CBSE Class 12 Chemistry Compartment Question Paper 2023 with Answer Key pdf (Set 1 - 56/C/1) is available for download here. The exam was conducted by Central Board of Secondary Education on July 17, 2023 in the Morning Session 10:30 AM to 1:30 PM. The question paper comprised a total of 35 questions divided among 5 sections.
CBSE Class 12 Chemistry (Set 1 - 56/C/1) Compartment Question Paper 2023 with Answer Key
| CBSE Class 12 Chemistry Question Paper With Answer Key | Download PDF | Check Solutions |

Choose the compound which is more acidic than phenol:
View Solution
Which of the following reactions is a halogenated exchange reaction:
View Solution
The most common oxidation state for all lanthanoids is:
View Solution
A reaction follows second order kinetics. How is the rate of reaction affected if the concentration of the reactant is reduced to half? Choose the correct value from the following:
View Solution
Solutions of two electrolytes X and Y are diluted. Molar conductivity of X increases 25 times whereas that of Y increases 1.5 times. Which one is a stronger electrolyte?
View Solution
Unit of rate constant for the zero order reaction is:
View Solution
Three compounds are given below:

The correct decreasing order of their basic strength is:
View Solution
Which type of isomerism is shown by the complexes \([Co(NH_3)_5Br]SO_4\) and \([Co(NH_3)_5SO_4]Br\) ?
View Solution
What would be the major product of the given reaction?
\[ HCHO + CH_3MgI \xrightarrow{H_2O} \; ? \]
View Solution
Pentan-2-one and Pentan-3-one can be distinguished by:
View Solution
Hoffmann Bromamide Degradation reaction is given by:
View Solution
Given below is the decomposition of hydrogen peroxide in alkaline medium, which is catalysed by iodide ions:
\[ 2H_2O_2 \xrightarrow{I^-, \; alkaline medium} 2H_2O + O_2 \]
The above reaction takes place in two steps:
Step I: \( H_2O_2 + I^- \longrightarrow H_2O + IO^- \) (slow)
Step II: \( H_2O_2 + IO^- \longrightarrow H_2O + I^- + O_2 \)
Molecularity of Step I and Step II is:
View Solution
Oxidation state of central metal atom in the given complex is:
\[ [Co(NH_3)_4(H_2O)_2]Cl_3 \]
View Solution
A galvanic cell can behave as an electrolytic cell when:
View Solution
Assertion (A): Phenol gives 2,4,6-trinitrophenol on treatment with concentrated \( HNO_3 \) and concentrated \( H_2SO_4 \).
Reason (R): –OH group in phenol is \( m \)-directing.
View Solution
Assertion (A): D (+)-Glucose is dextrorotatory.
Reason (R): Symbol ‘D’ represents its dextrorotatory nature.
View Solution
Assertion (A): Zn, Cd and Hg are not regarded as transition elements.
Reason (R): Zn, Cd and Hg do not have partially filled d-orbitals in their ground state or in any one of their common oxidation states.
View Solution
Assertion (A): Benzene diazonium salt is stable and can be easily stored.
Reason (R): Benzene diazonium chloride decomposes easily.
View Solution
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction, assuming that it does not change with temperature.
\[ (\log 2 = 0.30,\; \log 4 = 0.60) \quad [R = 8.314\; J\, K^{-1}\, mol^{-1}] \]
View Solution
(b)i) Why do amino acids show amphoteric behaviour?
View Solution
(b)(ii) What happens when D-Glucose is treated with hydroxylamine?
View Solution
(i) Write the structure of 1-chloro-4-ethylcyclohexane.
View Solution
(ii) Why is sulphuric acid not used during the reactions of alcohols with KI?
View Solution
(i) Arrange the following in increasing order of their boiling points:
1-chloropropane, 2-chloropropane, 1-chlorobutane
(A) 2-chloropropane \(<\) 1-chloropropane \(<\) 1-chlorobutane
View Solution
(ii) What is an ambident nucleophile? Give one example.
