CBSE Class 12 Chemistry Question Paper 2024 PDF (Set 2 - 56/5/2) is available for download here. CBSE conducted the Chemistry exam on February 27, 2024, from 10:30 AM to 1:30 PM. The total marks for the theory paper are 70. The question paper contains 20% MCQ-based questions, 40% competency-based questions, and 40% short and long answer type questions.
CBSE Class 12 Chemistry Question Paper 2024 (Set 2 - 56/5/2) with Answer Key
| CBSE Class 12 2024 Chemistry Question Paper with Answer Key | Check Solution |
CBSE Class 12 Chemistry Question Paper 2024 with Solutions
Question 1:
Which of the following species can act as the strongest base?
View Solution
The basicity of a species is determined by its ability to donate electrons (i.e., its nucleophilicity and its tendency to accept a proton). The stronger the conjugate base, the stronger the base. Here’s the analysis of the species:
1. \(OH-\): Hydroxide is a strong base, but it is less basic than alkoxides because it is the conjugate base of water, which is a relatively weak acid.
2. \( C_6H_5O^- \): Phenoxide ion is a weaker base compared to alkoxides because the negative charge on the oxygen is delocalized into the aromatic ring, stabilizing the anion but decreasing the availability of the electron pair for protonation.
3. \(RO-\): Alkoxide ions (such as methoxide, \( CH_3O^- \)) are stronger bases than hydroxide ions because the negative charge is localized on oxygen, and alkyl groups are electron-donating, increasing the basicity.
4. \( O_2N^-\): Nitro group has a strong electron-withdrawing effect, making this species a very weak base.
Thus, \(RO-\) (alkoxide) is the strongest base among these species. Quick Tip: Alkoxide ions are stronger bases than hydroxide and phenoxide due to the electron-donating effect of alkyl groups, which increases the negative charge on oxygen, making it more likely to accept a proton.
Auto-oxidation of chloroform in air and light produces a poisonous gas known as:
View Solution
The auto-oxidation of chloroform \((CHCl_3)\) in the presence of air and light leads to the formation of phosgene \((COCl_2)\), a highly toxic gas. Phosgene is produced when chloroform is exposed to oxygen and ultraviolet light, which breaks down the chloroform and produces phosgene along with other by-products such as hydrogen chloride (HCl).
- Phosgene is used as a chemical warfare agent due to its toxicity, and it can cause severe damage to the lungs when inhaled. Quick Tip: Phosgene is a toxic gas and must be handled carefully, especially in the presence of chloroform and light. The reaction is a classic example of the dangers of auto-oxidation.
Isotonic solutions have the same:
View Solution
Isotonic solutions are solutions that have the same osmotic pressure. Osmotic pressure is the pressure exerted by a solvent when it moves through a semipermeable membrane to balance the concentrations of solute on both sides of the membrane. Isotonic solutions are crucial for maintaining the equilibrium in biological systems, especially for red blood cells.
For two solutions to be isotonic, the concentration of solute particles (ions, molecules) in the two solutions must be the same. This ensures that there is no net movement of water across the membrane, preventing cell shrinkage (crenation) or bursting (lysis).
For example, an isotonic saline solution used in medical treatments has the same osmotic pressure as the human blood plasma, ensuring that the cells do not undergo osmotic stress. Quick Tip: Osmotic pressure is the key property that defines isotonic solutions. This ensures that cells retain their shape by maintaining equal concentrations of solutes inside and outside the cell.
The specific sequence in which amino acids are arranged in a protein is called its:
View Solution
The primary structure of a protein refers to the linear sequence of amino acids in the polypeptide chain. This sequence is encoded by the genetic material (DNA) and is unique for each protein. The amino acids are connected by peptide bonds, which are covalent bonds formed through dehydration reactions between the amino group of one amino acid and the carboxyl group of another.
The sequence of amino acids in the primary structure determines the overall shape of the protein and, consequently, its function. Even a small change (mutation) in the amino acid sequence can drastically alter the function of a protein, leading to diseases such as sickle-cell anemia. Quick Tip: The primary structure is the foundation for all higher-level protein structures (secondary, tertiary, and quaternary). Any change in the amino acid sequence can potentially change the protein's function.
Transition metals are known to make interstitial compounds. Formation of interstitial compounds makes the transition metal:
View Solution
Interstitial compounds are formed when small atoms, such as carbon, hydrogen, or nitrogen, occupy the interstitial spaces between the larger metal atoms in the crystal lattice. This creates a more rigid structure as the smaller atoms obstruct the movement of the metal atoms.
For transition metals, which have relatively large atomic radii, the presence of smaller atoms in the interstitial sites restricts the mobility of the metal atoms, thus increasing the hardness of the metal. For example, the formation of steel (iron-carbon alloy) results in a much harder material than pure iron due to the carbon atoms occupying the interstitial spaces in the iron lattice.
The formation of interstitial compounds typically leads to increased hardness, but the material may become less ductile and more brittle, which makes it harder, not softer. Quick Tip: Interstitial compounds restrict the movement of metal atoms, increasing hardness but often decreasing ductility. This is why steel is harder than pure iron due to the carbon atoms filling interstitial spaces.
The correct name of the given reaction is:
\[ Ar - N_2^+ X^- \xrightarrow{HBr, Cu powder} Ar - Br + N_2 \]
View Solution
The reaction given in the question is an example of the Gatterman reaction. In this reaction, an aryl diazonium salt reacts with hydrobromic acid (\( HBr \)) in the presence of copper powder to yield the corresponding aryl bromide. This reaction is similar to the Sandmeyer reaction but differs in terms of the catalyst used.
Mechanism:
1. The aryl diazonium salt (\( Ar - N_2^+ \)) undergoes reduction in the presence of copper powder.
2. This results in the formation of the corresponding aryl halide (\( Ar - Br \)) with the release of nitrogen gas (\( N_2 \)).
Reaction equation: \[ Ar - N_2^+X^- + HBr + Cu \rightarrow Ar - Br + N_2 + CuX \] Quick Tip: The Gatterman reaction is used for the synthesis of aryl chlorides and bromides from aryl diazonium salts. It provides a convenient method for introducing halogens into the benzene ring.
Visha took 4 test-tubes namely A, B, C \& D containing \( CH_3CH = CH_2 \), \( CH_3CH_2CH = CH_2 \), \( CH_3CH = CH - CH_3 \) and \( (CH_3)_2C = CH_2 \) respectively and tried to convert them into tert-butyl alcohol. She carried out acid catalysed hydration reaction on every alkene. Out of the four test-tubes, the one which will give desired result is:
View Solution
The reaction involves the acid-catalyzed hydration of alkenes, which follows Markovnikov’s rule. According to this rule, the hydroxyl group (\(OH\)) from water will attach to the more substituted carbon in the double bond.
- Test-tube A (CH\(_3\)CH=CH\(_2\)): Propene, which forms isopropanol upon hydration.
- Test-tube B (CH\(_3\)CH\(_2\)CH=CH\(_2\)): But-1-ene, which forms butan-2-ol (not tert-butanol).
- Test-tube C (CH\(_3\)CH=CH-CH\(_3\)): But-2-ene, which forms butan-2-ol.
- Test-tube D ((CH\(_3\))\(_2\)C=CH\(_2\)): 2-Methylpropene, which forms tert-butanol upon hydration.
Since the desired product is tert-butanol, the correct answer is Test-tube D. Quick Tip: In acid-catalyzed hydration, the hydroxyl group attaches to the more substituted carbon in the double bond (Markovnikov’s addition), leading to the formation of the most stable carbocation.
