
Education Journalist | Study Abroad Strategy Lead
Bohrium is a synthetic chemical element with the symbol Bh and atomic number 107. Bohrium was first synthesized in 1981 by a team of German and Russian scientists at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia.
- The element is named after Danish physicist Niels Bohr.
- The atomic mass of the most stable isotope of bohrium, Bh-270, is 270 atomic mass units (amu).
- Bohrium is located in the seventh period of the periodic table, in the d-block of the transactinide elements.
- It is a member of group 7, which also includes manganese (Mn), technetium (Tc), and rhenium (Re).
| Table of Content |
Key Terms: Bohrium, Nuclear Fusion, Elements, Periodic Table, Conductor, Electron Configuration
What is Bohrium?
[Click Here for Sample Questions]
Bohrium is a member of the group 7 elements, which includes manganese, technetium, and rhenium. It is located in the middle of the periodic table, between Hassium and Seaborgium.
- Bohrium is a very heavy element.
- It is also highly unstable and radioactive, with a half-life of only a few seconds.
- Bohrium is a silvery-white metal that is solid at room temperature.
- It has a high melting point and boiling point and is a good conductor of electricity.
- Bohrium is classified as a transition metal, meaning that it has a partially filled d electron shell.
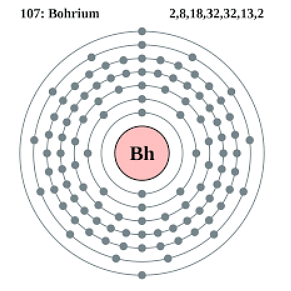
- The electron configuration of Bohrium is [Rn]5f14 6d5 7s2.
- It is produced by bombarding lighter elements with high-energy particles in a process called nuclear fusion.
Bohrium
Read More:
Chemical Properties of Bohrium
[Click Here for Sample Questions]
As a synthetic element, bohrium's chemical properties have not been extensively studied. However, based on its position in the periodic table, it is expected to exhibit properties that are similar to those of other group 7 elements such as technetium and rhenium. Some of the key chemical properties of bohrium are -
- It is predicted to belong to the group 7 elements and share similarities with its neighbours, technetium and rhenium.
- Bohrium is expected to have a high melting and boiling point due to its position in the periodic table.
- It may also exhibit a range of oxidation states, including +7, +5, and +3, with the +7 state being the most stable.
- It is expected to be highly radioactive and may have applications in nuclear research.
| Property | Value or Description |
|---|---|
| Group | 7 |
| Period | 7 |
| Block | d |
| Atomic number | 107 |
| Electron configuration | [Rn] 5f14 6d5 7s2 |
| Relative atomic mass | [270] |
| Key isotopes | Bh-267, Bh-270, Bh-271, Bh-272 |
General Properties of Bohrium
[Click Here for Sample Questions]
Due to its synthetic nature and its extreme instability, only small amounts of bohrium have been produced and studied, making its properties difficult to determine.
- Bohrium is a solid at room temperature, but it is likely to be highly unstable and reactive
- Its chemical properties are predicted to be similar to those of its lighter homologues, such as chromium and molybdenum.
- The density of bohrium is estimated to be around 37.1 g/cm3.
- The atomic radius of bohrium is estimated to be around 128 picometers.
- Bohrium is expected to exhibit oxidation states ranging from -1 to +7, with the +7 state being the most stable.
- Bohrium is predicted to be a highly reactive and unstable element, reacting easily with other elements and compounds
- The electronegativity of bohrium is not known, but it is expected to be similar to that of another group 7 elements.
Also Read:
Uses of Bohrium
[Click Here for Sample Questions]
The uses of bohrium are as follows -
- Its radioactive properties make it a valuable tool for studying nuclear reactions and atomic structures.
- It can be used in medical imaging and cancer treatment. However, more research is needed to explore its potential in this area.
- Bohrium's extreme rarity and short half-life make it unsuitable for most industrial applications. However, it could potentially be used in nuclear power plants or as a tracer in industrial processes.
- Bohrium's rarity and unique properties make it an interesting subject for science education and outreach programs. It can be used to teach students about the periodic table and the properties of elements.
- Bohrium is also of interest to researchers in the field of fundamental physics, as it can provide insights into the behavior of subatomic particles and the nature of matter.
- The primary use of bohrium is in the study of nuclear physics, where its unique properties are used to further our understanding of the behaviour of atomic nuclei and the fundamental forces that govern them.
Things to Remember
- Bohrium is a synthetic element with the symbol Bh and atomic number 107.
- It was first synthesized in 1981 by a team of scientists in Germany.
- The element is named after the Danish physicist Niels Bohr.
- Bohrium is a highly radioactive element that is not found naturally on Earth.
- Bohrium is classified as a transition metal and is expected to have properties similar to its lighter homologues, such as chromium and molybdenum.
- Bohrium is produced by bombarding lighter elements with heavy ions in particle accelerators.