View Solution
(a) Why is \([Ti(H_2O)_6]^{3+}\) coloured?
View Solution
(b) Write IUPAC name of the given complex: \( K_3[Cr(C_2O_4)_3] \)
View Solution
(a) Why are iron pipes usually coated with zinc?
View Solution
(b) Why does mercury cell give a constant voltage throughout its life?
View Solution
(a) Write the mathematical relation between rate constant and half-life period of a zero order reaction.
View Solution
(b) Define Pseudo first order reaction with an example.
View Solution
(a) Why is \( pK_a \) of ClCH\textsubscript{2COOH lower than the \( pK_a \) of CH\textsubscript{3COOH?
View Solution
(b) Write the chemical equation for Hell-Volhard-Zelinsky reaction.
View Solution
Write the chemical equation for the following:
(a) Hydration of propene in presence of an acid
(b) Reaction between Ethyl bromide and C\textsubscript{2H\textsubscript{5ONa
(c) Reaction between Dimethyl ether and Hydrogen iodide
View Solution
Using valence bond theory, predict:
(a) Hybridisation of central metal atom of the complex \([Co(NH_3)_6]^{3+}\)
(b) Its shape and magnetic behaviour
(c) Whether it is a high spin or a low spin complex
[Atomic number: \(CO\) = 27]
(b) Octahedral shape; diamagnetic
(c) Low spin complex
View Solution
Give reasons for the following:
(a) Reduction with iron scrap and HCl is preferred for the preparation of amines from nitro compounds.
(b) Lower aliphatic amines are soluble in water.
(c) Pyridine is used in the acylation reaction of amines.
(d) Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
View Solution
(i) Why do tertiary alkyl halides undergo S\textsubscript{N}1 reaction at a faster rate?
View Solution
(ii) Define Enantiomers.
View Solution
(iii) Why is chloroform stored in dark coloured air tight bottles?
View Solution
(i) Write the major alkene that would be formed by dehydrohalogenation of 2-Bromopentane.
View Solution
(ii) Which would undergo S\textsubscript{N}2 reaction at a faster rate and why?
Given compounds:
Compound A: CH\textsubscript{3–CH\textsubscript{2–Br
Compound B: \quad
\begin{tabular{c
CH\textsubscript{3
\quad|
CH\textsubscript{3–C–Br
\quad|
CH\textsubscript{3
\end{tabular
View Solution
(iii) Why is chlorobenzene less reactive towards nucleophilic substitution reaction?
View Solution
(a) Name the linkage which connects monosaccharide units in polysaccharides.
View Solution
(b) Carbohydrates are classified on the basis of their behaviour on hydrolysis. Write the hydrolysis products of sucrose.
View Solution
(c) Write two differences between Amylose and Amylopectin.
View Solution
What are reducing sugars?
View Solution
Sucrose is dextrorotatory but the mixture obtained after hydrolysis is laevorotatory. Why?
View Solution
(a) 20 mL of a liquid A was mixed with 20 mL of liquid B. The volume of resulting solution was found to be less than 40 mL. What do you conclude from the above data?
View Solution
(b) Which of the following show positive deviation from Raoult’s law?
Carbon disulphide and Acetone;
Phenol and Aniline;
Ethanol and Acetone
View Solution
(c) The vapour pressure of a solution of glucose in water is 750 mm Hg at 100°C. Calculate the mole fraction of solute.
(Vapour pressure of water at 373 K = 760 mm Hg)
View Solution
The boiling point of solution increases when 1 mol of NaCl is added to 1 litre of water while addition of 1 mol of methanol to one litre of water decreases its boiling point. Explain the above observations.
View Solution
(i) A cell and its emf is given below:
Pt (s) \(\mid\) H\(_2\)(g, 1 bar) \(\mid\) H\(^+\)(aq, 1 M) \(\parallel\) Cu\(^{2+}\)(aq, 1 M) \(\mid\) Cu (s)
emf of the cell = +0.34 V
Write the reduction half-reaction at cathode.