Van’t Hoff factor for \( KCl \) solution assuming the complete dissociation is:
View Solution
Van’t Hoff factor (\(i\)) is given by:
\[ i = \frac{Number of particles after dissociation}{Number of particles before dissociation} \]
For potassium chloride (KCl), it dissociates completely in water as follows:
\[ KCl \rightarrow K^+ + Cl^- \]
Initially, 1 mole of KCl is present, but after complete dissociation, it produces 2 moles of ions (1 mole of \( K^+ \) and 1 mole of \( Cl^- \)).
Thus,
\[ i = \frac{2}{1} = 2. \] Quick Tip: Van’t Hoff factor (\(i\)) represents the number of particles formed per formula unit of solute in solution. Electrolytes like NaCl and KCl dissociate completely in water, leading to \( i = 2 \), while non-electrolytes like glucose have \( i = 1 \).
Dilution affects both conductivity as well as molar conductivity. Effect of dilution on both is as follows:
View Solution
When a solution is diluted, the conductivity (which is the ability of the solution to conduct electricity) typically decreases because the ions are spread out in a larger volume, leading to fewer collisions between the ions and less efficient movement of charge.
However, molar conductivity (which is the conductivity per mole of ions) increases with dilution because as the ions are spread out, they experience less ion pairing, allowing for greater mobility and, therefore, a higher molar conductivity.
This behavior is described by the relation: \[ \Lambda_m = \frac{\kappa}{c} \]
where \( \Lambda_m \) is the molar conductivity, \( \kappa \) is the conductivity, and \( c \) is the concentration of the solution.
Thus, conductivity decreases, while molar conductivity increases as the solution is diluted. Quick Tip: Dilution results in the decrease of conductivity due to fewer ions per unit volume. However, molar conductivity increases because ions move more freely when diluted.
Which of the following cell is used in inverter?
View Solution
The lead storage cell is the most commonly used rechargeable battery in inverters and automobiles. It consists of a lead dioxide (\(PbO_2\)) cathode and a sponge lead (\(Pb\)) anode, both immersed in a sulfuric acid (\(H_2SO_4\)) electrolyte.
The overall reaction in a lead-acid battery during discharge is:
\[ Pb + PbO_2 + 2H_2SO_4 \rightarrow 2PbSO_4 + 2H_2O \]
- The lead storage cell is preferred in inverters due to its high efficiency, rechargeability, and capability to provide a stable power supply.
- Fuel cells are used in spacecraft and vehicles for continuous energy conversion.
- Mercury cells are used in watches and hearing aids due to their small size and stable voltage output.
- Dry cells are used in portable devices like torches and remote controls, but they are not suitable for inverters due to their non-rechargeable nature. Quick Tip: Lead storage cells are rechargeable, making them ideal for applications requiring continuous and stable power supply, such as inverters and automotive batteries.
In which of the following molecules, the C atom marked with an asterisk is chiral?
View Solution
A chiral carbon (asymmetric carbon) is a carbon atom that is bonded to four different groups. To determine which marked carbon atoms are chiral, we analyze each molecule:
- Molecule (I): The carbon marked with an asterisk is bonded to two identical methyl (\( CH_3 \)) groups, making it not chiral.
- Molecule (II): The marked carbon is attached to four different groups (H, Br, Cl, and \( CH_3 \)), making it chiral.
- Molecule (III): The marked carbon is attached to four different groups (\( CH_3CH_2 \), D, I, and \( CH_3 \)), making it chiral.
- Molecule (IV): The marked carbon is attached to four different groups (\( CH_3 \), \( H_3CCH_2 \), H, and D), making it chiral.
Thus, the correct answer is (B) II, III, IV. Quick Tip: A chiral carbon must have four different groups attached to it. If any two groups are the same, the carbon is not chiral.
For the reaction \( A + 2B \rightarrow C + D \), the order of the reaction is:
View Solution
The order of a reaction is defined as the sum of the powers of concentration terms in the rate law expression.
- The given reaction:
\[ A + 2B \rightarrow C + D \]
is only a stoichiometric equation and does not provide information about the rate law.
- The order of a reaction cannot be deduced directly from the balanced chemical equation; it must be determined experimentally through rate measurements.
- Some reactions may follow the stoichiometric coefficients, while others may have different reaction orders due to complex reaction mechanisms.
Thus, the correct answer is (C) can’t be predicted as order is determined experimentally. Quick Tip: The order of a reaction is not necessarily the same as the sum of reactant coefficients in the balanced equation. It must be determined experimentally using methods like the initial rate method.
Assertion (A): p-methoxyphenol is a stronger acid than p-nitrophenol.
Reason (R): Methoxy group shows +I effect whereas nitro group shows -I effect.
View Solution
- Assertion (A) is false: p-Nitrophenol is actually a stronger acid than p-methoxyphenol. The nitro group is an electron-withdrawing group, which stabilizes the negative charge on the conjugate base (phenoxide ion) more effectively, making it a stronger acid.
- Reason (R) is true: The methoxy group exhibits a +I effect (electron-donating), which increases the electron density on the aromatic ring and makes the phenol less acidic, whereas the nitro group exhibits a -I effect (electron-withdrawing), which makes the phenol more acidic. Quick Tip: Electron-withdrawing groups like the nitro group stabilize the conjugate base, making the compound more acidic, whereas electron-donating groups like methoxy make the compound less acidic.
Assertion (A): Inversion of configuration is observed in \(S_N2\) reaction.
Reason (R): The reaction proceeds with the formation of carbocation.
View Solution
- Assertion (A) is true: In an \(S_N2\) reaction, there is an inversion of configuration at the chiral carbon center due to the backside attack of the nucleophile. This is a defining characteristic of the \(S_N2\) mechanism.
- Reason (R) is false: The formation of a carbocation is not a feature of \(S_N2\) reactions. Carbocation formation is associated with the \(S_N1\) mechanism, where the leaving group departs before the nucleophile attacks. Quick Tip: In \(S_N2\) reactions, the reaction mechanism involves a single step where the nucleophile attacks the electrophilic carbon from the opposite side of the leaving group, causing inversion of configuration.
Assertion (A): The units of rate constant of a zero order reaction and rate of reaction are the same.
Reason (R): In zero order reaction, the rate of reaction is independent of concentration of reactants.
View Solution
- Assertion (A) is true: In a zero-order reaction, the rate of reaction is independent of the concentration of reactants. The rate constant and rate of the reaction share the same unit of \( mol L^{-1} s^{-1} \).
- Reason (R) is true: In a zero-order reaction, the rate does not depend on the concentration of the reactant, meaning the rate is constant, and the rate law is written as:
\[ Rate = k[A]^0 = k \]
Thus, the rate of reaction is independent of the concentration, confirming the reason. Quick Tip: In a zero-order reaction, the rate remains constant regardless of the concentration of reactants, and the units of the rate constant and the rate of reaction are the same.
Assertion (A): Zr and Hf are of almost similar atomic radii.
Reason (R): This is due to Lanthanoid contraction.
View Solution
- Assertion (A) is true: Zirconium (Zr) and Hafnium (Hf) are in the same group of the periodic table, and their atomic radii are almost the same.
- Reason (R) is also true: The reason for this similarity is the Lanthanoid contraction, a phenomenon where the atomic radii of the lanthanoids decrease due to poor shielding of the nuclear charge by the f-electrons. As a result, elements following the lanthanoids (like Zr and Hf) experience a similar effective nuclear charge, leading to nearly the same atomic radii. Quick Tip: Lanthanoid contraction refers to the gradual decrease in size of the elements in the lanthanide series, affecting the elements in the same period after them, such as Zr and Hf.
