JEE Advanced 2023 Question Paper is available for Paper 2. The paper pattern for JEE Advanced Paper 2 was similar to Paper 1. There were a total of 51 questions in JEE Advanced 2023 Question Paper 2 with 17 in each subject, Physics, Chemistry and Mathematics. The total weightage of JEE Advanced 2023 question paper 2 is 180 marks. JEE Advanced 2023 Paper 2 questions were based on MAMCQS, SAQS, Integer Type Questions and Paragraph-based questions.
| JEE Advanced 2023 Paper 2 Question Paper with Answer Key | Check Solution |
JEE Advanced 2023 June 4 Paper 2 Questions with Solutions
Mathematics
Question 1:
Let f: [1, ∞) → ℝ be a differentiable function such that f(1) = 1/3 and 3 ∫₁ˣ f(t) dt = x f(x) - x³/3, x ∈ [1, ∞). Let e denote the base of the natural logarithm. Then the value of f(e) is:
View Solution
Step 1: Rewrite the given equation. Using Newton-Leibniz theorem, we start with:
3f(x) = x f'(x) + f(x) - x².
Step 2: Simplify and rewrite as a differential equation. Rearranging terms:f'(x) + 1/x f(x) = x.
This is a first-order linear differential equation. Step 3: Determine the integrating factor (I.F.). The integrating factor is:I.F. = e∫ 1/x dx = elog x = x.
Step 4: Solve the differential equation. Multiply through by the integrating factor x:x f'(x) + f(x) = x².
This simplifies to:d/dx(x f(x)) = x².
Integrating both sides:x f(x) = ∫ x² dx = x³/3 + c,
where c is the constant of integration. Divide through by x:f(x) = x²/3 + c/x.
Step 5: Apply the initial condition f(1) = 1/3. Substitute x = 1 and f(1) = 1/3:f(1) = 1²/3 + c/1 = 1/3.
Simplify:1/3 + c = 1/3 → c = 0.
Thus, the solution becomes:f(x) = x²/3.
Step 6: Evaluate f(e). Substitute x = e into f(x):f(e) = 4e²/3.
Final Answer:f(e) = 4e²/3.
Quick Tip: For first-order linear differential equations, always compute the integrating factor and multiply through before solving.Question 2:
Consider an experiment of tossing a coin repeatedly until the outcomes of two consecutive tosses are the same. If the probability of a random toss resulting in a head is 1/3, then the probability that the experiment stops with a head is:
View Solution
Step 1: Define the probabilities for heads and tails.
P(H) = 1/3, P(T) = 2/3.
Step 2: Compute the probability that the experiment ends with two consecutive heads.P = P(HH) + (P(THH) + P(TTHH) + ...).
Expanding:P = 1/9 + 2/9 * 1/9 + 2²/9 * 1/9 + ...
Step 3: Simplify using the geometric series.P = (1/3 * 1/3) / (1 - 2/3 * 1/3) + (2/3 * 1/3 * 1/3) / (1 - 2/3 * 1/3) = 5/21
Quick Tip: For problems involving geometric probabilities, identify the repeating pattern and use the geometric series formula.Question 3:
For any y ∈ ℝ, let cot⁻¹(y) ∈ (0, π) and tan⁻¹(y) ∈ (-π/2, π/2). Then the sum of all the solutions of the equation
tan⁻¹(6y / (9 - y²)) + cot⁻¹((9 - y²) / 6y) = 2π/3,
for 0 < |y| < 3, is equal to:View Solution
Step 1: Simplify using trigonometric identities.
tan⁻¹(6y / (9 - y²)) + cot⁻¹((9 - y²) / 6y) = π (Sum of complementary angles).
Step 2: Solve for y using given conditions.tan⁻¹(6y / (9 - y²)) = π/3.
Simplifying:6y / (9 - y²) = √3.
Solve the quadratic:6√3y = -9 + y²
solve quadratic equation.Step 3: Sum of solutions.
Sum of solutions = 3√3 - 6 + √3 = 4√3 - 6.
Quick Tip: For trigonometric equations, use complementary angle identities to simplify complex terms.Question 4:
Let the position vectors of the points P, Q, R, and S be:
- P: \( \hat{i} + 2\hat{j} - 5\hat{k} \)
- Q: \( 3\hat{i} + 6\hat{j} + 3\hat{k} \)
- R: \( \frac{17}{5}\hat{i} + \frac{16}{5}\hat{j} + 7\hat{k} \)
- S: \( 2\hat{i} + \hat{j} + \hat{k} \)
Which of the following statements is true?
View Solution
Step 1: Calculate the vectors PQ, PR, and PS.
We are given the following points:
\( \vec{a} = \hat{i} + 2\hat{j} - 5\hat{k} \), \( \vec{b} = 3\hat{i} + 6\hat{j} + 3\hat{k} \), \( \vec{c} = \frac{17}{5}\hat{i} + \frac{16}{5}\hat{j} + 7\hat{k} \), \( \vec{d} = \hat{i} - \hat{j} + 6\hat{k} \).
Step 2: Verify coplanarity using the scalar triple product.
The vectors PQ, PR, and PS are coplanar if the scalar triple product is zero:
| 2 4 8 |
| 12/5 6/5 12 |
| 0 -3 11 |
Expanding along the first row and solving, we get a nonzero result, so the points are NOT coplanar.
Step 3: Verify if \( \frac{\vec{b} + 2\vec{d}}{3} \) divides PR internally.
\( \frac{\vec{b} + 2\vec{d}}{3} = \frac{(3 + 2)\hat{i} + (6 + 2(-1))\hat{j} + (3 + 2(6))\hat{k}}{3} \)
\( = \frac{5\hat{i} + 4\hat{j} + 15\hat{k}}{3} \)
Comparing with PR, it divides PR internally in the ratio 5:4. Hence, option (B) is correct.
Final Answer: Option B
Quick Tip: Use the scalar triple product to check coplanarity, and remember vector algebra for cross products and dividing line segments.
Question 5:
Let M = (aij), where i, j ∈ {1, 2, 3}, be the 3 × 3 matrix defined as:
- aij = 1 if (j + 1) is divisible by i
- aij = 0 otherwise
Which of the following statements is(are) true?
| a1 |
| a2 |
| a3 |
where M times this matrix results in:
| -a1 |
| -a2 |
| -a3 |
View Solution
Step 1: Given Matrix
M = | 1 1 1 |
| 1 0 1 |
| 0 1 0 |
Step 2: Determinant Calculation
The determinant of M is calculated as:
|M| = -1 + 1 + 1 = 0
Since the determinant is zero, M is non-invertible. Hence, option A is incorrect.
Step 3: Solving for the Null Space of M
Solving MX = 0:
| 1 1 1 | | a1 | | 0 |
| 1 0 1 | * | a2 | = | 0 |
| 0 1 0 | | a3 | | 0 |
From the equations:
a1 + a2 + a3 = 0
a1 + a3 = 0
a2 = 0
Solving gives a1 = -a3, confirming option B.
Step 4: Checking for M - 2I Invertibility
M - 2I =
| -1 1 1 |
| 1 -2 1 |
| 0 1 -2 |
Calculating determinant:
|M - 2I| = 0
Since the determinant is zero, M - 2I is not invertible, making option D incorrect.
Quick Tip: Use row reduction or determinant calculations to check invertibility.
Question 6:
Let f: (0, 1) → ℝ be the function defined as f(x) = ⌊4x⌋ (x - 1/4)² (x - 1/2), where ⌊x⌋ denotes the greatest integer less than or equal to x. Which of the following statements is(are) true?
, (B) There is exactly one point in (0, 1) at which the function f is continuous but NOT differentiable.
View Solution
Step 1: Define the piecewise function. The function f(x) is given as:
| f(x) = | 0, 0 < x < 1/4 1 (x - 1/4)² (x - 1/2), 1/4 ≤ x < 1/2 2 (x - 1/4)² (x - 1/2), 1/2 ≤ x < 3/4 3 (x - 1/4)² (x - 1/2), 3/4 ≤ x < 1 |
Step 2: Analyze continuity.
The function f(x) is defined piecewise with changes at x = 1/4, x = 1/2, and x = 3/4. To check continuity, evaluate the left-hand limit (LHL) and right-hand limit (RHL) at these points.
- At x = 1/4:
LHL: limx → 1/4⁻ f(x) = 0, RHL: limx → 1/4⁺ f(x) = 0.
Therefore, f(x) is continuous at x = 1/4. - At x = 1/2:
LHL: limx → 1/2⁻ f(x) = 0,
RHL: limx → 1/2⁺ f(x) = 0.
Thus, f(x) is continuous at x = 1/2. - At x = 3/4:
LHL: limx → 3/4⁻ f(x) = 2 (3/4 - 1/4)² (3/4 - 1/2),
RHL: limx → 3/4⁺ f(x) = 3 (3/4 - 1/4)² (3/4 - 1/2).
Since these values are not equal, f(x) is discontinuous at x = 3/4.
Step 3: Analyze differentiability.
For differentiability, calculate f'(x) within each interval and check at the boundary points x = 1/4, x = 1/2, and x = 3/4.
- f(x) in (1/4, 1/2):
f(x) = (x - 1/4)² (x - 1/2).
Differentiate:
f'(x) = 2(x - 1/4)(x - 1/2) + (x - 1/4)².
Step 4: Find the minimum value of f(x).
The minimum value occurs where f'(x) = 0. Solve:
(x - 1/4)(2x - 1/2 + x - 1/4) = 0.
x = 1/4 or x = 5/12.
At x = 5/12:
f(x) = (5/12 - 1/4)² (5/12 - 1/2) = -1/432.
