The GATE 2025 XL Question Paper PDF is available as the exam concludes. IIT Roorkee is conducting the GATE 2025 Life Sciences (XL) exam on February 16 Shift 2 took place from 2:30 PM to 5:30 PM. As per the updated exam pattern, the exam consists of 65 questions with 100 marks, with 10 from the General Aptitude section and 55 questions from Life Sciences topics.The difficulty level of GATE 2025 XL was moderate.
Also Check:
GATE 2025 XL Question Paper with Solutions PDF
| GATE 2025 XL Question Paper with Answer Key | Download PDF | Check Solutions |

Question 1:
Even though I had planned to go skiing with my friends, I had to ............. at the last moment because of an injury.
Select the most appropriate option to complete the above sentence.
The President, along with the Council of Ministers, ............. to visit India next week.
Select the most appropriate option to complete the above sentence.
An electricity utility company charges Rs.7 per kWh. If a 40-watt desk light is left on for 10 hours each night for 180 days, what would be the cost of energy consumption? If the desk light is on for 2 more hours each night for the 180 days, what would be the percentage-increase in the cost of energy consumption?
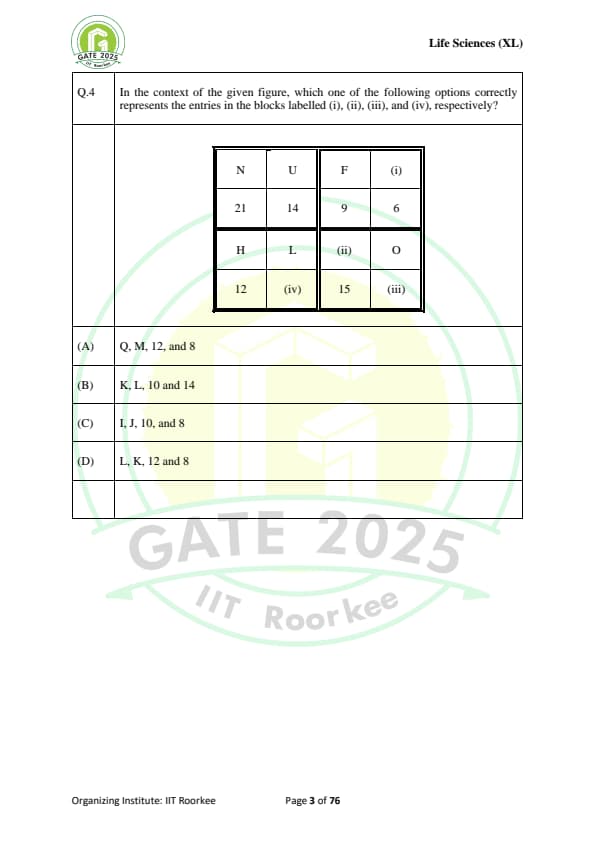
In the context of the given figure, which one of the following options correctly represents the entries in the blocks labelled (i), (ii), (iii), and (iv), respectively?
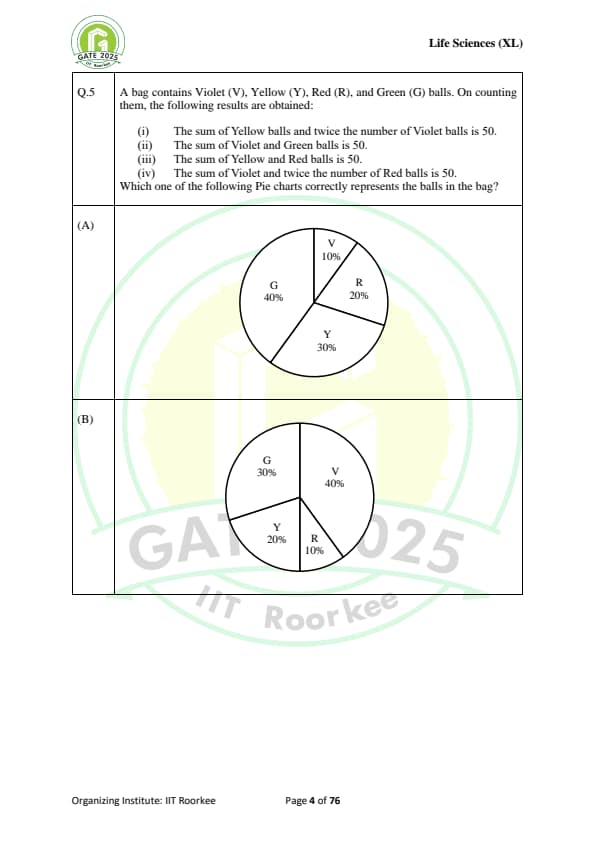
A bag contains Violet (V), Yellow (Y), Red (R), and Green (G) balls. On counting them, the following results are obtained:
(i) The sum of Yellow balls and twice the number of Violet balls is 50.
(ii) The sum of Violet and Green balls is 50.
(iii) The sum of Yellow and Red balls is 50.
(iv) The sum of Violet and twice the number of Red balls is 50.
Which one of the following Pie charts correctly represents the balls in the bag?
“His life was divided between the books, his friends, and long walks. A solitary man, he worked at all hours without much method, and probably courted his fatal illness in this way. To his own name there is not much to show; but such was his liberality that he was continually helping others, and fruits of his erudition are widely scattered, and have gone to increase many a comparative stranger’s reputation.”
(From E.V. Lucas’s “A Funeral”)
Based only on the information provided in the above passage, which one of the following statements is true?
For the clock shown in the figure, if
O = O Q S Z P R T, and
X = X Z P W Y O Q,
then which one among the given options is most appropriate for P?
Consider a five-digit number PQRST that has distinct digits P, Q, R, S, and T, and satisfies the following conditions:
1. \( P < Q \)
2. \( S > P > T \)
3. \( R < T \)
If integers 1 through 5 are used to construct such a number, the value of P is:
A business person buys potatoes of two different varieties P and Q, mixes them in a certain ratio and sells them at Rs.192 per kg.
The cost of the variety P is Rs.800 for 5 kg.
The cost of the variety Q is Rs.800 for 4 kg.
If the person gets 8% profit, what is the P : Q ratio (by weight)?
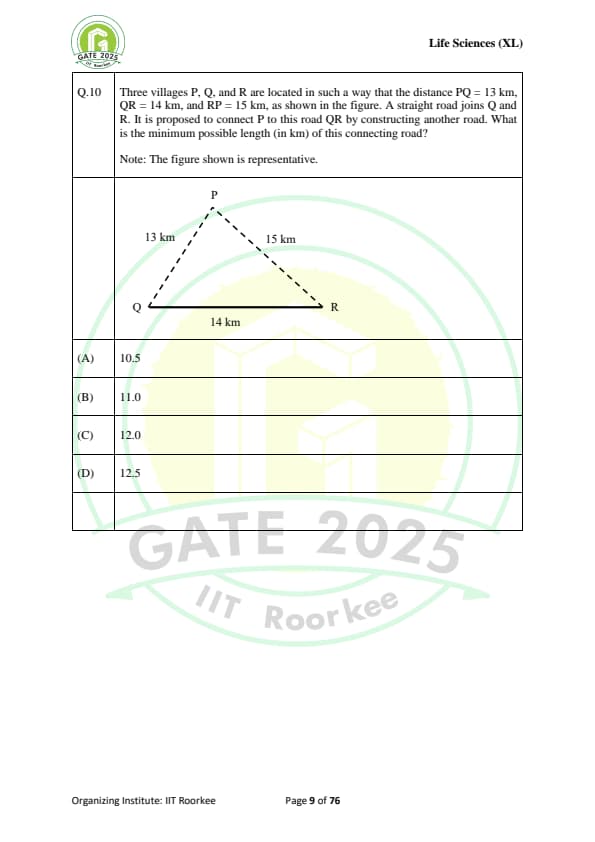
Three villages P, Q, and R are located in such a way that the distance PQ = 13 km, QR = 14 km, and RP = 15 km, as shown in the figure. A straight road joins Q and R. It is proposed to connect P to this road QR by constructing another road. What is the minimum possible length (in km) of this connecting road?
Note: The figure shown is representative.
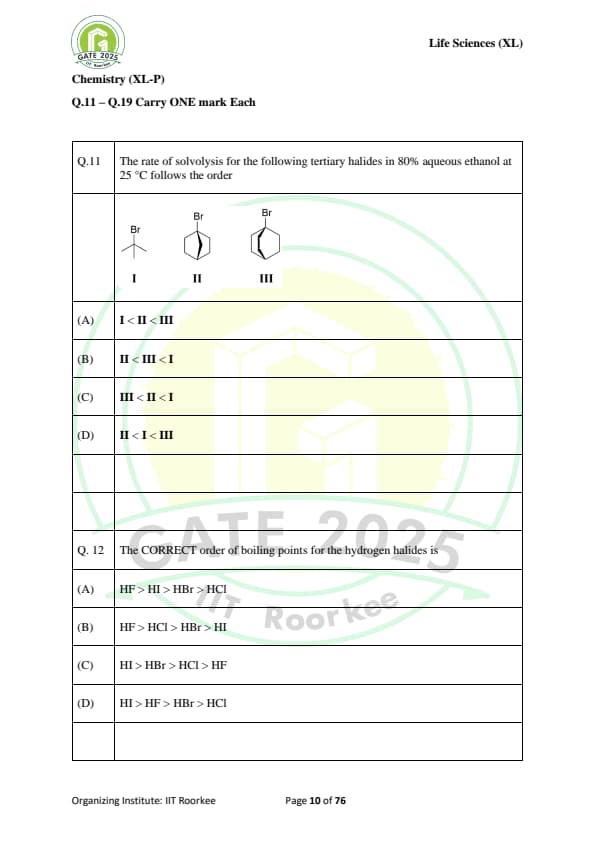
The rate of solvolysis for the following tertiary halides in 80% aqueous ethanol at 25°C follows the order:
The CORRECT order of boiling points for the hydrogen halides is:
The bond order in \( N_2^{2-} \) species is:
The standard enthalpy of the reaction,
\[ C (graphite) + H_2O (g) \rightarrow CO (g) + H_2 (g) is found to be +131.3 kJ mol^{-1} \]
and the \( \Delta_f H^\circ \) value for CO (g) is -110.5 kJ mol^{-1.
