CBSE Class 12 Chemistry Question Paper 2024 PDF (Set 3 - 56/3/3) is available for download here. CBSE conducted the Chemistry exam on February 27, 2024, from 10:30 AM to 1:30 PM. The total marks for the theory paper are 70. The question paper contains 20% MCQ-based questions, 40% competency-based questions, and 40% short and long answer type questions.
CBSE Class 12 Chemistry Question Paper 2024 (Set 3 - 56/3/3) with Answer Key
| CBSE Class 12 2024 Chemistry Question Paper with Answer Key | Check Solution |
CBSE Class 12 Chemistry Question Paper 2024 with Solution
Section A
Which of the following ligands is an ambidentate ligand?
View Solution
An ambidentate ligand is one that has two or more donor atoms capable of forming coordinate bonds with the central metal ion, but it can coordinate through either one of them. In this case, SCN\(^-\) (thiocyanate) is an ambidentate ligand because it can bind through either the sulfur atom or the nitrogen atom. Other options, such as CO, NH\(_3\), and H\(_2\)O, are monodentate ligands, which can only form a single coordinate bond with the metal ion. Quick Tip: Ambidentate ligands can coordinate to the central metal ion via more than one donor atom. SCN\(^-\) is a classic example where the ligand can bind via either the sulfur or nitrogen atom.
On adding AgNO\(_3\) solution to 1 mole of CoCl\(_3\)·4NH\(_3\), one mole of AgCl is precipitated. The secondary valency of Co is:
View Solution
In this case, CoCl\(_3\)·4NH\(_3\) represents a coordination compound with cobalt as the central metal ion. The primary valency of the metal ion is given by the chloride ions (Cl\(^-\)), which are directly bonded to the metal ion. Since there are three chloride ions, the primary valency of Co is 3.
The secondary valency is the number of coordinate bonds that the metal ion forms with other ligands, which are the ammonia molecules (NH\(_3\)) in this case. Since the compound has 4 NH\(_3\) molecules, the secondary valency of Co is 6 (3 from Cl\(^-\) and 3 from NH\(_3\)).
The formation of AgCl upon addition of AgNO\(_3\) indicates that the chloride ions are in the primary coordination sphere, confirming that the secondary valency is 6.
Quick Tip: In coordination compounds, the primary valency is typically the charge on the metal ion, and the secondary valency is the number of ligands directly coordinated to the metal ion. In this case, ammonia ligands satisfy the secondary valency.
Which of the following elements of the 3d series of transition elements has the lowest \(\Delta_{a} H^\circ\)?
View Solution
\(\Delta H^\circ\) refers to the enthalpy change or heat of formation for a particular element in its standard state. For transition elements, the enthalpy change is affected by the electron configuration, the stability of the element, and its ionization energy.
- Scandium (Sc) and chromium (Cr) have relatively high ionization energies and enthalpy changes.
- Copper (Cu) has a relatively stable electron configuration due to its completely filled d-subshell, making it less reactive and having a moderate \(\Delta H^\circ\).
- Zinc (Zn), on the other hand, has a completely filled 3d subshell and a stable electron configuration, making it the least reactive and having the lowest enthalpy change among the 3d transition elements. This is why Zn has the lowest \(\Delta H^\circ\).
Thus, the correct answer is Zn. Quick Tip: The elements with a completely filled d-subshell, such as Zn, exhibit greater stability and lower enthalpy change, while other transition elements with partially filled d-subshells tend to have higher enthalpy changes.
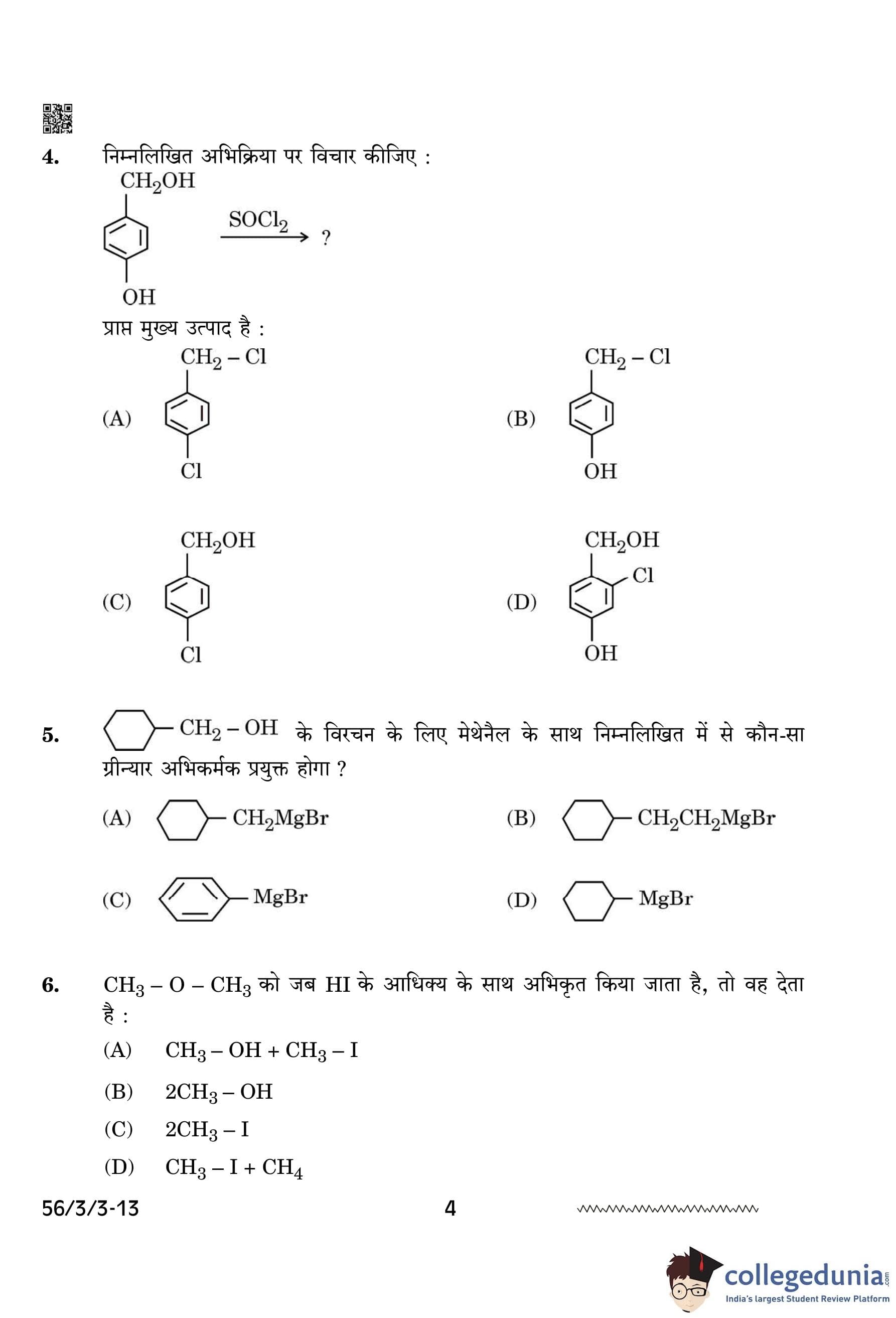
Consider the following reaction:
The major product obtained is:
View Solution
When benzyl alcohol (C\(_6\)H\(_5\)CH\(_2\)OH) reacts with thionyl chloride (SOCl\(_2\)), the hydroxyl group (OH) is replaced by a chlorine atom, resulting in the formation of benzyl chloride (C\(_6\)H\(_5\)CH\(_2\)Cl). This is a typical reaction for alcohols with SOCl\(_2\), where the alcohol undergoes nucleophilic substitution.
