CBSE Class 12 Chemistry exam is being conducted today on February 27, 2025, from 10:30 AM to 1:30 PM.The Chemistry theory paper has 70 marks, while 30 marks are allocated for the practical assessment. The paper is divided into Physical, Organic, and Inorganic Chemistry, and it includes numerical, conceptual, and application-based problems. The question paper includes multiple-choice questions (1 mark each), short-answer questions (3 marks each), and long-answer questions (5 marks each).
CBSE Class 12 Chemistry Question Paper 2025 PDF (Set 3) is available for download here.
CBSE Class 12 Chemistry Question Paper 2025 (Set 3) with Answer Key
| CBSE Class 12 2025 Chemistry Question Paper with Answer Key | Download | Check Solution |

Standard electrode potential for Sn\(^{4+}\)/Sn\(^{2+}\) couple is +0.15 V and that for the Cr\(^{3+}\)/Cr couple is –0.74 V. The two couples in their standard states are connected to make a cell. The cell potential will be
View Solution
The magnetic moment is associated with its spin angular momentum and orbital angular momentum. Spin only magnetic moment value of Cr\(^{3+}\) ion (Atomic no. : Cr = 24) is ____ .
View Solution
In case of association, abnormal molar mass of solute will
View Solution
Alkyl halides undergoing nucleophilic bimolecular substitution reaction involve
View Solution
Arrange the following compounds in increasing order of their boiling points:
The correct order is
View Solution
The correct IUPAC name of [Pt(NH\(_3\))\(_2\)Cl\(_2\)]\(^{2+}\) is
View Solution
The acid formed when propyl magnesium bromide is treated with CO\(_2\) followed by acid hydrolysis is :
View Solution
Acidified KMnO\(_4\) oxidises sulphite to
View Solution
Which is the correct order of acid strength from the following?
View Solution
An unripe mango placed in a concentrated salt solution to prepare pickle, shrivels because
View Solution
The best reagent for converting propanamide into propanamine is ___.
View Solution
Which of the following statements is not true about glucose?
View Solution
Assertion (A) : All naturally occurring \(\alpha\)-amino acids except glycine are optically active.
Reason (R) : Most naturally occurring amino acids have L-configuration.
View Solution
Assertion (A) : Naturally occurring amino acids except glycine are optically active.
Reason (R) : Glycine has no chiral center.
View Solution
Assertion (A) : The boiling points of alkyl halides decrease in the order: RI \(>\) RBr \(>\) RCl \(>\) RF.
Reason (R) : The boiling points of alkyl chlorides, bromides, and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
View Solution
Assertion (A) : [Cr(H\(_2\)O)\(_6\)]Cl\(_2\) and [Fe(H\(_2\)O)\(_6\)]Cl\(_2\) are examples of homoleptic complexes.
Reason (R) : All the ligands attached to the metal are the same.
View Solution
Would you expect benzaldehyde to be more reactive or less reactive in nucleophilic addition reactions than propanal? Justify your answer.
View Solution
Complete and balance the following chemical equations:
(a)
8MnO\(_4^-\) + 3S\(_2\)O\(_3^{2-}\) + H\(_2\)O \(\rightarrow\) 8Mn\(^{2+}\) + 3SO\(_4^{2-}\) + 2H\(^+\)
(b)
Cr\(_2\)O\(_7^{2-}\) + 3Sn\(^{2+}\) + 14H\(^+\) \(\rightarrow\) 2Cr\(^{3+}\) + 3Sn\(^{4+}\) + 7H\(_2\)O
View Solution
(A) Give reasons:
(a) Cooking is faster in a pressure cooker than in an open pan.
(b) On mixing liquid X and liquid Y, volume of the resulting solution decreases. What type of deviation from Raoult’s law is shown by the resulting solution? What change in temperature would you observe after mixing liquids X and Y?
View Solution
Define Azeotrope. What type of Azeotrope is formed by negative deviation from Raoult’s law? Give an example.
View Solution
Identify A and B in each of the following reaction sequences:
(a)
CH\(_3\)CH\(_2\)Cl \(\xrightarrow{NaCN}\) A \(\xrightarrow{H_2/Ni}\) B
(b)
C\(_6\)H\(_5\)NH\(_2\) \(\xrightarrow{NaNO_2/HCl, 0-5^\circ C}\) A \(\xrightarrow{H^+}\) B
View Solution
What are the hydrolysis products of:
(a)Sucrose
(b)Lactose
View Solution
- (a) The hydrolysis of sucrose (C\(_{12}\)H\(_{22}\)O\(_{11}\)) results in the formation of glucose (C\(_6\)H\(_{12}\)O\(_6\)) and fructose (C\(_6\)H\(_{12}\)O\(_6\)).
