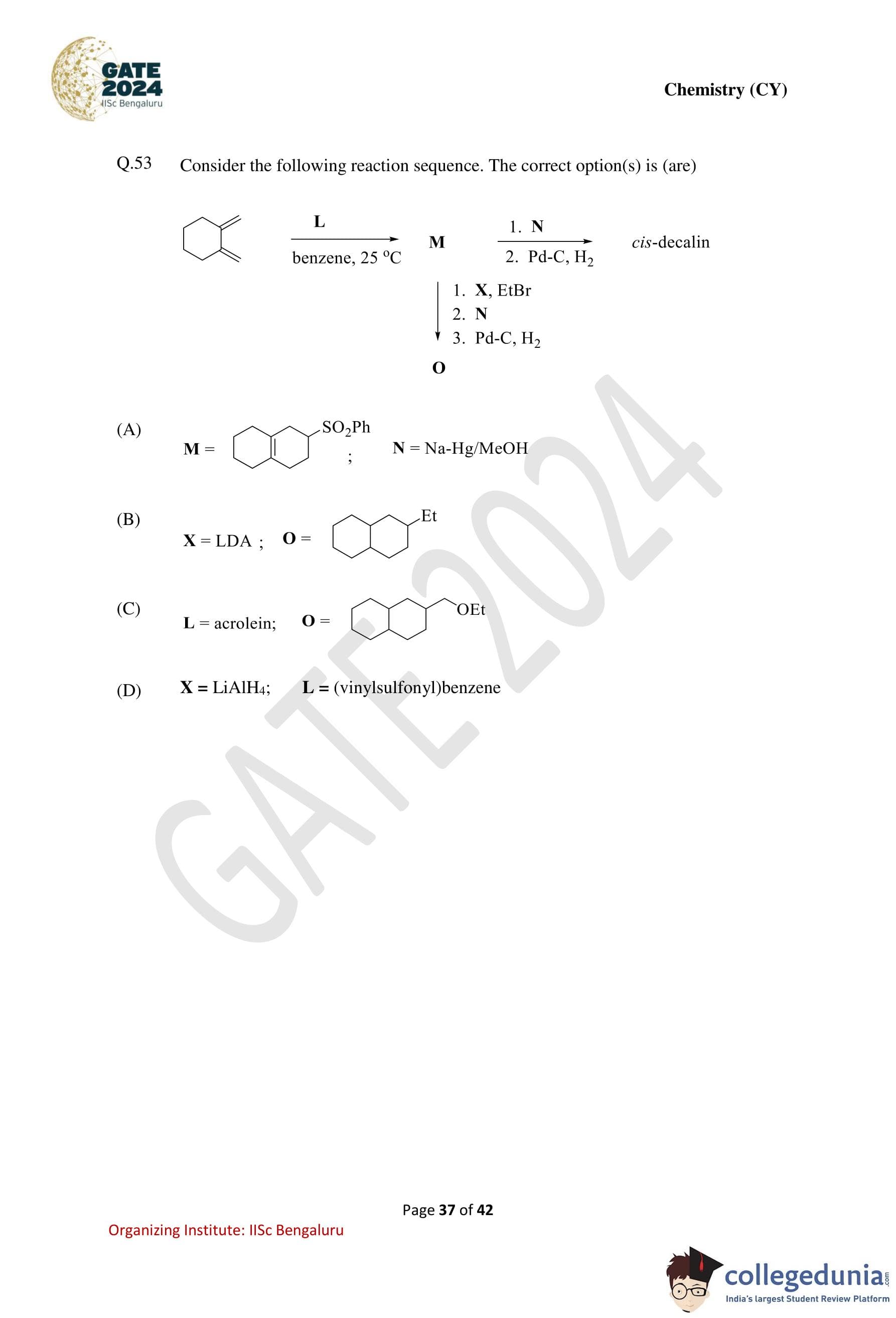
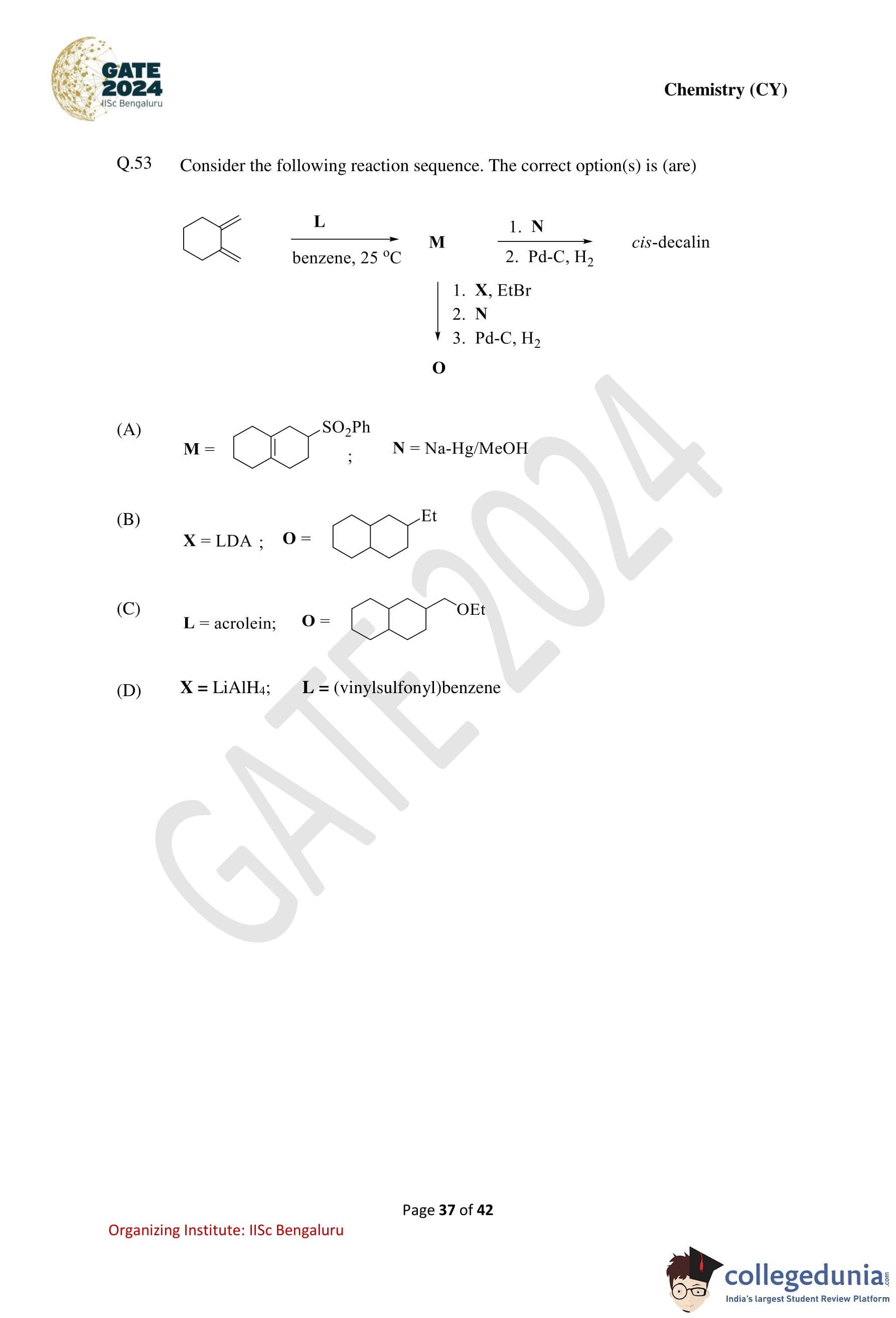
Consider the following reaction sequence. The correct option(s) is (are):
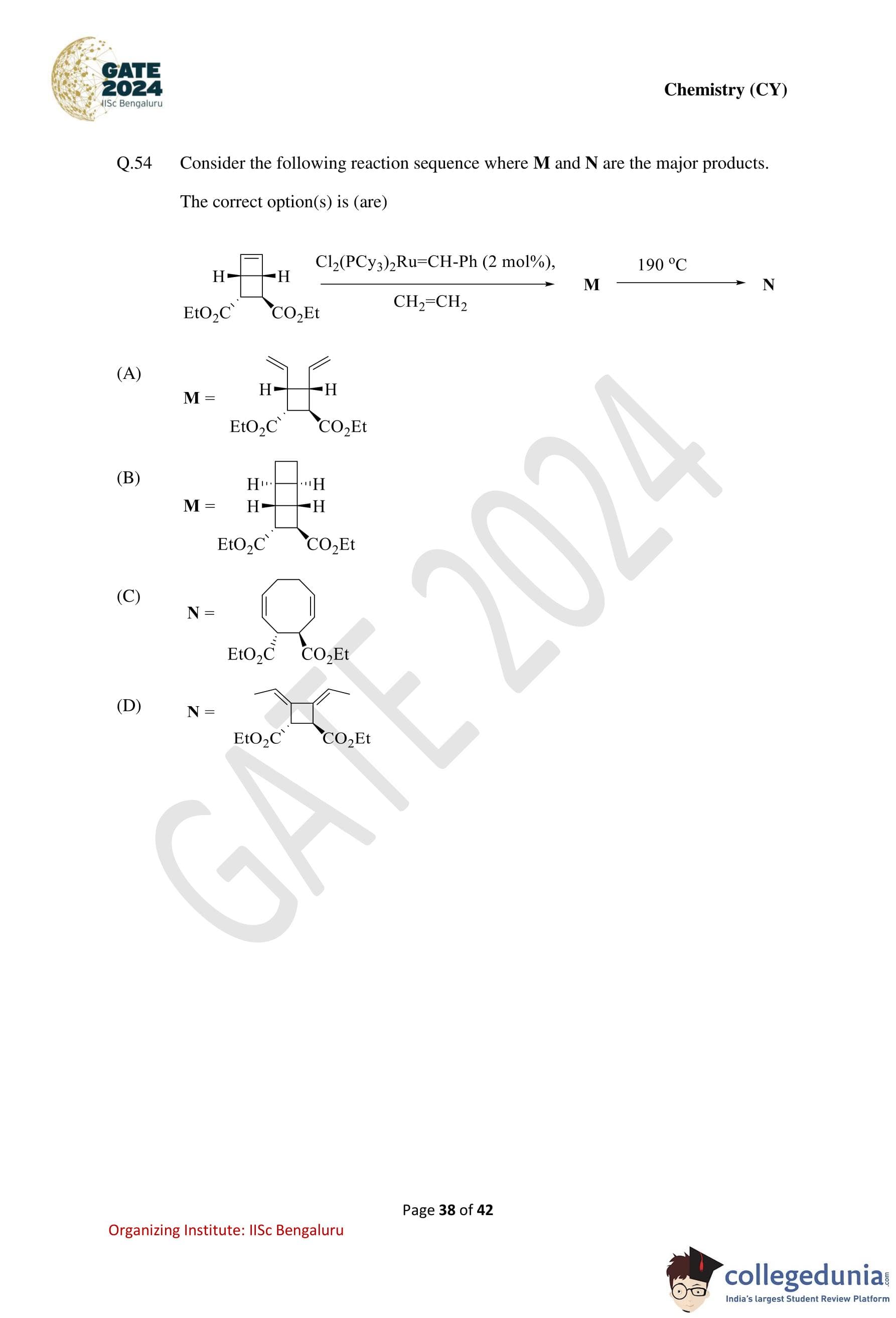
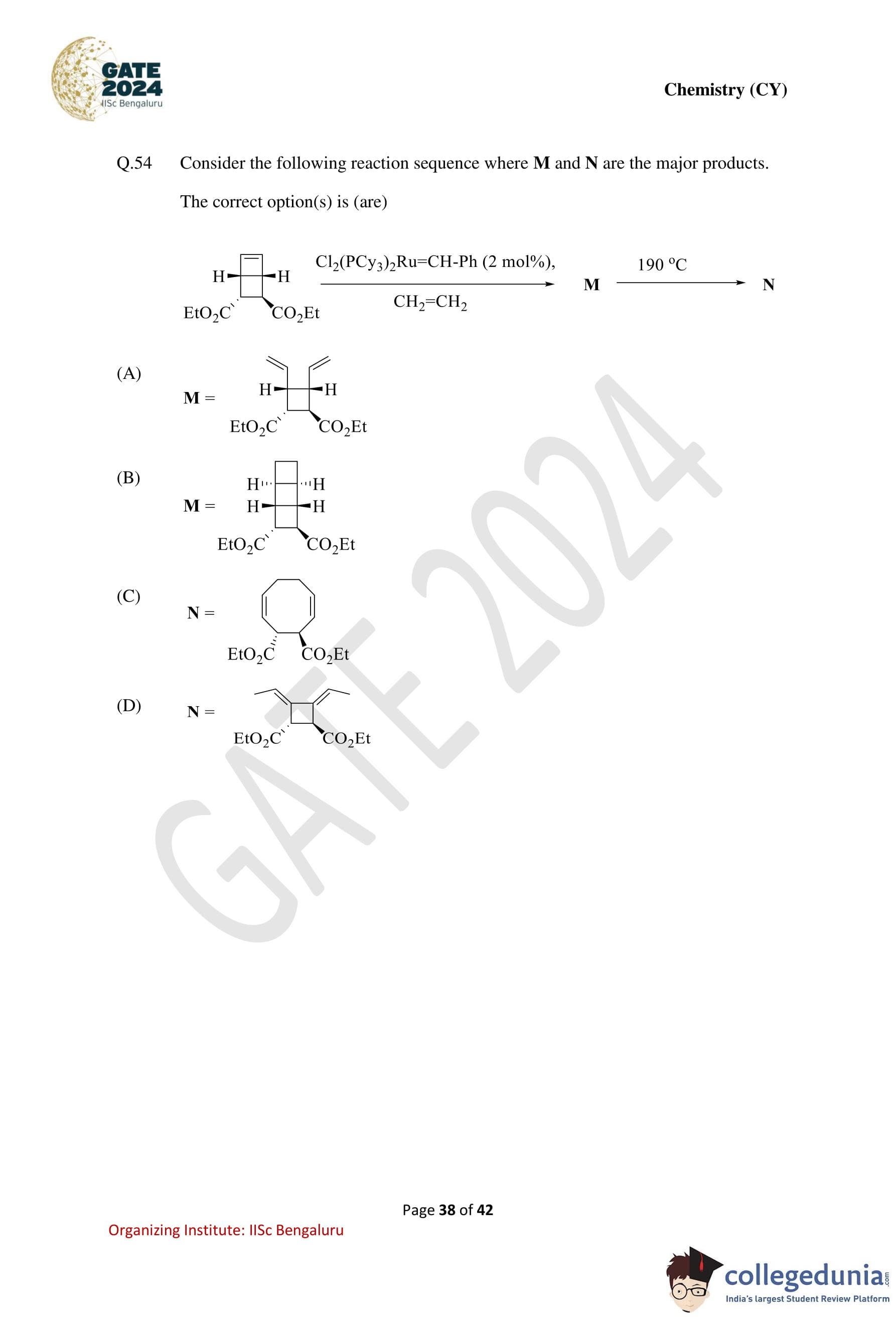
Consider the following reaction sequence where M and N are the major products.
The correct option(s) is (are):
The correct statement(s) about the relationship for the H-atoms in the following compounds is (are):
Among the following, the correct statement(s) is (are):