Final Answer:
The function f(x) is discontinuous at x = 3/4, non-differentiable at x = 1/2 and x = 3/4, and achieves its minimum value of -1/432 at x = 5/12.
Quick Tip: Break down piecewise functions and analyze continuity and differentiability carefully.Question 7:
Let S be the set of all twice differentiable functions f from ℝ to ℝ such that d²f/dx²(x) > 0 for all x ∈ (-1, 1). For f ∈ S, let Xf be the number of points x ∈ (-1, 1) for which f(x) = x. Then which of the following statements is(are) true?
View Solution
Step 1: Analyze the condition d²f/dx² > 0.
The condition implies that f(x) is concave upward in the interval (-1, 1). The graph of f(x) must be a parabola-like curve opening upward.
Step 2: Analyze intersection with y = x.
The function f(x) intersects y = x at points where f(x) - x = 0.
- Xf = 0: Possible when f(x) lies entirely above y = x (e.g., f(x) = x² + 1).
- Xf = 2: Possible when f(x) intersects y = x at two points (e.g., f(x) = 2x²).
Final Answer: Xf is at most 2 for all functions in S. Option (D) is incorrect.
Quick Tip: For intersection problems, set f(x) - x = 0 and analyze the roots based on the function's curvature.Question 8:
For x ∈ ℝ, let tan⁻¹(x) ∈ (-π/2, π/2). Then the minimum value of the function f: ℝ → ℝ defined by
f(x) = ∫0x tan⁻¹(x) (e(t - cos t) / (1 + t2023)) dt
is:
View Solution
Step 1: Write the function and use the Fundamental Theorem of Calculus. The given function is:
f(x) = ∫0x tan⁻¹(x) (e-cos t / (1 + t2023)) dt.
Using the Fundamental Theorem of Calculus for derivatives of integrals:
f'(x) = (e-cos(x tan⁻¹ x) / (1 + (x tan⁻¹ x)2023)) × d/dx (x tan⁻¹ x).
Step 2: Differentiate x tan⁻¹ x.
Let g(x) = x tan⁻¹ x. Differentiate using the product rule:
g'(x) = tan⁻¹ x + x / (1 + x²).
Substitute g'(x) into the expression for f'(x):
f'(x) = (e-cos(x tan⁻¹ x) / (1 + (x tan⁻¹ x)2023)) × (tan⁻¹ x + x / (1 + x²)).
Step 3: Analyze f'(x).
The term e-cos(x tan⁻¹ x) is always positive, and 1 + (x tan⁻¹ x)2023 > 0 for all x. Thus, f'(x) = 0 only when:
tan⁻¹ x + x / (1 + x²) = 0.
Step 4: Solve tan⁻¹ x + x / (1 + x²) = 0.
For x = 0:
tan⁻¹(0) + (0 / (1 + 0²)) = 0.
Thus, x = 0 is the only critical point.
Step 5: Evaluate f(x) at x = 0.
Substituting x = 0 into f(x):
f(0) = ∫00 (e-cos t / (1 + t2023)) dt = 0.
Step 6: Conclusion.
Since f'(x) > 0 for x > 0 and f'(x) < 0 for x < 0, x = 0 is the global minimum.
Final Answer: The minimum value of f(x) is:
0.
Quick Tip: The minimum or maximum value of an integral function occurs at critical points where f'(x) = 0.
Question 9:
For x ∈ ℝ, let y(x) be a solution of the differential equation
(x² - 5) (dy/dx) - 2xy = -2x(x² - 5)²,
such that y(2) = 7. Then the maximum value of the function y(x) is:
View Solution
Step 1: Rewrite the differential equation.
(dy/dx) - (2x / (x² - 5)) y = -2x(x² - 5).
This is a first-order linear differential equation.
Step 2: Find the integrating factor (I.F.).
I.F. = e∫ (-2x / (x² - 5)) dx = e-ln(x² - 5) = 1 / (x² - 5).
Step 3: Solve for y(x).
Multiply through by the I.F.:
(1 / (x² - 5)) (dy/dx) - (2x / (x² - 5)²) y = -2x.
This simplifies to:
d/dx (y / (x² - 5)) = -2x.
Integrate both sides:
(y / (x² - 5)) = -x² + c.
Solve for y:
y = -x²(x² - 5) + c(x² - 5).
Step 4: Apply initial condition y(2) = 7.
7 = -4(-1) + c(-1) ⇒ c = -3.
Step 5: Maximum value of y(x).
y = -(x⁴ - 2x² - 15) = -(x² - 1)² - 16.
The maximum value is 16.
Quick Tip: For first-order linear differential equations, use the integrating factor method to find the general solution.
Question 10:
Let X be the set of all five-digit numbers formed using 1, 2, 2, 2, 4, 4, 0. For example, 22240 is in X while 02244 and 44422 are not in X. Suppose that each element of X has an equal chance of being chosen. Let p be the conditional probability that an element chosen at random is a multiple of 20 given that it is a multiple of 5. Then the value of 38p is equal to:
View Solution
Step 1: Count the total number of elements in X that are multiples of 5.
We are given subsets of X, and we need to calculate the number of elements divisible by 5 across different ranges.
Number of multiples of 5 in each subset:
- 4
- 12
- 4
- 6
- 12
Total number of multiples of 5: 4 + 12 + 4 + 6 + 12 = 38.
Step 2: Count the elements divisible by 5 but not by 20.
Among the 38 elements divisible by 5, we calculate those not divisible by 20 as follows:
- 1
- 3
- 3
Total multiples not divisible by 20: 1 + 3 + 3 = 7.
Step 3: Calculate the probability.
The probability p is the ratio of elements divisible by 5 and also divisible by 20 to the total elements divisible by 5:
p = (Total divisible by 5 - Not divisible by 20) / (Total divisible by 5).
Substituting the values:
p = (38 - 7) / 38 = 31/38.
Final Answer:
38p = 31
Quick Tip: For combinatorics problems, consider symmetries and constraints in the number construction.
Question 11:
Let A1, A2, A3, ..., A8 be the vertices of a regular octagon that lie on a circle of radius 2. Let P be a point on the circle, and let P Ak denote the distance between the points P and Ak, for k = 1, 2, ..., 8. If P varies over the circle, then the maximum value of the product P A1 · P A2 · ... · P A8 is:
View Solution
Step 1: Represent point P and vertices of the octagon.
Let P = 2eiθ, and the vertices of the octagon are given as:
Ak = 2ei(2kπ/8) = 2ei(kπ/4), for k = 0, 1, 2, ..., 7.
Step 2: Compute PAk.
The distance between P and Ak is:
PAk = |P - Ak| = |2eiθ - 2ei(kπ/4)| = 2 |eiθ - ei(kπ/4)|.
Using the identity |eix - eiy| = 2 |sin((x - y) / 2)|, we get:
PAk = 4 |sin((θ - kπ/4) / 2)|.
Step 3: Product of all distances PAk.
The product of all distances is:
∏k=07 PAk = ∏k=07 [4 |sin((θ - kπ/4) / 2)|].
Simplify:
∏k=07 PAk = 48 ∏k=07 |sin((θ - kπ/4) / 2)|.
Step 4: Symmetry and roots of unity.
The roots of unity identity implies symmetry in the angles:
∏k=07 sin((θ - kπ/4) / 2) = sin(4θ) / 8.
Thus:
∏k=07 PAk = 48 · (|sin(4θ)| / 8).
Simplify further:
∏k=07 PAk = 29 |sin(4θ)|.
Step 5: Maximum value of ∏k=07 PAk.
The maximum value occurs when |sin(4θ)| = 1, which happens at:
4θ = π/2 ⇒ θ = π/8.
Substituting:
∏k=07 PAk = 29 = 512.
Final Answer:
The maximum value of ∏k=07 PAk is:
512.
Quick Tip: Use symmetry and trigonometric simplifications when working with regular polygons inscribed in circles.
Question 12:
Let \( R = \begin{bmatrix} a & 3 & b \\ c & 2 & d \\ 0 & 5 & 0 \end{bmatrix} \), where \( a, b, c, d \in \{0, 3, 5, 7, 11, 13, 17, 19\} \). Then the number of invertible matrices in \( R \) is:
View Solution
Step 1: Total number of matrices. Since \( a, b, c, d \) can each take 8 values:
Total matrices = \( 8^4 = 4096 \).
Step 2: Compute the determinant of \( R \).
\( \det(R) = 5(bc - ad) \).
For \( R \) to be invertible, \( bc - ad \neq 0 \).
Step 3: Calculate non-invertible cases.
- Case 1: If \( a, b, c, d \neq 0 \), then \( bc - ad = 0 \) leads to 91 cases.
- Case 2: If \( a = 0 \) or \( b = 0 \), \( bc = ad = 0 \), leading to 225 cases.
Step 4: Calculate invertible matrices.
Number of invertible matrices = \( 4096 - (91 + 225) = 3780 \).
Quick Tip: For matrix invertibility, ensure the determinant is non-zero by analyzing all possible cases.
Question 13:
Let C1 be the circle of radius 1 with center at the origin. Let C2 be the circle of radius r with center at the point A = (4, 1) , where 1 < r < 3 . Two distinct common tangents PQ and ST of C1 and C2 are drawn. The tangent PQ touches C1 at P and C2 at Q . The tangent ST touches C1 at S and C2 at T . Midpoints of the line segments PQ and ST are joined to form a line which meets the x-axis at a point B . If AB = √5 , then the value of r2 is:
View Solution
Step 1: Write the equations of the circles. The equations of the given circles are:
C1: x2 + y2 = 1, C2: (x - 4)2 + (y - 1)2 = r2. Step 2: Find the radical axis of the two circles. The radical axis is obtained by subtracting the equations of the circles:
(x2 + y2 - 1) - [(x - 4)2 + (y - 1)2 - r2] = 0.