The value of \( \Delta_f H^\circ \) (in kJ mol^{-1) for H_2\text{O (g) is:
The temperature dependence of reaction rates is generally given by the Arrhenius equation. A plot of \( \ln k_r \) against \( 1/T \) is a straight line from which the pre-exponential factor ‘\( A \)’ and the activation energy ‘\( E_a \)’ can be determined.
The CORRECT option regarding this plot is:
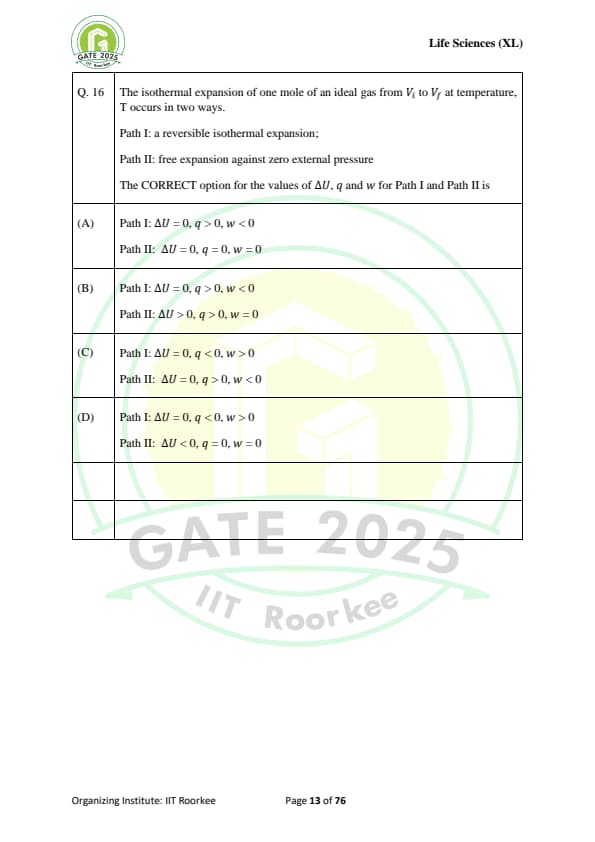
The isothermal expansion of one mole of an ideal gas from \( V_i \) to \( V_f \) at temperature \( T \) occurs in two ways:
Path I: a reversible isothermal expansion;
Path II: free expansion against zero external pressure.
The CORRECT option for the values of \( \Delta U \), \( q \), and \( w \) for Path I and Path II is:
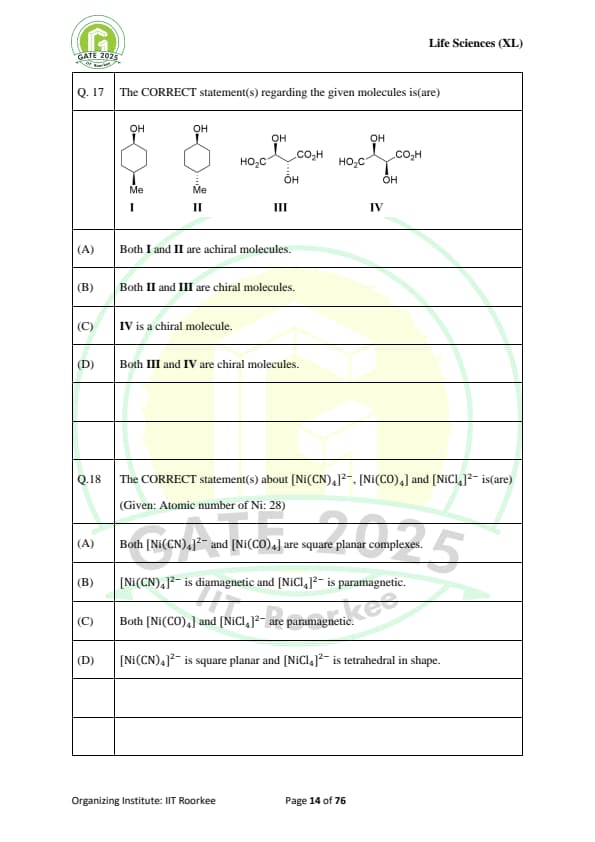
The CORRECT statement(s) regarding the given molecules is(are):
The CORRECT statement(s) about \([Ni(CN)_4]^{2-}, [Ni(CO)_4]\) and \([NiCl_4]^{2-}\) is(are):
(Given: Atomic number of Ni = 28)
Consider the two \( pK_a \) values of valine as 2.32 and 9.62. The isoelectric point (pI) of this amino acid is ....... (rounded off to two decimal places)
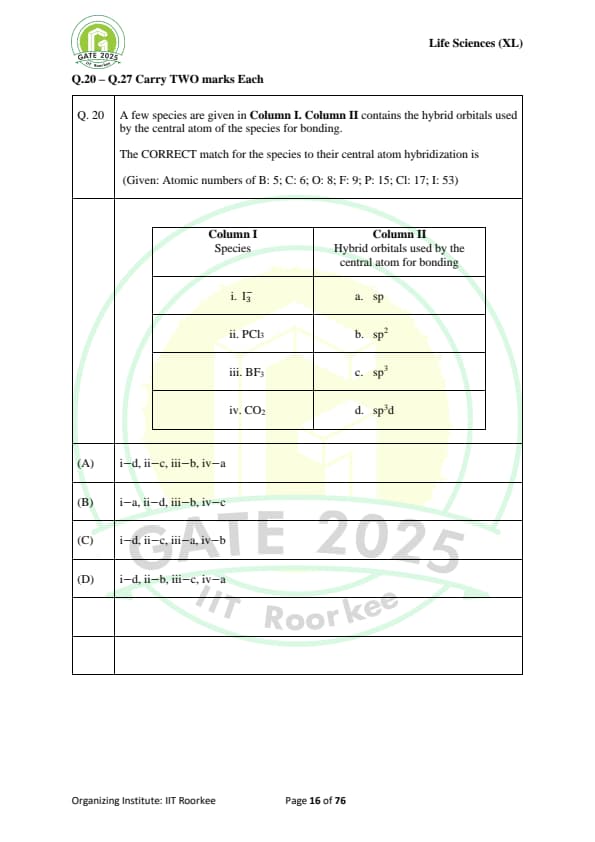
A few species are given in Column I. Column II contains the hybrid orbitals used by the central atom of the species for bonding.
The CORRECT match for the species to their central atom hybridization is:
\text{(Given: Atomic numbers of B: 5; C: 6; O: 8; F: 9; P: 15; Cl: 17; I: 53)
\begin{tabular{|l|l|
\hline
Column I Species & Column II Hybrid orbitals used by the central atom for bonding
\hline
i. \( I_3^- \) & a. sp
ii. \( PCl_3 \) & b. sp²
iii. \( BF_3 \) & c. sp³
iv. \( CO_2 \) & d. sp³d
\hline
\end{tabular
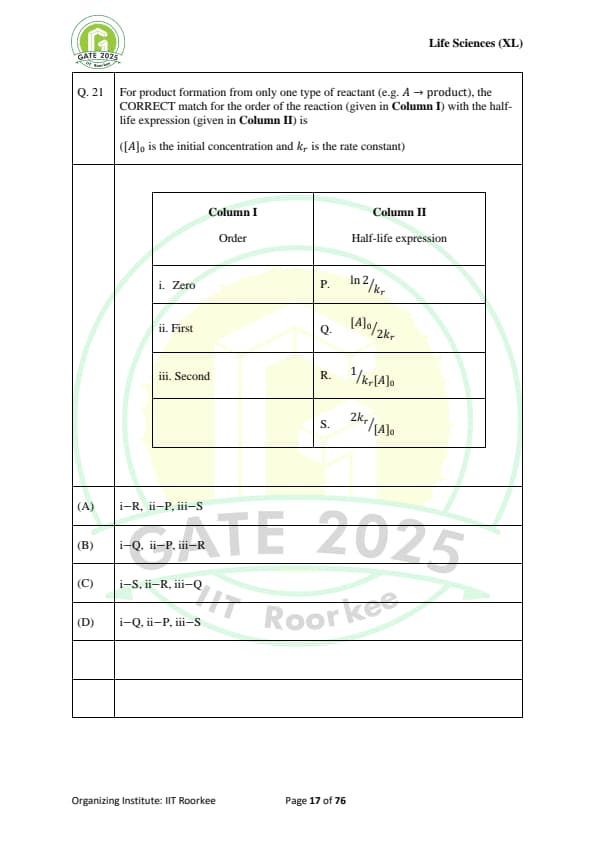
For product formation from only one type of reactant (e.g. A \(\rightarrow\) product), the CORRECT match for the order of the reaction (given in Column I) with the half-life expression (given in Column II) is:
(\([A]_0 \) is the initial concentration and \( k_r \) is the rate constant)

The CORRECT statement(s) for the given reactions is(are):
Addition of a few drops of concentrated HCl to an aqueous solution of CoCl₂ forms a dark blue complex X.
The CORRECT statement(s) for this reaction is(are):
(Given: Atomic number of Co: 27)
The CORRECT statement(s) regarding biomolecules is(are):
Energy of the transition from \( n_h = 4 \) to \( n_l = 2 \) for hydrogen atom is \( E \times 10^3 \) cm\(^{-1}\).
Given: Rydberg constant for hydrogen: \( 1.097 \times 10^7 \, m^{-1} \)
Value of E is ............... (rounded off to two decimal places)
A non-volatile solute has a molecular weight of 180 g mol\(^{-1}\). Assume that the solute does not associate or dissociate in water, and the boiling-point constant (ebullioscopic constant) of water is 0.51 K kg mol\(^{-1}\).
The amount (in g) of solute added to 500 g of water to elevate the boiling point by 0.153 K is ............... (answer in integer)
The standard potentials (\( E^\circ \)) for the Fe\(^{3+}\)/Fe and Fe\(^{3+}\)/Fe\(^{2+}\) couples are -0.04 V and +0.76 V, respectively.