Among the following, the correct statement(s) is (are):
\(\Delta S^\circ\) (in J mol\(^{-1}\) K\(^{-1}\)) for the given reaction at 298 K is .......................
\[ [\mathrm{Cu(H_2O)_6}]^{2+} + \mathrm{en} \rightleftharpoons [\mathrm{Cu(H_2O)_4(en)}]^{2+} + 2\mathrm{H_2O} \]
(Given: \(\log K_1 = 10.6\), \(\Delta H^\circ = -54\) kJ mol\(^{-1}\), \(R = 8.314\) J mol\(^{-1}\) K\(^{-1}\))
(rounded off to two decimal places)
The turnover frequency (in h\(^{-1}\)) of a reaction where 5 mol% of a catalyst is required for 90% conversion in 3 h is .......................
(rounded off to the nearest integer)
In thermogravimetric analysis, 12.45 mg of CuSO\(_4\cdot\)5H\(_2\)O was subjected to heating under N\(_2\) atmosphere. At a particular temperature, there was a weight loss of 3.6 mg. The number of water molecule(s) lost per formula unit is .......................
(rounded off to the nearest integer)
In the given reaction sequence, the amount of R produced (in g) is .......................
\[ Benzene (7.8 g) \xrightarrow[\;200^\circC\;]{oleum (excess), 80%} P \xrightarrow[\;75%\;]{NaOH, heat then H\(_3\)O\(^+\)} Q \xrightarrow[\;50%\;]{HNO\(_3\) (excess)/H\(_2\)SO\(_4\) (excess)} R \]
(rounded off to two decimal places)
The wave function of a particle in a cubic box (of side \(L\)) is given by
\[ \psi(x,y,z) = \sqrt{\frac{32}{L^3}} \sin\frac{\pi x}{L}\, \cos\frac{\pi y}{L}\, \sin\frac{2\pi y}{L}\, \sin\frac{\pi z}{L} \]
The ratio of the energy of the state corresponding to the above wave function to the ground state energy is .......................