- (b) The hydrolysis of lactose (C\(_{12}\)H\(_{22}\)O\(_{11}\)) produces glucose (C\(_6\)H\(_{12}\)O\(_6\)) and galactose (C\(_6\)H\(_{12}\)O\(_6\)).
Quick Tip: To identify hydrolysis products, remember that sucrose is a disaccharide made of glucose and fructose, while lactose consists of glucose and galactose. Hydrolysis breaks the glycosidic bond in both sugars.
Henry's law constant for CO\(_2\) in water is 1.67 \(\times\) 10\(^8\) Pa at 298 K. Calculate the number of moles of CO\(_2\) in 500 mL of soda water when packed under 2.53 \(\times\) 10\(^5\) Pa at the same temperature.
View Solution
Henry's law states that the concentration of a gas in a liquid is directly proportional to the pressure of the gas above the liquid: \[ C = k_H \times P \]
Where:
- \(C\) is the concentration of the gas (mol/L),
- \(k_H\) is Henry's law constant (Pa),
- \(P\) is the partial pressure of the gas (Pa).
Given:
- \(k_H = 1.67 \times 10^8\) Pa,
- \(P = 2.53 \times 10^5\) Pa,
- Volume = 500 mL = 0.5 L,
Using Henry’s law: \[ C = (1.67 \times 10^8) \times (2.53 \times 10^5) = 4.23 \times 10^{13} \, mol/L \, Pa \]
Now, to calculate the moles of CO\(_2\), use the relation: \[ moles of CO_2 = C \times V = (4.23 \times 10^{-8} \, mol/L) \times (0.5 \, L) = 2.12 \times 10^{-8} \, mol \] Quick Tip: In Henry’s law, remember to use the pressure in the same units as the constant to ensure correct results.
Calculate \(\Delta_r G^\circ\) and log \(K_C\) of the reaction:
2Cr(s) + 3Cd\(^{2+}\)(aq) \(\rightarrow\) 2Cr\(^{3+}\)(aq) + 3Cd(s)
% Given data:
Given:
\(E^\circ_{Cr^{3+}/Cr} = -0.74\) V
\(E^\circ_{Cd^{2+}/Cd} = -0.40\) V
\(R = 8.314 \, J K^{-1} mol^{-1}\)
\(F = 96500 \, C mol^{-1}\)
View Solution
First, calculate the cell potential \(E^\circ_cell\) using the formula: \[ E^\circ_cell = E^\circ_{cathode} - E^\circ_{anode} \]
Here, Cr is oxidized (anode) and Cd\(^{2+}\) is reduced (cathode), so: \[ E^\circ_cell = (-0.40) - (-0.74) = 0.34 \, V \]
Next, calculate the Gibbs free energy change \(\Delta_r G^\circ\) using the relation: \[ \Delta_r G^\circ = -nFE^\circ_cell \]
Since the reaction involves 6 electrons (from 2 Cr\(^{3+}\) to Cr and 3 Cd\(^{2+}\) to Cd), we get: \[ \Delta_r G^\circ = -6 \times (96500) \times (0.34) = -197220 \, J/mol = -197.22 \, kJ/mol \]
To calculate \(K_C\), use the relation: \[ \Delta_r G^\circ = -RT \ln K_C \] \[ K_C = e^{-\Delta_r G^\circ / RT} = e^{(197220) / (8.314 \times 298)} = e^{80} \approx 10^{38} \]
Thus, log \(K_C\) = 38.
Quick Tip: To convert from Gibbs free energy to the equilibrium constant, use the equation: \[ \Delta_r G^\circ = -RT \ln K_C \] This equation relates the spontaneity of a reaction to the equilibrium constant.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
% Given data:
Given:
Rate increases by a factor of 4 when temperature changes from 293 K to 313 K.
\(R = 8.314 \, J K^{-1} mol^{-1}\), log 4 = 0.602, log 2 = 0.301.