Given: Faraday constant = 96500 C mol\(^{-1}\)
The value for \( E^\circ \) (Fe\(^{2+}\)/Fe) in V is ............... (rounded off to two decimal places)
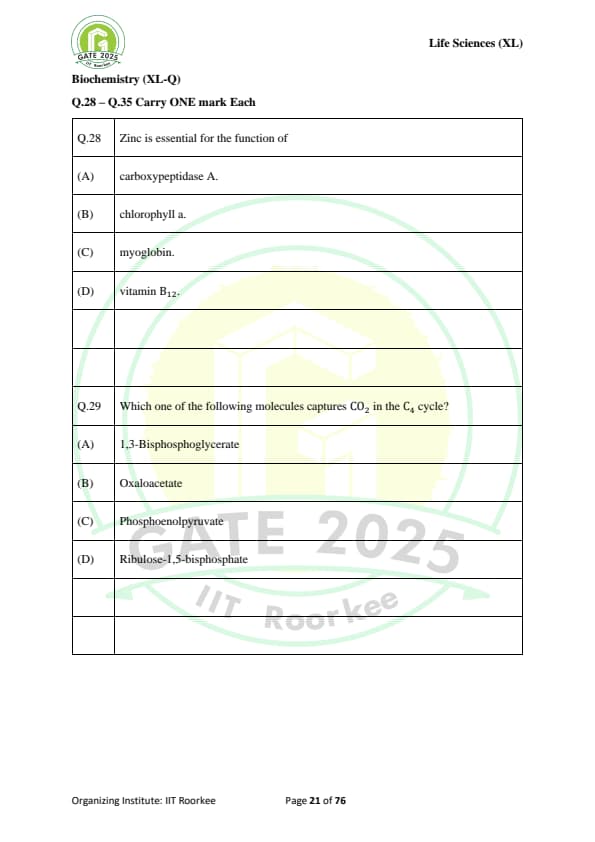
Zinc is essential for the function of
Which one of the following molecules captures \(CO_2\) in the \(CO_4\) cycle?
Which one of the following methods separates biomolecules based on their hydrodynamic volumes?
Which one of the following restriction endonucleases is a blunt cutter?
Which one of the following DNA repair systems requires DNA glycosylases?
Which one of the following ion channels opens to repolarize the neuronal membrane when an action potential is generated?
Which one of the following is the most sensitive immunoassay?
Which of the following statements about antibodies is/are correct?
Which one of the following molecules does NOT contain phosphoanhydride bond(s)?
For an enzyme that follows Michaelis-Menten kinetics, a competitive inhibitor
Förster Resonance Energy Transfer does NOT depend on the
Phospholipid vesicles prepared in 50 mM KCl were diluted in water. Based on this information, statements P and Q are made.
P: The diluted vesicles will develop membrane potential.
Q: There is a K\textsuperscript{+} concentration difference across the vesicular membrane.
Which one of the following options is correct?
Peptide-binding cleft in MHC-I is formed by
Which of the following peptides do/does NOT absorb ultraviolet light above 250 nm wavelength?
Which of the following is/are peptide hormone(s)?
Which of the following is/are heteropolysaccharide(s)?
The equilibrium dissociation constant of acetic acid is \( 1.74 \times 10^{-5} \) M. The \( pK_a \) of acetic acid (rounded off to one decimal place) is ......
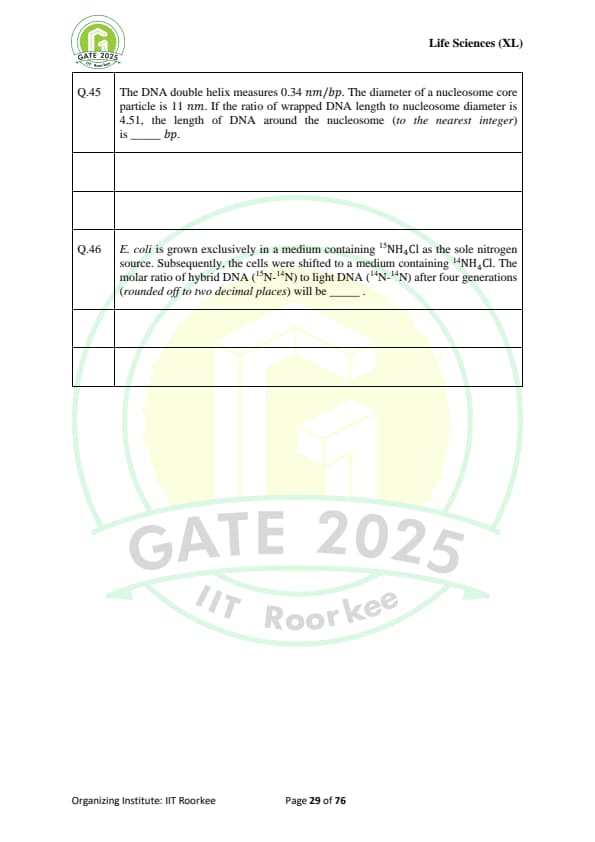
The DNA double helix measures 0.34 nm/bp. The diameter of a nucleosome core particle is 11 nm. If the ratio of wrapped DNA length to nucleosome diameter is 4.51, the length of DNA around the nucleosome (to the nearest integer) is ...... bp.
E. coli is grown exclusively in a medium containing \(^{15}NH_4Cl\) as the sole nitrogen source. Subsequently, the cells were shifted to a medium containing \(^{14}NH_4Cl\). The molar ratio of hybrid DNA (\(^{15}N-^{14}N\)) to light DNA (\(^{14}N-^{14}N\)) after four generations (rounded off to two decimal places) will be .......
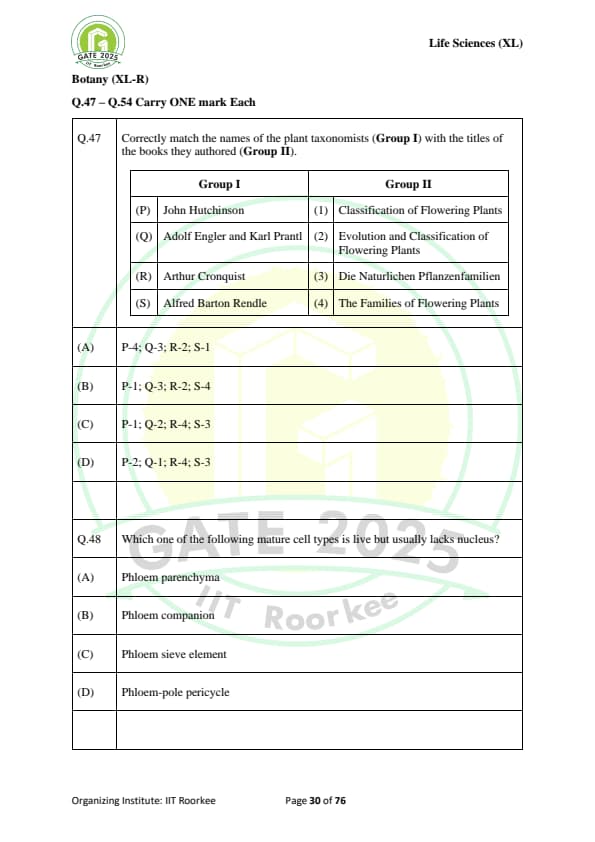
Correctly match the names of the plant taxonomists (Group I) with the titles of the books they authored (Group II):
\begin{tabular{|l|l|
\hline
Group I & Group II
\hline
(P) John Hutchinson & (1) Classification of Flowering Plants
(Q) Adolf Engler and Karl Prantl & (2) Evolution and Classification of Flowering Plants
(R) Arthur Cronquist & (3) Die Natürlichen Pflanzenfamilien
(S) Alfred Barton Rendle & (4) The Families of Flowering Plants
\hline
\end{tabular
Which one of the following mature cell types is live but usually lacks nucleus?
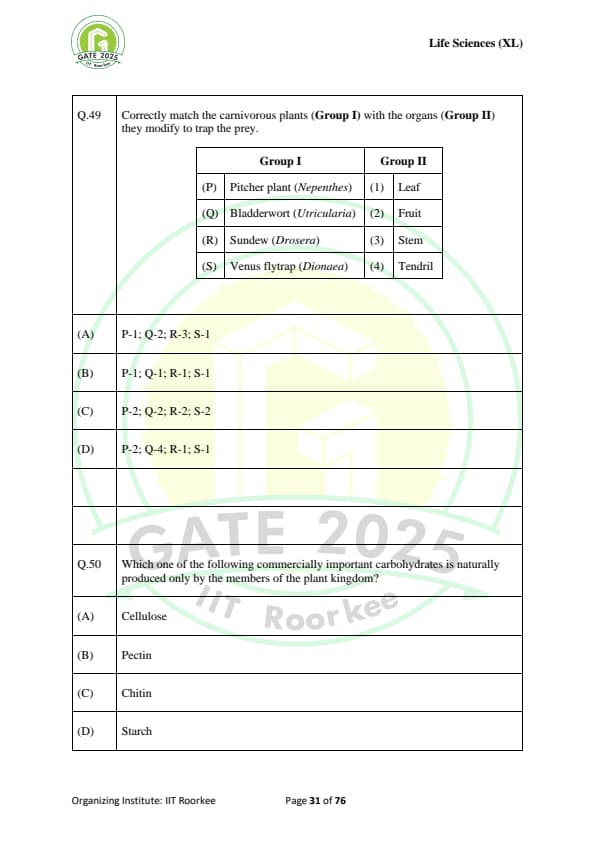
Correctly match the carnivorous plants (Group I) with the organs (Group II) they modify to trap the prey:
\begin{tabular{|l|l|
\hline
Group I & Group II
\hline
(P) Pitcher plant (Nepenthes) & (1) Leaf
(Q) Bladderwort (Utricularia) & (2) Fruit
(R) Sundew (Drosera) & (3) Stem
(S) Venus flytrap (Dionaea) & (4) Tendril
\hline
\end{tabular
Which one of the following commercially important carbohydrates is naturally produced only by the members of the plant kingdom?
Which one of the following agents causes the necrotic ring spot disease in stone fruits?
Identify the correct statement(s) with respect to plant disease:
Which of the following molecular approaches can be used to generate complete knock-out of a target gene in plants?
If an egg cell of a diploid plant species has 10 chromosomes, the expected number of chromosomes in a double trisomic somatic cell of this species would be ............. (Answer in integer).