Expand C2 :
(x - 4)2 + (y - 1)2 = x2 - 8x + 16 + y2 - 2y + 1 = x2 + y2 - 8x - 2y + 17.
Substitute back:
(x2 + y2 - 1) - (x2 + y2 - 8x - 2y + 17 - r2) = 0.
Simplify:
8x + 2y - (18 - r2) = 0.
The radical axis is:
4x + y = (18 - r2)/2. Step 3: Determine point B on the radical axis. Point B is where the radical axis meets the x -axis ( y = 0 ):
4x + 0 = (18 - r2)/2.
Solve for x :
x = (18 - r2)/8.
Thus, B = ((18 - r2)/8, 0). Step 4: Apply the distance condition. Given AB = √5 , where A = (4, 1) is the center of C2 , compute the distance:
AB = √((4 - (18 - r2)/8)2 + (1 - 0)2).
Simplify:
AB = √(((32 - (18 - r2))/8)2 + 1).
AB = √(((14 + r2)/8)2 + 1).
Equating to √5 :
√(((14 + r2)/8)2 + 1) = √5.
Square both sides:
((14 + r2)/8)2 + 1 = 5.
Simplify:
((14 + r2)/8)2 = 4.
Take the square root:
(14 + r2)/8 = 2 (only positive root is valid).
Solve for r2 :
14 + r2 = 16 ⇒ r2 = 2. Step 5: Verify the solution. Substitute r2 = 2 back into the equations. The distance AB = √5 is satisfied, confirming the solution. Final Answer:
r2 = 2. Quick Tip: For problems involving tangents and circles, use radical axes and distance formulas to derive key relationships.
Question 14:
Let a be the area of the triangle ABC . Then the value of (64a)2 is:
View Solution
Step 1: Analyze the problem. The triangle ABC is an obtuse-angled triangle with vertices on a circle of radius R = 1 . The difference between the largest and smallest angles is π/2 , and the sides are in arithmetic progression. Step 2: Use trigonometric relations. Let A > B > C be the angles, where A - C = π/2 . Using the property of arithmetic progression:
a + c = 2b.
Since A + B + C = π :
π/2 + B + C = π ⇒ B + C = π/2. Step 3: Calculate sine values. Using the given conditions:
sin C = (-1 + √7)/4, sin A = (√7 + 1)/4, sin B = √7/4. Step 4: Compute the area. The area of the triangle is:
a = 2R2 sin A sin B sin C = 2(1)2 · (√7 + 1)/4 · √7/4 · (-1 + √7)/4.
Simplify:
a = 2/64 (6√7)
Thus:
⇒ (64a)2 = 1008. Quick Tip: For obtuse triangles in a circle, use sine rules and trigonometric identities to compute the area.
Question 15:
Then the inradius of the triangle ABC is:
View Solution
Step 1: Recall the formula for the inradius. The inradius r of a triangle is given by:
r = Δ/s,
where Δ is the area of the triangle and s is the semi-perimeter. Step 2: Express Δ and s in terms of given parameters. The area Δ of the triangle is given by:
Δ = 1/2 · base · height.
Let the sides of the triangle be n, n+d, and n+d' , where d and d' are offsets. The semi-perimeter s is:
s = (n + (n+d) + (n+d'))/2 = (3n + d + d')/2. Step 3: Use the angle to simplify Δ . The height can be expressed as:
height = (n + d) sinα,
where α is the included angle. Thus:
Δ = 1/2 · n · (n + d) sinα. Step 4: Substitute into the inradius formula. The inradius is:
r = Δ/s = (1/2 n (n + d) sinα)/((3n + d + d')/2).
Simplify:
r = (n (n + d) sinα)/(3n + d + d'). Step 5: Relate to trigonometric identities. If sin(2α) = 2 cosα sinα , we can express sinα in terms of given parameters. Substitute this into r :
r = sin(2α)/3. Step 6: Given r = 1/4 , solve for α . From:
sin(2α) = 1/4,
we have:
sin(2α) = 1/4. Quick Tip: The inradius depends on the area and the semi-perimeter, requiring correct computations of both.
Question 16:
Let pi be the probability that a randomly chosen point has i many friends, i = 0, 1, 2, 3, 4 . Let X be a random variable such that for i = 0, 1, 2, 3, 4 , the probability P(X = i) = pi . Then the value of 7E(X) is:
View Solution
Step 1: Construct the probability distribution. From the problem:
P(0) = 0, P(1) = 0, P(2) = 4/49, P(3) = 20/49, P(4) = 25/49. Step 2: Compute E(X) .
E(X) = ∑i=04 i · P(i) = 0 · 0 + 0 · 0 + 2 · 4/49 + 3 · 20/49 + 4 · 25/49.
On Simplifying:
E(X) = (8 + 60 + 100)/49 = 168/49 = 24/7. Step 3: Calculating 7E(X) .
7E(X) = 7 · 24/7 = 24. Quick Tip: For expected value problems, multiply probabilities by outcomes and sum them up systematically.
Question 17:
Two distinct points are chosen randomly out of the points A1, A2, ..., A49 . Let p be the probability that they are friends. Then the value of 7p is:
View Solution
Step 1: Compute total pairs. The total number of ways to choose two points is:
49C2 Step 2: Compute favorable pairs. Consecutive points are friends. There are 2 · 6 · 7 = 84 such pairs. Step 3: Compute probability p .
p = 84/49C2. Step 4: Compute 7p .
Simplify:
7p = 0.5. Quick Tip: For combinatorics problems, carefully count total and favorable outcomes to calculate probabilities.
Physics
Question 1:
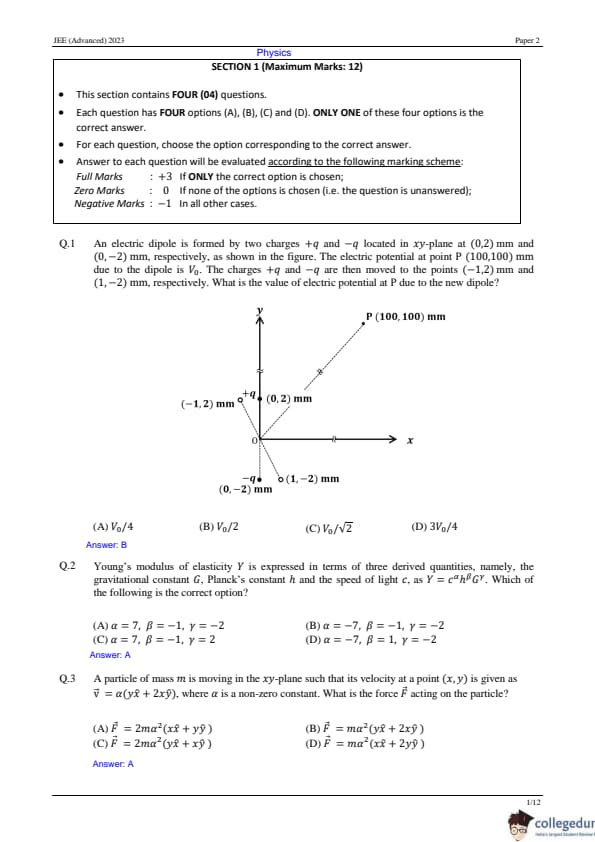
An electric dipole is formed by two charges +q and -q located in the xy -plane at (0, 2) mm and (0, -2) mm , respectively, as shown in the figure. The electric potential at point P(100, 100) mm due to the dipole is V0 . The charges +q and -q are then moved to the points (-1, 2) mm and (1, -2) mm , respectively. What is the value of the electric potential at P due to the new dipole?

View Solution
Step 1: Testing the dipole configuration.
The electric potential at P due to the initial dipole is given as:
V0 = (kp0/r2) cos 45°,
where p0 is the dipole moment. Step 2: With new dipole moment.
After the charges are moved, the new dipole moment is:
the Component P_0' along xaxis = -P0/2 i
the Component P_0' along yaxis = P0 j
Potential due to P_0 j
⇒ (kP0 cos 45°)/r2 = V0
Potential due to -P0/2 i
The new potential at P is:
(k(P0/2) cos 135°)/r2 = -V0/2
Thus, the new potential is V0 - V0/2 = V0/2 Quick Tip: For electric dipoles, the potential at a point is proportional to the dipole moment and the cosine of the angle.
Question 2:
Young's modulus of elasticity Y is expressed in terms of three derived quantities, namely, the gravitational constant G , Planck's constant h , and the speed of light c , as Y = cαhβGγ . Which of the following is the correct option?
View Solution
Step 1: Perform dimensional analysis for Y . The dimensional formula of Y is:
[M L-1 T-2] = [cαhβGγ],
where c , h , and G are constants with specific dimensional formulas. Step 2: Write the dimensional formulas for c , h , and G . The dimensional formulas are:
[c] = [L T-1], [h] = [M L2 T-1], [G] = [M-1 L3 T-2]. Step 3: Expand the dimensions of the right-hand side. Using the given dimensions:
[cα] = [Lα T-α], [hβ] = [Mβ L2β T-β], [Gγ] = [M-γ L3γ T-2γ].
The overall dimensions of cαhβGγ are:
[Mβ M-γ] [Lα L2β L3γ] [T-α T-β T-2γ].