Thus, the major product is benzyl chloride (C\(_6\)H\(_5\)CH\(_2\)Cl). Quick Tip: When reacting alcohols with thionyl chloride (SOCl\(_2\)), the hydroxyl group is replaced by a chlorine atom, forming an alkyl chloride. This reaction typically proceeds via nucleophilic substitution (S\(_N\)2 mechanism) with the elimination of sulfur dioxide (SO\(_2\)) and hydrogen chloride (HCl).
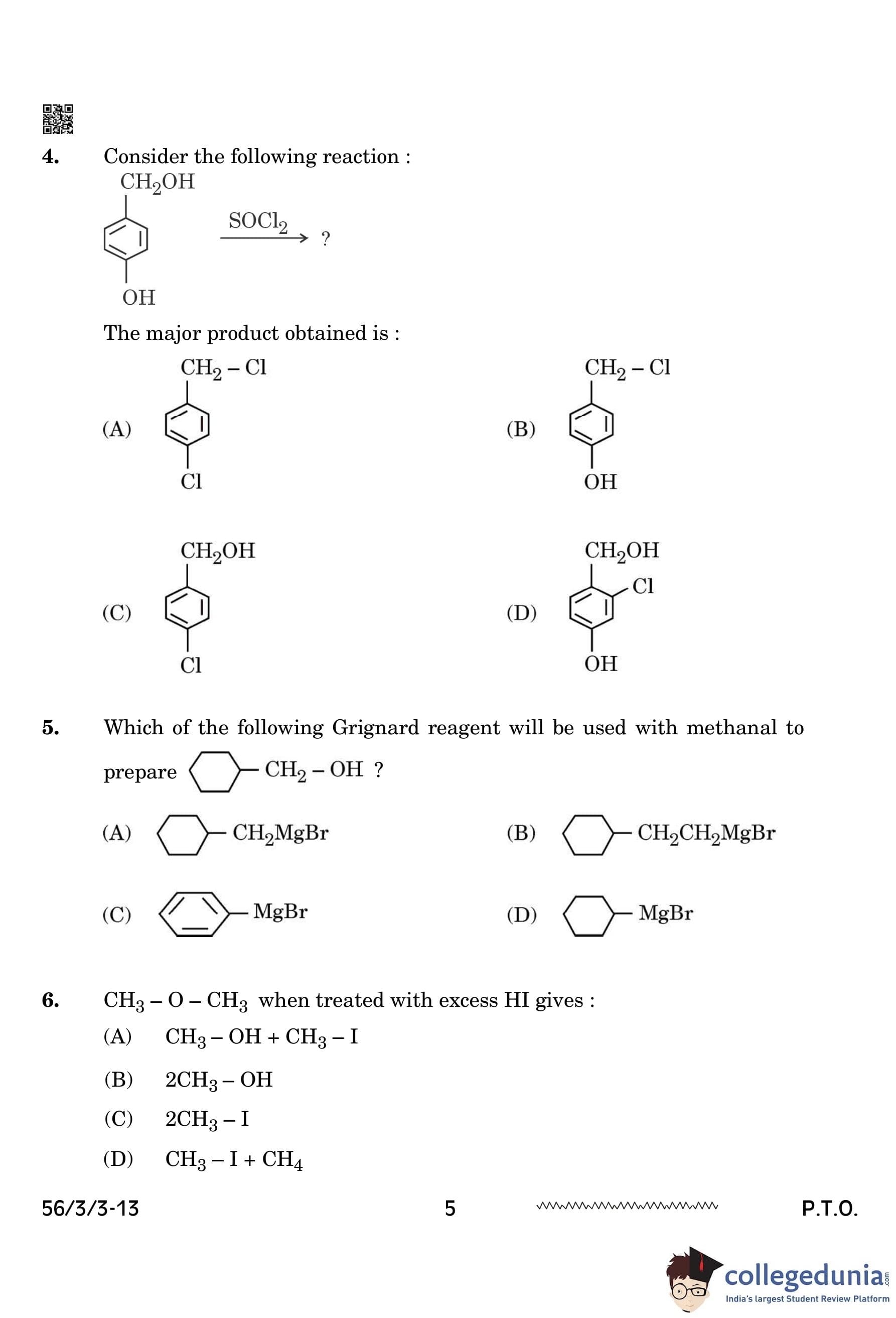
Which of the following Grignard reagents will be used with methanol to prepare
View Solution
The reaction of Grignard reagents with aldehydes (such as methanal) leads to the formation of alcohols after hydrolysis. In this case, CH\(_2\)OH is an alcohol, and the correct Grignard reagent for methanal would be a methyl group (CH\(_3\)-) attached to magnesium bromide (MgBr).
Therefore, the correct answer is MgBr (option D), which reacts with methanol to form CH\(_2\)OH. Quick Tip: Grignard reagents react with carbonyl compounds such as aldehydes and ketones. The reaction forms an alkoxide intermediate, which, upon hydrolysis, gives the corresponding alcohol.
CH\(_3\)-O-CH\(_3\) when treated with excess HI gives:
View Solution
The reaction of ether (CH\(_3\)-O-CH\(_3\)) with excess hydroiodic acid (HI) results in the cleavage of the ether bond. This cleavage produces methyl iodide (CH\(_3\)-I) and methyl alcohol (CH\(_3\)-OH). However, with excess HI, both ether bonds are cleaved, producing 2 moles of CH\(_3\)-I and CH\(_3\)-OH.
Thus, the correct answer is (C), where two moles of CH\(_3\)-I are formed. Quick Tip: Ethers undergo cleavage when treated with excess HI. The bond between the oxygen and the alkyl group is broken, leading to the formation of alkyl iodide and alcohol.
Which of the following compounds will not react with benzene sulphonyl chloride?
View Solution
Step 1: Understanding the Hinsberg Test
Benzene sulphonyl chloride (\(C_6H_5SO_2Cl\)) is used in the Hinsberg test to differentiate primary, secondary, and tertiary amines.
- Primary amines (\(RNH_2\)) react with benzene sulphonyl chloride to form a sulphonamide (\(RNHSO_2C_6H_5\)), which dissolves in alkali.
- Secondary amines (\(R_2NH\)) form an insoluble sulphonamide (\(R_2NSO_2C_6H_5\)), which does not dissolve in alkali.
- Tertiary amines (\(R_3N\)) do not have an available hydrogen on nitrogen, so they do not react with benzene sulphonyl chloride.
Step 2: Identifying the Type of Amines
- Option (A) \( (C_2H_5)_3N \) (Triethylamine): Tertiary amine → No reaction.
- Option (B) \( C_2H_5 - NH_2 \) (Ethylamine): Primary amine → Reacts.
- Option (C) \( (C_2H_5)_2NH \) (Diethylamine): Secondary amine → Reacts.
- Option (D) (Aniline): Primary aromatic amine → Reacts.
Step 3: Conclusion
Since tertiary amines do not react with benzene sulphonyl chloride, the correct answer is (A) \( (C_2H_5)_3N \). Quick Tip: The Hinsberg test is useful for distinguishing primary, secondary, and tertiary amines based on their reactivity with benzene sulphonyl chloride.
‘Scurvy’ is caused by the deficiency of vitamin:
View Solution
Step 1: Scurvy is caused by a deficiency of Vitamin C (ascorbic acid).
Step 2: Vitamin C is essential for collagen synthesis and helps maintain healthy connective tissues.
Step 3: A deficiency leads to symptoms such as bleeding gums, joint pain, and weakness. Quick Tip: Citrus fruits like oranges and lemons are rich sources of Vitamin C and help prevent scurvy.
A 1% solution of solute ‘X’ is isotonic with a 6% solution of sucrose (molar mass = \( 342 \) g mol\(^{-1}\)). The molar mass of solute ‘X’ is:
View Solution
Step 1: Understanding Isotonic Solutions
Two solutions are isotonic if they have the same osmotic pressure, which is given by the formula: \[ \Pi = \frac{w}{M} \times \frac{1000}{V} \]
where:
- \( \Pi \) = osmotic pressure,
- \( w \) = mass of solute in grams,
- \( M \) = molar mass of solute,
- \( V \) = volume of solution in mL.