View Solution
The relation between the rate constant and temperature is given by the Arrhenius equation: \[ \ln \left(\frac{k_2}{k_1}\right) = \frac{E_a}{R} \left(\frac{1}{T_1} - \frac{1}{T_2}\right) \]
Where:
- \(k_1\) and \(k_2\) are the rate constants at temperatures \(T_1\) and \(T_2\),
- \(E_a\) is the activation energy,
- \(R\) is the gas constant,
- \(T_1 = 293\) K, \(T_2 = 313\) K.
Given that the rate quadruples, \[ \frac{k_2}{k_1} = 4 \Rightarrow \ln 4 = \frac{E_a}{8.314} \left(\frac{1}{293} - \frac{1}{313}\right) \] \[ 0.602 = \frac{E_a}{8.314} \left(\frac{1}{293} - \frac{1}{313}\right) = \frac{E_a}{8.314} \times \left(0.00342 - 0.00319\right) \] \[ 0.602 = \frac{E_a}{8.314} \times 0.00023 \] \[ E_a = \frac{0.602 \times 8.314}{0.00023} = 21936 \, J/mol = 21.94 \, kJ/mol \] Quick Tip: For a temperature increase that leads to a change in reaction rate, use the Arrhenius equation to calculate the activation energy. Remember that rate constants are related to temperature exponentially.
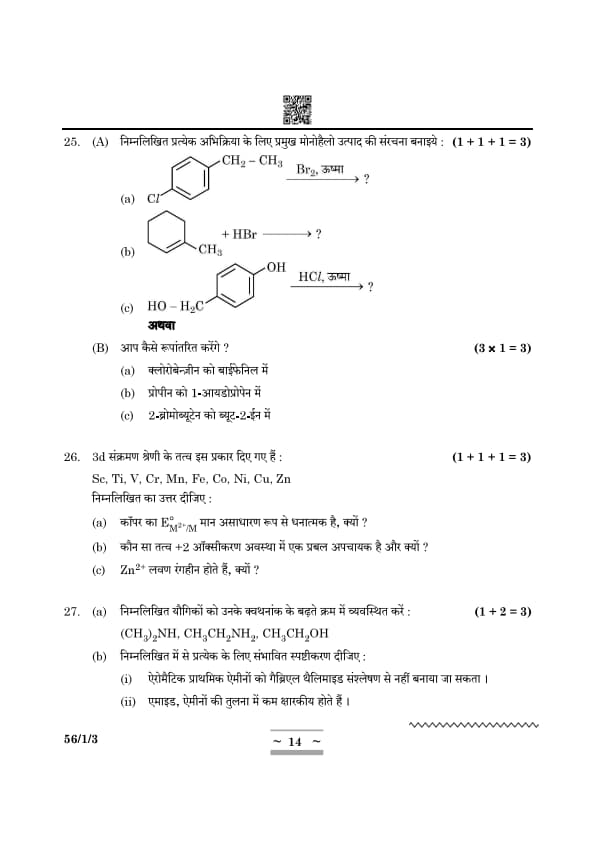
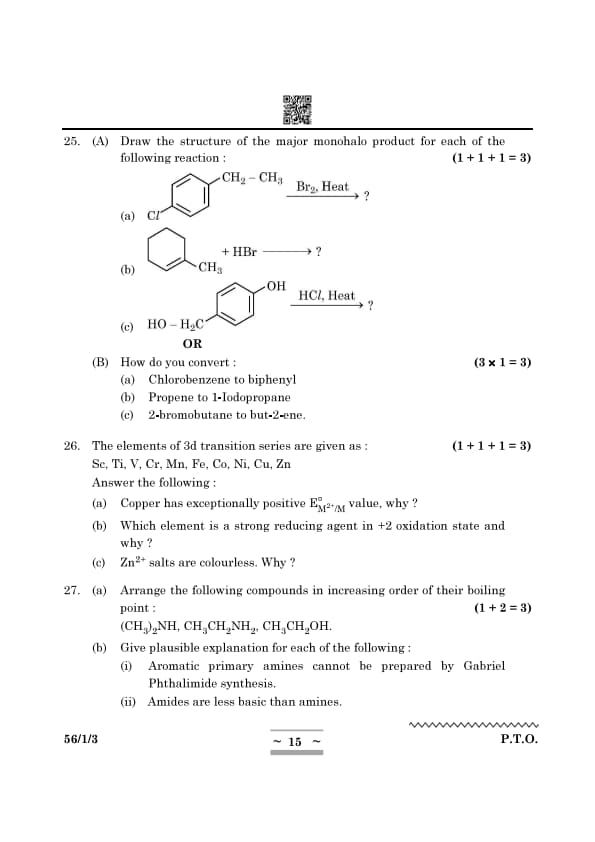
Draw the structure of the major monohalo product for each of the following reactions:
\[ (a) \quad CH_2 – CH_3 \xrightarrow{Br_2, Heat} \ ? \]
\[ (b) \quad CH_3 – HBr \xrightarrow{} \ ? \]
\[ (c) \quad HO – H_2 C \xrightarrow{HCl, Heat} \ ? \]
View Solution
How do you convert:
[(a)] Chlorobenzene to biphenyl
[(b)] Propene to 1-Iodopropane
[(c)] 2-Bromobutane to but-2-ene
View Solution
The elements of the 3d transition series are given as: Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn
Answer the following:
[(a)] Copper has exceptionally positive \(E^\circ_{M^{2+}/M}\) value, why?
