JEE Advanced 2024 Paper 1 Question Paper with Answer Key and Solution PDF is available for download here. Candidates can use the link provided by Collegedunia here to download JEE Advanced 2024 Answer Key for Paper 1. NTA has released the official JEE Advanced 2024 Answer Key PDF on the official website jeeadv.ac.in.
Download JEE Advanced 2025 Question Paper with Solution PDF
JEE Advanced 2024 Paper 1 Answer Key PDF Download
| JEE Advanced 2024 Paper 1 Question Paper with Answer Key | Check Solution |

Mathematics
Question 1:
Let \( f(x) \) be a continuously differentiable function on the interval \( (0, \infty) \) such that \( f(1) = 2 \) and
\[ \lim_{t \to x} \frac{t^{10} f(x) - x^{10} f(t)}{t^9 - x^9} = 1 \quad \text{for each } x > 0. \]
Then, for all \( x > 0 \), \( f(x) \) is equal to:
View Solution
Question 2:
A student appears for a quiz consisting of only true-false type questions and answers all the questions. The student knows the answers of some questions and guesses the answers for the remaining questions. Whenever the student knows the answer of a question, he gives the correct answer. Assume that the probability of the student giving the correct answer for a question, given that he has guessed it, is \( \frac{1}{2} \).
Also assume that the probability of the answer for a question being guessed, given that the student's answer is correct, is \( \frac{5}{6} \). Then the probability that the student knows the answer of a randomly chosen question is:
View Solution
Question 3:
Let \( \frac{\pi}{2} < x < \pi \) be such that \( \cot x = -\frac{5}{\sqrt{11}} \). Then \[ \sin \frac{11x}{2} (\sin 6x - \cos 6x) + \cos \frac{11x}{2} (\sin 6x + \cos 6x) \] is equal to:
View Solution
Question 4:
Consider the ellipse \[ \frac{x^2}{9} + \frac{y^2}{4} = 1. \] Let \( S(p, q) \) be a point in the first quadrant such that \( \frac{p^2}{9} + \frac{q^2}{4} > 1 \). Two tangents are drawn from \( S \) to the ellipse, one of which meets the ellipse at one endpoint of the minor axis, and the other meets the ellipse at a point \( T \) in the fourth quadrant. Let \( R \) be the vertex of the ellipse with positive \( x \)-coordinate, and \( O \) be the center of the ellipse. If the area of the triangle \( \Delta ORT \) is \( \frac{3}{2} \), which of the following options is correct?
View Solution
Question 5:
Let \( S = \{a + b\sqrt{2} : a, b \in \mathbb{Z}\} \), \( T_1 = \{(-1 + n\sqrt{2})^2 : n \in \mathbb{N}\} \), and \( T_2 = \{(1 + n\sqrt{2})^2 : n \in \mathbb{N}\} \). Then which of the following statements is (are) TRUE?
Question 6:
Let \( \mathbb{R}^2 \) denote \( \mathbb{R} \times \mathbb{R} \). Let \( S = \{(a, b, c) : a, b, c \in \mathbb{R} \text{ and } ax^2 + 2bxy + cy^2 > 0 \text{ for all } (x, y) \in \mathbb{R}^2 \setminus \{(0, 0)\}\} \). Then which of the following statements is (are) TRUE?
Question 7:
Let \( \mathbb{R}^3 \) denote the three-dimensional space. Take two points \( P = (1, 2, 3) \) and \( Q = (4, 2, 7) \). Let \( \text{dist}(X, Y) \) denote the distance between two points \( X \) and \( Y \) in \( \mathbb{R}^3 \). Let: \[ S = \{X \in \mathbb{R}^3 : (\text{dist}(X, P))^2 - (\text{dist}(X, Q))^2 = 50\}, \] \[ T = \{Y \in \mathbb{R}^3 : (\text{dist}(Y, Q))^2 - (\text{dist}(Y, P))^2 = 50\}. \] Then which of the following statements is (are) TRUE?