Define the following terms:
(a) Faraday’s second law of electrolysis
(b) Corrosion
View Solution
(a) Faraday’s second law of electrolysis:
Faraday’s second law of electrolysis states that when the same quantity of electricity is passed through different electrolytes, the mass of substances deposited at the electrodes is directly proportional to their equivalent weights.
This law can be expressed mathematically as: \[ \frac{m_1}{m_2} = \frac{E_1}{E_2} \]
where:
- \( m_1 \) and \( m_2 \) are the masses of substances deposited,
- \( E_1 \) and \( E_2 \) are the equivalent weights of the substances.
Explanation:
- Faraday’s laws of electrolysis are based on the principle that the amount of a substance deposited during electrolysis is proportional to the charge passed.
- The second law specifically relates to different substances, stating that for the same quantity of charge, heavier substances deposit in proportion to their equivalent weights.
- This law is particularly useful in electroplating and purification of metals.
Example:
If the same amount of charge is passed through copper and silver solutions, the mass of silver deposited will be in the ratio of their equivalent weights to copper. Quick Tip: Faraday’s second law is useful in electroplating industries and metal refining, where different substances need to be deposited in exact proportions.
Resistance of a conductivity cell filled with 0.2 mol L\(^{-1}\) KCl solution is 200 ohm. If the resistance of the same cell when filled with 0.05 mol L\(^{-1}\) KCl solution is 620 ohm, calculate the conductivity and molar conductivity of 0.05 mol L\(^{-1}\) KCl solution. The conductivity of 0.2 mol L\(^{-1}\) KCl solution is 0.0248 S cm\(^{-1}\).
View Solution
Step 1: Calculate the Cell Constant
The cell constant (G*) is given by: \[ Cell constant = Conductivity \times Resistance \]
Substituting values for 0.2 mol L\(^{-1}\) KCl solution: \[ G^* = 0.0248 \times 200 = 4.96 \, cm^{-1} \]
Step 2: Calculate Conductivity of 0.05 mol L\(^{-1}\) KCl Solution
Using the relation: \[ Conductivity (\kappa) = \frac{Cell Constant (G*)}{Resistance (R)} \] \[ \kappa = \frac{4.96}{620} = 0.008 \, S cm^{-1} \]
Step 3: Calculate Molar Conductivity
Molar conductivity \( \Lambda_m \) is given by: \[ \Lambda_m = \kappa \times \frac{1000}{C} \]
where \( C = 0.05 \) mol L\(^{-1}\): \[ \Lambda_m = 0.008 \times \frac{1000}{0.05} = 160 \, S cm^2 mol^{-1} \] Quick Tip: The cell constant remains the same for different solutions in the same conductivity cell. Conductivity decreases with dilution, but molar conductivity increases as the ions move more freely.
Show that in case of a first-order reaction, the time taken for completion of 99% reaction is twice the time required for 90% completion of the reaction. (log 10 = 1)
View Solution
The integrated rate equation for a first-order reaction is: \[ t = \frac{2.303}{k} \log \frac{[A]_0}{[A]} \]
Time required for 99% completion:
For 99% completion, the remaining concentration is 1% of the initial concentration, so: \[ t_{99%} = \frac{2.303}{k} \log \frac{100}{1} \] \[ t_{99%} = \frac{2.303}{k} \times 2 \]
Time required for 90% completion:
For 90% completion, the remaining concentration is 10% of the initial concentration, so: \[ t_{90%} = \frac{2.303}{k} \log \frac{100}{10} \] \[ t_{90%} = \frac{2.303}{k} \times 1 \]
Thus, \[ \frac{t_{99%}}{t_{90%}} = \frac{\frac{2.303}{k} \times 2}{\frac{2.303}{k} \times 1} = 2 \]
which proves that the time taken for 99% completion is twice the time required for 90% completion. Quick Tip: For a first-order reaction, the time required for a higher percentage completion follows a logarithmic relationship, leading to a predictable ratio of completion times.
(a) Carry out the following conversions:
(i) Nitrobenzene to Aniline.
(ii) Aniline to Phenol.
View Solution
(i) Nitrobenzene to Aniline:
To convert nitrobenzene to aniline, a reduction reaction is required. This can be achieved by reducing the nitro group (-NO\(_2\)) using reducing agents such as iron (Fe) and hydrochloric acid (HCl) or catalytic hydrogenation: \[ C_6H_5NO_2 + 3H_2 \xrightarrow{Fe/HCl} C_6H_5NH_2 \]
This process reduces the nitro group \((-NO_2)\) to an amino group \((-NH_2)\), resulting in the formation of aniline.
(ii) Aniline to Phenol:
To convert aniline \((C_6H_5NH_2)\) to phenol \((C_6H_5OH)\), the amino group must be oxidized to a hydroxyl group. This can be done using an oxidizing agent such as bromine in the presence of water: \[ C_6H_5NH_2 + Br_2 \xrightarrow{H_2O} C_6H_5OH + 2HBr \]
The reaction replaces the amino group \((-NH_2)\) with a hydroxyl group (-OH), forming phenol. Quick Tip: Reduction of nitrobenzene to aniline requires an electron-donating reduction, while the conversion of aniline to phenol involves oxidation using bromine in water.
(b) (i) Write a chemical test to distinguish between Dimethyl amine and Ethanamine.
View Solution
To distinguish Dimethylamine \((CH_3NH_2)\) from Ethanamine \((C_2H_5NH_2)\), we can use the Nitrous acid test.
- Test for Dimethylamine:
When Dimethylamine is treated with nitrous acid \((HNO_2)\), it forms a blue solution due to the formation of a methyl diazonium salt. The reaction is as follows:
\[ CH_3NH_2 + HNO_2 \to CH_3N_2^+ + H_2O \]
The blue color indicates the presence of Dimethylamine.
- Test for Ethanamine:
When Ethanamine is treated with nitrous acid, it forms ethanol and nitrogen gas, and there is no color formation:
\[ C_2H_5NH_2 + HNO_2 \to C_2H_5OH + N_2 + H_2O \]
Hence, Ethanamine does not produce a color change.
Thus, Dimethylamine gives a blue color with nitrous acid, while Ethanamine does not. Quick Tip: Dimethylamine forms a blue-colored solution with nitrous acid, distinguishing it from Ethanamine, which produces no such color.
(b) (ii) Write the product formed when benzene diazonium chloride is treated with KI.
View Solution
When benzene diazonium chloride \((C_6H_5N_2^+Cl^-)\) is treated with potassium iodide (KI), the diazonium group undergoes a nucleophilic substitution reaction with the iodide ion \((I^-)\). The product formed is iodobenzene \((C_6H_5I)\). The reaction is as follows:
\[ C_6H_5N_2^+Cl^- + KI \to C_6H_5I + N_2 + KCl \]
This reaction is an example of a diazonium substitution reaction, where the diazonium ion is replaced by an iodine atom, forming iodobenzene. Quick Tip: Benzene diazonium chloride reacts with potassium iodide to form iodobenzene, which is a useful halogenation reaction in organic synthesis.
Classify the following sugars into monosaccharides and disaccharides: Fructose, Glucose, Lactose, and Maltose.
View Solution
- Monosaccharides:
- Fructose and Glucose are monosaccharides, meaning they consist of a single sugar unit and cannot be hydrolyzed into simpler sugars. Both are important sources of energy in the body and are involved in the synthesis of larger carbohydrates.