Simplify:
[Mβ - γ] [Lα + 2β + 3γ] [T-α - β - 2γ]. Step 4: Match the dimensions of Y . Equating the dimensions on both sides:
[M L-1 T-2] = [Mβ - γ Lα + 2β + 3γ T-α - β - 2γ].
Equate the powers of M, L, T :
1. For M :
β - γ = 1 ... (i).
2. For L :
α + 2β + 3γ = -1 ... (ii).
3. For T :
-α - β - 2γ = -2 ... (iii).
Step 5: Solve the equations. From equation (i):
β = 1 + γ ... (iv).
Substitute β = 1 + γ into equations (ii) and (iii):
From (ii):
α + 2(1 + γ) + 3γ = -1 ⇒ α + 2 + 2γ + 3γ = -1.
α + 5γ = -3 ... (v).
From (iii):
-α - (1 + γ) - 2γ = -2 ⇒ -α - 1 - γ - 2γ = -2.
-α - 3γ = -1 ⇒ α + 3γ = 1 ... (vi).
Solve equations (v) and (vi):
From (vi):
α = 1 - 3γ ... (vii).
Substitute α = 1 - 3γ into (v):
1 - 3γ + 5γ = -3 ⇒ 1 + 2γ = -3.
2γ = -4 ⇒ γ = -2.
Substitute γ = -2 into (iv):
β = 1 + (-2) = -1.
Substitute γ = -2 into (vii):
α = 1 - 3(-2) = 1 + 6 = 7.
Step 6: Final result. The values of α, β, γ are:
α = 7, β = -1, γ = -2.
Final Answer:
α = 7, β = -1, γ = -2. Quick Tip: Dimensional analysis is a powerful method to determine relationships between physical quantities.
Question 3:
A particle of mass m is moving in the xy -plane such that its velocity at a point (x, y) is given as v→ = α(y x ^ + 2x y ^) , where α is a non-zero constant. What is the force F→ acting on the particle?
View Solution
Step 1: Write the velocity components. The velocity vector is given as:
V→ = α(yx ^ + 2xy ^).
From this:
vx = αy, vy = 2αx. Step 2: Differentiate to find acceleration components. The acceleration vector is:
a→ = (dV→)/dt = d/dt(vxx ^) + d/dt(vyy ^).
For vx = αy , differentiate with respect to t :
(dvx)/dt = α (dy)/dt.
Substitute (dy)/dt = vy = 2αx :
(dvx)/dt = α (2αx) = 2α2x.
For vy = 2αx , differentiate with respect to t :
(dvy)/dt = 2α (dx)/dt.
Substitute (dx)/dt = vx = αy :
(dvy)/dt = 2α (αy) = 2α2y.
Thus, the acceleration vector is:
a→ = (2α2x)x ^ + (2α2y)y ^.
Step 3: Calculate the force. Using Newton's second law:
F→ = ma→.
Substitute a→ :
F→ = m[(2α2x)x ^ + (2α2y)y ^].
Simplify:
F→ = 2mα2 (xx ^ + yy ^).
Final Answer:
F→ = 2mα2 (xx ^ + yy ^).
Quick Tip: For velocity-dependent motion, differentiate with respect to time to find the acceleration.
Question 4:
An ideal gas is in thermodynamic equilibrium. The number of degrees of freedom of a molecule of the gas is n . The internal energy of one mole of the gas is Un and the speed of sound in the gas is vn . At a fixed temperature and pressure, which of the following is the correct option?
View Solution
Step 1: Analyze internal energy. The internal energy is:
Un = n/2RT (proportional to n ). Step 2: Analyze speed of sound. The speed of sound is:
vn = √(γRT/M),
where γ = 1 + 2/n . As n increases, γ decreases, and hence vn decreases. Step 3: Conclusion. For n = 5, 7 , we have:
v5 > v7, U5 < U7. Quick Tip: Use degrees of freedom to calculate internal energy and speed of sound for ideal gases.
Question 5:
A monochromatic light wave is incident normally on a glass slab of thickness d , as shown in the figure. The refractive index of the slab increases linearly from n1 to n2 over the height h . Which of the following statements is/are true about the light wave emerging out of the slab?

, (D) The deflection angle depends only on n2 - n1 and not on the individual values of n1 and n2.
View Solution
Step 1: Analyze the refractive index gradient. The refractive index increases linearly from n1 to n2 over a height h . This causes the wavefront to tilt as the speed of light decreases with increasing refractive index. Step 2: Calculate the tilt of the wavefront. At time t , the wavefront PQ is given by:
n2d/c = n1d/c + x/c.
Simplifying for x :
x = d(n2 - n1). Step 3: Determine the angle of deflection. The angle of deflection θ is:
tan θ = x/h = (n2 - n1)d/h.
Thus:
θ = tan-1[(n2 - n1)d/h]. Step 4: Validate the dependency on n2 - n1 . The deflection angle depends only on the difference n2 - n1 , as seen in the expression for θ , and not on the individual values of n1 or n2. Quick Tip: In problems involving gradient refractive indices, the deflection angle can be found using basic trigonometric relations between wavefront displacement and the slab's geometry.
Question 6:
An annular disk of mass M , inner radius a , and outer radius b is placed on a horizontal surface with a coefficient of friction μ , as shown in the figure. At some time, an impulse J0 x ^ is applied at a height h above the center of the disk. If h = hm , then the disk rolls without slipping along the x -axis. Which of the following statement(s) is/are correct?

(B) For μ ≠ 0 and a → b , hm = b
(C) For h = hm , the initial angular velocity does not depend on the inner radius a .
(D) For μ = 0 and h = 0 , the wheel always slides without rolling.
View Solution
Step 1: Angular impulse-momentum relation. The angular impulse-momentum relation gives:
Jh = Icmω,
where J is the impulse and Icm is the moment of inertia about the center of mass. Step 2: Rolling condition. For rolling without slipping, the linear velocity vcm and angular velocity ω are related by:
vcm = bω. Step 3: Calculate hm . The critical height hm is found by equating:
hm = Icm/Mb.
For different cases:
1. For a → 0 , hm = b/2.
2. For a → b , hm = b. Step 4: Additional observations.
1. For h = 0 and μ = 0 , rolling does not occur due to the absence of torque.
2. When h = hm , rolling without slipping occurs.
Quick Tip: In rolling motion, the relationship between linear velocity, angular velocity, and radius is key to determining rolling conditions.
Question 7:
The electric field associated with an electromagnetic wave propagating in a dielectric medium is given by
E→ = 30 (2x ^ + y ^) sin[2π(5 × 1014t - 107z/3)] V m-1.
Which of the following option(s) is/are correct?
[Given: The speed of light in vacuum, c = 3 × 108 m s–1]
, (D) The refractive index of the medium is 2.
View Solution
Step 1: Calculate the wave speed in the dielectric medium. The wave number k is related to the wavelength λ as:
k = 2π/λ.
From the given data, the wave number is:
k = 107/3.
The angular frequency ω is given as:
ω = 2π · 5 × 1014.
The speed of the wave v is:
v = ω/k.
Substitute the values of ω and k :
v = (2π · 5 × 1014)/(107/3) = (6 · 5 × 1014)/107 = 1.5 × 108 m/s. Step 2: Calculate the refractive index of the medium. The refractive index n is related to the speed of light c and the speed of the wave v as:
n = c/v.
Substitute c = 3 × 108 m/s and v = 1.5 × 108 m/s :
n = (3 × 108)/(1.5 × 108) = 2. Step 3: Relate the magnetic field amplitude to the electric field amplitude. The amplitude of the magnetic field B is related to the electric field E and the speed of the wave v by:
B = E/v.
Substitute E = 30 V/m and v = 1.5 × 108 m/s :
B = 30/(1.5 × 108) = 2 × 10-7 T. Step 4: Express the magnetic field components. The magnetic field components are sinusoidal and can be expressed as:
Bx = -Bsin[2π(5 × 1014t - 107z/3)],
By = 2B sin[2π(5 × 1014t - 107z/3)].
Substitute B = 2 × 10-7 :
Bx = -2 × 10-7 sin[2π(5 × 1014t - 107z/3)],
By = 4 × 10-7 sin[2π(5 × 1014t - 107z/3)].
Final Answer: The magnetic field components are:
Bx = -2 × 10-7 sin[2π(5 × 1014t - 107z/3)],
By = 4 × 10-7 sin[2π(5 × 1014t - 107z/3)]. Quick Tip: In EM waves, the electric and magnetic fields are perpendicular to each other and the direction of propagation, and their magnitudes are related by the wave speed.
Question 8:
A thin circular coin of mass 5 gm and radius 4/3 cm is initially in a horizontal xy -plane. The coin is tossed vertically up (+ z -direction) by applying an impulse √(π/2) × 10-2 N-s at a distance 2/3 cm from its center. The coin spins about its diameter and moves along the + z -direction. By the time the coin reaches back to its initial position, it completes n rotations. The value of n is _____.

View Solution
Step 1: Angular impulse and linear momentum. The impulse J is given as:
J = Mvcm.
For angular motion:
J · R/2 = Iω,
where I = MR2/4 is the moment of inertia about the center of mass. Step 2: Calculate vcm and ω . From the above equations:
vcm = J/M, ω = 2J/MR. Step 3: Time to return to the initial position. The time for the coin to return is:
T = 2vcm/g = 2J/gM. Step 4: Total angle rotated. The angle rotated in time T is:
θ = ωT = 2J2/M2gR.
Substituting J = √(π/2) × 10-2 , M = 5 gm , and R = 4/3 cm :
θ = 60π. Step 5: Number of rotations. The number of rotations is:
n = θ/2π = 60π/2π = 30. Quick Tip: In problems involving rotational motion, angular momentum conservation is key to relating linear impulse and rotational variables.