Question 8:
Let \( a = 3\sqrt{2} \) and \( b = \frac{1}{5^{1/6}\sqrt{6}} \). If \( x, y \in \mathbb{R} \) are such that: \[ 3x + 2y = \log_a\left(18^{5/4}\right) \quad \text{and} \quad 2x - y = \log_b\left(\sqrt{1080}\right), \] then \( 4x + 5y \) is equal to _____.
Question 9:
Let \( f(x) = x^4 + ax^3 + bx^2 + c \) be a polynomial with real coefficients such that \( f(1) = -9 \). Suppose that \( i\sqrt{3} \) is a root of the equation \( 4x^3 + 3ax^2 + 2bx = 0 \), where \( i = \sqrt{-1} \). If \( \alpha_1, \alpha_2, \alpha_3, \alpha_4 \) are all the roots of the equation \( f(x) = 0 \), then \( |\alpha_1|^2 + |\alpha_2|^2 + |\alpha_3|^2 + |\alpha_4|^2 \) is equal to ____.
Question 10:
Let \( S = \{A = \begin{pmatrix} 0 & 1 & c
1 & a & d
1 & b & e \end{pmatrix} : a, b, c, d, e \in \{0, 1\}\} \) and \( |A| \in \{-1, 1\} \), where \( |A| \) denotes the determinant of \( A \). Then the number of elements in \( S \) is _____.
Question 11:
Let \( s_1, s_2, \ldots, s_9 \) be 9 students. They are to be divided into three teams \( X \), \( Y \), and \( Z \) of sizes 2, 3, and 4, respectively. Suppose that \( s_1 \) cannot be selected for team \( X \), and \( s_2 \) cannot be selected for team \( Y \). Then the number of ways to form such teams is ____.
Question 12:
Let \( \overrightarrow{OP} = \frac{\alpha - 1}{\alpha} \, \mathbf{i} + \mathbf{j} + \mathbf{k} \), \( \overrightarrow{OQ} = \mathbf{i} + \frac{\beta - 1}{\beta} \, \mathbf{j} + \mathbf{k} \), and \( \overrightarrow{OR} = \mathbf{i} + \mathbf{j} + \frac{1}{2} \mathbf{k} \) be three vectors, where \( \alpha, \beta \in \mathbb{R} - \{0\} \) and \( O \) denotes the origin. If \( (\overrightarrow{OP} \times \overrightarrow{OQ}) \cdot \overrightarrow{OR} = 0 \) and the point \( (\alpha, \beta, 2) \) lies on the plane \[ 3x + 3y - z + \ell = 0, \] then the value of \( \ell \) is ____.
Question 13:
Let \(X\) be a random variable, and let \(P(X = x)\) denote the probability that \(X\) takes the value \(x\). Suppose that points \((x, P(X = x))\), \(x = 0, 1, 2, 3, 4\), lie on a fixed straight line in the \(xy\)-plane, and \(P(X = x) > 0\) for all \(x \in \{0, 1, 2, 3, 4\}\). If the mean of \(X\) is \(\frac{5}{2}\) and the variance of \(X\) is \(\alpha\), then the value of \(24\alpha\) is ____.
Question 14:
Let \(\alpha\) and \(\beta\) be the distinct roots of the equation \(x^2 + x - 1 = 0\). Consider the set \(T = \{\alpha, \beta\}\). For a \(3 \times 3\) matrix \(M = (a_{ij})_{3 \times 3}\), define \(R_i = a_{i1} + a_{i2} + a_{i3}\) and \(C_j = a_{1j} + a_{2j} + a_{3j}\) for \(i = 1, 2, 3\) and \(j = 1, 2, 3\).