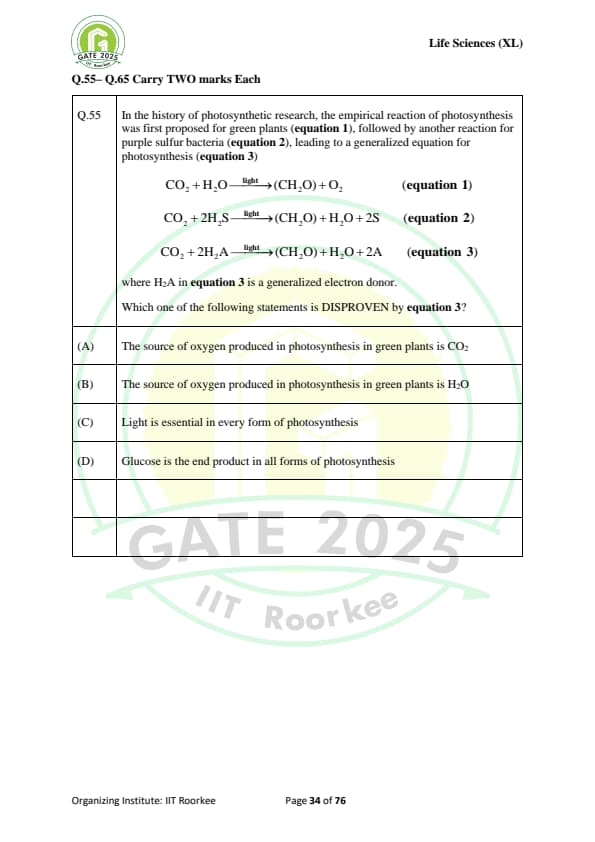
In the history of photosynthetic research, the empirical reaction of photosynthesis was first proposed for green plants (equation 1), followed by another reaction for purple sulfur bacteria (equation 2), leading to a generalized equation for photosynthesis (equation 3), where \( H_2A \) in equation 3 is a generalized electron donor.
\[ CO_2 + H_2O \xrightarrow{light} (CH_2O) + O_2 \quad (equation 1) \] \[ CO_2 + 2H_2S \xrightarrow{light} (CH_2O) + H_2O + 2S \quad (equation 2) \] \[ CO_2 + 2H_2A \xrightarrow{light} (CH_2O) + H_2O + 2A \quad (equation 3) \]
Where \( H_2A \) in equation 3 is a generalized electron donor.
Which one of the following statements is DISPROVEN by equation 3?
Consider a diploid plant species where the cells in the epidermis (the outermost single cell layer) always divide in the anticlinal orientation. If one such cell within the central zone of the shoot apical meristem (SAM) spontaneously becomes tetraploid at the seedling stage, which one of the following cellular arrangements would be most likely observed if the meristem is examined at the adult stage?
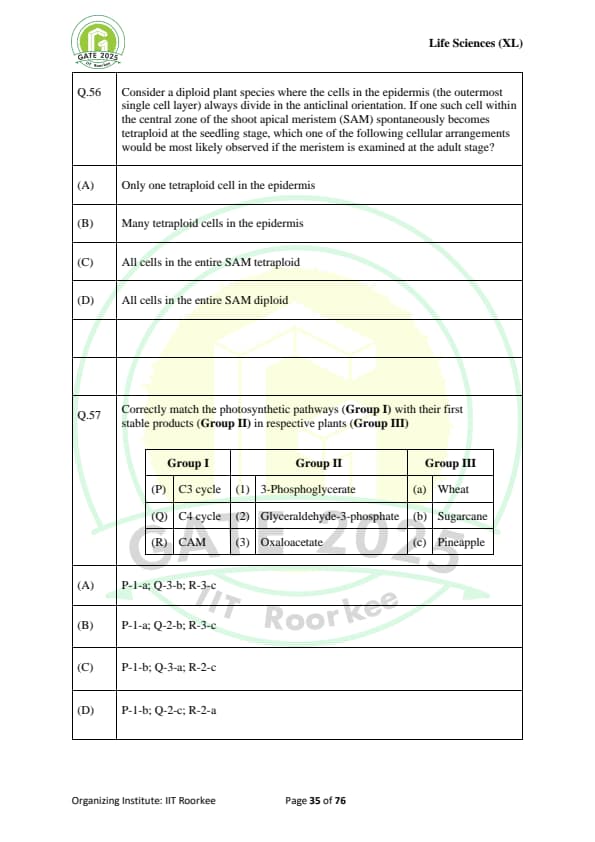
Correctly match the photosynthetic pathways (Group I) with their first stable products (Group II) in respective plants (Group III):
\begin{tabular{|l|l|l|
\hline
Group I & Group II & Group III
\hline
(P) C3 cycle & (1) 3-Phosphoglycerate & (a) Wheat
(Q) C4 cycle & (2) Glyceraldehyde-3-phosphate & (b) Sugarcane
(R) CAM & (3) Oxaloacetate & (c) Pineapple
\hline
\end{tabular
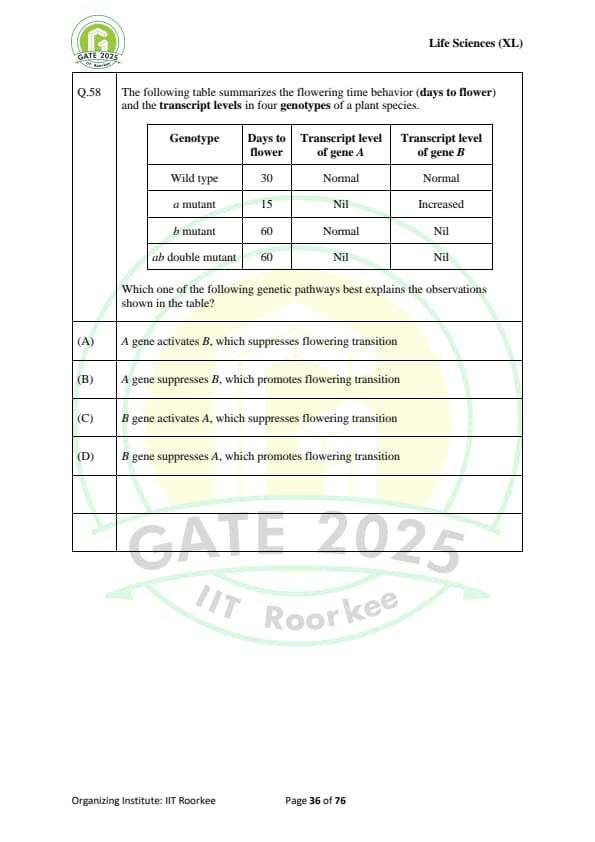
The following table summarizes the flowering time behavior (days to flower) and the transcript levels in four genotypes of a plant species.
\begin{tabular{|l|l|l|l|
\hline
Genotype & Days to flower & Transcript level of gene A & Transcript level of gene B
\hline
Wild type & 30 & Normal & Normal
a mutant & 15 & Nil & Increased
b mutant & 60 & Normal & Nil
ab double mutant & 60 & Nil & Nil
\hline
\end{tabular
Which one of the following genetic pathways best explains the observations shown in the table?
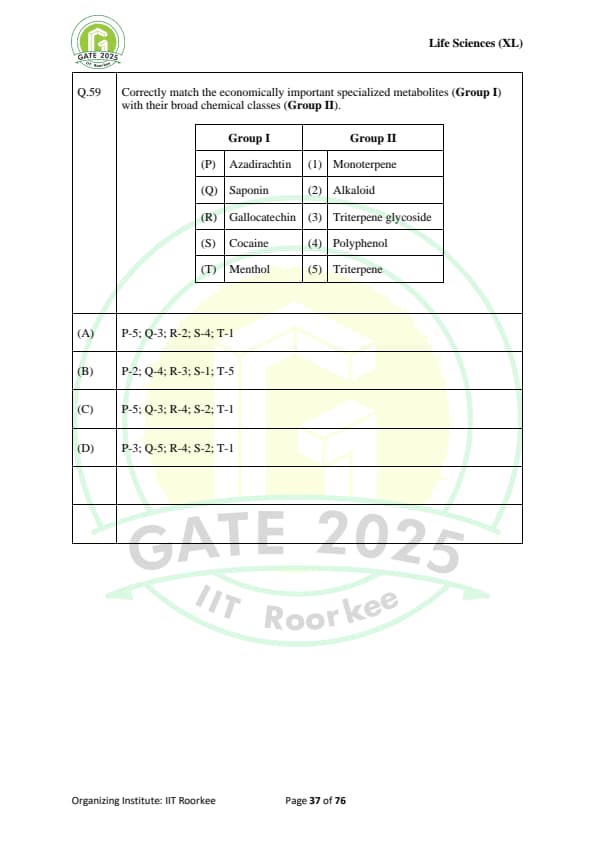
Correctly match the economically important specialized metabolites (Group I) with their broad chemical classes (Group II):
\begin{tabular{|l|l|
\hline
Group I & Group II
\hline
(P) Azadirachtin & (1) Monoterpene
(Q) Saponin & (2) Alkaloid
(R) Gallocatechin & (3) Triterpene glycoside
(S) Cocaine & (4) Polyphenol
(T) Menthol & (5) Triterpene
\hline
\end{tabular
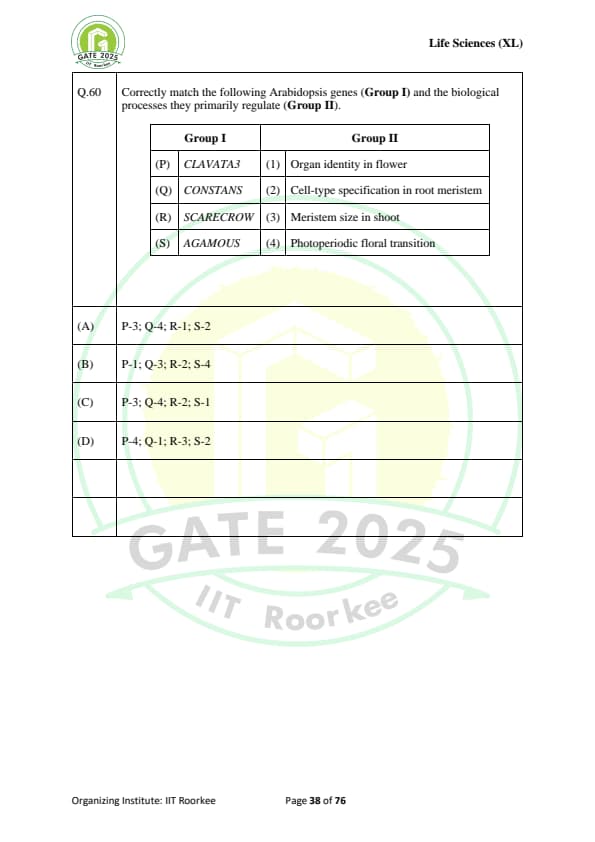
Correctly match the following Arabidopsis genes (Group I) and the biological processes they primarily regulate (Group II):
\begin{tabular{|l|l|
\hline
Group I & Group II
\hline
(P) CLAVATA3 & (1) Organ identity in flower
(Q) CONSTANS & (2) Cell-type specification in root meristem
(R) SCARECROW & (3) Meristem size in shoot
(S) AGAMOUS & (4) Photoperiodic floral transition
\hline
\end{tabular
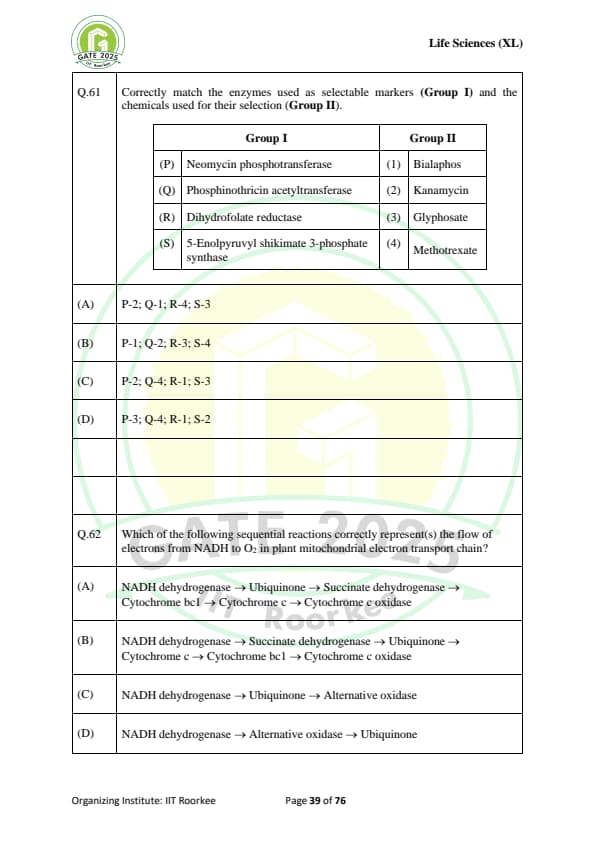
Correctly match the enzymes used as selectable markers (Group I) and the chemicals used for their selection (Group II):
\begin{tabular{|l|l|
\hline
Group I & Group II
\hline
(P) Neomycin phosphotransferase & (1) Bialaphos
(Q) Phosphinothricin acetyltransferase & (2) Kanamycin
(R) Dihydrofolate reductase & (3) Glyphosate
(S) 5-Enolpyruvyl shikimate 3-phosphate synthase & (4) Methotrexate
\hline
\end{tabular
Which of the following sequential reactions correctly represent(s) the flow of electrons from NADH to O\textsubscript{2} in plant mitochondrial electron transport chain?