For isotonic solutions, their osmotic pressures are equal: \[ \frac{w_1}{M_1} = \frac{w_2}{M_2} \]
Step 2: Applying Given Data
- Sucrose solution: \( 6% \) solution means \( 6 \) g of sucrose in \( 100 \) mL solution.
- Solute ‘X’ solution: \( 1% \) solution means \( 1 \) g of solute ‘X’ in \( 100 \) mL solution.
- Molar mass of sucrose: \( M_1 = 342 \) g mol\(^{-1}\).
- Molar mass of solute ‘X’: \( M_2 \) (to be determined).
Using the isotonic equation: \[ \frac{6}{342} = \frac{1}{M_2} \]
Step 3: Solving for \( M_2 \)
Rearranging: \[ M_2 = \frac{1 \times 342}{6} \]
\[ M_2 = 57 g mol^{-1} \]
Step 4: Conclusion
The molar mass of solute ‘X’ is \( 57 \) g mol\(^{-1}\), so the correct answer is (B) \( 57 \) g mol\(^{-1}\). Quick Tip: For isotonic solutions, use the formula \( \frac{w_1}{M_1} = \frac{w_2}{M_2} \). If one solution is a percentage solution, express it as grams per 100 mL.
The half-life of a first-order reaction with a rate constant (\( k \)) of 3 min\(^{-1}\) is:
View Solution
Step 1: The half-life (\( t_{1/2} \)) of a first-order reaction is given by the formula: \[ t_{1/2} = \frac{0.693}{k} \]
Step 2: Substituting \( k = 3 \) min\(^{-1}\): \[ t_{1/2} = \frac{0.693}{3} = 0.231 min \] Quick Tip: For first-order reactions, half-life is independent of initial concentration and is calculated as \( t_{1/2} = \frac{0.693}{k} \).
Which of the following cells is used in hearing aids?
View Solution
Mercury cells are commonly used in hearing aids because they provide a stable and long-lasting voltage. They operate based on the reaction:
\[ Zn + HgO \rightarrow ZnO + Hg \]
These cells have a small size and deliver a constant potential, making them ideal for sensitive electronic devices like hearing aids. Quick Tip: Mercury cells are used in hearing aids, watches, and calculators due to their stable voltage and compact size.
Aniline on reaction with Bromine water gives:
View Solution
Aniline (\(C_6H_5NH_2\)) is highly reactive towards bromination due to the strong activating effect of the \(-NH_2\) group. When aniline is treated with bromine water, a white precipitate of 2,4,6-tribromoaniline forms:
\[ C_6H_5NH_2 + 3Br_2 \rightarrow C_6H_2Br_3NH_2 + 3HBr \]
Since the \(-NH_2\) group is an electron-donating group, it directs substitution to the ortho and para positions, leading to tribromoaniline formation. Quick Tip: Aromatic amines like aniline undergo electrophilic substitution easily due to the strong activating effect of the \(-NH_2\) group.
For Questions number 13 to 16, two statements are given one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (A), (B), (C) and (D) as given below.
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is notthe correct explanation of the Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
Question 13:
Assertion (A): \( \Delta_{mix} H \) and \( \Delta_{mix} V \) are zero for an ideal solution.
Reason (R): The solution which obeys Raoult’s law over the entire range of concentration is called an ideal solution.
View Solution
For an ideal solution:
- \( \Delta_{mix} H = 0 \) because there is no enthalpy change upon mixing, meaning the interactions between solute and solvent are similar to those in the pure components.
- \( \Delta_{mix} V = 0 \) because there is no volume change when components mix ideally.
Although Raoult’s law describes an ideal solution, it does not directly explain why \( \Delta H \) and \( \Delta V \) are zero. Quick Tip: Reaction rates usually increase with temperature due to an increase in the number of molecules with energy greater than the activation energy.
Assertion (A): Rate of reaction decreases with increase in temperature.
Reason (R): Number of effective collisions increases with increase in temperature.
View Solution
Increasing temperature generally increases the rate of reaction because more molecules gain sufficient energy to overcome the activation barrier. The assertion is incorrect, but the reason is correct.
\[ Rate \propto e^{-\frac{E_a}{RT}} \]
where \( E_a \) is the activation energy, \( R \) is the gas constant, and \( T \) is temperature. A higher \( T \) leads to a higher reaction rate. Quick Tip: Reaction rates usually increase with temperature due to an increase in the number of molecules with energy greater than the activation energy.
Assertion (A): Phenol on reaction with aqueous NaOH gives sodium phenoxide.
Reason (R): This reaction supports the acidic nature of phenol.
View Solution
Phenol reacts with NaOH to form sodium phenoxide, confirming its weakly acidic nature:
\[ C_6H_5OH + NaOH \rightarrow C_6H_5ONa + H_2O \]
This reaction occurs because phenol can donate a proton (\( H^+ \)), showing its acidic behavior. Quick Tip: Phenol is weakly acidic and reacts with NaOH to form sodium phenoxide, a reaction that supports the acidic nature of phenol. This is a key characteristic of phenols, distinguishing them from alcohols.
Assertion (A): Boiling point of butan-1-ol is higher than that of butan-1-amine.
Reason (R): Being more polar, butan-1-ol forms stronger intermolecular hydrogen bonds as compared to butan-1-amine.
View Solution
Hydrogen bonding is stronger in alcohols (\(-OH\) group) than in amines (\(-NH_2\) group) due to the higher electronegativity of oxygen compared to nitrogen. As a result, butan-1-ol exhibits stronger hydrogen bonding and thus a higher boiling point. Quick Tip: Hydrogen bonding strength follows the trend: \[ Alcohols > Amines > Ethers \] due to electronegativity differences.
Section B
Write the reactions of glucose with
(a) HI
(b) \((CH_3CO_2)O\)
View Solution
Step 1: Reaction of glucose with HI
Glucose undergoes complete reduction when treated with hydroiodic acid (HI) in the presence of heat. This reaction removes all oxygen functionality from glucose, converting it into \( n \)-hexane. The reaction is as follows:
\[ CHO - (CHOH)_4 - CH_2OH \xrightarrow{HI, \Delta} CH_3 - CH_2 - CH_2 - CH_2 - CH_3 \]
Step 2: Reaction of glucose with Acetic Anhydride
When glucose is treated with acetic anhydride \((CH_3CO)_2O\), all the hydroxyl groups (-OH) get acetylated, leading to the formation of glucose pentaacetate. This reaction is shown below:
Quick Tip: Glucose can undergo reduction and acetylation reactions. Reduction with HI removes oxygen functionality, yielding \( n \)-hexane. Acetic anhydride reacts with all hydroxyl (-OH) groups, forming pentaacetate derivatives.
(a) Define molal depression constant. How is it related to enthalpy of fusion?
View Solution
Step 1: Definition of Molal Depression Constant
Molal depression constant (\( K_f \)) is the depression in freezing point observed when one mole of a non-volatile solute is dissolved in 1 kg (1000 g) of solvent.
Step 2: Relationship with Enthalpy of Fusion
The molal depression constant is mathematically expressed as:
\[ K_f = \frac{R \times M_{solvent} \times T_f^0}{1000 \times \Delta_{fus} H^0} \]
where:
- \( R \) is the universal gas constant,
- \( M_{solvent} \) is the molar mass of the solvent,
- \( T_f^0 \) is the freezing point of the pure solvent,
- \( \Delta_{fus} H^0 \) is the enthalpy of fusion of the solvent. Quick Tip: Molal depression constant helps determine freezing point depression. It depends on the solvent properties, including its enthalpy of fusion and molar mass.
(b) What type of deviation is shown by ethanol and acetone mixture ? Give reason. What type of azeotropic mixture is formed by that deviation ?