- Disaccharides:
- Lactose and Maltose are disaccharides, consisting of two monosaccharide units linked by a glycosidic bond. Lactose is composed of one molecule of glucose and one of galactose, while maltose is made up of two glucose molecules. Quick Tip: Monosaccharides are simple sugars like glucose and fructose, while disaccharides are made of two monosaccharide units, such as maltose (glucose-glucose) and lactose (glucose-fructose).
Give the structure of the major product expected from the following reactions:
(a) Reaction of Ethanal with methyl-magnesium bromide followed by hydrolysis.
(b) Hydration of But-1-ene in the presence of dilute sulfuric acid.
(c) Reaction of phenol with bromine water.
View Solution
(a) Reaction of Ethanal with Methyl-Magnesium Bromide:
- Ethanal (CH\(_3\)CHO) reacts with methylmagnesium bromide (CH\(_3\)MgBr) in an addition reaction to form an intermediate.
- Upon acidic hydrolysis, the intermediate gives 2-propanol (CH\(_3\)CH(OH)CH\(_3\)).
- The reaction follows: \[ CH_3CHO + CH_3MgBr \rightarrow CH_3CH(OMgBr)CH_3 \] \[ CH_3CH(OMgBr)CH_3 + H_2O \rightarrow CH_3CH(OH)CH_3 + Mg(OH)Br \]
(b) Hydration of But-1-ene in Dilute Sulfuric Acid:
- But-1-ene (CH\(_3\)CH\(_2\)CH=CH\(_2\)) undergoes acid-catalyzed hydration, following Markovnikov’s rule.
- The product is butan-2-ol (CH\(_3\)CH(OH)CH\(_2\)CH\(_3\)).
- The reaction follows: \[ CH_3CH_2CH=CH_2 + H_2O \xrightarrow{H_2SO_4} CH_3CH(OH)CH_2CH_3 \]
(c) Reaction of Phenol with Bromine Water:
- Phenol (C\(_6\)H\(_5\)OH) reacts with bromine water (Br\(_2\)) in a substitution reaction.
- The hydroxyl (-OH) group activates the benzene ring, directing bromine to the ortho and para positions.
- The product is 2,4,6-tribromophenol with the structure:
\[ C_6H_2(OH)Br_3 \]
Quick Tip: 1. Grignard reagents react with aldehydes/ketones to form alcohols.
2. Hydration of alkenes follows Markovnikov's rule (H attaches to the carbon with more hydrogens).
3. Phenol reacts with bromine water to form 2,4,6-tribromophenol.
A compound ‘X’ with molecular formula \( C_3H_9N \) reacts with \( C_6H_5SO_2Cl \) to give a solid, insoluble in alkali. Identify ‘X’ and give the IUPAC name of the product. Write the reaction involved.
View Solution
The given molecular formula \( C_3H_9N \) corresponds to an amine. The reaction with benzene sulfonyl chloride (\( C_6H_5SO_2Cl \)) leads to the formation of a sulfonamide derivative, which is insoluble in alkali.
The reaction follows:
\[ C_2H_5-NH-CH_3 + C_6H_5SO_2Cl \rightarrow C_6H_5SO_2N(C_2H_5)(CH_3) + HCl \]
- Here, \( X \) is identified as N-Ethylmethylamine.
- The IUPAC name of the product is N-Ethyl-N-methylbenzenesulfonamide.
- The sulfonamide formed is insoluble in alkali due to the absence of an acidic hydrogen. Quick Tip: Sulfonamides are generally \textbf{insoluble in alkali} due to the strong electron-withdrawing nature of the sulfonyl group, making the nitrogen non-basic.
The rate constant of a reaction quadruples when the temperature changes from 300 K to 320 K. Calculate the activation energy for this reaction. \[ [log 2 = 0.30, log 4 = 0.60, 2.303 R = 19.15 J K^{-1} mol^{-1}] \]
View Solution
Using the Arrhenius equation: \[ \log \left( \frac{k_2}{k_1} \right) = \frac{E_a}{2.303 R} \left( \frac{1}{T_1} - \frac{1}{T_2} \right) \]
Given: \[ \frac{k_2}{k_1} = 4, \quad T_1 = 300 \, K, \quad T_2 = 320 \, K, \quad \log 4 = 0.60 \]
Substituting the given values into the equation: \[ \log 4 = \frac{E_a}{2.303 \times 19.15} \left( \frac{1}{300} - \frac{1}{320} \right) \] \[ 0.60 = \frac{E_a}{2.303 \times 19.15} \times \left( \frac{320 - 300}{300 \times 320} \right) \] \[ 0.60 = \frac{E_a}{19.15 \times 2.303} \times \frac{20}{96000} \] \[ E_a = \frac{0.60 \times 19.15 \times 2.303 \times 96000}{20} \] \[ E_a = 55.152 \, kJ mol^{-1} \] Quick Tip: The Arrhenius equation relates rate constant (k) to activation energy (E\(_a\)).
For reactions where k quadruples, use log 4 = 0.60 to simplify calculations.
Write IUPAC names of the following coordination compounds:
(a) \( K_3[Fe(CN)_6] \)
(b) \( [Pt(en)_2Cl_2]^{2+} \)
(c) \( [Co(NH_3)_4Cl(ONO)]Cl \)
(d) \( [Zn(OH)_4]^{2-} \)
View Solution
(a) The complex ion consists of an iron metal center coordinated with six cyanide ligands. Since cyanide is a negative ligand, the complex is named as "hexacyanidoferrate." The oxidation state of iron is determined by balancing charges: \[ K_3[Fe(CN)_6] \] \[ 3(+1) + x + 6(-1) = 0 \] \[ 3 + x - 6 = 0 \Rightarrow x = +3 \]
Thus, the name is Potassium hexacyanidoferrate(III).
(b) The complex contains platinum as the central metal coordinated with two ethane-1,2-diamine (en) ligands and two chloride ligands. Since ethane-1,2-diamine is a neutral ligand, and chloride contributes a charge of -1 each, the oxidation state of platinum is: \[ x + 2(0) + 2(-1) = +2 \] \[ x - 2 = +2 \Rightarrow x = +4 \]
Thus, the name is Dichloridobis(ethane-1,2-diamine)platinum(IV) ion.
(c) The cobalt complex contains four ammonia ligands (neutral), one chloride ligand (-1), and one nitrito ligand (-1). The oxidation state of cobalt is: \[ x + 4(0) + (-1) + (-1) = 0 \] \[ x - 2 = 0 \Rightarrow x = +3 \]
The ONO ligand is written as nitrito-O, indicating the oxygen donor form. Thus, the name is Tetraamminechloridonitrito-O-cobalt(III) chloride.
(d) The zinc complex contains four hydroxide ligands (-1 each). The oxidation state of zinc is: \[ x + 4(-1) = -2 \] \[ x - 4 = -2 \Rightarrow x = +2 \]
Thus, the name is Tetrahydroxidozincate(II) ion. Quick Tip: When naming coordination compounds:
- Ligands are named in alphabetical order.
- Anionic ligands end with "-o" (e.g., cyanide → cyanido).
- Neutral ligands retain their usual names except for aqua \((H_2O)\), ammine \((NH_3)\), and carbonyl (CO). - The oxidation state of the central metal is given in Roman numerals in parentheses.