Question 9:
A rectangular conducting loop of length 4 cm and width 2 cm is in the xy -plane, as shown in the figure. It is being moved away from a thin and long conducting wire along the direction √3/2 x ^ + 1/2 y ^ with a constant speed v . The wire is carrying a steady current I = 10 A in the positive x -direction. A current of 10 μA flows through the loop when it is at a distance d = 4 cm from the wire. If the resistance of the loop is 0.1 Ω , then the value of v is _____.
[Given: The permeability of free space μ0 = 4π × 10-7 N A-2]

View Solution
Step 1: Calculate the induced emf in the loop. The induced emf ε in the loop is generated due to the change in magnetic flux. Using the Biot-Savart law, the magnetic field at a distance r from the wire is:
B(r) = μ0I/2πr.
For a small strip of width b , the flux change due to motion of the loop with velocity v is:
ε = dΦ/dt.
The change in flux for two sides of the loop at distances d and d + ℓ (from the current-carrying wire) is:
ε = ∫dd + ℓ (μ0Ivb/2πr)dr.
Simplify:
ε = (μ0Ivb/2π)(1/d - 1/(d + ℓ)). Step 2: Current in the loop. The induced current I0 in the loop is given by Ohm's law:
I0 = ε/R.
Substitute ε :
I0 = ((μ0Ivb/2π)(1/d - 1/(d + ℓ)))/R.
Simplify:
I0 = (μ0Ivb/2πR)(ℓ/d(d + ℓ)). Step 3: Solve for v . Rearrange to solve for v :
v = 2πRI0d(d + ℓ)/μ0Ibℓ. Step 4: Substitute the given values. Given:
I0 = 10 μA, b = 2 cm = 0.02 m, ℓ = 4 cm = 0.04 m,
d = 4 cm = 0.04 m, R = 10 Ω, I = 10 A.
Substitute:
v = (2π(10)(10 × 10-6)(0.04)(0.04 + 0.04))/((4π × 10-7)(10)(0.02)(0.04)).
Simplify:
v = (2π(10)(10-6)(0.04)(0.08))/((4π × 10-7)(0.02)(0.04)). v = (2 · 10 · 0.04 · 0.08)/(4 · 10-7 · 0.02 · 0.04).
v = 0.064/(3.2 × 10-8) = 2 × 106 m/s.
Final Answer: The velocity of the loop is:
v = 2 m/s. Quick Tip: In electromagnetic induction, the induced emf depends on the rate of change of magnetic flux through the loop.
Question 10:
A string of length 1 m and mass 2 × 10-5 kg is under tension T . When the string vibrates, two successive harmonics are found to occur at frequencies 750 Hz and 1000 Hz . The value of tension T is _____ Newton.
View Solution
The frequency of vibration of a string is given by:
f = nv/2ℓ,
where:
- n is the harmonic number,
- v is the speed of the wave,
- ℓ = 1 m is the length of the string.
For two successive harmonics:
fn = nv/2ℓ and fn+1 = (n+1)v/2ℓ.
The difference in frequencies is:
fn+1 - fn = v/2ℓ.
Given:
fn+1 - fn = 1000 - 750 = 250 Hz.
Substitute values:
v/(2 · 1) = 250 ⇒ v = 500 m/s.
The speed v is related to tension T and linear mass density μ by:
v = √(T/μ),
where μ = mass/length = (2 × 10-5)/1 = 2 × 10-5 kg/m . Substitute v = 500 :
500 = √(T/(2 × 10-5)).
Square both sides:
250000 = T/(2 × 10-5) ⇒ T = 250000 × 2 × 10-5 = 5 N. Quick Tip: The difference between successive harmonics is constant for a vibrating string and is determined by the wave speed and string length.
Question 11:
An incompressible liquid is kept in a container having a weightless piston with a hole. A capillary tube of inner radius 0.1 mm is dipped vertically into the liquid through the airtight piston hole, as shown in the figure. The air in the container is isothermally compressed from its original volume V0 to 100/101 V0 with the movable piston. Considering air as an ideal gas, the height ( h ) of the liquid column in the capillary above the liquid level in cm is _____.
[Given: Surface tension of the liquid is 0.075 N m-1, atmospheric pressure is 105 N m-2, acceleration due to gravity (g) is 10 m s-2, density of the liquid is 103 kg m-3 and contact angle of capillary surface with the liquid is zero]

View Solution
Step 1: Apply Boyle's Law for isothermal compression. The gas is compressed isothermally, so:
P0V0 = PAVA.
Substitute VA = 100/101 V0 :
P0V0 = PA(100/101 V0).
Simplify to find PA :
PA = P0 101/100. Step 2: Calculate the pressure transmitted to the liquid. The pressure PA is transmitted to the liquid, so:
PD = PA = P0 101/100. Step 3: Capillary pressure equation. The pressure at the capillary is given by:
PB = PC + 2T/r,
where:
T = 0.075 N/m (surface tension),
r = 0.1 mm = 0.0001 m (capillary radius).
Rearranging:
PC = PB - 2T/r. Step 4: Use hydrostatic pressure to relate PD and PC . The pressure difference between PD and PC causes the liquid to rise in the capillary:
PD = PC + ρgh.
Substitute PC = PB - 2T/r :
PD = (PB - 2T/r) + ρgh. Step 5: Relate PD to PB . From Boyle's law, we know PD = 101/100 P0 , so:
101/100 P0 = (P0 - 2T/r) + ρgh.
Rearrange to solve for h :
ρgh = 101/100 P0 - P0 + 2T/r.
Simplify:
ρgh = P0/100 + 2T/r. Step 6: Solve for h .
h = (P0/100 + 2T/r)/ρg. Step 7: Substitute the given values. Given:
P0 = 105 Pa, T = 0.075 N/m, r = 0.0001 m, ρ = 103 kg/m3, g = 10 m/s2.
Substitute:
h = (105/100 + (2 · 0.075)/0.0001)/(103 · 10).
h = (1000 + 1500)/10000.
Simplify:
h = 2500/10000 = 0.25 m.
Convert to cm:
h = 25 cm.
Final Answer: The height of the liquid column is:
25 cm. Quick Tip: The height of the liquid column in a capillary depends on surface tension, pressure difference, and the radius of the capillary tube.
Question 12:
In a radioactive decay process, the activity is defined as A = -dN/dt , where N(t) is the number of radioactive nuclei at time t . Two radioactive sources, S1 and S2 , have the same activity at time t = 0 . At a later time, the activities of S1 and S2 are A1 and A2 , respectively. When S1 and S2 have just completed their 3rd and 7th half-lives, respectively, the ratio A1/A2 is ______.
View Solution
The activity of a radioactive source is proportional to the number of radioactive nuclei present:
A(t) = A0(1/2)n,
where A0 is the initial activity, and n is the number of half-lives.
For S1 after 3 half-lives:
A1 = A0(1/2)3 = A0/8.
For S2 after 7 half-lives:
A2 = A0(1/2)7 = A0/128.
The ratio A1/A2 is:
A1/A2 = (A0/8)/(A0/128) = 128/8 = 16. Quick Tip: In radioactive decay, the activity decreases exponentially with time. Each half-life reduces the activity to half its previous value.
Question 13:
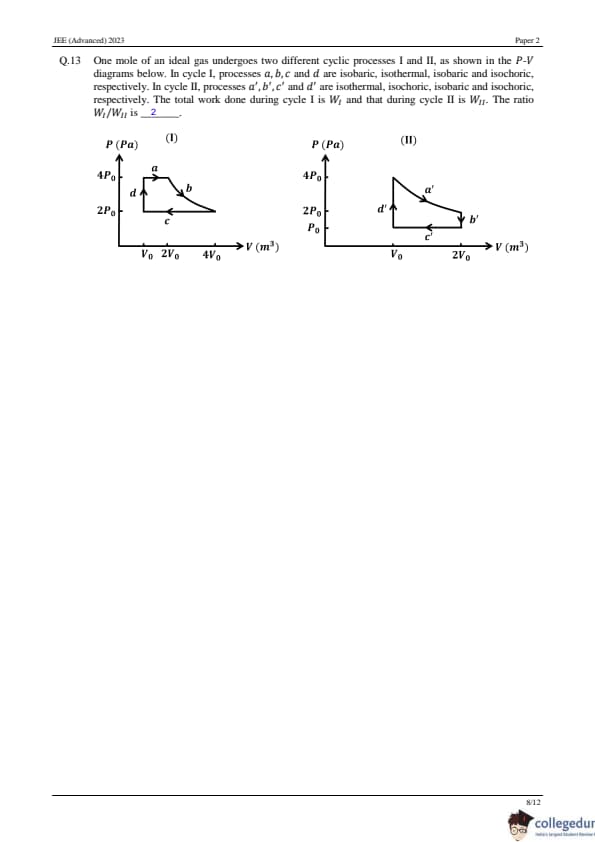
One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the P-V diagrams below. In cycle I, processes a, b, c, d are isobaric, isothermal, isobaric, and isochoric, respectively. In cycle II, processes a', b', c', d' are isothermal, isochoric, isobaric, and isochoric, respectively. The total work done during cycle I is WI and that during cycle II is WII . The ratio WI/WII is ______.
View Solution
Calculation of Work Done in Two Thermodynamic Cycles The work done in cycle I is given by:
WI = 4P0V0 + 8P0V0 ln 2 - 6P0V0
The work done in cycle II is:
WII = 4P0V0 ln 2 - P0V0
Step 1: Work Ratio Calculation
WI/WII = 2
Conclusion: The ratio of work done in cycle I to cycle II is 2, indicating that cycle I requires twice the work compared to cycle II. Quick Tip: For cyclic processes, work done is equal to the area enclosed by the P-V curve. Isothermal processes depend on ln terms for work calculations.