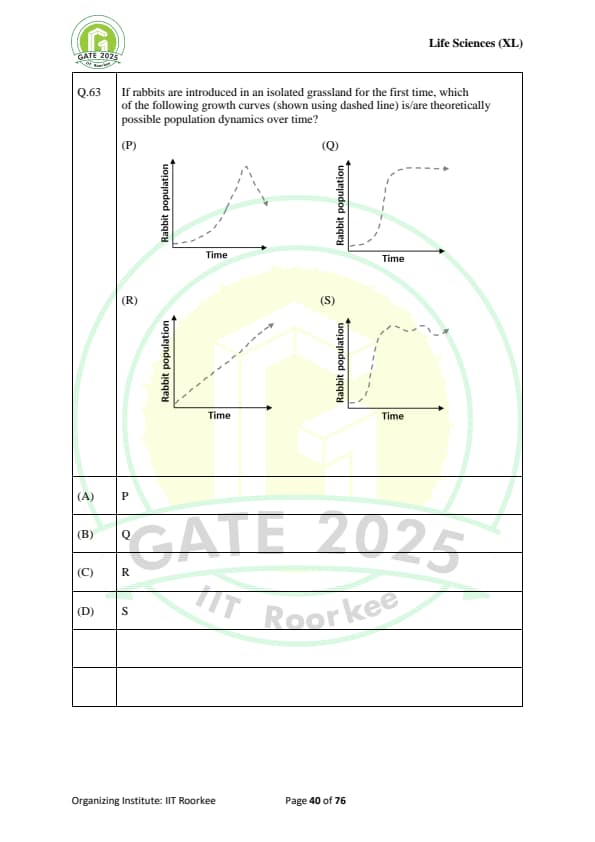
If rabbits are introduced in an isolated grassland for the first time, which of the following growth curves (shown using dashed line) is/are theoretically possible population dynamics over time?
Which of the following reactions in plants is/are catalyzed by the malic enzymes?
In a genetic cross between a true-breeding tall parent bearing red flowers and a true-breeding dwarf parent bearing white flowers, only tall plants with red flowers are obtained in the F\(_1\) population. Considering these two traits segregate independently, if one tall individual is selected from the F\(_2\) population, the probability that it would be genotypically homozygous for plant height and make red flowers is ........... \textit{(Round off to two decimal places)
Which one of the following metabolites is associated with bacterial stringent response?
India is aiming to be free of tuberculosis by 2025. One of the key approaches for this program is DOTS. Which one of the following options is the full form of DOTS?
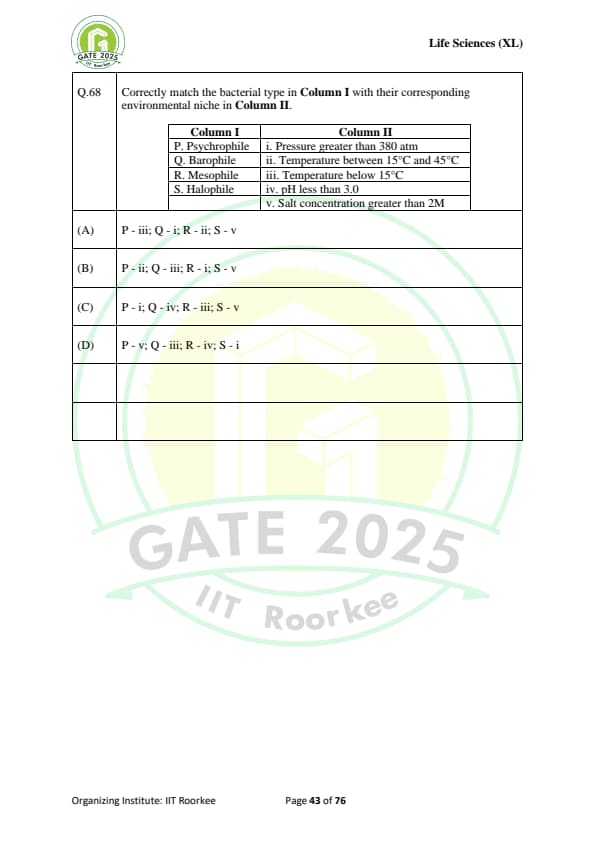
Correctly match the bacterial type in Column I with their corresponding environmental niche in Column II:
\begin{tabular{|l|l|
\hline
Column I & Column II
\hline
P. Psychrophile & i. Pressure greater than 380 atm
Q. Barophile & ii. Temperature between 15°C and 45°C
R. Mesophile & iii. Temperature below 15°C
S. Halophile & iv. pH less than 3.0
& v. Salt concentration greater than 2M
\hline
\end{tabular
Robert Koch used a meat-infused nutrient medium for which one of the following purposes?
A penicillin sensitive Escherichia coli population is exposed to a lethal dose (200 µg/ml) of penicillin. Assuming density-independent mortality, which one of the following relationships would describe the number of surviving bacteria (N) over time (T)?
A bacterium obtains energy from a chemical source by the oxidation of reduced \(NO_2^{-}\), with \(CO_2\) as the principal carbon source. Which one of the following nutritional groups does this bacterium belong to?
The origin of the Escherichia coli chromosome on the genetic map is shown below.
Bidirectional replication is a feature of this system and both replication forks move at the same rate. Which one of the following sequences of replication of the genes is correct?
Which of the following sites is/are the location(s) of ATP generation through oxidative phosphorylation in Escherichia coli?
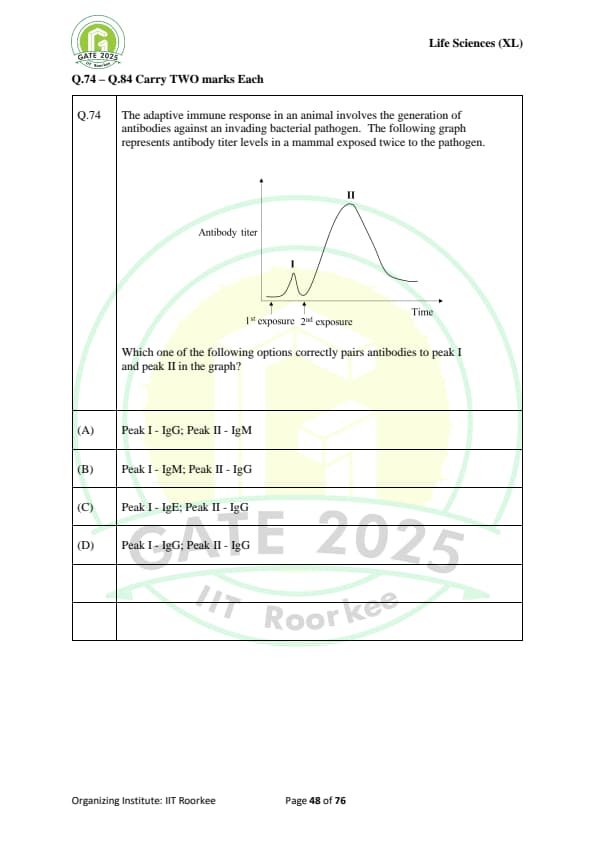
The adaptive immune response in an animal involves the generation of antibodies against an invading bacterial pathogen. The following graph represents antibody titer levels in a mammal exposed twice to the pathogen.