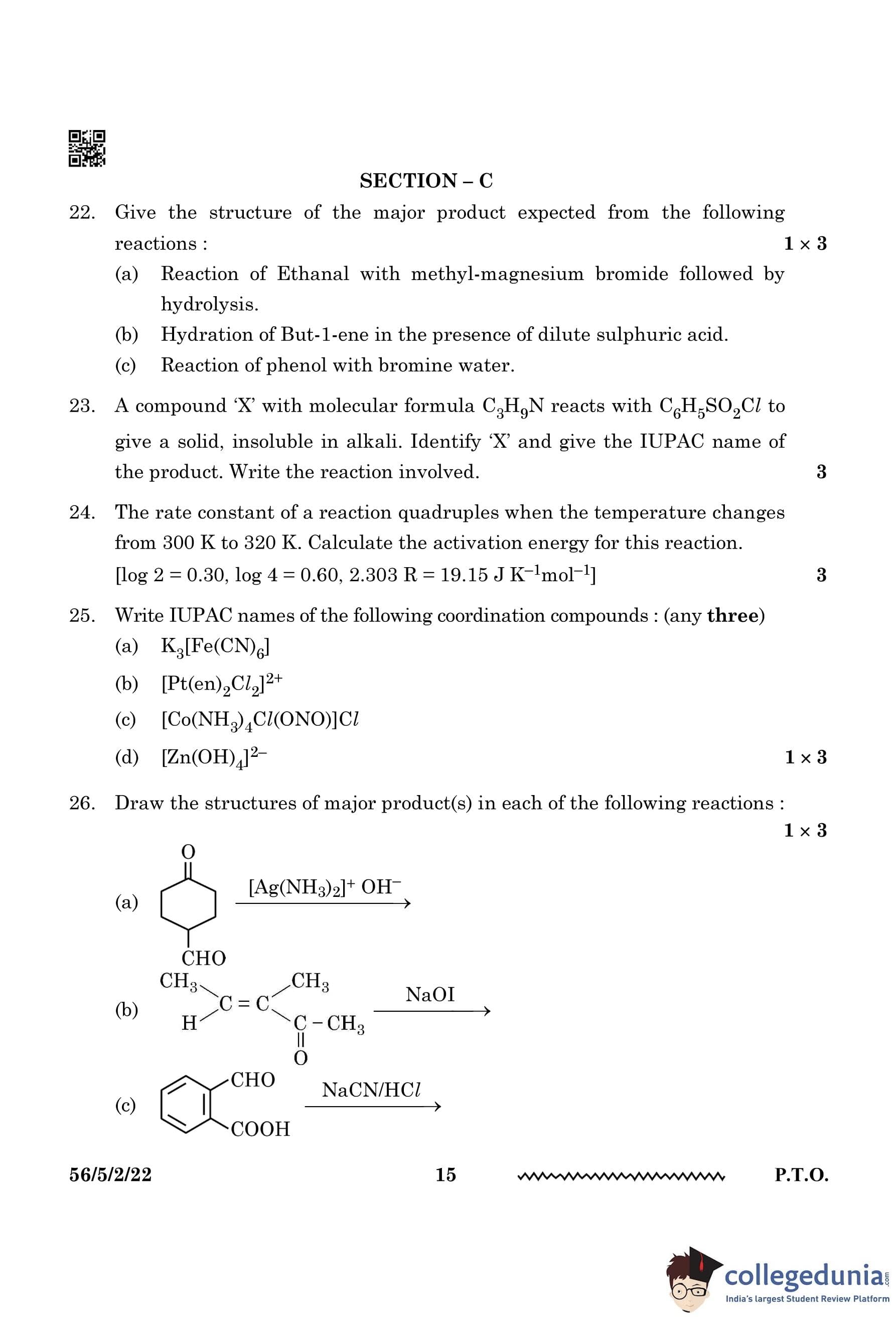
Draw the structures of major product(s) in each of the following reactions:
View Solution
(a) Reaction with \([Ag(NH_3)_2]^+ OH^-\) (Tollens' Test)
- The given compound Cyclohexanone undergoes oxidation by Tollens’ reagent.
- The aldehyde group in Cyclohexanone is oxidized to form a carboxylate anion.
- The reaction mechanism follows: \[ Cyclohexanone + [Ag(NH_3)_2]^+ + OH^- \rightarrow Cyclohexanone carboxylate + Ag + NH_3 + H_2O \]
Major Product: Cyclohexanone carboxylate ion.
(b) Iodoform Reaction with NaOI
- The given compound 4-Methylpent-3-en-2-one contains a methyl ketone \((-COCH_3)\) group.
- In the presence of NaOI, the iodoform test occurs, where the methyl ketone group is cleaved.
- This leads to the formation of Sodium propionate \((CH_3CH=CHCOONa)\) and Iodoform \((CHI_3)\).
Major Products: \[ Sodium propionate (CH_3CH=CHCOONa) + Iodoform (CHI_3) \]
(c) Cyanohydrin Formation with NaCN/HCl
- The given compound p-Carboxybenzaldehyde reacts with NaCN/HCl.
- The cyanohydrin reaction occurs, where the cyanide ion \((CN^)\) attacks the carbonyl group.
- The aldehyde (-CHO) is converted into a cyanohydrin (-CH(OH)CN).
Major Product: \[ 4-Carboxybenzaldehyde cyanohydrin (p-Carboxybenzylcyanohydrin) \] Quick Tip: 1. Tollens' test oxidizes aldehydes to carboxylate ions.
2. Iodoform test cleaves methyl ketones, forming carboxylates and iodoform \((CHI_3)\).
3. Cyanohydrin formation adds a cyanide (-CN) to an aldehyde, producing a cyanohydrin (-CH(OH)CN).
Calculate the emf of the following cell: \[ Ni(s) + 2Ag^+(0.01 M) \rightarrow Ni^{2+} (0.1 M) + 2Ag(s) \]
Given that \(E^\circ_{cell}\) = 1.05 V, log 10 = 1.
View Solution
Using the Nernst equation: \[ E_{cell} = E^\circ_{cell} - \frac{0.059}{n} \log \frac{[Ni^{2+}]}{[Ag^{+}]^2} \]
Substituting the given values: \[ E_{cell} = 1.05 - \frac{0.059}{2} \log \frac{(0.1)}{(0.01)^2} \] \[ E_{cell} = 1.05 - \frac{0.059}{2} \times \log (0.1/0.0001) \] \[ E_{cell} = 1.05 - \frac{0.059}{2} \times \log (1000) \] \[ E_{cell} = 1.05 - \frac{0.059}{2} \times 3 \] \[ E_{cell} = 1.05 - (0.059 \times 1.5) \] \[ E_{cell} = 1.05 - 0.0885 \] \[ E_{cell} = 0.9615 V \] Quick Tip: 1. The Nernst equation relates cell potential to ion concentration.
2. The logarithmic term uses log base 10, which simplifies calculations.
3. For correct units, express the final answer in volts (V).
Account for the following:
(a) Haloalkanes react with NaCN to form both cyanides and isocyanides.
View Solution
When haloalkanes react with sodium cyanide (NaCN), cyanide (\( CN^- \)) acts as a nucleophile. Cyanide is an ambidentate nucleophile, meaning it has two possible atoms (carbon and nitrogen) that can donate electrons. Depending on the conditions, the cyanide ion can attack the carbon atom (forming a nitrile, i.e., cyanide) or the nitrogen atom (forming an isocyanide, i.e., isonitrile). The reaction typically proceeds through an \( S_N2 \)-type mechanism, where the nucleophile attacks the carbon of the alkyl halide, leading to the substitution of the halide with the cyanide group. The product formed depends on which atom (carbon or nitrogen) is involved in the nucleophilic attack. Quick Tip: - Cyanide is an ambidentate nucleophile, meaning it can attack through either the carbon or nitrogen atom. The product depends on which atom is involved in the attack.
- The type of product (cyanide or isocyanide) formed depends on the reaction conditions and the nucleophile's choice of atom.