Question 14:
When only S2 is emitting sound and it is at Q , the frequency of sound measured by the detector in Hz is:
View Solution
Step 1: Formula for apparent frequency. The Doppler effect formula for apparent frequency is:
f = (v/(v + vs cos θ)) f0,
where:
v = 324 m/s (speed of sound),
vs = 4√2 m/s (speed of the source),
θ = 45° (angle of motion),
f0 = 656 Hz (original frequency). Step 2: Simplify the velocity term. The component of the source velocity along the line of sight is:
vs cos θ = 4√2 cos 45° = 4√2 · 1/√2 = 4 m/s.
Substitute vs cos θ = 4 into the formula:
f = (v/(v + 4)) f0. Step 3: Substitute the given values. Substitute v = 324 m/s and f0 = 656 Hz :
f = (324/(324 + 4)) 656.
Simplify:
f = (324/328) 656. Step 4: Calculate the apparent frequency. Divide:
324/328 ≈ 0.9878.
Thus:
f = 0.9878 · 656 ≈ 648 Hz.
Final Answer: The apparent frequency is:
648 Hz. Quick Tip: For Doppler effect problems, identify the source and observer's relative velocities and use the appropriate frequency shift formula.
Question 15:
Consider both sources emitting sound. When S2 is at R and S1 approaches the detector with a speed of 4 m/s , the beat frequency measured by the detector is:
View Solution
Step 1: Calculate f1 for S1 . The apparent frequency is given by:
f1 = (v/(v - vs))f0,
where v = 324 m/s , vs = 4 m/s , and f0 = 656 Hz . Substituting the values:
f1 = (324/(324 - 4)) 656 = 324/320 × 656 = 664.2 Hz. Step 2: Calculate f2 for S2 . Since S2 is stationary relative to the detector:
f2 = f0 = 656 Hz. Step 3: Calculate the beat frequency. The beat frequency is given by:
fb = |f1 - f2| = |664.2 - 656| = 8.2 Hz. Quick Tip: The beat frequency is the absolute difference between the frequencies of two overlapping sound waves.
Question 16:
Considering the airflow to be streamline, the steady mass flow rate of air exiting the chimney is:
View Solution
Step 1: Calculate air density. From the ideal gas law:
ρ = ρ0(Ta/T) = 1.2 × 300/360 = 1 kg/m3. Step 2: Apply Bernoulli's equation. At points 1 and 2 :
Pa + 0 + 0 = Pa - ρgh + 0 + ρv2/2,
where h = 9 m . Solving for v :
v = √(2(ρa - ρ)gh/ρ) = √40 m/s. Step 3: Calculate mass flow rate. The mass flow rate is given by:
Q = ρAv = πd2/4 × v = π(0.1)2/4 × √40 × 1000 = 49.61 gm/s. Quick Tip: For streamline flow, apply Bernoulli's principle and calculate density changes using the ideal gas law.
Question 17:
When the chimney is closed using a cap at the top, a pressure difference ΔP develops between the top and the bottom surfaces of the cap. If the changes in temperature and density of the hot air, due to the stoppage of airflow, are negligible, then ΔP is:
View Solution
Step 1: Write expressions for pressure at the surfaces. At the bottom surface inside the fluid, the pressure is:
Pinside = Pa + ρgh,
where:
Pa is the atmospheric pressure,
ρ is the fluid density,
g = 10 m/s2 is the acceleration due to gravity,
h = 10 m is the depth.
At the top surface outside the fluid, the pressure is:
Poutside = Pa + ρagh,
where ρa is the density of air. Step 2: Compute the pressure difference. The pressure difference between the inside and outside is:
ΔP = Pinside - Poutside.
Substitute the expressions:
ΔP = (Pa + ρgh) - (Pa + ρagh).
Simplify:
ΔP = (ρ - ρa)gh. Step 3: Substitute the given values. Given:
ρ = 1 kg/m3, ρa = 1.2 kg/m3, g = 10 m/s2, h = 10 m.
Substitute:
ΔP = (1.2 - 1) · 10 · 10.
Simplify:
ΔP = 0.2 · 100 = 20 N/m2.
Final Answer: The pressure difference is:
20 N/m2. Quick Tip: For pressure difference problems, consider the density variations and height difference using hydrostatic principles.
Chemistry
Question 1:
The correct molecular orbital diagram for F2 molecule in the ground state is


View Solution
Step 1: Analyze the atomic orbitals of fluorine. Each fluorine atom has 9 electrons, with the electronic configuration:
1s2 2s2 2p5.
The valence electrons are in the 2s and 2p orbitals. Step 2: Molecular orbital formation in F₂. When two fluorine atoms combine to form F2 , their atomic orbitals overlap to form molecular orbitals:
2s orbitals form σ2s and σ*2s molecular orbitals.
2p orbitals form:
σ2pz and σ*2pz from head-on overlap,
π2px, π2py and π*2px, π*2py from side-on overlap. Step 3: Fill molecular orbitals. The total number of valence electrons in F2 is:
7 + 7 = 14.
The molecular orbitals are filled in order of increasing energy:
1. σ2s, σ*2s (each with 2 electrons).
2. σ2pz (2 electrons).
3. π2px, π2py (4 electrons total).
4. π*2px, π*2py (4 electrons total).
5. σ*2pz (2 electrons). Step 4: Key points of the molecular orbital diagram.
σ2s and σ*2s are completely filled.
σ2pz, π2px, and π2py are also completely filled.
π*2px and π*2py each have two electrons. Step 5: Bond order calculation. The bond order is:
Bond order = (Number of bonding electrons - Number of antibonding electrons)/2.
Substitute:
Bond order = (8 - 6)/2 = 1. Step 6: Conclusion. The correct molecular orbital diagram is consistent with Option C, where:
Bonding orbitals ( σ2s, π2px, π2py, σ2pz ) are filled,
Antibonding orbitals ( σ*2s, π*2px, π*2py ) are partially filled.
Final Answer: The correct molecular orbital diagram is:
Option C. Quick Tip: Explanation for Incorrect Options:
Option A: Incorrectly shows the σ2pz orbital lower in energy than the π2px and π2py orbitals.
Option B: Incorrectly shows the σ2pz orbital lower in energy than the π2px and π*2py orbitals.
Option D: Incorrectly shows the π2px and π2py orbitals filled with two electrons each, violating Hund's rule.
Question 2:
Consider the following statements related to colloids:
(I) Lyophobic colloids are not formed by simple mixing of dispersed phase and dispersion medium.
(II) For emulsions, both the dispersed phase and the dispersion medium are liquid.
(III) Micelles are produced by dissolving a surfactant in any solvent at any temperature.
(IV) Tyndall effect can be observed from a colloidal solution with dispersed phase having the same refractive index as that of the dispersion medium.
The option with the correct set of statements is:
View Solution
Step 1: Analyze each statement.
(I) Lyophobic colloids are indeed not formed by simple mixing. They require stabilizing agents.
(II) In emulsions, both the dispersed phase and the dispersion medium are liquids. This is correct.
(III) Micelles form at the critical micelle concentration (CMC) and specific temperatures, like the Kraft temperature. This makes the statement incorrect.
(IV) Tyndall effect requires different refractive indices of the dispersed phase and medium, so this statement is incorrect.
Quick Tip: Colloids exhibit unique properties such as the Tyndall effect, which depends on differences in refractive indices. Lyophobic colloids require stabilizers for formation.
Question 3:
In the following reactions, P, Q, R, and S are the major products.
The correct statement about P, Q, R, and S is:


% Reactions (Insert reaction diagrams here)
View Solution
Step 1: Analyze each reaction step.
- P : Treating the compound with Mg/dry ether followed by hydrolysis ( H2O ) yields a primary alcohol containing four carbon atoms.
- Q : The reaction sequence produces a carboxylic acid, which undergoes Kolbe’s electrolysis to form an eight-carbon compound.
- R : The reaction forms an aldehyde that does not participate in the Cannizzaro reaction.
- S : The reaction results in the formation of a primary amine with six carbon atoms. Quick Tip: Understand the mechanism of Grignard reagents and the conditions required for reactions like Kolbe’s electrolysis and Cannizzaro reactions.
Question 4:
A disaccharide X cannot be oxidized by bromine water. The acid hydrolysis of X leads to a laevorotatory solution. The disaccharide X is:


View Solution
Step 1: Analyze the nature of sucrose. Sucrose is a non-reducing disaccharide because its anomeric carbons (C1 of glucose and C2 of fructose) are involved in a glycosidic bond, leaving no free aldehyde or keto group. Hence, sucrose does not react with bromine water. Step 2: Hydrolysis of sucrose. When sucrose is hydrolyzed by acid or the enzyme sucrase, it produces equimolar amounts of glucose and fructose:
sucrose + H2O → glucose + fructose.
Glucose is dextrorotatory (+52.7°) and fructose is laevorotatory (-92.4°). The resulting solution exhibits a net laevorotation because fructose’s negative optical rotation dominates. Step 3: Conclusion.
The key properties of sucrose are:
- It is a non-reducing sugar and does not react with bromine water.
- On hydrolysis, it gives glucose and fructose, with the solution becoming laevorotatory.
Question 5:
The complex(es) which can exhibit the type of isomerism shown by [Pt(NH3)2Br2] is(are):
View Solution
Step 1: Analyze the given complexes. [Pt(NH3)2Br2]: This complex is square planar and exhibits cis-trans isomerism.