Which one of the following options correctly pairs antibodies to peak I and peak II in the graph?
Carl Woese established that short subunit rRNA sequences can be used to reveal evolutionary relationships between various organisms. Based on this, which one of the following options is the established phylogenetic arrangement of the three domains of life?
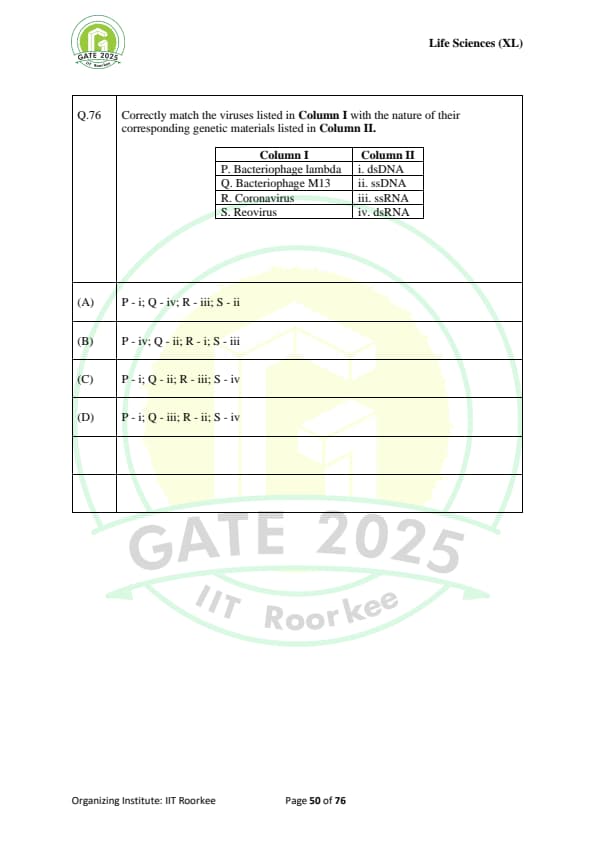
Correctly match the viruses listed in Column I with the nature of their corresponding genetic materials listed in Column II:
\begin{tabular{|l|l|
\hline
Column I & Column II
\hline
P. Bacteriophage lambda & i. dsDNA
Q. Bacteriophage M13 & ii. ssDNA
R. Coronavirus & iii. ssRNA
S. Reovirus & iv. dsRNA
\hline
\end{tabular
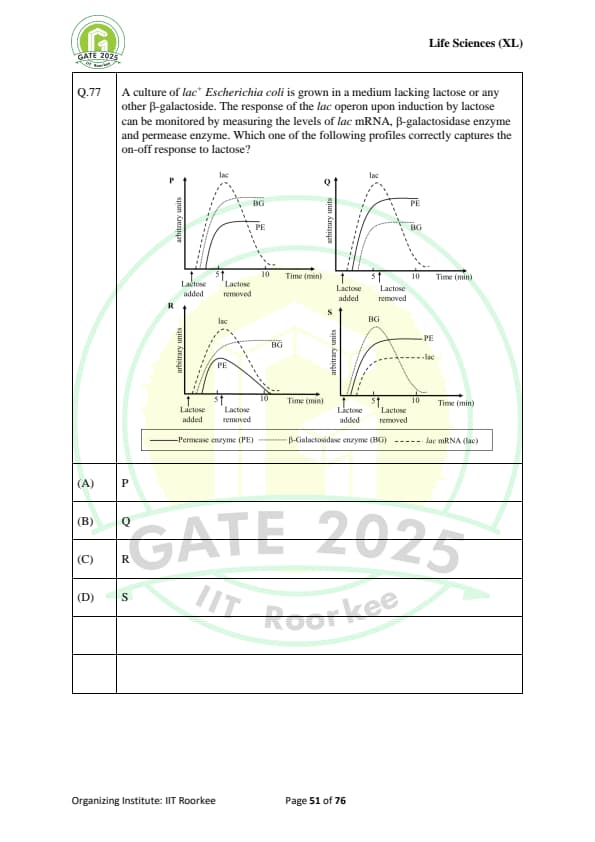
A culture of lac Escherichia coli is grown in a medium lacking lactose or any other \(\beta\)-galactoside. The response of the lac operon upon induction by lactose can be monitored by measuring the levels of lac mRNA, \(\beta\)-galactosidase enzyme and permease enzyme. Which one of the following profiles correctly captures the on-off response to lactose?
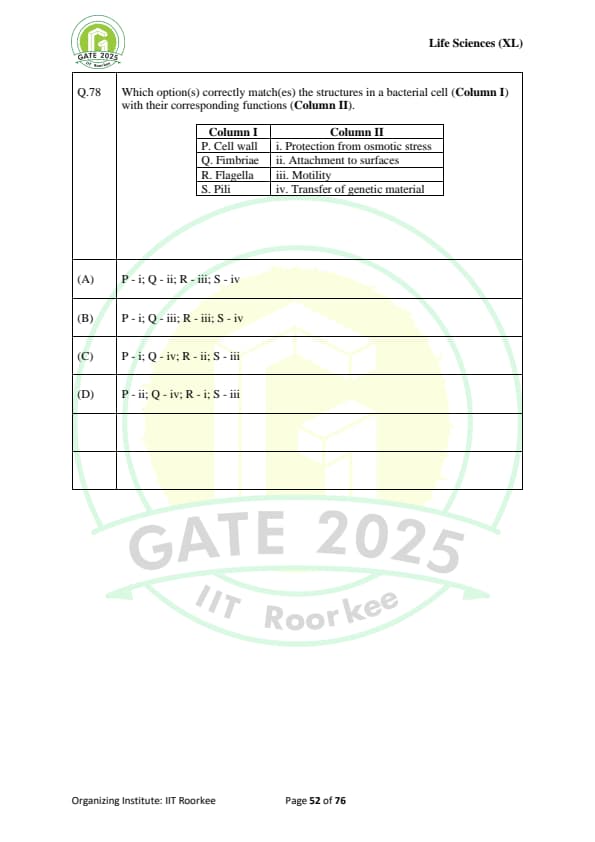
Which option(s) correctly match(es) the structures in a bacterial cell (Column I) with their corresponding functions (Column II)?
\begin{tabular{|l|l|
\hline
Column I & Column II
\hline
P. Cell wall & i. Protection from osmotic stress
Q. Fimbriae & ii. Attachment to surfaces
R. Flagella & iii. Motility
S. Pili & iv. Transfer of genetic material
\hline
\end{tabular
Which of the following statements regarding micro-organisms is/are correct?
Which of the following is/are example(s) of animal-microbe mutualism?
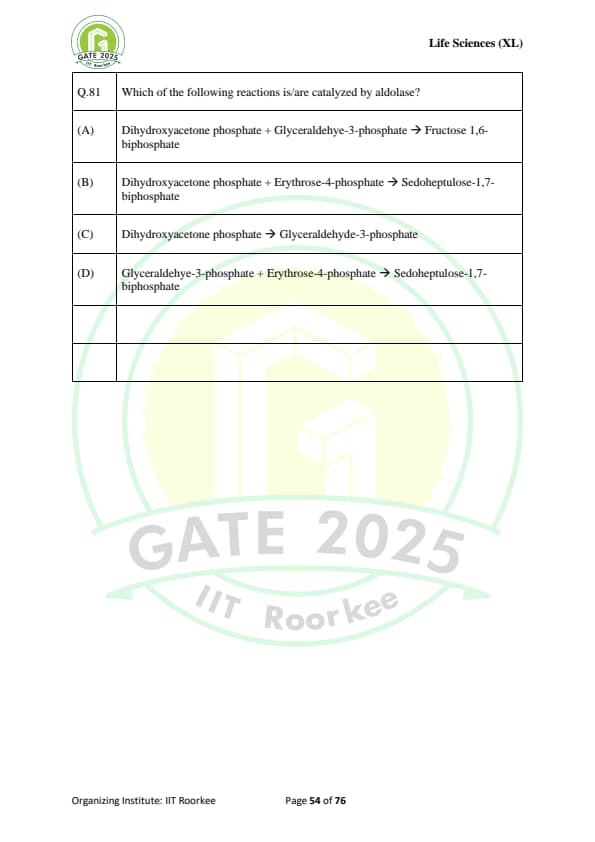
Which of the following reactions is/are catalyzed by aldolase?
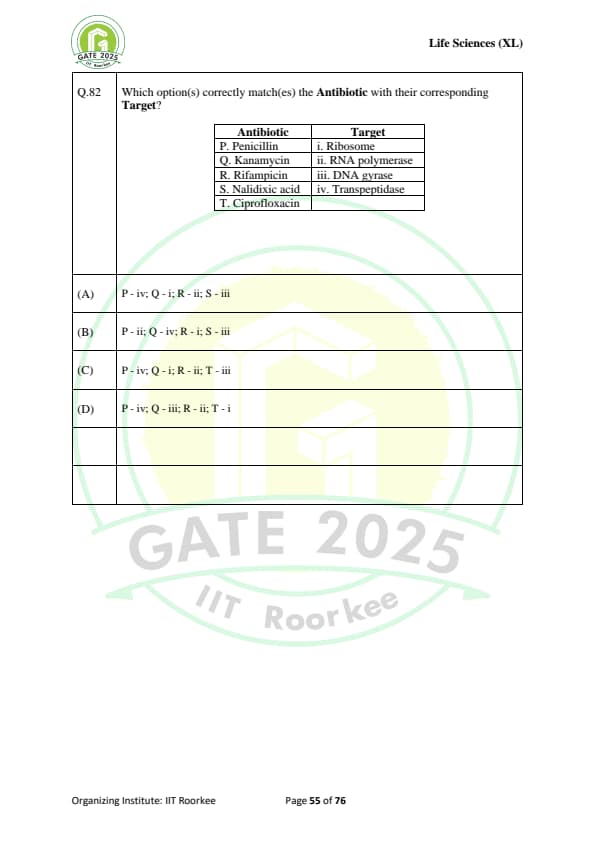
Which option(s) correctly match(es) the Antibiotic with their corresponding Target?