View Solution
Ethanol and acetone mixture shows a positive deviation from Raoult's law. In this case, the intermolecular forces between ethanol and acetone molecules are weaker than the forces between ethanol molecules and between acetone molecules. This leads to an increase in vapor pressure of the mixture compared to what is predicted by Raoult's law for an ideal solution.
This deviation leads to the formation of an azeotropic mixture. In this case, the azeotrope formed is a minimum boiling azeotrope, where the mixture boils at a lower temperature than either of the pure components. The azeotrope consists of approximately 89% ethanol and 11% acetone by mole. Quick Tip: Positive deviation occurs when the interactions between molecules in the mixture are weaker than those in the pure components, leading to an increase in vapor pressure.
(a) In a reaction, if the concentration of reactant \( X \) is tripled, the rate of reaction becomes twenty-seven times. What is the order of the reaction?
View Solution
Step 1: Determining the Order of Reaction
The rate law for a reaction is generally given by:
\[ Rate = k [X]^p \]
Given that when \( [X] \) is tripled, the rate increases by a factor of 27:
\[ 27 \times Rate = k [3X]^p \]
\[ (3)^p = 27 \]
Since \( 27 = 3^3 \), we get \( p = 3 \).
Thus, the order of the reaction is Third order. Quick Tip: For determining reaction order, observe how the rate changes with concentration. If a bimolecular reaction has an excess reactant, it behaves like a first-order reaction.
(b) State a condition under which a bimolecular reaction is kinetically a first-order reaction and give an example.
View Solution
Condition for Bimolecular Reaction to Follow First-Order Kinetics
A bimolecular reaction follows first-order kinetics when one of the reactants is in excess. In such cases, the reaction appears to be first-order because the concentration of the excess reactant remains nearly constant.
Example:
Hydrolysis of ester:
\[ CH_3COOCH_3 + H_2O \rightarrow CH_3COOH + CH_3OH \]
Here, water is in large excess, making the reaction effectively first-order. Quick Tip: For determining reaction order, observe how the rate changes with concentration. If a bimolecular reaction has an excess reactant, it behaves like a first-order reaction.
Complete and balance the following chemical equations:
(a) \( 3 MnO_4^- + 4 H^+ \longrightarrow ? \)
(b) \( Cr_2 O_7^{2-} + 14 H^+ + 6 Fe^{2+} \longrightarrow ? \)
View Solution
(a) The first equation represents the reduction of permanganate ions (\( MnO_4^- \)) in acidic medium. The balanced equation is:
\[ 3 MnO_4^- + 4 H^+ \longrightarrow 2 MnO_4^- + MnO_2 + 2 H_2O \]
Explanation:
- Permanganate (\( MnO_4^- \)) is reduced to manganese dioxide (\( MnO_2 \)) in acidic medium.
- The oxidation state of manganese in \( MnO_4^- \) is +7, and it is reduced to +4 in \( MnO_2 \), releasing oxygen and water.
(b) The second equation represents the reaction of dichromate ions (\( Cr_2 O_7^{2-} \)) in acidic medium, where it is reduced by \( Fe^{2+} \) to form \( Cr^{3+} \) ions. The balanced equation is:
\[ Cr_2 O_7^{2-} + 14 H^+ + 6 Fe^{2+} \longrightarrow 2 Cr^{3+} + 6 Fe^{3+} + 7 H_2O \]
Explanation:
- Dichromate (\( Cr_2 O_7^{2-} \)) is reduced to \( Cr^{3+} \) in the presence of acidic conditions.
- The iron (\( Fe^{2+} \)) acts as a reducing agent and is oxidized to \( Fe^{3+} \).
- The reduction of dichromate involves the transfer of electrons and the formation of water as a product. Quick Tip: In redox reactions, balancing involves ensuring that both mass and charge are conserved. For example, in the reduction of permanganate to manganese dioxide, balance oxygen atoms with water and hydrogen ions to balance charge.
(a) Which halogen compound in the following pair will react faster in \( S_N2 \) reaction and why?
View Solution
(a) The compound \( CH_3CH_2Cl \) (ethyl chloride) will react faster in an \( S_N2 \) reaction than \( C_6H_5CH_2Cl \) (benzyl chloride). The main reasons are:
1. Steric Hindrance:
- \( CH_3CH_2Cl \) is a primary halide with less steric hindrance around the carbon bonded to the halogen. This allows the nucleophile to attack the electrophilic carbon more easily.
- \( C_6H_5CH_2Cl \) (benzyl chloride) is a benzyl halide. Although benzyl halides are generally reactive in \( S_N2 \), they still experience more steric hindrance than a simple alkyl halide like ethyl chloride.
2. Nucleophilic Substitution Mechanism:
- \( S_N2 \) reactions involve a backside attack of the nucleophile on the carbon bonded to the halogen, resulting in the inversion of configuration. Less steric hindrance on the carbon results in a faster reaction.
Therefore, \( CH_3CH_2Cl \) reacts faster in an \( S_N2 \) reaction than \( C_6H_5CH_2Cl \). Quick Tip: In \( S_N2 \) reactions, primary halides react faster than secondary or tertiary halides due to less steric hindrance. The nucleophile attacks the electrophilic carbon directly, leading to an inversion of configuration. Less bulky groups around the carbon make this attack easier and faster.
(b) Why does the presence of nitro groups at ortho- and para- positions in haloarenes increase their reactivity towards nucleophilic substitution reactions?
View Solution
(b) The presence of nitro groups at the ortho- and para- positions in haloarenes increases their reactivity towards nucleophilic substitution reactions due to the following reasons:
1. Electron-Withdrawing Effect:
- Nitro groups (\( NO_2 \)) are strong electron-withdrawing groups due to their resonance and inductive effects. They pull electron density away from the aromatic ring, particularly at the ortho- and para- positions, making the carbon attached to the halogen more electrophilic.
2. Stabilization of the Transition State:
- In nucleophilic substitution reactions, the intermediate or transition state is stabilized by electron-withdrawing groups. The nitro group at the ortho- or para- position stabilizes the negative charge on the transition state, making it easier for the nucleophile to attack the electrophilic carbon.
3. Increased Reactivity:
- This increased electrophilicity of the carbon to which the halogen is attached makes the molecule more reactive towards nucleophilic substitution reactions, especially in the \( S_N2 \) mechanism.
Thus, nitro groups at ortho- and para- positions in haloarenes increase their reactivity towards nucleophilic substitution reactions by making the carbon more electrophilic and stabilizing the transition state. Quick Tip: In \( S_N2 \) reactions, primary halides react faster than secondary or tertiary halides due to less steric hindrance. In haloarenes, nitro groups at ortho- and para- positions increase reactivity by stabilizing the transition state through their electron-withdrawing nature.
Section C
(a) What type of nucleophilic substitution (\( S_N1 \) or \( S_N2 \)) occurs in the hydrolysis of 2-Bromobutane to form \( (+-) \)-Butan-2-ol? Give reason.
View Solution
Step 1: The reaction follows an \( S_N1 \) mechanism.
This is due to the formation of a planar carbocation, which can be attacked by the nucleophile from either side. This leads to racemization of the product.
\[ CH_3CH_2C(Br)H_2 \rightarrow CH_3C(+)H_2 \]
The reaction is accompanied by racemization, and the intermediate carbocation is formed in an \( S_N1 \) reaction. Quick Tip: In \( S_N1 \) reactions, the rate-determining step is the formation of a carbocation intermediate. This intermediate can be attacked from either side, leading to racemization of the product.
(b) What happens when chlorobenzene and methyl chloride are treated with sodium metal in dry ether?
View Solution
When chlorobenzene and methyl chloride are treated with sodium metal in dry ether, a reaction known as the Wurtz-Fittig reaction occurs, leading to the formation of toluene.
