UP Board Class 12 Chemistry Question Paper 2025 PDF (Code 347 KA) is available for download here. The Mathematics exam was conducted on March 8, 2025 in the Morning Shift from 2:00 PM to 5:15 PM. The total marks for the theory paper are 100. Students reported the paper to be easy to moderate.
UP Board Class 12 Chemistry Question Paper 2025 (Code 347 KA) with Solutions
| UP Board Class Physics Question Paper with Answer Key | Check Solutions |

UP Board Class 12 Chemistry Questions with Solutions
Example of gaseous solution is
Compound of which transition metal ion is colourless?
Oxidation number of cobalt in K[Co(CO)\(_4\)] is
The correct order of increasing acid strength is
Which of the following gives a positive Fehling's solution test?
Best method of preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is
In a solution 30 mass percent of benzene is dissolved in carbon tetrachloride. Calculate mole fraction of benzene.
Explain Raoult's Law.
Transition metals and their maximum compounds are paramagnetic. Explain it.
The spin-only magnetic moment value of \([\mathrm{MnBr}_4]^{2-}\) is \(5.9\,\mathrm{BM}\). What will be the geometry of the complex ion?
Write the uses of carbon tetrachloride and chloroform.
Boiling point of propanol is greater than butane. Explain.
Explain Cannizzaro reaction with chemical equation.
What is nucleic acid? Write two important properties of them.
A cell in which the following reaction occurs:
\[ Zn_{(s)} + Cu^{2+}_{(aq)} \rightarrow Zn^{2+}_{(aq)} + Cu_{(s)} \]
has standard electrode potential \(E^\circ_{cell} = 1.1~V\) at \(298~K\). Calculate the standard Gibbs energy and the equilibrium constant of the cell reaction.
How do the following factors affect the velocity of reaction?
(i) Concentration
(ii) Temperature
(iii) Catalyst
Describe the method of preparation of potassium permanganate. How does acidic potassium permanganate react with the following? Give ionic equations: (i) H\(_2\)S \quad (ii) Fe \quad (iii) iodide ion.
What is elevation of boiling point? A liquid has boiling point 353.23 K. The boiling point of solution becomes 354.11 K after dissolving 1.8 g non-volatile solute of molar mass 58 g mol\(^{-1}\) to 90 g liquid. Calculate boiling point elevation constant for the liquid.
Explain in brief the Faraday's laws of Electrolysis. CuSO\(_4\) solution was electrolysed for 20 minutes with 2.0 ampere current. What will be the mass of precipitated copper at cathode?