[(b)] Which element is a strong reducing agent in +2 oxidation state and why?
[(c)] Zn\(^{2+}\) salts are colourless. Why?
View Solution
(a) Arrange the following compounds in increasing order of their boiling point: \[ (CH_3)_2NH, CH_3CH_2NH_2, CH_3CH_2OH. \]
(b) Give plausible explanation for each of the following:
[(i)] Aromatic primary amines cannot be prepared by Gabriel Phthalimide synthesis.
[(ii)] Amides are less basic than amines.
View Solution
Define the following terms:
[(a)] Native protein
[(b)] Nucleotide
[(c)] Essential amino acid
View Solution
(a) A native protein refers to a protein that is in its natural, functional form, with its specific three-dimensional structure intact. This form allows the protein to perform its biological function.
(b) A nucleotide is a basic building block of nucleic acids (DNA and RNA). It consists of a nitrogenous base, a sugar molecule, and a phosphate group. Nucleotides link together to form the backbone of nucleic acids.
(c) An essential amino acid is an amino acid that cannot be synthesized by the body and must be obtained through the diet. Examples include lysine, leucine, and tryptophan.
Quick Tip: In biochemistry, understanding the structure and function of proteins, nucleotides, and amino acids is crucial for exploring metabolic processes.
The rate of a chemical reaction is expressed either in terms of decrease in the concentration of reactants or increase in the concentration of a product per unit time. Rate of the reaction depends upon the nature of reactants, concentration of reactants, temperature, presence of catalyst, surface area of the reactants and presence of light. Rate of reaction is directly related to the concentration of reactant. Rate law states that the rate of reaction depends upon the concentration terms on which the rate of reaction actually depends, as observed experimentally. The sum of powers of the concentration of the reactants in the Rate law expression is called order of reaction while the number of reacting species taking part in an elementary reaction which must collide simultaneously in order to bring about a chemical reaction is called molecularity of the reaction.
Answer the following questions:
(a) (i) What is a rate determining step?
View Solution
Phenols undergo electrophilic substitution reactions readily due to the strong activating effect of the OH group attached to the benzene ring. Since, the OH group increases the electron density more to o– and p– positions therefore OH group is ortho, para-directing. Reimer-Tiemann reaction is one of the examples of aldehyde group being introduced on the aromatic ring of phenol, ortho to the hydroxyl group. This is a general method used for the ortho-formylation of phenols.
Answer the following questions:
(a) What happens when phenol reacts with:
(i) Br\(_2\)/CS\(_2\)
View Solution
(a) Carry out the following conversions:
(i) Ethanol to But-2-enal
View Solution
An organic compound (A) (molecular formula C\(_8\)H\(_{16}\)O\(_2\)) was hydrolyzed with dilute sulphuric acid to get a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) on dehydration gives But-1-ene. Identify (A), (B) and (C) and write chemical equations for the reactions involved.
View Solution
1. Identification of (A):
The molecular formula of (A) is C\(_8\)H\(_{16}\)O\(_2\). A compound with this formula could be 1-octanol (CH\(_3\)(CH\(_2\))\(_6\)CH\(_2\)OH), as it fits the molecular weight and structure.
2. Reaction of (A) with dilute sulphuric acid:
When 1-octanol reacts with dilute sulphuric acid, it undergoes hydrolysis, breaking the molecule into a carboxylic acid and an alcohol. In this case, it forms octanoic acid (CH\(_3\)CH\(_2\)CH\(_2\)CH\(_2\)CH\(_2\)COOH) and 1-propanol (CH\(_3\)CH\(_2\)OH) as products.