(b) Haloarenes do not undergo nucleophilic substitution reaction easily:
View Solution
Haloarenes are relatively less reactive towards nucleophilic substitution reactions. This is primarily due to resonance effects. The lone pair of electrons on the halogen in the aryl group participates in resonance with the aromatic ring, which creates partial double bonds between the carbon and halogen. This resonance effect reduces the partial positive charge on the carbon atom attached to the halogen, making it less susceptible to attack by nucleophiles. Furthermore, the bond between the carbon and halogen in haloarenes is stronger due to the resonance stabilization of the aryl group. As a result, haloarenes are generally less reactive than alkyl halides when it comes to nucleophilic substitution reactions. Quick Tip: - The resonance stabilization in haloarenes makes the carbon-halogen bond stronger and less likely to undergo nucleophilic substitution.
- Resonance reduces the partial positive charge on the carbon atom, making it less susceptible to attack by nucleophiles.
(c) Benzyl chloride gives \( S_N1 \) reaction:
View Solution
Benzyl chloride undergoes the \( S_N1 \) (unimolecular nucleophilic substitution) reaction because the benzyl carbocation (formed by the departure of the chloride ion) is resonance-stabilized. The positive charge formed on the benzyl carbocation is delocalized onto the aromatic ring, which significantly stabilizes the intermediate. This stabilization makes the carbocation relatively stable and more likely to form, making the reaction proceed through the \( S_N1 \) mechanism. In the \( S_N1 \) mechanism, the leaving group (in this case, chloride) departs first, forming a carbocation intermediate. Then, the nucleophile (such as water or an alcohol) attacks the carbocation, leading to the formation of the final product. Quick Tip: - The stability of the carbocation is key in \( S_N1 \) reactions. Benzyl carbocation is stabilized by resonance with the aromatic ring, making the \( S_N1 \) pathway more favorable.
- The \( S_N1 \) reaction involves the formation of a carbocation intermediate before the nucleophile attacks, while in \( S_N2 \), the nucleophile attacks simultaneously with the leaving group.
Certain organic compounds are required in small amounts in our diet but their deficiency causes specific disease. These compounds are called vitamins. Most of the vitamins cannot be synthesized in our body but plants can synthesize almost all of them. So they are considered as essential food factors. However, the bacteria of the gut can produce some of the vitamins required by us. All the vitamins are generally available in our diet. The term ‘vitamin’ was coined from the words vital + amine, since the earlier identified compounds had amino group. Vitamins are classified into two groups depending upon their solubility in water or fat namely-fat soluble vitamins and water soluble vitamins.
Question 29:
(a) What is the other name of vitamin B\textsubscript{6}?
View Solution
Understanding Vitamin B\textsubscript{6}:
Vitamin B\textsubscript{6, also known as Pyridoxine, is a water-soluble vitamin that plays a crucial role in protein metabolism, neurotransmitter synthesis, and hemoglobin production. It is essential for brain development and function, as well as for the immune system. Quick Tip: Vitamin B\textsubscript{6} is found in foods such as bananas, poultry, fish, and fortified cereals. It helps prevent anemia and supports neurological function.
(b) Name the vitamin whose deficiency causes increased blood clotting time.
View Solution
Role of Vitamin K in Blood Clotting:
Vitamin K is a fat-soluble vitamin essential for the synthesis of clotting factors in the liver. It plays a vital role in blood coagulation by activating proteins that help in clot formation.
Deficiency and Its Effect:
A deficiency in Vitamin K leads to prolonged blood clotting time, which can cause excessive bleeding (hemorrhaging). This condition is particularly dangerous for newborns, as they have low vitamin K stores at birth. Quick Tip: Vitamin K is found in green leafy vegetables such as spinach and kale. It is also synthesized by gut bacteria.
(c) Xerophthalmia is caused by the deficiency of which vitamin? Give two sources of this vitamin.
View Solution
Understanding Vitamin A and Its Role:
Vitamin A is a fat-soluble vitamin that plays a key role in maintaining vision, immune function, and cell growth. It is present in two forms:
- Retinol (animal-based): Found in liver, fish oils, and dairy products.
- Beta-carotene (plant-based): Found in carrots, sweet potatoes, and spinach.
Vitamin A Deficiency and Xerophthalmia:
Xerophthalmia is a severe eye disorder caused by Vitamin A deficiency. It leads to:
- Night blindness (difficulty seeing in low light).
- Dryness of the conjunctiva and cornea, which can lead to irreversible blindness if untreated.
Food Sources of Vitamin A:
- Animal sources: Fish liver oil, egg yolk, dairy products.
- Plant sources: Carrots, sweet potatoes, spinach. Quick Tip: Vitamin A is crucial for eye health and immune function. A diet rich in carrots, spinach, and dairy products can help prevent deficiencies.
Question 29:
(c) Why can’t vitamin C be stored in our body? Name the disease caused by the deficiency of this vitamin.
View Solution
Water-Solubility of Vitamin C:
Vitamin C (Ascorbic Acid) is a water-soluble vitamin, meaning it dissolves in water and is not stored in the body. Since it is not retained for long, it must be regularly consumed through diet.
Deficiency and Scurvy:
Scurvy is caused by a deficiency of Vitamin C and is characterized by:
- Bleeding gums and loose teeth.
- Joint pain and muscle weakness.
- Delayed wound healing and anemia.
Why Vitamin C is Not Stored:
Being water-soluble, excess Vitamin C is excreted through urine. This prevents toxicity but also means that regular dietary intake is essential. Quick Tip: Vitamin C is an antioxidant that supports the immune system and collagen formation. Citrus fruits, strawberries, and bell peppers are rich sources.
Question 30:
The oxidation number of the central atom in a complex is defined as the charge it would carry if all the ligands are removed along with the electron pairs that are shared with the central atom. Similarly, the charge on the complex is the sum of the charges of the constituent parts i.e., the sum of the charges on the central metal ion and its surrounding ligands. Based on this, the complex is called neutral if the sum of the charges of the constituents is equal to zero. However, for an anion or cationic complex, the sum of the charges of the constituents is equal to the charge on the coordination sphere.
Based on the above information, answer the following questions:
(a) Define ambidentate ligand with an example.
View Solution
(a) Ambidentate Ligand
- A ligand that has two different donor atoms and can bind through either of the two is called an ambidentate ligand.
- Examples:
- NO\(_2^-\) (Nitrito) – Can coordinate via N or O.
- SCN\(^-\) (Thiocyanato) – Can coordinate via S or N.
- CN\(^-\) (Cyanide) – Can coordinate via C or N. Quick Tip: Ambidentate ligands have two possible donor atoms, allowing them to coordinate in multiple ways.
Examples: NO\(_2^-\) (N/O), SCN\(^-\) (S/N), CN\(^-\) (C/N).
(b) What type of isomerism is shown by [Co(NH\(_3\))\(_5\)Cl]SO\(_4\) and [Co(NH\(_3\))\(_5\)SO\(_4\)]Cl ?
View Solution
(b) Type of Isomerism
- The given complexes [Co(NH\(_3\))\(_5\)Cl]SO\(_4\) and [Co(NH\(_3\))\(_5\)SO\(_4\)]Cl show ionization isomerism.
- Reason: In one complex, SO\(_4^{2-}\) is present inside the coordination sphere, whereas in the other, Cl\(^-\) is inside. Quick Tip: Ionization Isomerism occurs when anions or cations swap positions inside and outside the coordination sphere.
(c) Define Chelate effect. How does it affect the stability of a complex?