Sodium metal reduces the chlorine atom in chlorobenzene, and the methyl group from methyl chloride is added to the benzene ring. This results in the formation of toluene (C\(_6\)H\(_5\)CH\(_3\)).
\[ C_6H_5Cl + CH_3Cl + Na \xrightarrow{dry ether} C_6H_5CH_3 + NaCl \] Quick Tip: The Wurtz-Fittig reaction is a coupling reaction where sodium metal reduces halides, leading to the formation of new carbon-carbon bonds. The reaction between chlorobenzene and methyl chloride forms toluene.
A first-order reaction is 25% complete in 40 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
[Given: \( \log 2 = 0.30 \), \( \log 3 = 0.48 \), \( \log 4 = 0.60 \), \( \log 5 = 0.69 \)]
View Solution
Step 1: The integrated rate law for a first-order reaction is given by:
\[ \log \left( \frac{[R]_0}{[R]} \right) = \frac{k \cdot t}{2.303} \]
Where:
- \( [R]_0 \) is the initial concentration,
- \( [R] \) is the concentration at time \( t \),
- \( k \) is the rate constant, and
- \( t \) is the time.
We are told that the reaction is 25% complete in 40 minutes, which means that 75% of the reactant remains. Therefore, we calculate:
\[ \frac{[R]_0}{[R]} = \frac{1}{0.75} = 1.33 \]
Taking the logarithm:
\[ \log 1.33 = 0.125 \]
Substitute into the rate law:
\[ 0.125 = \frac{k \cdot 40}{2.303} \]
Solving for \( k \):
\[ k = \frac{0.125 \times 2.303}{40} = 0.0069 \, min^{-1} \]
Step 2: To find the time required for the reaction to be 80% complete, i.e., \( \frac{[R]_0}{[R]} = \frac{1}{0.20} = 5 \), we use:
\[ \log 5 = 0.69 \]
Substitute into the rate law:
\[ 0.69 = \frac{k \cdot t}{2.303} \]
Substituting the value of \( k \):
\[ 0.69 = \frac{0.0069 \cdot t}{2.303} \]
Solving for \( t \):
\[ t = \frac{0.69 \times 2.303}{0.0069} = 230.3 \, min \]
Thus, the time required for the reaction to be 80% complete is 230.3 minutes. Quick Tip: For first-order reactions, the concentration decreases exponentially over time. Use the integrated rate law to find the rate constant and calculate the time for any given percentage completion.
(a) Write the reactions involved in the following:
(i) Reimer-Tiemann reaction
(ii) Kolbe's reaction
View Solution
(i) Reimer-Tiemann Reaction: The Reimer-Tiemann reaction involves the introduction of a formyl group (–CHO) to the aromatic ring using chloroform (\( CHCl_3 \)) and sodium hydroxide (\( NaOH \)):
\[ C_6H_5OH + CHCl_3 + NaOH \rightarrow C_6H_4CHO + NaCl + H_2O \]
(ii) Kolbe’s Reaction: This reaction involves the carboxylation of sodium phenoxide with carbon dioxide under high pressure, yielding salicylic acid:
\[ C_6H_5ONa + CO_2 \xrightarrow{High Pressure} C_6H_4COOH + NaOH \]
(b) Name the reagent used in the bromination of phenol to form 2,4,6-Tribromophenol.
View Solution
The reagent used in the bromination of phenol to form 2,4,6-Tribromophenol is Bromine water (Br\(_2\) in water).
### Detailed Explanation:
1. Electrophilic Aromatic Substitution:
- The bromination of phenol is an example of electrophilic aromatic substitution, where the phenol acts as an electron-rich aromatic compound due to the presence of the hydroxyl group (-OH). The hydroxyl group is an activating group and directs incoming electrophiles to the ortho- and para- positions of the aromatic ring.
2. Action of Bromine:
- Bromine water (a solution of Br\(_2\) in water) acts as the electrophile in this reaction. The bromine molecule (Br\(_2\)) is polarized by the electron-rich benzene ring, and one of the bromine atoms becomes highly electrophilic, attacking the ortho- and para- positions relative to the hydroxyl group on the benzene ring.
3. Result:
- The reaction results in the formation of 2,4,6-Tribromophenol, where the three bromine atoms are attached to the ortho- and para- positions of the phenol ring, as shown below:
\[ C_6H_5OH + 3Br_2 \xrightarrow{H_2O} C_6H_2(Br)_3OH \]
- The bromine atoms occupy the 2, 4, and 6 positions (relative to the hydroxyl group), making the product 2,4,6-Tribromophenol.
Thus, the reagent used is Bromine water. Quick Tip: In electrophilic aromatic substitution reactions such as bromination and formylation, electron-rich aromatic rings are more reactive. The presence of activating groups like hydroxyl (–OH) increases the reactivity of phenol.
Give the structures of A, B, and C in the following reactions:
(a) \[ CH_3CH_2Cl \xrightarrow{KCN} \, A \xrightarrow{LiAlH_4} \, B \xrightarrow{HNO_2, \, 0^\circ C} \, C \]
View Solution
(a) The first reaction involves the conversion of chloroethane (CH\(_3\)CH\(_2\)Cl) to a nitrile (A) using potassium cyanide (KCN). The nitrile (A) is then reduced to an amine (B) using lithium aluminium hydride (LiAlH\(_4\)). Finally, the amine (B) undergoes diazotization with nitrous acid (HNO\(_2\)) to form an alcohol (C).
The reactions are as follows:
1. A: \(CH_3CH_2CN\) (Ethyl Cyanide)
2. B: \(CH_3CH_2NH_2\) (Ethylamine)
3. C: \(CH_3CH_2OH\) (Ethanol)
(b)
View Solution
(b) The second reaction involves the reduction of nitrobenzene to aniline (A) using iron and hydrochloric acid (Fe/HCl). Then, the aniline undergoes diazotization with sodium nitrite (NaNO\(_2\)) and hydrochloric acid (HCl) at 273K to form a diazonium salt (B). Finally, the diazonium salt undergoes hydrolysis to form phenol (C).
The reactions are as follows:
1. A: \(C_6H_5NH_2\) (Aniline)
2. B: \(C_6H_5N_2^+\) (Phenyl Diazonium Ion)
3. C: \(C_6H_5OH\) (Phenol) Quick Tip: - In the first reaction, the nitrile is reduced to an amine, which is then diazotized to form an alcohol.
- In the second reaction, nitrobenzene is reduced to aniline, which is then diazotized to form the diazonium ion, followed by hydrolysis to give phenol.
How will you bring about the following conversions? (any three)
(a) Benzoic acid to Benzaldehyde
(b) Ethanal to Propanone
(c) Acetophenone to Benzoic acid
(d) Bromobenzene to 1-Phenylethanol
View Solution
(a) Benzoic acid to Benzaldehyde:
This can be achieved by the reduction of benzoic acid using a selective reducing agent like diborane (B\(_2\)H\(_6\)) or Lithium aluminum hydride (LiAlH\(_4\)). This will reduce the carboxyl group (-COOH) to an aldehyde group (-CHO), forming benzaldehyde.
\[ C_6H_5COOH \xrightarrow{LiAlH_4} C_6H_5CHO \]
(b) Ethanal to Propanone:
Ethanal (acetaldehyde) can be oxidized to propanone (acetone) using a mild oxidizing agent like Chromium(VI) oxide (CrO\(_3\)). This oxidation introduces a second methyl group, forming propanone.
\[ CH_3CHO \xrightarrow{CrO_3} CH_3COCH_3 \]
(c) Acetophenone to Benzoic acid:
Acetophenone can be oxidized to benzoic acid using an oxidizing agent like potassium permanganate (KMnO\(_4\)). The methyl group (-CH\(_3\)) is oxidized to a carboxyl group (-COOH), forming benzoic acid.
\[ C_6H_5COCH_3 \xrightarrow{KMnO_4} C_6H_5COOH \]
(d) Bromobenzene to 1-Phenylethanol:
Bromobenzene can be converted to 1-phenylethanol via the Grignard reaction. First, magnesium is added to bromobenzene to form a phenylmagnesium bromide (Grignard reagent), which then reacts with ethyl formate followed by hydrolysis to form 1-phenylethanol.
\[ C_6H_5Br \xrightarrow{Mg} C_6H_5MgBr \xrightarrow{C_2H_5COO^-, H_2O} C_6H_5CH_2OH \] Quick Tip: For organic conversions, selective reagents such as LiAlH\(_4\), KMnO\(_4\), and Grignard reagents play a crucial role in controlling the specific transformation of functional groups.
