
Content Writer
Gattermann reaction in Chemistry is named after the German chemist Ludwig Gattermann, which is also termed as the Gattermann formylation and the Gattermann salicylaldehyde synthesis. This reaction is used in the synthesis of aromatic ring compounds like aromatic halides and aldehydes. The chemical reaction involves the formylation of aromatic compounds by a mixture of Hydrogen cyanide (HCN) and hydrogen chloride (HCl) in the presence of a Lewis acid catalyst.
| Table of Content |
Key Terms: Gattermann Reaction, Mechanism, Diazonium Salt, Gattermann-Koch, Hydrolysis, Diazotization, Aromatic Ring Compounds, Electrophile, Benzene
Gattermann Reaction
[Click Here for Sample Questions]
The Gattermann reaction is the formylation of aromatic ring compounds. Formylation is the process where the formyl group (-CH=O) is attached to the compound. It can be categorized as a substitution reaction which is very similar to the Friedel Crafts Reaction.
Reaction: Treatment of Benzene or its derivative with HCl and HCN in the presence of a Lewis acid catalyst like aluminium chloride, AlCl3, followed by hydrolysis gives benzaldehyde or substituted benzaldehyde.

Gattermann Reaction
Also Read:
Diazonium Salt
[Click Here for Previous Year's Questions]
Diazonium salt is formed as a result of reaction of aromatic Amine with Nitrous Acid leaving water as a side product. This reaction is termed as Diazotization Reaction.
The reaction is given below:
ArNH2 + HNO2 + HX → RN2+X- + H2O
[Aromatic amine + Nitrous acid + Mineral acid → Diazonium salt + Water]
Diazotization of Aniline: It is done by treating aniline with sodium nitrate and HCl at the temperature of 273K.

Diazotization of Aniline
Gattermann Reaction Mechanism
[Click Here for Sample Questions]
Let us look at the Gattermann reaction mechanism step-by-step.
Step 1: Formation of Formimino Chloride
HCN Reacts with HCl to form formimino chloride.
The reaction is given below:

Formation of Formimino Chloride
Step 2: Formation of Electrophile
Formimino chloride reacts with Lewis acid catalyst (such as AlCl3) and forms formimino cation.
Reaction is given below:

Formation of Electrophile
Step 3: Attack of Electrophile on Benzene Ring
Formimino cation (electrophile) attacks the benzene rings and forms benzylamine.
Reaction is given below:

Attack of Electrophile on Benzene Ring
Step 4: Hydrolysis of Benzylamine
Hydrolysis of benzylamine takes place in this step which results in the formation of benzaldehyde.
Reaction is given below:
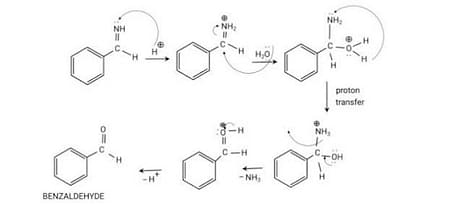
Hydrolysis of Benzylamine
Examples of Gattermann Reaction
[Click Here for Previous Year's Questions]
- Example 1: This reaction is used for obtaining chlorobenzene or bromobenzene from benzene-diazonium chloride by treating it with Cu/HCl or Cu/HBr respectively.

Examples of Gattermann Reaction
- Example 2:

Examples of Gattermann Reaction
Key points for the above reaction:
- The reaction can be simplified by replacing the HCN/AlCl3 combination with zinc cyanide.
- Although it is extremely toxic, Zn(CN)2 is a solid, making it safer to work with than gaseous HCN.
- The Zn(CN)2 reacts with the HCl to form the key HCN reactant and Zn(Cl)2 which serves as the Lewis-acid catalyst in-situ.
- An example of the Zn(CN)2 method is the synthesis of mesitaldehyde from mesitylene.
Gattermann-Koch Reaction
[Click Here for Sample Questions]
The Gattermann–Koch reaction, named after the German chemists Ludwig Gattermann and Julius Arnold Koch, is a variant of the Gattermann reaction in which carbon monoxide (CO) is used instead of hydrogen cyanide. The reaction can be represented as follows:

Gattermann-Koch Reaction
Gattermann Reaction & Gattermann-Koch Reaction: Differences
[Click Here for Previous Year's Questions]
| Gattermann Reaction | Gattermann-Koch Reaction |
|---|---|
| It is the organic substitution reaction in which we can formulate aromatic ring compounds. | A variant of the Gattermann-Koch reaction which uses carbon monoxide instead of hydrogen cyanide(HCN). |
| Involves mixture of hydrogen cyanide and hydrochloric acid. | This reaction uses carbon monoxide instead of HCN. |
| This reaction can be applied in phenol and phenol ether substrates as well. | This reaction cannot be applied to phenol and phenol ether substrates. |
| This reaction is named after one German scientist, Ludwig Gattermann | This reaction is named after 2 scientists, Ludwig Gattermann and Julius Koch. |
Applications of Gattermann Reaction
[Click Here for Sample Questions]
- It is used in obtaining Aromatic halides like Chlorobenzene and Bromobenzene.
- It is used in making Aromatic aldehydes like Benzaldehyde.
- Products of Gattermann reaction such as benzaldehydes, haloarenes, chlorobenzene and others are used in various fields such as pharmaceuticals, agricultural, medicinal etc.
Things to Remember
- Formyl Chloride is very unstable, hence it is produced and reacted simultaneously.
- Rings having withdrawing groups do not undergo Gattermann reaction.
- Another term used for Gattermann reaction is Gattermann Formylation.
- The reaction is used for the synthesis of aromatic ring compounds.
- The catalyst used in the formation of benzaldehyde by the Gattermann reaction is Aluminium Chloride.
- Gattermann reaction is a substitution reaction
- Gattermann reaction is similar to Friedel Crafts Reaction.
Also Read:
Sample Questions
Ques. What is the Gattermann Reaction? Explain with an example. (2 marks)
Ans: Aromatic compounds are synthesised using Gattermann Reaction. Aromatic aldehyde and aromatic halide are also product of this reaction. It is also known as Gattermann formylation or Gattermann salicylaldehyde synthesis. This reaction is named after German Chemist Ludwig Gattermann. This reaction is similar to the Friedel Craft reaction. It is a substitution reaction. An example is as follows:

Ques. What are the other names of the Gattermann reaction? (1 mark)
Ans: It is also known as Gattermann formylation and Gattermann salicylaldehyde synthesis.
Ques. The Gattermann reaction is similar to (1 mark)
a) Cannizaro reaction
b) Claisen schmidt reaction
c) Aldol condensation
d) Esterification
Ans: Esterification Reaction
Ques. The Gattermann reaction can be defined as a method of formylation of (1 mark)
a) Aromatic Ring compounds
b) hetro cyclic ring compounds
c) alicyclic compounds
d) aliphatic compounds
Ans: Aromatic Ring Compounds
Ques. Gattermann reaction is used for obtaining chlorobenzene from benzene-diazonium chloride by treating it with (1 mark)
a) Cu/HCl
b) Cu/KCl
c) Br/KBr
d) Ag/AgCl
Ans: Cu/HCl
Ques. What is the catalyst used in the formation of benzaldehyde by the Gattermann reaction? (1 mark)
Ans: Aluminium chloride (AlCl3).
Ques: What type of reaction is Gattermann? Give an example. (2 marks)
Ans: Gattermann reaction is primarily used for the formylation of aromatic ring compounds and is also known as Gattermann Formylation. the following reaction is an example.

Ques: Mention the steps involved in Gattermann Reaction mechanism. (3 marks)
Ans: The steps involved in Gattermann reaction mechanisms can be summarised as:
Step 1- Formation of Formimino Chloride
Step 2 - Formation of Electrophile
Step 3 - Attack of Electrophile on Benzene Ring
Step 4 - Hydrolysis of Benzylamine
Previous Year Questions
|
For Latest Updates on Upcoming Board Exams, Click Here: https://t.me/class_10_12_board_updates
Do Check Out:






Comments