\begin{array{|l|l|
\hline
Antibiotic & Target
\hline
P. \text{Penicillin & i. \text{Ribosome
Q. \text{Kanamycin & ii. \text{RNA polymerase
R. \text{Rifampicin & iii. \text{DNA gyrase
S. \text{Nalidixic acid & iv. \text{Transpeptidase
T. \text{Ciprofloxacin &
\hline
\end{array
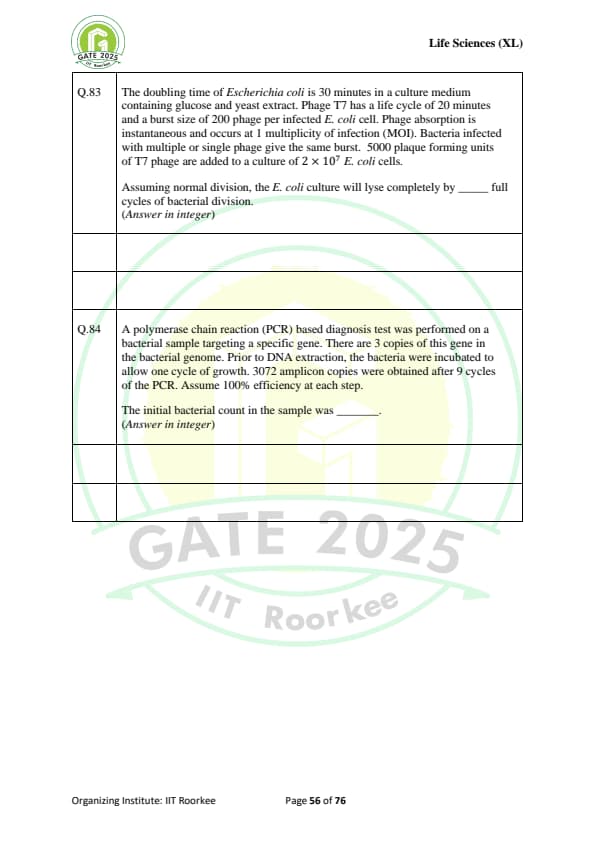
The doubling time of Escherichia coli is 30 minutes in a culture medium containing glucose and yeast extract. Phage T7 has a life cycle of 20 minutes and a burst size of 200 phage per infected E. coli cell. Phage absorption is instantaneous and occurs at 1 multiplicity of infection (MOI). Bacteria infected with multiple or single phage give the same burst. 5000 plaque forming units of T7 phage are added to a culture of \( 2 \times 10^7 \) \textit{E. coli cells. Assuming normal division, the \textit{E. coli culture will lyse completely by ........ full cycles of bacterial division. (Answer in integer)
A polymerase chain reaction (PCR) based diagnosis test was performed on a bacterial sample targeting a specific gene. There are 3 copies of this gene in the bacterial genome. Prior to DNA extraction, the bacteria were incubated to allow one cycle of growth. 3072 amplicon copies were obtained after 9 cycles of the PCR. Assume 100% efficiency at each step. The initial bacterial count in the sample was ....... (Answer in integer)
Which one of the following is a "brood parasite"?
During the development of a mammalian embryo, "yolk sac" is formed by which one of the following?
The animals belonging to which one of the following phyla are characterized by "segmented body"?
Which one of the following is a "post-zygotic" isolating mechanism of speciation?
Desmosomes are
The "foramen of Panizza" is found in which one of the following groups of animals?
Imagine a population of diploid species in Hardy-Weinberg equilibrium. The population has two alleles for a gene which are ‘a’ and ‘A’. The number of individuals with ‘aa’ genotype in this population is 1 in 10000. The frequency of the allele ‘A’ in the population is ............ (up to two decimal places)
A PCR was setup to amplify a 500 nucleotides-long DNA. The dNTPs in the reaction mixture were radiolabeled. The percentage (%) of radiolabeled single-stranded DNA after three cycles will be ............ (up to one decimal place)
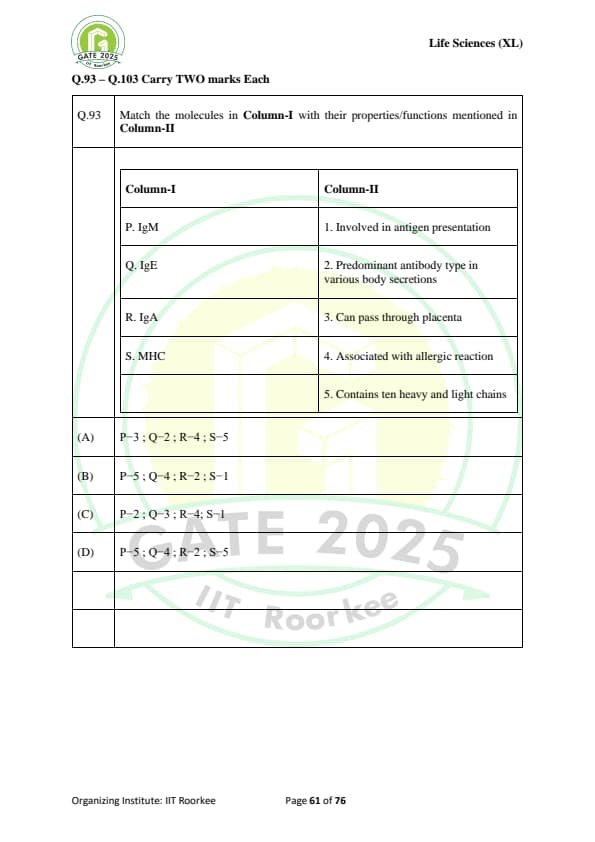
Match the molecules in Column-I with their properties/functions mentioned in Column-II
\begin{array{|l|l|
\hline
Column-I & Column-II
\hline
P. \text{IgM & 1. \text{Involved in antigen presentation
Q. \text{IgE & 2. \text{Predominant antibody type in various body secretions
R. \text{IgA & 3. \text{Can pass through placenta
S. \text{MHC & 4. \text{Associated with allergic reaction
\hline
\end{array
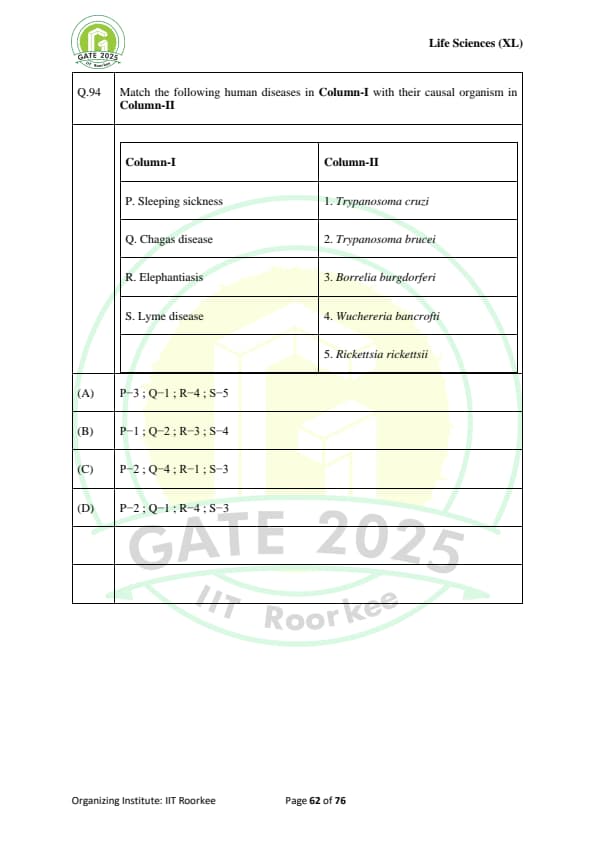
Match the following human diseases in Column-I with their causal organism in Column-II
\begin{array{|l|l|
\hline
Column-I & Column-II
\hline
P. \text{Sleeping sickness & 1. \text{Trypanosoma cruzi
Q. \text{Chagas disease & 2. \text{Trypanosoma brucei
R. \text{Elephantiasis & 3. \text{Borrelia burgdorferi
S. \text{Lyme disease & 4. \text{Wuchereria bancrofti
\hline
\end{array
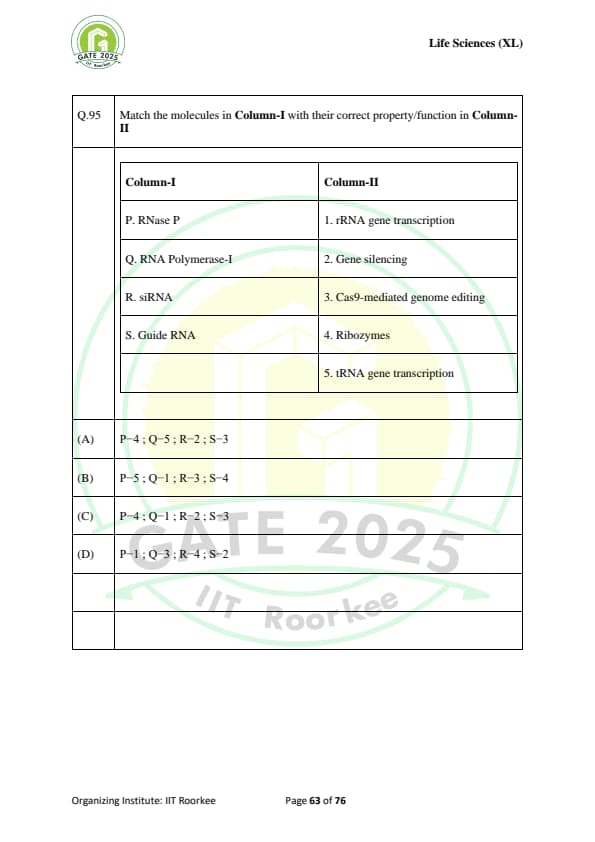
Match the molecules in Column-I with their correct property/function in Column-II
\begin{array{|l|l|
\hline
Column-I & Column-II
\hline
P. \text{RNase P & 1. \text{rRNA gene transcription
Q. \text{RNA Polymerase-I & 2. \text{Gene silencing
R. \text{siRNA & 3. \text{Cas9-mediated genome editing
S. \text{Guide RNA & 4. \text{Ribozymes
\hline
\end{array
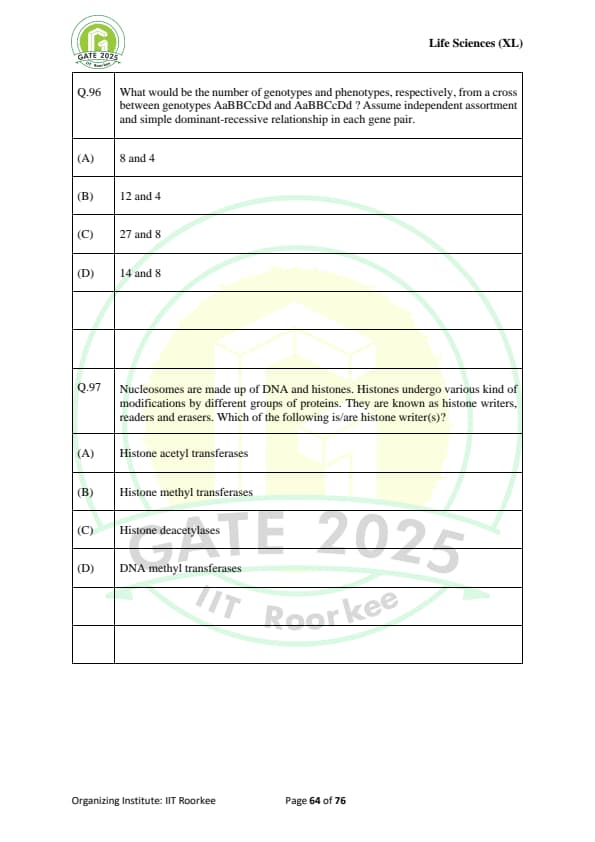
What would be the number of genotypes and phenotypes, respectively, from a cross between genotypes AaBBCcDd and AaBBCcDd? Assume independent assortment and simple dominant-recessive relationship in each gene pair.