3. Oxidation of (C) (1-propanol) with chromic acid:
1-Propanol (C) is oxidized by chromic acid (H\(_2\)CrO\(_4\)) to form propanoic acid (CH\(_3\)CH\(_2\)COOH), which is compound (B). The oxidation reaction is:
\[ CH_3CH_2OH + [O] \rightarrow CH_3CH_2COOH \]
4. Dehydration of (C) (1-propanol) to But-1-ene:
On dehydration, 1-propanol undergoes elimination to form But-1-ene. The reaction proceeds as follows:
\[ CH_3CH_2OH \xrightarrow{H_2SO_4} CH_3CH=CH_2 \]
Thus, the identified compounds are:
- (A) = 1-Octanol
- (B) = Propanoic acid
- (C) = 1-Propanol Quick Tip: Dehydration reactions of alcohols generally lead to the formation of alkenes, with the elimination of a water molecule.
In the following complex ions, explain the type of hybridization, shape, and magnetic property: (2\(\frac{1}{2}\) x 2 = 5)
(a) [Fe(H\(_2\)O)\(_6\)]\(^{2+}\)
(b) [NiCl\(_4\)]\(^{2-}\)
View Solution
1. [Fe(H\(_2\)O)\(_6\)]\(^{2+}\):
- Hybridization: The central iron ion (Fe\(^{2+}\)) is surrounded by six water molecules. This corresponds to an octahedral geometry, which is formed due to sp\(^3\)d\(^2\) hybridization.
- Shape: Octahedral.
- Magnetic property: Since Fe\(^{2+}\) has unpaired electrons, the complex is paramagnetic.
2. [NiCl\(_4\)]\(^{2-}\):
- Hybridization: The central nickel ion (Ni\(^{2+}\)) is surrounded by four chloride ions, resulting in a tetrahedral geometry. This geometry arises from sp\(^3\) hybridization.
- Shape: Tetrahedral.
- Magnetic property: Ni\(^{2+}\) has unpaired electrons, so this complex is paramagnetic. Quick Tip: Octahedral complexes often have sp\(^3\)d\(^2\) hybridization, while tetrahedral complexes typically use sp\(^3\) hybridization. Paramagnetism is common in complexes with unpaired electrons.
Write IUPAC names of the following: (3 + 2 = 5)
(i) [Co(H\(_2\)O)(CN)(en)\(_2\)]\(^{2+}\)
(ii) [PtCl\(_4\)]\(^{2-}\)
(iii) [Cr(NH\(_3\))\(_4\)Cl(ONO)]\(^+\)
View Solution
1. [Co(H\(_2\)O)(CN)(en)\(_2\)]\(^{2+}\):
This is a cobalt complex with water, cyanide, and ethylenediamine (en) ligands. The IUPAC name is:
Aqua-cyanido-bis(ethylenediamine)cobalt(III) ion.
2. [PtCl\(_4\)]\(^{2-}\):
This is a platinum(II) complex with four chloride ions. The IUPAC name is:
Tetrachloroplatinate(II) ion.
3. [Cr(NH\(_3\))\(_4\)Cl(ONO)]\(^+\):
This is a chromium(III) complex with ammonia, chloride, and nitrito (ONO) ligands. The IUPAC name is:
Tetraamminochloro(nitrito)chromium(III) ion. Quick Tip: In naming coordination compounds, the ligands are named first, followed by the central metal and its oxidation state in parentheses. Prefixes are used for indicating the number of each type of ligand.
(a) Write the cell reaction and calculate the e.m.f. of the following cell at 298 K:
\(\text{Sn(s)}\) | \(Sn^{2+}\text{(0.004 M)}\)|| \(\text{H^+}\text{(0.02 M)}\) | \(\text{H_2(g)} \text{(1 Bar)}\) | Pt(s)
View Solution
(a) Account for the following:
(i) On the basis of E\(^0\) values, O\(_2\) gas should be liberated at the anode, but it is Cl\(_2\) gas which is liberated in the electrolysis of aqueous NaCl.
View Solution
Write the anode and cathode reactions and the overall cell reaction occurring in a lead storage battery during its use.
View Solution





























Comments