View Solution
(c) Chelate Effect and Stability
- When a bi-dentate or polydentate ligand binds to a metal ion, it forms a ring structure, known as a chelate.
- Chelate Effect: The formation of a chelate ring increases the stability of the complex due to:
1. Entropic advantage – Reduces the number of free molecules, increasing disorder.
2. Stronger binding – Multi-point attachment prevents ligand displacement. Quick Tip: Chelation enhances stability by forming ring structures around the metal ion. The greater the chelation, the more stable the complex.
Question 30:
(c) Find the coordination number and oxidation state of chromium in Na\(_3\)[Cr(C\(_2\)O\(_4\))\(_3\)].
View Solution
(c) Coordination Number and Oxidation State of Chromium
- Given complex: Na\(_3\)[Cr(C\(_2\)O\(_4\))\(_3\)]
- Step 1: Coordination Number
- C\(_2\)O\(_4^{2-}\) (Oxalate) is a bidentate ligand (each binds at 2 sites).
- There are three oxalate ligands, contributing \(3 \times 2 = 6\) coordination sites.
- Thus, the coordination number of Cr = 6.
- Step 2: Oxidation State of Cr
- The total charge on the complex ion [Cr(C\(_2\)O\(_4\))\(_3\)]\(^{3-}\) is -3.
- The charge on each oxalate ligand (C\(_2\)O\(_4^{2-}\)) is -2.
- Let the oxidation state of Cr be x.
\[ x + 3(-2) = -3 \]
\[ x - 6 = -3 \]
\[ x = +3 \]
Answer: \[ Coordination Number = 6, \quad Oxidation State = +3 \] Quick Tip: - Oxalate (C\(_2\)O\(_4^{2-}\)) is a bidentate ligand → Each contributes two coordination sites.
- Charge balancing helps determine oxidation states.
(a) An organic compound (A) with the molecular formula \( C_9H_{10}O \) forms 2, 4-DNP derivative, reduces Fehling solution and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzene dicarboxylic acid.
(i) Identify the compound (A) and write its IUPAC name.
View Solution
(i) The compound (A) has the molecular formula \( C_9H_{10}O \) and shows characteristics of an aromatic aldehyde. Since it undergoes Cannizzaro reaction (which occurs in non-enolizable aldehydes), it must be an aldehyde without \(\alpha\)-hydrogen atoms. The structure that fits these properties is 2-ethylbenzaldehyde.
(a) (ii) Write the reaction of compound (A) with
(1) 2, 4-Dinitrophenyl hydrazine and
(2) Fehling solution.
View Solution
(ii) (1) Reaction with 2,4-Dinitrophenylhydrazine:
\[ C_9H_{10}O + H_2N-NH(NO_2)_2 \rightarrow C_9H_{10}=N-N(NO_2)_2 + H_2O \]
2-Ethylbenzaldehyde reacts with 2,4-DNP reagent to form a yellow-orange precipitate of 2,4-DNP derivative.
(2) Fehling’s Solution Test:
Aromatic aldehydes generally do not give Fehling's test, but if attempted, some aliphatic aldehyde impurities may react. Fehling’s solution is reduced by aliphatic aldehydes, forming a brick-red precipitate of \( Cu_2O \).
(a) (iii) Write the equation of compound (A) when it undergoes Cannizzaro reaction.
View Solution
(iii) Cannizzaro Reaction:
Since 2-ethylbenzaldehyde lacks an \(\alpha\)-hydrogen, it undergoes disproportionation in a strong base via the Cannizzaro reaction, forming 2-ethylbenzoic acid and 2-ethylbenzyl alcohol. Quick Tip: Aldehydes with no \(\alpha\)-hydrogen undergo Cannizzaro reaction in the presence of a strong base. Aromatic aldehydes generally do not give Fehling’s test, but aliphatic aldehydes do.
(b) (i) Account for the following:
(1) The alpha (\(\alpha\))-hydrogens of aldehydes and ketones are acidic in nature.
View Solution
(1) The acidity of \(\alpha\)-hydrogens in aldehydes and ketones is due to the electron-withdrawing nature of the carbonyl group, which stabilizes the enolate ion formed after deprotonation. Additionally, resonance stabilization of the conjugate base further enhances the acidity.
(b) (i) (2) Oxidation of aldehydes is easier than ketones.
View Solution
(2) Aldehydes undergo oxidation more readily than ketones because they have a hydrogen atom attached to the carbonyl carbon, making the cleavage of the C-H bond easier. In contrast, ketones require the breaking of a C-C bond, which is more difficult. Quick Tip: The presence of an \(\alpha\)-hydrogen in aldehydes and ketones allows for tautomerization to the enol form, which plays a crucial role in their reactivity.
(b) (ii) Arrange the following in:
(1) Decreasing reactivity towards nucleophilic addition reaction: Propanal, Acetone, Benzaldehyde.
(2) Increasing order of boiling point: Propane, Ethanol, Dimethylether, Propanal.
View Solution
(1) The correct order of decreasing reactivity towards nucleophilic addition reaction is: \[ Propanal > Benzaldehyde > Acetone \]
Aldehydes are more reactive than ketones due to less steric hindrance and higher electrophilicity. Benzaldehyde is less reactive than aliphatic aldehydes due to the resonance stabilization of the carbonyl carbon.
(2) The increasing order of boiling point is: \[ Propane < Dimethyl ether < Propanal < Ethanol \]
Ethanol has the highest boiling point due to extensive hydrogen bonding, while propane has the lowest due to weak van der Waals forces. Quick Tip: For nucleophilic addition reactions, aldehydes are more reactive than ketones because of the absence of electron-donating alkyl groups that reduce the electrophilicity of the carbonyl carbon.
(b) (iii) Give a simple chemical test to distinguish between Benzoic acid and Benzaldehyde.
View Solution
Benzoic acid and benzaldehyde can be distinguished using sodium bicarbonate (\( NaHCO_3 \)) test.
- Benzoic acid reacts with sodium bicarbonate to produce carbon dioxide gas with effervescence:
\[ C_6H_5COOH + NaHCO_3 \rightarrow C_6H_5COONa + CO_2 + H_2O \]
- Benzaldehyde does not react with sodium bicarbonate, so no effervescence is observed. Quick Tip: Benzoic acid can also be distinguished from benzaldehyde using Fehling’s test. Benzaldehyde does not reduce Fehling’s solution, whereas carboxylic acids do not react.
(a) Ce(III) is easily oxidised to Ce(IV). Comment.
View Solution
Cerium (\( Ce \)) is a lanthanide element with the electronic configuration \([Xe]4f^1 5d^1 6s^2\). The presence of a single electron in the 4f orbital makes Ce(III) stable. However, the stability of Ce(IV) arises due to the completely empty 4f orbital, which leads to a noble gas-like configuration (\([Xe]\)). This stability provides a strong driving force for the oxidation of Ce(III) to Ce(IV). Furthermore, Ce(IV) forms stable compounds, especially with oxygen, such as \( CeO_2 \), making its oxidation favorable in aqueous solutions. Quick Tip: Cerium is one of the few lanthanides that exists in a stable \( +4 \) oxidation state due to the stability of its empty 4f orbital.
(b) \(E^0(Mn^{2+}/Mn)\) is -1.18 V. Why is this value highly negative in comparison to neighbouring d-block elements?