(A) [Pt(en)(SCN)2]: This complex does not exhibit isomerism because of the symmetrical arrangement of ligands.
(B) [Zn(NH3)2Cl2]: Being a tetrahedral complex, it does not exhibit geometrical isomerism.
(C) [Pt(NH3)2Cl4]: This octahedral complex shows geometrical isomerism due to the possibility of cis and trans arrangements of ligands.
(D) [Cr(en)2(H2O)(SO4)]+: This complex is octahedral and exhibits geometrical isomerism due to different spatial arrangements of the ligands. Quick Tip: For geometrical isomerism, the arrangement of ligands in octahedral or square planar complexes is key. Tetrahedral complexes do not exhibit this type of isomerism.
Question 6:
Atoms of metals x , y , and z form face-centred cubic (fcc) unit cell of edge length Lx , body-centred cubic (bcc) unit cell of edge length Ly , and simple cubic unit cell of edge length Lz , respectively. If rz = √3/2ry; ry = 8/√3rx ; Mz = 3/2My and Mz = 3Mx , then the correct statement(s) is(are): Given: Mx, My, and Mz are molar masses of metals x, y, and z, respectively. rx, ry, and rz are atomic radii of metals x, y, and z, respectively
(B) Ly > Lz .
, (D) Density of x > Density of y .
View Solution
Step 1: Analyze edge lengths and radii. For metal x (FCC):
Lx = 2√2rx.
For metal y (BCC):
Ly = 4ry/√3.
For metal z (SC):
Lz = 2rz.
Step 2: Packing efficiencies. (i) For FCC (Z = 4) metal 'x', 4rx = √2Lx
P.E. = (Z × 4/3π(rx)3)/ax3 = (4 × 4/3π(rx)3)/(Lx)3 = (4 × 4/3π(rx)3)/((4rx/√2)3 = 0.24π
(ii) For BCC (Z = 2) metal 'y', 4ry = √3Ly
P.E. = (Z × 4/3π(ry)3)/ay3 = (2 × 4/3π(ry)3)/(Ly)3 = (2 × 4/3π(ry)3)/((4ry/√3)3 = 0.22π
(iii) For SC (Z = 1) metal 'z', 2rz = Lz
P.E. = (Z × 4/3π(rz)3)/az3 = (1 × 4/3π(rz)3)/(Lz)3 = (1 × 4/3π(rz)3)/(2rz)3 = π/6 = 0.17π
Thus, P.E.FCC > P.E.BCC > P.E.SC . Step 3: Density comparison.
4rx = √2Lx, 4ry = √3Ly, 2rz = Lz
Lx = 4rx/√2, Ly = 4ry/√3
Ly/Lx = (4ry/√3)/(4rx/√2) = √2ry/√3rx = (√2ry)/(√3·(√3/2ry)) = 4/3
Therefore, Ly > Lx
For option (C)
4rx = √2Lx, 4ry = √3Ly
Lx = 4rx/√2, Ly = 4ry/√3
Ly/Lx = (4ry/√3)/(4rx/√2) = √2ry/√3rx = (√2 × 8/√3rx)/(√3rx) = 8√2/3
Therefore, Lx < Ly (incorrect)
For option (D)
dx = (4 × Mx)/((4rx/√2)3 × NA)
dy = (4 × My)/((4ry/√3)3 × NA)
ry = 8/√3rx, Mx/My = 1/2
dx/dy = 512/2√2 = 256/√2
Therefore, dx > dy (correct)
ρx > ρy > ρz. Quick Tip: Packing efficiency decreases from FCC to BCC to SC. Use relations between edge length and radius to solve such problems.
Question 7:
In the following reactions, P, Q, R, and S are the major products. The correct statement(s) about P, Q, R, and S is(are):

(D) R does not undergo aldol condensation and S does not undergo Cannizzaro reaction.
View Solution
Step 1: Analyze the reactions for P, Q, R, and S .
P: Oxidation of the alkyl group results in the formation of a dicarboxylic acid.
Q: Hydrolysis of the acyl chloride produces the same dicarboxylic acid as in P .
R: The product lacks an alpha-hydrogen, making it incapable of undergoing aldol condensation.
S: The product cannot undergo the Cannizzaro reaction since it is not a non-enolizable aldehyde. Quick Tip: Use the structural features of the products to analyze their reactivity towards condensation and Cannizzaro reactions.
Question 8:
H2S (5 moles) reacts completely with acidified aqueous potassium permanganate solution. In this reaction, the number of moles of water produced is x , and the number of moles of electrons involved is y . The value of x + y is:
View Solution
The balanced half-reactions are:
For permanganate ion reduction:
MnO4- + 8H+ + 5e- → Mn2+ + 4H2O × 2.
For hydrogen sulfide oxidation:
H2S → S + 2H+ + 2e- × 5.
Combine the two half-reactions:
2MnO4- + 16H+ + 5H2S → 2Mn2+ + 5S + 8H2O + 10H+.
Simplify:
2MnO4- + 6H+ + 5H2S → 2Mn2+ + 5S + 8H2O.
Here:
x = 8 (moles of water), y = 10 (moles of electrons transferred).
The sum is:
x + y = 8 + 10 = 18. Quick Tip: Balance redox reactions by ensuring conservation of both mass and charge.
Question 9:
Among [I3]+, [SiO4]4-, SO2Cl2, XeF2, SF4, ClF3, Ni(CO)4, XeO2F2, [PtCl4]2-, XeF4,
and SOCl2 , the total number of species having sp3 -hybridised central atom is:
View Solution
The species with sp3 -hybridized central atoms are as follows: [SiO4]4-: The central silicon atom exhibits tetrahedral geometry with sp3 hybridization. SOCl2: The central sulfur atom has tetrahedral geometry with sp3 hybridization. Ni(CO)4: The central nickel atom forms a tetrahedral structure with sp3 hybridization. XeO2F2: The central xenon atom exhibits a distorted tetrahedral geometry with sp3 hybridization. SF4: The central sulfur atom has a see-saw geometry and sp3 hybridization. Thus, the total number of species with sp3 -hybridized central atoms is 5. Quick Tip: Determine the hybridization by considering the steric number (sum of bonded atoms and lone pairs).
Question 10:
Consider the following molecules: Br3O8, F2O, H2S4O6, H2S5O6, and C3O2 . Count the number of atoms existing in their zero oxidation state in each molecule. Their sum is:
View Solution
Step 1: Determine the number of atoms in the zero oxidation state for each molecule. (i) Br3O8: In this molecule, one bromine atom is in the zero oxidation state. (ii) F2O: Both fluorine atoms are in the zero oxidation state. (iii) H2S4O6: Among the sulfur atoms, one is in the zero oxidation state. (iv) H2S5O6: Two sulfur atoms are in the zero oxidation state. (v) C3O2: All three carbon atoms are in the zero oxidation state. Step 2: Add the counts.
1 (from Br3O8) + 2 (from F2O) + 1 (from H2S4O6) + 2 (from H2S5O6) = 6.
Final Answer: The total number of atoms in the zero oxidation state is:
6. Quick Tip: Identify zero oxidation states by analyzing the bonding and oxidation state rules for each element.
Question 11:
For He+ , a transition takes place from the orbit of radius 105.8 pm to the orbit of radius 26.45 pm . The wavelength (in nm) of the emitted photon during the transition is: [Use:
Bohr radius, a = 52.9 pm
Rydberg constant, RH = 2.2 × 10-18 J
Planck's constant, h = 6.6 × 10-34 J s
Speed of light, c = 3 × 108 m s-1]
View Solution
The radius of an orbit is given by:
rn = (52.9 · n2)/Z pm.
For n1 :
105.8 = (52.9 · n12)/2 ⇒ n12 = 4 ⇒ n1 = 2.
For n2 :
26.45 = (52.9 · n22)/2 ⇒ n22 = 1 ⇒ n2 = 1.
Energy difference:
1/λ = 109677 · Z2 (1/n12 - 1/n22).
Substituting Z = 2 :
1/λ = 109677 · 4 (1/1 - 1/4) = 3 · 109677.
λ = 107/(3 · 109677) = 30.3 nm = 30 nm. Quick Tip: For hydrogen-like atoms, use the radius formula and the Rydberg equation for energy transitions.
Question 12:
50 mL of 0.2 molal urea solution (ρ = 1.012 g/mL at 300 K) is mixed with 250 mL
of a solution containing 0.06 g urea. Both the solutions were prepared in the same solvent. The osmotic pressure (in Torr) of the resulting solution at 300 K is: [Use: Molar mass of urea = 60 g mol-1; gas constant, R = 62 L Torr K-1 mol-1;
ΔHmix = 0, ΔVmix = 0]
View Solution
Step 1: Calculate the volume of the solution using its density. The mass of the solution is given as 1012 g. The density (d) is 1.012 g/ml. Using the formula Volume = Mass/Density, we get Volume = 1012 g / 1.012 g/ml = 1000.00 ml. Step 2: Determine the number of moles of solute in 1000 ml of solution. Since 1000 ml of solution contains 0.2 moles, the number of moles in 50 ml of solution is (0.2/1000) × 50 = 0.01 moles. Thus, nurea = 0.01 moles . Step 3: Calculate the moles of solute in the second solution. In the second solution, the mass of solute is 0.06 g. Using the molar mass of urea (60 g/mol), nurea = 0.06/60 = 0.001 moles . Step 4: Calculate the final molarity (M) of the mixed solution. The final molarity is given by M = (n1 + n2)/(V1 + V2) . Substituting the values, we get M = (0.01 + 0.001)/((50 + 250)/1000) = 0.011/(300/1000) = 11/300 . Step 5: Calculate the osmotic pressure (π). The osmotic pressure is given by π = CRT . Substituting the values, we get π = (11/300) × 62 × 300 = 682 torr . Final Answer: The osmotic pressure of the solution is π = 682 torr. Quick Tip: To calculate osmotic pressure, use π = MRT , ensuring proper conversion of moles and volumes.