View Solution
How is standard Gibbs energy for a reaction related to equilibrium constant?
View Solution
Calculate emf of the given cell:
Mg(s) \(\mid\) Mg\(^{2+}\)(0.1 M) \(\parallel\) Cu\(^{2+}\)(1.0 × 10\(^{-3}\) M) \(\mid\) Cu(s)
Given:
\(E^\circ_{Cu^{2+}/Cu} = +0.34\) V, \quad \(E^\circ_{Mg^{2+}/Mg} = -2.37\) V
(log 100 = 2)
View Solution
State Kohlrausch’s law of independent migration of ions.
Kohlrausch’s law states that:
\textit{At infinite dilution, the molar conductivity of an electrolyte is equal to the sum of the individual contributions of the cation and anion.}
Mathematically:
\[\Lambda_m^\circ = \lambda^\circ_+ + \lambda^\circ_-\]
View Solution
How much electricity in terms of Faraday is required to produce 40 g of Al from molten Al\(_2\)O\(_3\)?
(Given: Atomic mass of Al = 27 u)
View Solution
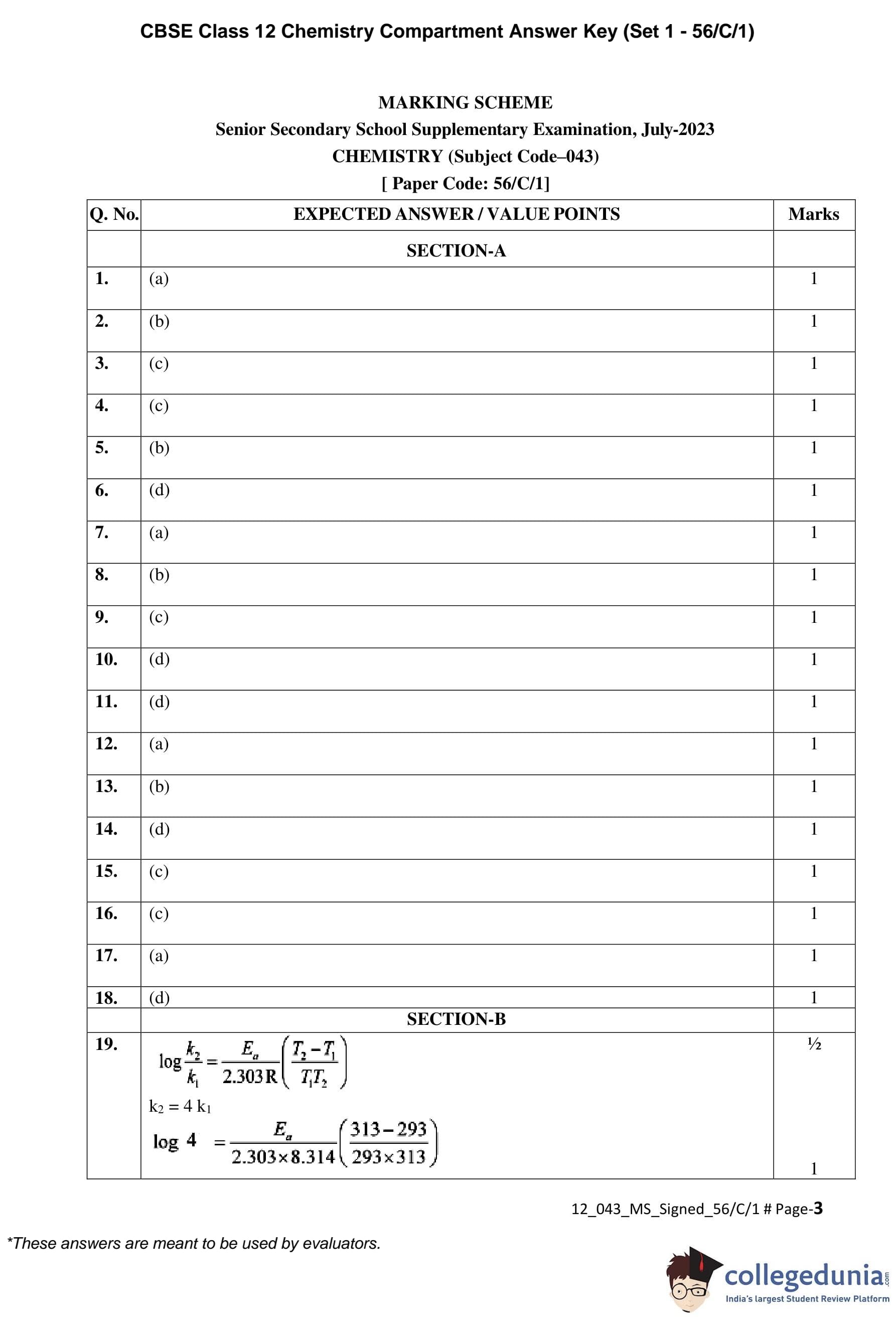
Calculate log \(K_c\) for the following reaction at 298 K:
Zn(s) + Cu\(^{2+}\)(aq) \(\rightleftharpoons\) Zn\(^{2+}\)(aq) + Cu(s)
Given:
\(E^\circ_{Zn^{2+}/Zn} = -0.76 \, V\), \quad \(E^\circ_{Cu^{2+}/Cu} = +0.34 \, V\)
View Solution