View Solution
The standard reduction potential of Mn\(^ {2+}\)/Mn is highly negative due to the extra stability of the Mn\(^ {2+}\) ion, which has a half-filled \( 3d^5 \) configuration. This half-filled d-orbital provides additional exchange energy, which stabilizes Mn\(^ {2+}\) and makes its reduction to metallic manganese thermodynamically less favorable. In contrast, neighboring elements like Fe and Cr have more favorable reduction potentials because they do not possess the same level of half-filled stability in their \( +2 \) oxidation states. Quick Tip: The extra stability of the \(3d^5\) half-filled configuration in Mn\(^ {2+}\) makes its reduction less favorable, resulting in a highly negative standard reduction potential.
(c) Which element of the 3d series has the lowest enthalpy of atomisation and why?
View Solution
Zinc (\( Zn \)) has the lowest enthalpy of atomisation among the 3d series elements. This is due to its completely filled \( 3d^{10} \) electronic configuration, which results in weak metallic bonding. Metallic bonding strength is primarily influenced by the number of unpaired electrons available for delocalization, and since zinc lacks unpaired d-electrons, the metallic bonding is weaker. This weak bonding reduces the energy required to separate Zn atoms in the metallic lattice, leading to a lower enthalpy of atomisation. Quick Tip: Zinc has a completely filled \( 3d^{10} \) configuration, leading to weak metallic bonding and low enthalpy of atomisation.
(d) What happens when sodium chromate is acidified?
View Solution
Sodium chromate (\( Na_2CrO_4 \)) is yellow in color and exists in basic medium. When acidified, chromate ions (\( CrO_4^{2-} \)) undergo a chemical equilibrium shift towards dichromate formation: \[ 2CrO_4^{2-} + 2H^+ \rightarrow Cr_2O_7^{2-} + H_2O \]
Dichromate ions (\( Cr_2O_7^{2-} \)) are orange in color. This color change from yellow to orange is a characteristic reaction used in analytical chemistry to distinguish between basic and acidic environments. Quick Tip: Chromate ions (\( CrO_4^{2-} \)) convert to dichromate (\( Cr_2O_7^{2-} \)) in acidic medium, changing color from yellow to orange.
(e) Zn, Cd, and Hg are soft metals. Why?
View Solution
Zinc, cadmium, and mercury belong to group 12 of the periodic table and have completely filled \( d^{10} \) electronic configurations. Due to the lack of unpaired d-electrons, the metallic bonding in these elements is weak, leading to their characteristic softness. Additionally, mercury is liquid at room temperature due to the very weak interatomic forces in its metallic lattice. Quick Tip: Zinc, cadmium, and mercury have completely filled \( d^{10} \) configurations, leading to weak metallic bonds and making them soft metals.
(f) Why is permanganate titration not carried out in the presence of \(HCl\)?
View Solution
Permanganate (\( MnO_4^- \)) is a strong oxidizing agent in acidic medium. If \( HCl \) is used instead of \( H_2SO_4 \), the chloride ions (\( Cl^- \)) present in \( HCl \) get oxidized to chlorine gas (\( Cl_2 \)), leading to side reactions. This interference makes the titration inaccurate. Instead, sulfuric acid (\( H_2SO_4 \)) is preferred as it does not undergo oxidation under these conditions. Quick Tip: \( HCl \) cannot be used in permanganate titrations because \( Cl^- \) gets oxidized to \( Cl_2 \), affecting the accuracy of the titration.
(g) The lower oxides of transition metals are basic whereas the highest are amphoteric/acidic. Give reason.
View Solution
The oxidation state of a transition metal determines the nature of its oxide. In lower oxidation states, transition metal oxides exhibit more ionic character, making them basic. In contrast, higher oxidation state oxides show increased covalent character, making them acidic or amphoteric. For example, \( MnO \) (manganese(II) oxide) is basic, while \( Mn_2O_7 \) (manganese(VII) oxide) is acidic. This behavior is due to the increasing oxidation state causing a greater degree of covalent bonding and polarization. Quick Tip: Oxides in lower oxidation states are basic due to their ionic character, while those in higher oxidation states are acidic due to increased covalent character.
(a) (i) Ishan’s automobile radiator is filled with 1.0 kg of water. How many grams of ethylene glycol (Molar mass = 62 g mol\(^{-1}\)) must Ishan add to get the freezing point of the solution lowered to \(-2.8^\circ C\). \( K_f \) for water is 1.86 K kg \(\cdot\) mol\(^{-1}\).
View Solution
The depression in freezing point is given by the formula: \[ \Delta T_f = K_f \times \frac{w_B}{M_B \times w_A} \]
Substituting the given values: \[ 2.8 = 1.86 \times \frac{w_B}{62 \times 1} \]
Solving for \( w_B \): \[ w_B = \frac{2.8 \times 62}{1.86} = 93.33 g \]
Thus, the required amount of ethylene glycol is 93.33 g. Quick Tip: For colligative properties, use the formula \( \Delta T_f = K_f \times \frac{w_B}{M_B \times w_A} \) to determine the mass of solute required to achieve a desired freezing point depression.
(a) (ii) What type of deviation from Raoult’s law is shown by ethanol and acetone mixture? Give reason.
View Solution
The ethanol-acetone mixture exhibits positive deviation from Raoult’s law. This occurs because the intermolecular forces in pure ethanol (hydrogen bonding) are stronger than those in the ethanol-acetone mixture. When acetone is added, it disrupts ethanol’s hydrogen bonding, leading to an increase in vapor pressure. Quick Tip: A positive deviation from Raoult’s law occurs when the intermolecular forces in the mixture are weaker than in the pure components, leading to higher vapor pressure than expected.
(b) (i) Boiling point of water at 750 mm Hg pressure is 99.68\( ^\circ C \). How much sucrose (Molar mass = 342 g mol\(^{-1}\)) is to be added to 500 g of water such that it boils at 100\( ^\circ C \)? (\( K_b \) for water = 0.52 K kg mol\(^{-1}\)).
View Solution
The change in boiling point (\( \Delta T_b \)) is given by the formula: \[ \Delta T_b = K_b \times m \]
where \( K_b \) is the ebullioscopic constant of water and \( m \) is the molality of the solution.
We are given that the boiling point increases from 99.68\( ^\circ C \) to 100\( ^\circ C \), so: \[ \Delta T_b = 100^\circ C - 99.68^\circ C = 0.32^\circ C \]
Now, we can calculate the molality of the solution using the equation: \[ m = \frac{\Delta T_b}{K_b} = \frac{0.32}{0.52} = 0.615 \, mol/kg \]
Molality (\( m \)) is defined as the number of moles of solute per kilogram of solvent. We are adding sucrose to 500 g (or 0.5 kg) of water. Therefore, the number of moles of sucrose required is: \[ moles of sucrose = m \times mass of solvent in kg = 0.615 \times 0.5 = 0.3075 \, mol \]
Finally, we calculate the mass of sucrose required using the molar mass of sucrose (342 g/mol): \[ mass of sucrose = moles of sucrose \times molar mass = 0.3075 \times 342 = 105.23 \, g \] Quick Tip: For boiling point elevation, use the formula \( \Delta T_b = K_b \times \frac{w_B}{M_B \times w_A} \) to calculate the required mass of solute.
(b) (ii) State Henry’s law and write its any one application.
View Solution
Henry’s law states that at a constant temperature, the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the solution. Mathematically, it is expressed as: \[ C = k_H \cdot P \]
where \( C \) is the concentration of the gas in the liquid, \( P \) is the partial pressure of the gas, and \( k_H \) is Henry’s constant.
Application: To increase the solubility of \( CO_2 \) in soft drinks and soda water, the bottle is sealed under high pressure. Quick Tip: Henry’s law explains the solubility of gases in liquids and is widely used in industries like carbonated beverage production and scuba diving safety.






























Comments