(rounded off to the nearest integer)
\(\phi_1\) and \(\phi_2\) are normalized eigenfunctions of a Hermitian operator.
\[ |\psi\rangle = 3i|\phi_1\rangle + 2|\phi_2\rangle, \quad |\chi\rangle = -2i|\phi_1\rangle + 5|\phi_2\rangle \]
The value of \(\langle \psi | \chi \rangle + \langle \chi | \psi \rangle\) is .......................
(rounded off to the nearest integer)
2 mol of a monoatomic ideal gas with initial volume of 5 L and pressure 10 bar undergoes an irreversible adiabatic expansion against a constant final pressure of 1 bar. The final volume (in L) is .......................
(Given: \(R = 8.314 \times 10^{-2}\) L bar mol\(^{-1}\) K\(^{-1}\))
(rounded off to one decimal place)
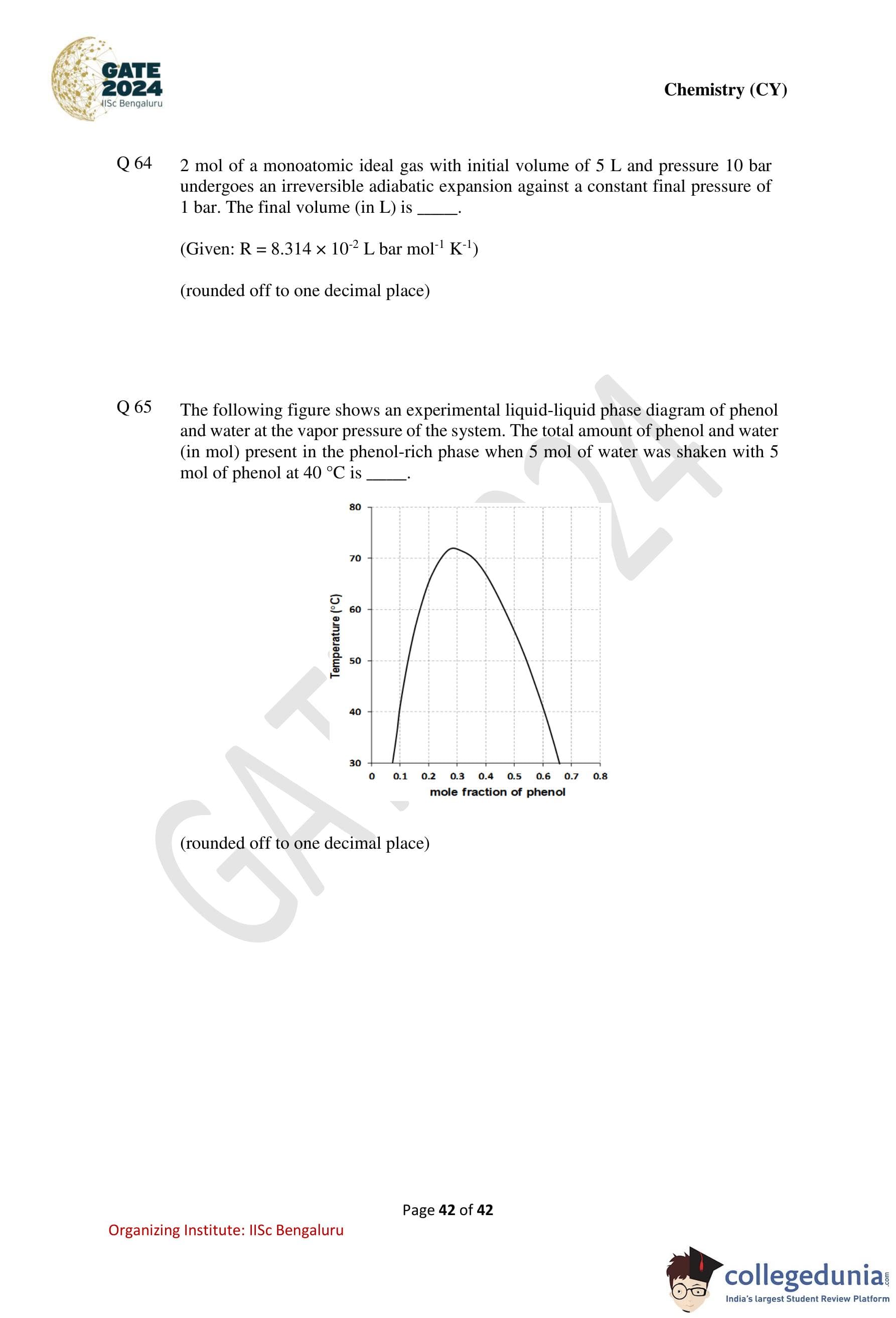
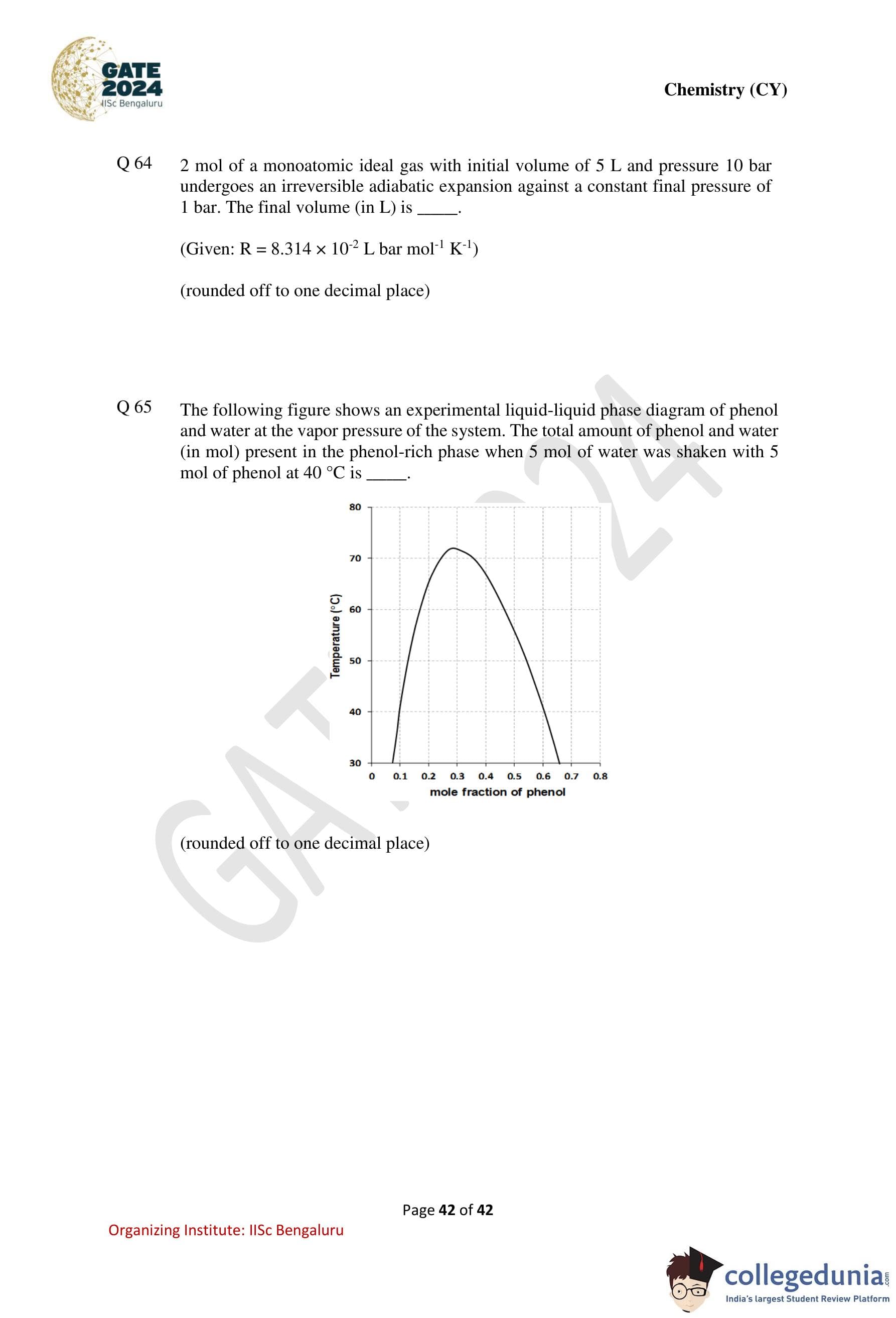
The following figure shows an experimental liquid–liquid phase diagram of phenol and water at the vapor pressure of the system.
The total amount of phenol and water (in mol) present in the phenol-rich phase when 5 mol of water was shaken with 5 mol of phenol at 40 \(^\circ\)C is .......................
(rounded off to one decimal place)





























































Comments