[15]
Question 15:
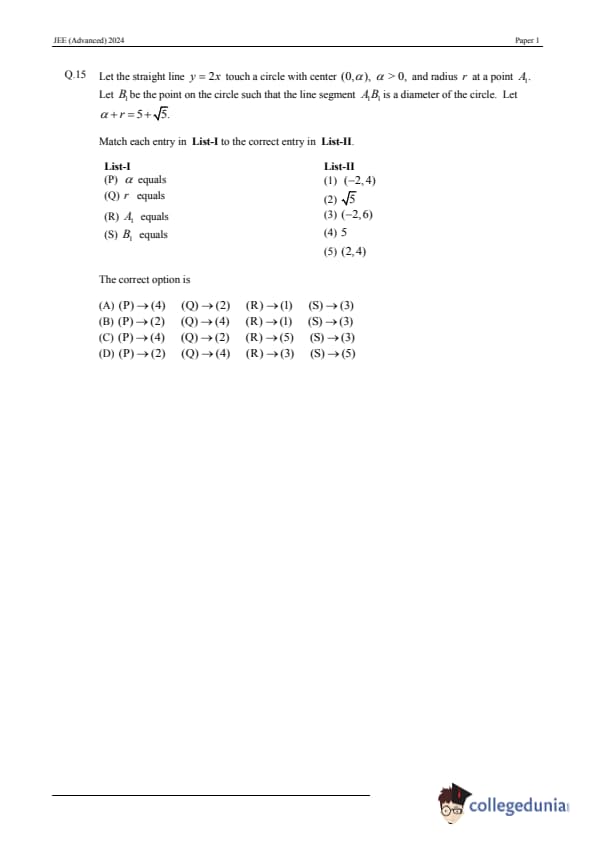
Let the straight line \(y = 2x\) touch a circle with center \((0, \alpha)\), \(\alpha > 0\), and radius \(r\) at a point \(A_1\). Let \(B_1\) be the point on the circle such that the line segment \(A_1B_1\) is a diameter of the circle. Let \(\alpha + r = 5 + \sqrt{5}\). Match each entry in List-I to the correct entry in List-II.

[(A)] (P) \(\to\) (4), (Q) \(\to\) (2), (R) \(\to\) (1), (S) \(\to\) (3)
[(B)] (P) \(\to\) (2), (Q) \(\to\) (4), (R) \(\to\) (1), (S) \(\to\) (3)
[(C)] (P) \(\to\) (4), (Q) \(\to\) (2), (R) \(\to\) (5), (S) \(\to\) (3)
[(D)] (P) \(\to\) (2), (Q) \(\to\) (4), (R) \(\to\) (3), (S) \(\to\) (5)
Question 16:
Let \(\gamma \in \mathbb{R}\) be such that the lines \(L_1: \frac{x+11}{1} = \frac{y+21}{2} = \frac{z+29}{3}\) and \(L_2: \frac{x+16}{3} = \frac{y+11}{2} = \frac{z+4}{7}\) intersect. Let \(R_1\) be the point of intersection of \(L_1\) and \(L_2\). Let \(O = (0, 0, 0)\), and \(\mathbf{\hat{n}}\) denote a unit normal vector to the plane containing both the lines \(L_1\) and \(L_2\). Match each entry in List-I to the correct entry in List-II.

The correct option is:
[(A)] (P) \(\to\) (3), (Q) \(\to\) (4), (R) \(\to\) (1), (S) \(\to\) (2)
[(B)] (P) \(\to\) (5), (Q) \(\to\) (4), (R) \(\to\) (1), (S) \(\to\) (2)
[(C)] (P) \(\to\) (3), (Q) \(\to\) (4), (R) \(\to\) (1), (S) \(\to\) (5)
[(D)] (P) \(\to\) (3), (Q) \(\to\) (1), (R) \(\to\) (4), (S) \(\to\) (5)
Question 17:
Let \(f: \mathbb{R} \to \mathbb{R}\) and \(g: \mathbb{R} \to \mathbb{R}\) be functions defined by: Let \(a, b, c, d \in \mathbb{R}\). Define the function \(h: \mathbb{R} \to \mathbb{R}\) by: \[ h(x) = a f(x) + b g(x) + c g\left(\frac{1}{2} - x\right) + d (x - g(x)) + d g(x), \quad x \in \mathbb{R}. \] Match each entry in List-I to the correct entry in List-II.