Read More:
Sample Questions
Ques: What is Bohrium? (1 mark)
Ans: Bohrium is a synthetic element with the symbol Bh and atomic number 107. It was first synthesized in 1981 by a team of German scientists led by Peter Armbruster and Gottfried Münzenberg.
Ques: What is the origin of the name Bohrium? (1 mark)
Ans: The name Bohrium is derived from the name of Danish physicist Niels Bohr.
Ques: What is the atomic mass of Bohrium? (1 mark)
Ans: The atomic mass of Bohrium is 270.
Ques: What is the electron configuration of Bohrium? (1 mark)
Ans: The electron configuration of Bohrium is [Rn] 5f14 6d5 7s2.
Ques: What are D Block Elements? (3 Marks)
Ans: D block elements are elements that have 1 to 10 electrons in the d-orbital of the penultimate energy level and 1 to 2 electrons in the s-orbital. D block elements exhibit metallic properties.
There are four series under the d block elements:
- 3d series, also called the first transition
- 4d series, also called the second transition
- 5d series, also called the third transition
- 6d series, also called the fourth transition
Ques: What are F Block Elements? (3 Marks)
Ans: F block elements are elements that have anywhere between 1 to 14 electrons in the f orbital and 0 or 1 electron in the d orbital. F block elements are a subset of the 6th and 7th periods. They are also called inner transition metals.
There are two series under f block elements:
- 4f series
- 5f series
Ques: Mention all the D Block Elements of the periodic table. (5 Marks)
Ans: The D block elements of the periodic table are as follows:
3d series (first transition)
Scandium (Sc), Titanium (Ti), Vanadium (V), Chromium (Cr), Manganese (Mn), Iron (Fe), Cobalt (Co), Nickel (Ni), Copper (Cu), and Zinc (Zn).
4d series (second transition)
Yttrium (Y), Zirconium (Zr), Niobium (Nb), Molybdenum (Mo), Technetium (Tc), Ruthenium (Ru), Rhodium (Rh), Palladium (Pd), Silver (Ag), and Cadmium (Cd).
5d series (third transition)
Lanthanum (La), Hafnium (Hf), Tantalum (Ta), Tungsten (W), Rhenium (Re), Osmium (Os), Iridium (Ir), Platinum (Pt), Gold (Au), and Mercury (Hg).
6d series (fourth transition)
Actinium (Ac), Rutherfordium (Rf), Hahnium (Ha), Seaborgium (Sg), Bohrium (Bh), Hassium (Hs) and Meitnerium (Mt). This series is still considered to be incomplete.
Ques: Mention the F Block Elements in the periodic table. (5 Marks)
Ans: The f block elements of the periodic table are as follows:
4f series (Lanthanides)
Lanthanum (La), Cerium (Ce), Praseodymium (Pr), Neodymium (Nd), Promethium (Pm), Samarium (Sm), Europium (Eu), Gadolinium (Gd), Terbium (Tb), Dysprosium (Dy), Holmium (Ho), Erbium (Er), Thulium (Tm), Ytterbium (Yb), and Lutetium (Lu).
5f series (Actinides)
Actinium (Ac), Thorium (Th), Protactinium (Pa), Uranium (U), Neptunium (Np), Plutonium (Pu), Americium (Am), Curium (Cm), Berkelium (Bk), Californium (Cf), Einsteinium (Es), Fermium (Fr), Mendelevium (Md), Nobelium (No), and Lawrencium (Lw).
Ques: Is Bohrium a metal or non-metal? (1 mark)
Ans: Bohrium is a metal.
Ques: What is the classification of Bohrium in the periodic table? (1 mark)
Ans: Bohrium is a transactinide element and is located in period 7 and group 7 of the periodic table.
Ques: What are the isotopes of Bohrium? (1 mark)
Ans: Bohrium has only one known isotope, 270Bh, which has a half-life of approximately 61 seconds.
Ques: What are the chemical properties of Bohrium? (1 mark)
Ans: Bohrium is expected to have chemical properties similar to those of other Group 7 elements, such as manganese and technetium.
Ques: How is Bohrium synthesized? (1 mark)
Ans: Bohrium is synthesized by bombarding a target material, usually bismuth, with high-energy projectiles in a particle accelerator.
Ques: What are the health effects of Bohrium? (1 mark)
Ans: Because Bohrium has such a short half-life, it is unlikely to have any significant health effects.
Ques: Who discovered Bohrium? (1 mark)
Ans: Bohrium was discovered in 1981 by a team of German scientists led by Peter Armbruster and Gottfried Münzenberg.
Ques: What is the future of Bohrium research? (1 mark)
Ans: The future of Bohrium research is likely to focus on developing new methods for synthesizing and studying this rare element and on exploring its potential applications in advanced technologies such as nuclear medicine and energy production.
For Latest Updates on Upcoming Board Exams, Click Here: https://t.me/class_10_12_board_updates
Check-Out:





Comments