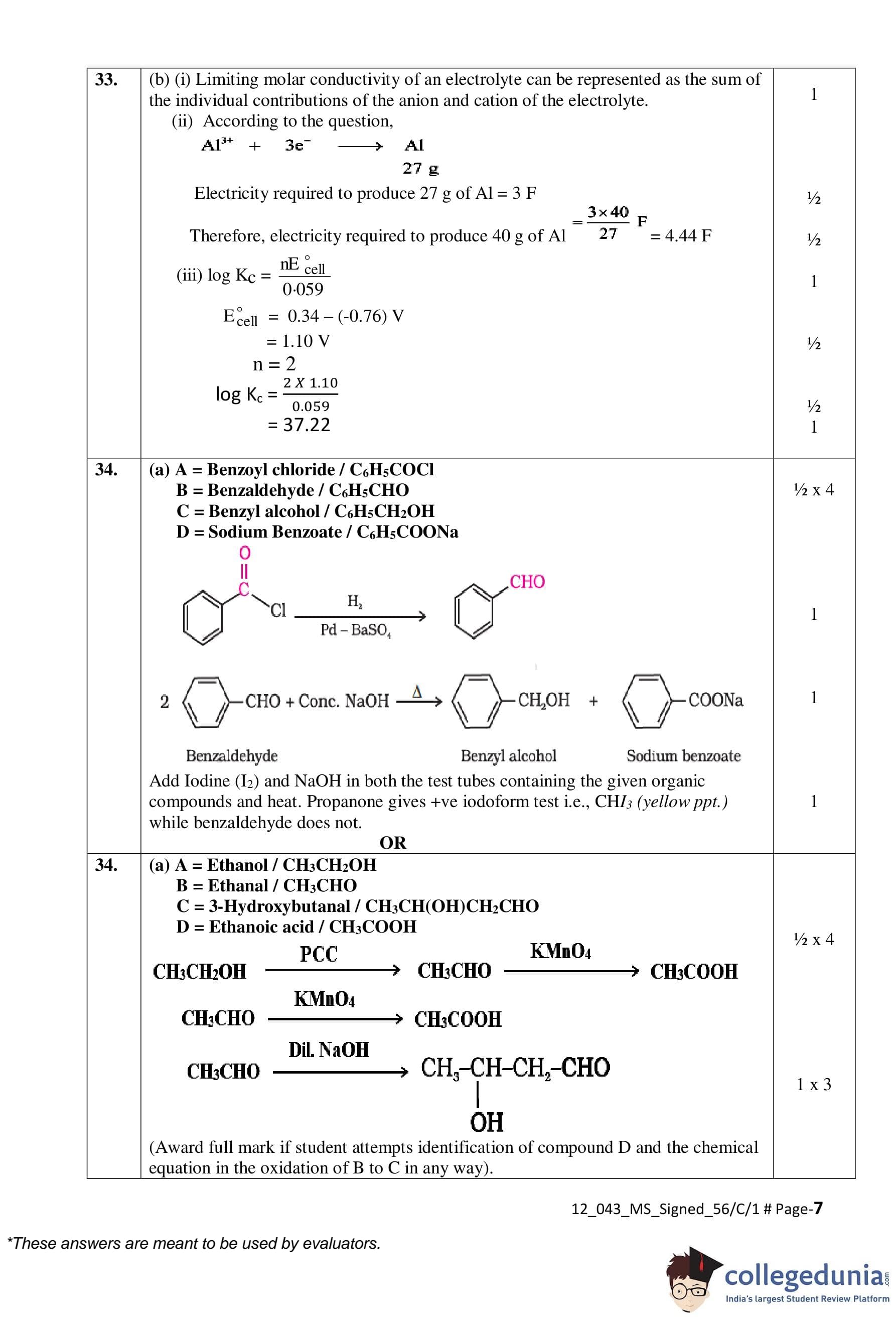
(a) Compound A undergoes Rosenmund reduction to give compound B with molecular formula C\textsubscript{7}H\textsubscript{6}O. Compound B does not give Fehling’s test but reacts with conc. NaOH to give C and D.
Identify A, B, C and D and write all the reactions involved.
Write one chemical test to distinguish between compound B and propanone.
A = Benzoyl chloride (C\textsubscript{6}H\textsubscript{5}COCl)
B = Benzaldehyde (C\textsubscript{6}H\textsubscript{5}CHO)
C = Benzoic acid (C\textsubscript{6}H\textsubscript{5}COOH)
D = Benzyl alcohol (C\textsubscript{6}H\textsubscript{5}CH\textsubscript{2}OH)
View Solution
Compound A with molecular formula (C\textsubscript{2}H\textsubscript{6}O) on oxidation by PCC gives compound B, which on treatment with dilute alkali forms compound C (a β-hydroxy aldehyde). B on oxidation by potassium permanganate forms C.
Identify A, B, C and D and write all the chemical equations involved.
A = Ethanol (CH\textsubscript{3}CH\textsubscript{2}OH)
B = Acetaldehyde (CH\textsubscript{3}CHO)
C = 3-Hydroxybutanal (CH\textsubscript{3}CH(OH)CH\textsubscript{2}CHO)
D = Acetic acid (CH\textsubscript{3}COOH)
View Solution
The chemistry of the actinoids is more complex as compared to the lanthanoids. Why?
Actinoids exhibit more complex chemistry due to the greater number of oxidation states and greater tendency to form coordination compounds than lanthanoids.
View Solution
Why is E\textdegree{} for Mn\textsuperscript{3+}/Mn\textsuperscript{2+} redox couple more positive?
Because Mn\textsuperscript{3+} is unstable in aqueous solution and readily reduces to Mn\textsuperscript{2+}.
View Solution
Why do transition metals form large numbers of complex compounds?
Due to small size, high charge, and availability of vacant d-orbitals for bonding.
View Solution
How does acidified potassium permanganate solution react with Fe\textsuperscript{2+} ions? Write ionic equation.
\[ \text{MnO}_4^- + 8H^+ + 5Fe^{2+} \rightarrow Mn^{2+} + 5Fe^{3+} + 4H_2O \]
View Solution
Calculate the ‘spin only’ magnetic moment of a divalent ion of a metal M in aqueous solution. The atomic number of the metal M is 25.
View Solution



































Comments