A solution is prepared by dissolving 5 g of a non-volatile solute in 200 g of water. It has a vapor pressure of 31.84 mm Hg at 300 K. Calculate the molar mass of the solute.
(Vapor pressure of pure water at 300 K = 32 mm Hg)
View Solution
We can use Raoult's Law to find the molar mass of the solute. Raoult's Law states:
\[ \frac{P_{solvent}}{P_{solvent, pure}} = \frac{n_{solute}}{n_{solution}} \]
Where:
- \( P_{solvent} \) is the vapor pressure of the solution,
- \( P_{solvent, pure} \) is the vapor pressure of the pure solvent,
- \( n_{solute} \) is the number of moles of solute,
- \( n_{solution} \) is the number of moles of the solution.
Step 1: Calculate the mole fraction of the solute:
\[ \frac{P_{solvent}}{P_{solvent, pure}} = 1 - \frac{31.84}{32} = 0.005 \]
Step 2: Use this value to find the moles of solute:
\[ \frac{n_{solute}}{n_{solution}} = 0.005 \quad \Rightarrow \quad n_{solute} = 0.005 \times \left( \frac{200}{18} \right) \]
This gives:
\[ n_{solute} = 0.005 \times 11.11 = 0.0555 mol \]
Step 3: Calculate the molar mass:
\[ Molar mass of solute = \frac{mass of solute}{n_{solute}} = \frac{5 \, g}{0.0555 \, mol} = 90.05 \, g/mol \]
So, the molar mass of the solute is 90.05 g/mol. Quick Tip: Raoult's Law allows the calculation of molar mass of non-volatile solutes by using vapor pressure lowering. Ensure to use proper unit conversions and balance the mole fraction equations carefully.
The conductivity of a 0.2 M solution of KCl is \( 2.48 \times 10^{-2} \) S cm\(^{-1}\). Calculate its molar conductivity and degree of dissociation (\( \alpha \)).
Given: \( \lambda^0_{K^+} = 73.5 \, S cm^2 mol^{-1} \), \( \lambda^0_{Cl^-} = 76.5 \, S cm^2 mol^{-1} \)
View Solution
Step 1: Molar conductivity \( \Lambda_m \)
The molar conductivity \( \Lambda_m \) is given by:
\[ \Lambda_m = \frac{k}{C} \]
Where:
- \( k = 2.48 \times 10^{-2} \, S cm^{-1} \) is the conductivity of the solution,
- \( C = 0.2 \, mol/L \) is the concentration of the solution.
First, we multiply the conductivity by 1000 to convert the units from \( S cm^{-1} \) to \( S L mol^{-1} \):
\[ \Lambda_m = \frac{k \times 1000}{C} = \frac{1000 \times 2.48 \times 10^{-2}}{0.2} \]
This gives:
\[ \Lambda_m = 124 \, S cm^2 mol^{-1} \]
Step 2: Calculate the molar conductivity at infinite dilution \( \Lambda^0 \)
The molar conductivity at infinite dilution is the sum of the molar conductivities of the individual ions:
\[ \Lambda^0_m = \lambda^0_{K^+} + \lambda^0_{Cl^-} \]
Substitute the given values:
\[ \Lambda^0_m = 73.5 + 76.5 = 150 \, S cm^2 mol^{-1} \]
Step 3: Calculate the degree of dissociation \( \alpha \)
The degree of dissociation \( \alpha \) is given by the ratio of the observed molar conductivity to the molar conductivity at infinite dilution:
\[ \alpha = \frac{\Lambda_m}{\Lambda^0_m} = \frac{124}{150} = 0.826 \]
Thus, the degree of dissociation is approximately 0.826. Quick Tip: Molar conductivity can be calculated using the solution's conductivity and concentration. The degree of dissociation can be determined by comparing the observed molar conductivity to the molar conductivity at infinite dilution.
Question 29:
The involvement of (n 1)d electrons in the behaviour of transition elements impart certain distinct characteristics to these elements. Thus, in addition to variable oxidation states, they exhibit paramagnetic behaviour, catalytic properties and tendency for the formation of coloured ions. The transition metals react with a number of non-metals like oxygen, nitrogen and halogens. KMnO4 and K2Cr2O7 are common examples. The two series of inner transition elements, lanthanoids and actinoids, constitute the f-block of the periodic table. In the lanthanoids, there is regular decrease in atomic size with increase in atomic number due to the imperfect shielding effect of 4f-orbital electrons which causes contraction.
(a) Why do transition metals and their compounds act as good catalysts?
View Solution
Transition metals and their compounds act as good catalysts because they have multiple oxidation states, which allows them to participate in electron transfer during reactions. Additionally, their ability to form complex intermediates and adsorb reactants on their surface reduces the activation energy, facilitating the reaction. Quick Tip: Catalysts work by lowering the activation energy for a reaction, often by providing an alternative reaction pathway. Transition metals are especially effective because of their variable oxidation states.
(b) Define lanthanoid contraction. How does it affect the atomic radii of the third transition series and the second transition series?
View Solution
Lanthanoid Contraction refers to the gradual decrease in atomic size across the lanthanoid series, due to the ineffective shielding of the 4f-electrons. This contraction affects the atomic radii of the third transition series, which are smaller than expected compared to the second transition series. The increased nuclear charge in the lanthanoids results in a stronger pull on the electrons, reducing their size. Quick Tip: Lanthanoid contraction is caused by the poor shielding effect of 4f-electrons, leading to a consistent decrease in atomic size as we move across the lanthanoid series.
Question 29:
(c) In aqueous media, which is a stronger reducing agent — Cr³⁺ or Fe³⁺ and why?
View Solution
Cr³⁺ is a stronger reducing agent than Fe³⁺ because Cr³⁺ is more easily reduced to Cr²⁺, which is relatively stable. Fe³⁺, on the other hand, is more stable in its +3 oxidation state and is less readily reduced to Fe²⁺. Quick Tip: The strength of a reducing agent is determined by how easily it can lose electrons. Cr³⁺ is more readily reduced to Cr²⁺, making it a stronger reducing agent compared to Fe³⁺.
Question 30:
Proteins are the most abundant biomolecules of the living system. Proteins are the polymers of about twenty different -amino acids which are linked by peptide bonds. Ten amino acids are called essential amino acids. In zwitter ionic form, amino acids show amphoteric behaviour as they react both with acids and bases.
On the basis of their molecular shape, proteins are classified into two types : Fibrous and Globular proteins. Structure and shape of proteins can be studied at four different levels i.e., primary, secondary, tertiary and quaternary, each level being more complex than the previous one. The secondary or tertiary structure of proteins get disturbed on change of pH or temperature and they are not able to perform their functions. This is called denaturation of proteins.
Question 30:
(a) What are essential amino acids?
View Solution
Essential amino acids are amino acids that the body cannot synthesize on its own and must be obtained through diet. Examples include leucine, lysine, and tryptophan. Quick Tip: Essential amino acids are those that cannot be synthesized by the body and must be consumed through diet to maintain proper metabolic function.
(b) What is meant by zwitter ionic form of amino acids?
View Solution
In the zwitter ionic form, amino acids have both a positive and a negative charge within the same molecule. The amino group (-NH₂) is protonated to form -NH₃⁺, and the carboxyl group (-COOH) is deprotonated to form -COO⁻. Quick Tip: Amino acids exist in a zwitterionic form at physiological pH, where they have both a positive and negative charge, making them amphoteric.
(c) (i) Give one example each for Fibrous protein and Globular protein.