Nucleosomes are made up of DNA and histones. Histones undergo various kinds of modifications by different groups of proteins. They are known as histone writers, readers, and erasers. Which of the following is/are histone writer(s)?
The expression of a gene is regulated by a transcription factor. Which of the following techniques can be used to identify the region in its promoter where the transcription factor binds?
Which of the following animals in India are included under "critically endangered" threat category as per the Red Data List of IUCN?
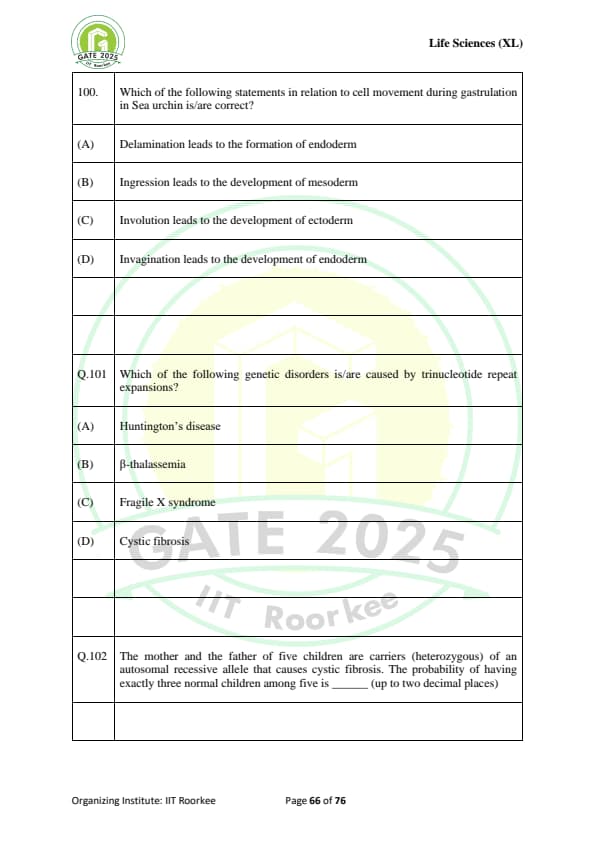
Which of the following statements in relation to cell movement during gastrulation in Sea urchin is/are correct?
Which of the following genetic disorders is/are caused by trinucleotide repeat expansions?
The mother and the father of five children are carriers (heterozygous) of an autosomal recessive allele that causes cystic fibrosis. The probability of having exactly three normal children among five is ........ (up to two decimal places).
An enzyme, which follows Michaelis-Menten equation, catalyzes the reaction A\(\rightarrow\)B. When enzyme and substrate concentrations are 15 nM and 10 \(\mu\)M, respectively, the reaction velocity is 5 \(\mu\)M s\(^{-1}\). If \(K_m\) for the substrate A is 5 \(\mu\)M, the kinetic efficiency of the enzyme will be ______ \(\times 10^6\) M\(^{-1}\) s\(^{-1}\) (in integer).
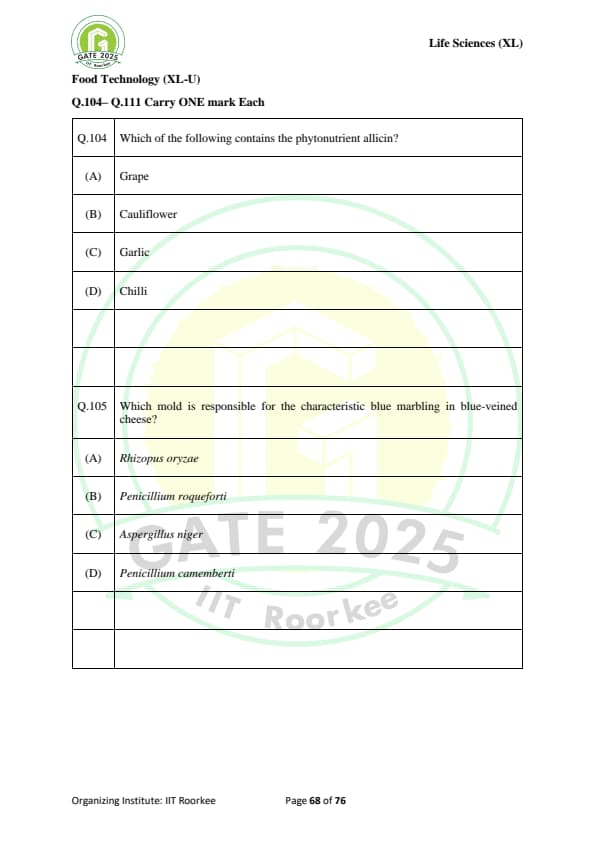
Which of the following contains the phytonutrient allicin?
Which mold is responsible for the characteristic blue marbling in blue-veined cheese?
Which genus of bacteria does NOT have a cell wall?
Which of the following pigment does NOT have pro-vitamin A activity?
Identify the analysis that must be performed FIRST to judge 'cleanliness' of spice/herb powders.
If there is a delay in oil extraction after bran is separated from the brown rice, the quality of rice bran oil deteriorates. Identify the suitable CAUSE and EFFECT for the deterioration in oil quality.
Among the following, which is/are the process(es) that lead to generation of new fats from existing ones?
The true density and bulk density of wheat grains are 1280 kg/m\(^3\) and 740 kg/m\(^3\), respectively. The porosity of the grains is ......... (rounded off to 2 decimal places).
Identify the gas composition (in percent) suitable for packaging cured meat under MAP conditions.
Which of the following sequence of events occurs during the formation of egg-white gel?
In canning and retorting of foods, which of the following is the correct expression of Ball process time (B)?
Which of the following is the most suitable flexible packaging laminate for dry fruits?
Identify the CORRECT sequence of operations for dressing of poultry.
Which of the following statement(s) is/are TRUE for a package of gamma-irradiated (7.5 kGy) whole chicken?
Match the following food products in Column I with their corresponding processes in Column II.
Which of the following is/are inhibitors of enzymatic browning in peeled potatoes?
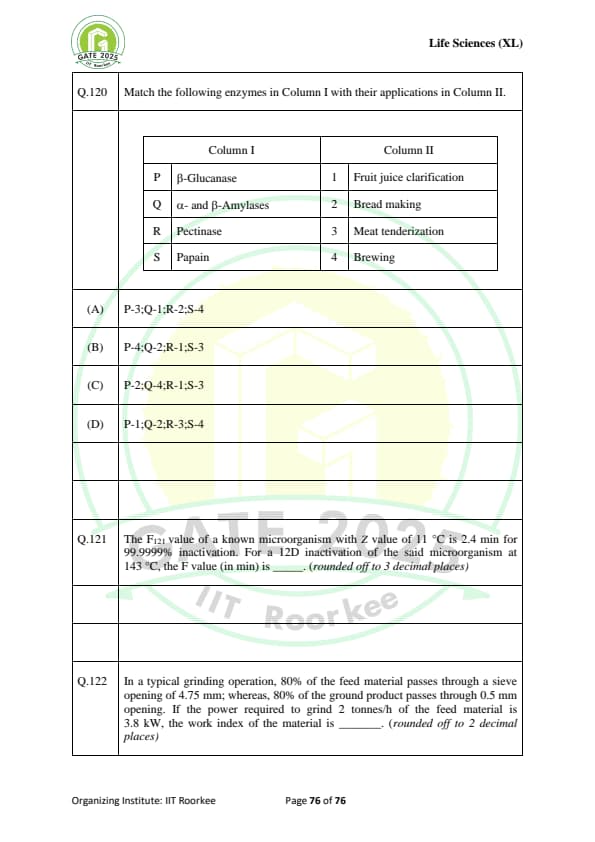
Match the following enzymes in Column I with their applications in Column II.
\begin{array{|l|l|
\hline
Column I (Enzyme) & Column II (Application)
\hline
P. \text{\(\beta\)-Glucanase & 1. \text{Fruit juice clarification
Q. \text{\(\alpha\)- and \(\beta\)-Amylases & 2. \text{Bread making
R. \text{Pectinase & 3. \text{Meat tenderization
S. \text{Papain & 4. \text{Brewing
\hline
\end{array
The \( F_{121} \) value of a known microorganism with \( Z \) value of \( 11^\circ C \) is 2.4 min for 99.9999% inactivation. For a 12D inactivation of the said microorganism at \( 143^\circ C \), the \( F \) value (in min) is .......... \textit{(rounded off to 3 decimal places)
In a typical grinding operation, 80% of the feed material passes through a sieve opening of 4.75 mm; whereas, 80% of the ground product passes through 0.5 mm opening. If the power required to grind 2 tonnes/h of the feed material is 3.8 kW, the work index of the material is ........ (rounded off to 2 decimal places)


































Comments