A first order reaction completes 20% in 10 minutes. Calculate the time taken for 75% completion.
Write formula of the following coordination compounds:
(x) Tetraammine diaqua cobalt (III) chloride
(y) Potassium trioxalato chromate (III)
The colour of dilute solutions of [Fe(CN)\(_6\)]\(^{4-}\) and [Fe(H\(_2\)O)\(_6\)]\(^{2+}\) are different. Explain.
Explain the following terms:
(i) Zwitter ion
(ii) Peptide bond
(iii) Primary structure of protein
(iv) Polysaccharides
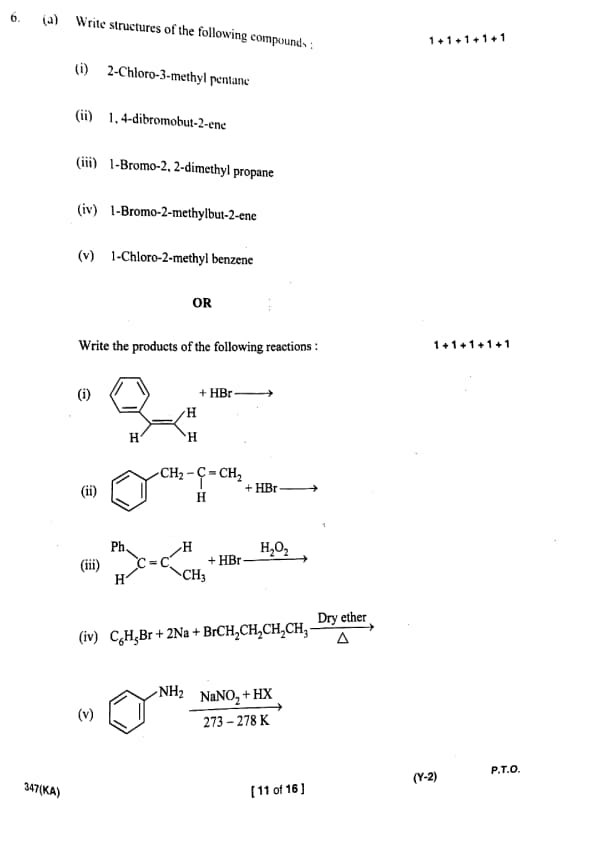
Write structures of the following compounds: (i) 2-Chloro-3-methylpentane
(ii) 1,4-dibromobut-2-ene
(iii) 1-Bromo-2,2-dimethylpropane
(iv) 1-Bromo-2-methylbut-2-ene
(v) 1-Chloro-2-methylbenzene
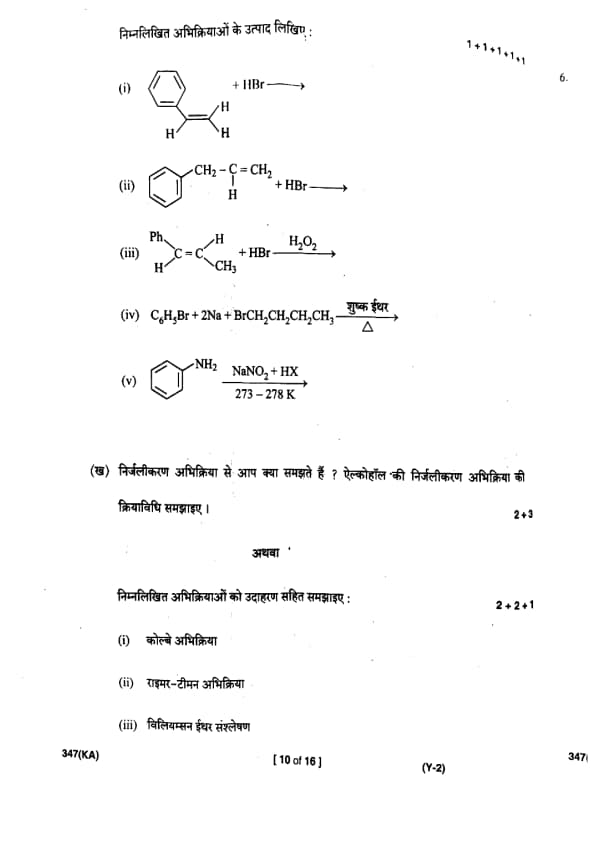
Write the products of the following reactions:
What do you understand by dehydration reaction? Write the mechanism of dehydration reaction of alcohol.
Explain the following reactions with examples:
(i) Kolbe reaction
(ii) Reimer–Tiemann reaction
(iii) Williamson Ether synthesis
Write IUPAC names of the following compounds:
(i)\; (ring with –CHO and adjacent –CH\(_3\))
(ii)\; (CH\(_3\))\(_2\)CHCOCH(CH\(_3\))\(_2\)
(iii)\; CH\(_3\)CH(OCH\(_3\))CHO
(iv)\; HOOC–(CH\(_2\))\(_4\)–COOH
(v)\; (benzene–CH\(_2\)COOH)
An organic compound (A) has molecular formula C\(_8\)H\(_8\)O. It gives an orange-red precipitate with 2,4-dinitrophenylhydrazine (2,4-DNP) and a yellow precipitate on heating with iodine in the presence of NaOH. It does not reduce Tollens’ reagent or Fehling’s solution and does not decolourise Br\(_2\)-water/Baeyer’s reagent. On strong oxidation with chromic acid it forms a carboxylic acid (B) of formula C\(_7\)H\(_6\)O\(_2\). Identify (A) and (B).
Give the structures of A, B, and C in the following reaction sequences:
(i)\; \(\mathrm{CH_3CH_2I} \xrightarrow[\ ]{\mathrm{NaCN}} A \xrightarrow[\ ]{\;\;HO^-\;/\;H_2O\;\;} B \xrightarrow[\;Br_2/NaOH\;]{} C\)
(ii)\; \(\mathrm{C_6H_5N_2^+Cl^-} \xrightarrow[\ ]{\mathrm{CuCN}} A \xrightarrow[\ ]{\;H_3O^+\;} B \xrightarrow[\ \Delta\ ]{\;NH_3\;} C\)
(iii)\; \(\mathrm{CH_3CH_2Br} \xrightarrow[\ ]{\mathrm{KCN}} A \xrightarrow[\ ]{\mathrm{LiAlH_4}} B \xrightarrow[\;273\,K\;]{\;HNO_2\;} C\)
Describe the method of identification of primary, secondary and tertiary amines. Write the chemical equations of these reactions also.
The boiling points of amines are lower than their comparable alcohols and carboxylic acids. Explain.




















Comments