View Solution
- Fibrous Protein: Collagen is an example of a fibrous protein. It is elongated and provides structural support in connective tissues.
- Globular Protein: Hemoglobin is an example of a globular protein. It is compact and involved in transporting oxygen in the blood.
(c) (ii) What type of linkages hold monomers of proteins together?
View Solution
Monomers of proteins are held together by peptide bonds, which form between the carboxyl group of one amino acid and the amino group of another.
Quick Tip: Peptide bonds are covalent bonds formed between the amino group of one amino acid and the carboxyl group of another, linking amino acids in a protein chain.
(c) (i) What is the structural feature that characterizes a reducing sugar?
View Solution
A reducing sugar contains a free aldehyde or ketone group, which allows it to act as a reducing agent. This group can undergo oxidation, as seen in glucose and maltose.
(c) (ii)What is the structural difference between nucleoside and nucleotide?
View Solution
A nucleoside consists of a nitrogenous base and a sugar (ribose or deoxyribose), while a nucleotide includes a nucleoside plus a phosphate group attached to the sugar. Quick Tip: Nucleotides are the building blocks of nucleic acids, and they consist of a nitrogenous base, a sugar, and a phosphate group. Nucleosides lack the phosphate group.
(a) (i) Write the major product(s) in the following reactions:
View Solution
(1) The reaction involves oxidation using KMnO₄ and KOH, which leads to the formation of a carboxylic acid. The product is Benzoic acid (C₆H₅COOH).
\[ C_6H_5CH_2CH_3 \xrightarrow{KMnO_4, KOH} C_6H_5COOH \]
(2) The reaction between benzaldehyde and acetone in the presence of dilute NaOH leads to the formation of Aldol product (4-hydroxy-4-phenylpent-3-en-2-one), followed by dehydration, which forms Cinnamon aldehyde.
\[ C_6H_5CHO + CH_3COCH_3 \xrightarrow{dil NaOH} C_6H_5CH= CHCOCH_3 \]
(3) Bromination of benzoic acid in the presence of FeBr₃ leads to the formation of 2,4,6-Tribromobenzoic acid.
\[ C_6H_5COOH \xrightarrow{Br_2 / FeBr_3} C_6H_2(Br)_3COOH \] Quick Tip: In oxidation reactions with KMnO₄, alkyl groups are converted into carboxylic acids, and in electrophilic aromatic substitution, bromination leads to substitution at the positions ortho and para to the existing functional group.
(ii)Give simple chemical tests to distinguish between the following pairs of compounds:
(1) \( C_6H_5COCH_3 and C_6H_5COCH_2CH_3 \) (3+2=5)
(2) Pentanal and Pentan-3-one
View Solution
Aldehydes react positively with Tollen's or Fehling's solution, while ketones do not react in this way. The Iodoform test helps distinguish methyl ketones from others by forming a yellow precipitate.
(b) (i) Give Reason for the following:
(1) In semicarbazide, only one -NH₂ group is involved in the formation of semicarbazone.
View Solution
(1) In semicarbazide, the carbonyl group (-C=O) in aldehydes or ketones reacts with the -NH
group. However, only the first -NH group is involved because the second -NH group is at-
tached to a nitrogen atom and is not available to react.
(b)(i) (2) Acetaldehyde is more reactive than acetone towards addition of HCN.
View Solution
Acetaldehyde is more reactive than acetone towards HCN addition because acetaldehyde has a smaller electron-donating alkyl group (-CH₃) compared to acetone, making its carbonyl carbon more electrophilic and susceptible to nucleophilic attack by HCN. Quick Tip: In semicarbazide, the reaction occurs via the available -NH₂ group attached to the carbonyl. Aldehydes are generally more reactive than ketones due to their less hindered electron distribution.
(b) (ii) (1) Arrange the following in decreasing order of their acidic strength:
\( CH_3COOH, \, O_2N-CH_2COOH, \, HCOOH \)
View Solution
The order of acidic strength is:
O2N − CH2COOH > HCOOH > CH3COOH.
This is due to the electron-withdrawing effect of the nitro group, which stabilizes the conjugate
base more, making O2N − CH2COOH the strongest acid.
(b) (ii) (2) Name the reagent in the following reaction:
\( CH_3CH = CH-CN \xrightarrow{} CH_3CH = CH-CHO \)
View Solution
The reagent is Boron Trifluoride (BF), which facilitates the conversion of the nitrile group to
an aldehyde group via hydrolysis Quick Tip: Electron-withdrawing groups increase the acidity of carboxylic acids, and BF₃ is commonly used in the hydrolysis of nitriles to aldehydes.
(iii) Write the reaction involved in Hell-Volhard-Zelinsky reaction.
View Solution
The Hell-Volhard-Zelinsky reaction involves the halogenation of carboxylic acids at the α-position using a halogen (usually bromine) and a catalyst (like PBr₃ or Br₂ with a catalyst):
\[ RCOOH \xrightarrow{Br_2, \, PBr_3} \, \alpha-Bromocarboxylic acid \] Quick Tip: The Hell-Volhard-Zelinsky reaction halogenates the α-carbon of carboxylic acids, providing a useful method for introducing halogen atoms into organic compounds.
(a) Write the IUPAC name of the complex:
\([Co(H_2O)(CN)(en_2)^{2+}]\)
View Solution
diaquacyanido-bis(ethylenediamine)cobalt(III) ion.
- ”aqua” represents the two water molecules (H2O) ligands,
- ”cyano” represents the cyanide (CN) ligand,
- ”ethylenediamine” represents the bidentate ligand (en),
- Cobalt is in the +3 oxidation state
Quick Tip: In naming coordination compounds, list the ligands alphabetically and indicate the oxidation state of the central metal ion in Roman numerals.
(b) Why is geometrical isomerism not possible in tetrahedral complexes having two different types of unidentate ligands coordinated with the central metal?
View Solution
Geometrical isomerism is not possible in tetrahedral complexes with two different uniden-
tate ligands because of the symmetry of the tetrahedral geometry. In a tetrahedral complex,
all four positions are equivalent, and swapping the two different ligands does not result in
distinct spatial arrangements. Therefore, no isomers with different spatial arrangements are
possible.
Quick Tip: Geometrical isomerism is possible in square planar or octahedral complexes, but tetrahedral complexes generally lack enough symmetry to support distinct isomers.
(c)Arrange the following complex ions in increasing order of their crystal field splitting energy (\( \Delta_0 \)):
\([Co(NH_3)_6]^{3+}, \, [CoF_6]^{3-}, \, [Co(CN)_6]^{3-}\)
View Solution
Ligand field strength influences the crystal field splitting energy (\( \Delta_0 \)). Cyanide (\( CN^- \)) is a strong field ligand, followed by ammonia (\( NH_3 \)), and fluoride (\( F^- \)) is a weak field ligand.
(d) Write the hybridization and magnetic character of the complex \([Ni(CO)_4]\) on the basis of valence bond theory.
[Atomic No. Ni = 28]
View Solution
For the complex \([Ni(CO)_4]\), nickel is in the +0 oxidation state. The electronic configuration of Ni is \( 3d^8 4s^2 \).
- Hybridization: In \([Ni(CO)_4]\), the CO ligands are strong field ligands that cause the pairing of the 3d electrons, resulting in the hybridization \( sp^3 \). The geometry of the complex is tetrahedral.
- Magnetic character: Since all electrons are paired in the complex, it is diamagnetic.
Explanation:
- In the presence of CO, which is a strong field ligand, the 3d electrons are paired, leading to a tetrahedral geometry with \( sp^3 \) hybridization and no unpaired electrons, thus the complex is diamagnetic. Quick Tip: The hybridization of a metal complex depends on the geometry and the nature of the ligands. CO is a strong field ligand that leads to electron pairing, resulting in a tetrahedral geometry.
(e) Out of \([CoF_6]^{3-}\) and \([Co(C_2O_4)_3]^{3-}\), which complex is:
(i) More stable?