Question 13:
The reaction of 4-methyloct-1-ene (P) (2.52 g) with HBr in the presence of (C6H5CO)2O2 gives two isomeric bromides in a 9:1 ratio, with a combined yield of 50%. Of these, the entire amount of the primary alkyl bromide was reacted with an appropriate amount of diethylamine followed by treatment with aq. K2CO3 to give a non-ionic product S in 100% yield. The mass (in mg) of S obtained is:
View Solution
Step 1: Given Reaction and Products Formation The given compound (P) undergoes an anti-Markovnikov addition reaction with HBr in the presence of a peroxide catalyst. The major and minor products formed are shown in the image. Step 2: Moles Calculation for Compound P Moles of P = 2.52/126 = 0.02 mole Step 3: Moles of Major Product A Formed Moles of A = 0.02 × (9/10) × (50/100) = 0.009 mole Step 4: Product Conversion and Mass Calculation The major product A undergoes further reaction with diethylamine (Et2NH) in the presence of K2CrO3, forming the final product with molecular formula C13H29N and molar mass 199 g/mol. Step 5: Final Mass Calculation Mass = 0.009 × 199 = 1.791 g = 1791 mg Conclusion: The final mass of the product obtained is 1791 mg. Quick Tip: Use stoichiometry and mole concept for calculating the yield in such reactions.
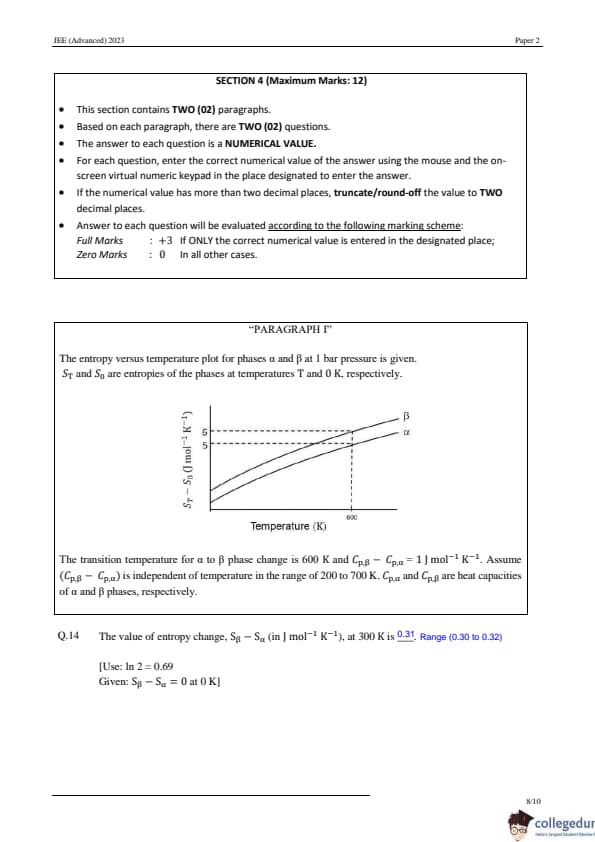
Question 14:
The value of entropy change, Sβ - Sα (in J mol-1 K-1), at 300 K is: [Use: ln 2 = 0.69, Given: Sβ - Sα = 0 at 0 K]
View Solution
Step 1: Write the entropy relation. The entropy S is given by:
S = S0 + ∫ Cp dT/T,
where Cp is the specific heat capacity at constant pressure. For the two phases, α and β:
Sα = S0 + ∫ (Cp)α dT/T, Sβ = S0 + ∫ (Cp)β dT/T. Step 2: Compute the entropy difference. The difference in entropy between the two phases is:
Sβ - Sα = ∫ [(Cp)β - (Cp)α] dT/T.
Given:
(Cp)β - (Cp)α = 1.
Substitute:
Sβ - Sα = ∫ 1/T dT.
Simplify:
Sβ - Sα = ln T + C,
where C is a constant of integration. Step 3: Evaluate the change in entropy at given temperatures. The change in Sβ - Sα between T2 = 600 K and T1 = 300 K is:
(Sβ - Sα)600 - (Sβ - Sα)300 = ln 600 - ln 300.
Simplify using the logarithmic property ln a - ln b = ln (a/b) :
(Sβ - Sα)600 - (Sβ - Sα)300 = ln (600/300) = ln 2.
ln 2 ≈ 0.69.
Step 4: Solve for (Sβ - Sα)300 . Using the relation:
(Sβ - Sα)600 = ln 600 + C, (Sβ - Sα)300 = ln 300 + C,
the difference at T1 = 300 K is:
(Sβ - Sα)300 = 1 - 0.69 = 0.31.
Final Answer: At T1 = 300 K , the entropy difference is:
0.31. Quick Tip: To calculate entropy change, use ΔS = ∫ Cp dT/T , considering given temperature limits.
Question 15:
The value of enthalpy change, Hβ - Hα (in J mol-1), at 300 K is:
View Solution
Step 1: Relation between enthalpy and entropy. The enthalpy change ΔH is related to the entropy change ΔS by:
ΔH = TΔS.
At T = 600 K and ΔS = 1 J mol-1 K-1 , the enthalpy change is:
ΔH600 = 600 × 1 = 600 J mol-1.
Step 2: Use Kirchhoff’s law to calculate ΔH300 . Kirchhoff’s law states:
ΔHT2 - ΔHT1 = ΔCp (T2 - T1),
where:
T1 = 300 K, T2 = 600 K,
ΔCp = 1 J mol-1 K-1,
ΔH600 = 600 J mol-1.
Substitute the values:
600 - ΔH300 = 1 · (600 - 300).
Simplify:
600 - ΔH300 = 300.
Solve for ΔH300 :
ΔH300 = 600 - 300 = 300 J mol-1.
Final Answer: The enthalpy change at T = 300 K is:
300 J mol-1. Quick Tip: Use Kirchhoff’s law to relate heat capacities and enthalpy changes across temperature ranges.
Question 16:
The number of heteroatoms present in one molecule of R is:
[Use: Molar mass (in g mol-1): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5. Atoms other than C and H are considered as heteroatoms.]
View Solution
The compound R contains:
3 oxygen atoms (O3),
3 nitrogen atoms (N3),
3 bromine atoms (Br3).
Total number of heteroatoms:
3 + 3 + 3 = 9. Quick Tip: Count heteroatoms explicitly by identifying atoms other than carbon and hydrogen in the molecule's structure.
Question 17:
The total number of carbon atoms and heteroatoms present in one molecule of S is:
[Use: Molar mass (in g mol-1): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5. Atoms other than C and H are considered as heteroatoms.]
View Solution
The compound S contains:
6 carbon atoms (C6),
3 nitrogen atoms (N3),
6 oxygen atoms (O6),
3 bromine atoms (Br3).
The structure of compound S is given as C24H18O3 . Let us count the total number of carbon atoms and heteroatoms. - Number of carbon atoms:
24 C atoms.
- Number of heteroatoms:
3 Oxygen atoms (O), 3 Nitrogen atoms (N), and 21 from aromatic rings (excluding hydrogens).
Total atoms (Carbon + Heteroatoms):
24 (C) + 27 (O + N) = 51. Quick Tip: Sum the total number of carbon atoms and heteroatoms based on molecular structure and stoichiometric data.



























JEE Advanced 2023 Paper 2 Questions
| Question Paper | Answer Key |
|---|---|
| Download PDF | Download PDF |
JEE Advanced 2023 Paper 2 Question Paper Pattern
| Question Type | Details | Questions | Total Marks |
|---|---|---|---|
| Section 1- MAMCQS | Questions with four options in which ONE OR MORE THAN ONE option(s) are correct | 3 | 12 marks |
| Section 2- SAQS | Questions with four options in which only ONE option was correct | 4 | 12 marks |
| Section 3- Integer Type Questions | Numerical Based (Non-Negative Integer Type) | 6 | 24 marks |
| Section 4- Paragraph-based Questions | Four questions based on two Paragraphs with two questions in each paragraph (Numerical based Decimal Type with answer correct to 2 decimal places) | 4 | 12 marks |
JEE Advanced 2023 Paper 2 Marking Scheme
Just like Paper 1, JEE Advanced 2023 Paper 2 total marks for each subject was 60 marks.
- Marking Scheme for MAMCQs
- Full marks: +4 only if all the correct options have been chosen.
- Partial Marks:
- +3 If all four options are correct but ONLY three correct options are chosen
- +2 If three or more options are correct but only two correct options are chosen
- +1 If two or more options are correct but only one correct option is chosen
- Zero Marks: 0 for unattempted/ unanswered questions
- Negative Marks: -2 in all other cases
- Marking Scheme for SAQs
- Full marks: +3 If only the correct option is chosen
- Zero Marks: 0 for unattempted/ unanswered questions
- Negative Marks: -1 In all other cases
- Marking Scheme for Integer Type Questions
- Full marks: +4 for the correct answer
- Zero Marks: 0 for unattempted/ unanswered questions
- Marking Scheme for Paragraph-based Questions
- Full marks: +3 in case all options are correct,
- Zero Marks: 0 for unattempted/ unanswered questions
Quick Links:
Also Check:








Comments