(ii) The high-spin complex?
View Solution
(i) More stable: The [Co(C2O4)3]3− complex is more stable than [CoF6]3− because oxalate
(C2O2−
4 ) is a chelating ligand, which provides additional stability through chelation. - (ii)
High-spin complex: The [CoF6]3− complex is a high-spin complex because fluoride is a weak
field ligand that does not cause significant pairing of electrons.
Explanation:
- Chelating ligands such as oxalate lead to greater stability due to their ability to form multiple
bonds with the central metal.
- Weak field ligands, such as fluoride, lead to high-spin complexes where electrons occupy
higher-energy orbitals to minimize electron-electron repulsion.
Quick Tip: Chelating ligands like oxalate form more stable complexes due to their ability to bind to the metal at multiple points, resulting in greater stability.
(f) What is the difference between an ambidentate ligand and a bidentate ligand?
View Solution
- Ambidentate ligand: A ligand that can coordinate to the central metal through two different atoms, but not simultaneously, is called an ambidentate ligand. For example, the cyanide ion (\( CN^- \)) can bind through either the carbon or nitrogen atom.
- Bidentate ligand: A ligand that can form two bonds with the central metal ion through two different donor atoms is called a bidentate ligand. For example, ethylenediamine (\( en \)) can form two bonds with the central metal.
Explanation:
- Ambidentate ligands bind through two possible donor atoms, while bidentate ligands always bind through two atoms, resulting in a more stable complex due to chelation. Quick Tip: Ambidentate ligands can bind through multiple donor atoms, while bidentate ligands always bind through two atoms.
(g) Write the electronic configuration of \(d^5\) in terms of \( t_{2g} \) and \( e_g \) in an octahedral field as:
(i) \( \Delta_o > P
(ii) \Delta_o < P \)
View Solution
- (i) \( \Delta_o > P \): When the crystal field splitting energy (\( \Delta_o \)) is greater than the pairing energy (\( P \)), the electrons will occupy the lower-energy \( t_{2g} \) orbitals first, leading to the configuration \( t_{2g}^6 e_g^0 \). This corresponds to a low-spin configuration.
- (ii) \( \Delta_o < P \): When \( \Delta_o \) is smaller than \( P \), the electrons will prefer to occupy the higher-energy \( e_g \) orbitals to minimize repulsion, leading to the configuration \( t_{2g}^4 e_g^1 \), a high-spin configuration.
Explanation:
- Crystal field splitting energy and pairing energy determine whether a complex is high-spin or low-spin. A larger \( \Delta_o \) favors low-spin, while a smaller \( \Delta_o \) favors high-spin. Quick Tip: The relative strength of \( \Delta_o \) and \( P \) determines whether a complex is high-spin or low-spin. A larger \( \Delta_o \) favors low-spin, while a smaller \( \Delta_o \) favors high-spin.
(a) (i) Calculate emf of the following cell at 25°C:
\(Zn (s) | Zn^{2+} (0.001M) || Cd^{2+} (0.1M) | Cd (s)\)
Given: \( E^\circ_{Zn^{2+}/Zn} = -0.76 \, V, E^\circ_{Cd^{2+}/Cd} = -0.40 \, V \)
\([ \log 10 = 1 ]\)
View Solution
The emf of the cell can be calculated using the Nernst equation:
\[ E = E^\circ - \frac{0.0591}{n} \log Q \]
Where:
- \( E^\circ = E^\circ_{cathode} - E^\circ_{anode} \),
- \( n \) is the number of moles of electrons transferred (here, \(n = 2\) for the zinc and cadmium half-reactions),
- \( Q \) is the reaction quotient, which for the given cell is:
\[ Q = \frac{[Zn^{2+}][Cd^{2+}]}{[Cd^{2+}][Zn^{2+}]} \]
Given concentrations are \( [Zn^{2+}] = 0.001M \) and \( [Cd^{2+}] = 0.1M \). Therefore,
\[ Q = \frac{0.001 \times 0.1}{1} = 0.0001 \]
Now, substituting the values into the Nernst equation:
\[ E = (-0.40 - (-0.76)) - \frac{0.0591}{2} \log (0.0001) \]
\[ E = 0.36 - \frac{0.0591}{2} \times (-4) \]
\[ E = 0.36 + 0.1182 \]
\[ E = 0.4782 \, V \]
So, the emf of the cell is \( 0.4782 \, V \).
Quick Tip: The Nernst equation is essential for calculating the emf of a cell under non-standard conditions. Ensure to substitute the correct values of concentrations and standard electrode potentials.
(a) (ii)State Faraday's second law of electrolysis. How will the pH of aqueous NaCl solution be affected when it is electrolyzed?
View Solution
N/A Quick Tip: Faraday's second law relates the quantity of substance deposited to the amount of electricity passed. Electrolysis of NaCl leads to an increase in pH at the cathode and a decrease at the anode due to the generation of hydroxide and hydrogen ions.
(b)(i) Calculate the \( \Delta G^\circ \) and \( \log K_c \) for the following cell reaction:
\(Fe (s) + Ag^+ (aq) \rightleftharpoons Fe^{2+} (aq) + Ag (s)\)
Given: \( E^\circ_{Fe^{2+}/Fe} = -0.44 \, V, E^\circ_{Ag^+/ Ag} = +0.80 \, V \)
\( 1 F = 96500 \, C mol^{-1} \)
View Solution
First, calculate the standard cell potential (\( E^\circ_{cell} \)):
\[ E^\circ_{cell} = E^\circ_{cathode} - E^\circ_{anode} = 0.80 - (-0.44) = 1.24 \, V \]
Next, calculate \( \Delta G^\circ \) using the equation:
\[ \Delta G^\circ = -nFE^\circ_{cell} \]
Since the number of electrons transferred \( n = 2 \):
\[ \Delta G^\circ = -2 \times 96500 \times 1.24 = -239,360 \, J/mol = -239.36 \, kJ/mol \]
Now, calculate \( \log K_c \) using the relationship:
\[ \Delta G^\circ = -RT \ln K_c \]
At 25°C, \( R = 8.314 \, J/mol·K, T = 298 \, K \):
\[ -239,360 = -(8.314)(298) \ln K_c \]
Solving for \( \ln K_c \):
\[ \ln K_c = \frac{239,360}{(8.314)(298)} = 96.5 \]
Therefore:
\[ K_c = e^{96.5} \quad \Rightarrow \quad \log K_c = 96.5 \, (approx.) \]
Thus, \( \Delta G^\circ = -239.36 \, kJ/mol \) and \( \log K_c = 96.5 \). Quick Tip: The Nernst equation can help relate the cell potential to equilibrium constants. A more positive standard electrode potential indicates a more spontaneous reaction.
(b)(ii) Write any two advantages of fuel cells over primary and secondary batteries?
View Solution
1. Higher efficiency: Fuel cells produce energy continuously as long as fuel is supplied, and they have higher efficiency than conventional batteries, which store limited energy.
2. Environmentally friendly: The only by-products of hydrogen fuel cells are water and heat, making them cleaner than batteries that involve toxic or harmful chemicals. Quick Tip: Fuel cells are advantageous over conventional batteries because of their higher efficiency and environmental friendliness.
(b)(iii) How many Faradays are required for the oxidation of one mole of \( H_2O \) to \( O_2 \)?
View Solution
The oxidation of \( H_2O \) to \( O_2 \) involves the following reaction:
\[ 2 H_2O \rightarrow O_2 + 4 H^+ + 4e^- \]
Since 4 electrons are involved in the oxidation of 2 moles of water, the number of Faradays required for 1 mole of \( H_2O \) to produce 1 mole of \( O_2 \) is 4.
Thus, 4 Faradays are required. Quick Tip: The number of Faradays required for a reaction is determined by the number of electrons transferred during the reaction. One mole of electrons corresponds to one Faraday.
































Comments