Physics
Question 1:
A dimensionless quantity is constructed in terms of electronic charge \(e\), permittivity of free space \(\varepsilon_0\), Planck’s constant \(h\), and speed of light \(c\). If the dimensionless quantity is written as \(e^{\alpha} \varepsilon_0^{\beta} h^{\gamma} c^{\delta}\) and \(n\) is a non-zero integer, then \((\alpha, \beta, \gamma, \delta)\) is given by:
(A) \((2n, -n, -n, -n)\)
(B) \((n, -n, -2n, -n)\)
(C) \((n, -n, -n, -2n)\)
(D) \((2n, -n, -2n, -2n)\)
Question 2:
An infinitely long wire, located on the \(z\)-axis, carries a current \(I\) along the \(+z\)-direction and produces the magnetic field \(\vec{B}\). The magnitude of the line integral \(\int \vec{B} \cdot d\vec{l}\) along a straight line from the point \((- \sqrt{3} \, a, a, 0)\) to \((a, a, 0)\) is given by:
(A) \(7\mu_0 I / 24\)
(B) \(7\mu_0 I / 12\)
(C) \(\mu_0 I / 8\)
(D) \(\mu_0 I / 6\)
Question 3:
Two beads, each with charge \(q\) and mass \(m\), are on a horizontal, frictionless, non-conducting, circular hoop of radius \(R\). One of the beads is glued to the hoop at some point, while the other one performs small oscillations about its equilibrium position along the hoop. The square of the angular frequency of the small oscillations is given by:
(A) \(\frac{q^2}{4 \pi \varepsilon_0 R^3 m}\)
(B) \(\frac{q^2}{32 \pi \varepsilon_0 R^3 m}\)
(C) \(\frac{q^2}{8 \pi \varepsilon_0 R^3 m}\)
(D) \(\frac{q^2}{16 \pi \varepsilon_0 R^3 m}\)
Question 4:
A block of mass \(5 \, \mathrm{kg}\) moves along the \(x\)-direction subject to the force \(F = (-20x + 10) \, \mathrm{N}\), with the value of \(x\) in metres. At time \(t = 0 \, \mathrm{s}\), it is at rest at position \(x = 1 \, \mathrm{m}\). The position and momentum of the block at \(t = \pi / 4 \, \mathrm{s}\) are:
(A) \(-0.5 \, \mathrm{m}, 5 \, \mathrm{kg \, m/s}\)
(B) \(0.5 \, \mathrm{m}, 0 \, \mathrm{kg \, m/s}\)
(C) \(0.5 \, \mathrm{m}, -5 \, \mathrm{kg \, m/s}\)
(D) \(-1 \, \mathrm{m}, 5 \, \mathrm{kg \, m/s}\)
Question 5:
A particle of mass \(m\) is moving in a circular orbit under the influence of the central force \(F(r) = -kr\), corresponding to the potential energy \(V(r) = \frac{kr^2}{2}\), where \(k\) is a positive force constant and \(r\) is the radial distance from the origin. According to Bohr’s quantization rule, the angular momentum of the particle is given by \(L = n \hbar\), where \(\hbar = h / (2 \pi)\), \(h\) is Planck's constant, and \(n\) a positive integer. If \(v\) and \(E\) are the speed and total energy of the particle, respectively, then which of the following expression(s) is (are) correct? (A) \(r^2 = n \hbar \sqrt{\frac{1}{mk}}\)
(B) \(v^2 = n \hbar \sqrt{\frac{k}{m^3}}\)
(C) \(\frac{L}{m r^2} = \sqrt{\frac{k}{m}}\)
(D) \(E = \frac{n \hbar}{2} \sqrt{\frac{k}{m}}\)
Question 6:
Two uniform strings of mass per unit length \(\mu\) and \(4\mu\), and length \(L\) and \(2L\), respectively, are joined at point \(O\), and tied at two fixed ends \(P\) and \(Q\), as shown in the figure. The strings are under a uniform tension \(T\). If we define the frequency \(v_0 = \frac{1}{2L} \sqrt{\frac{T}{\mu}}\), which of the following statement(s) is (are) correct?
(A) With a node at \(O\), the minimum frequency of vibration of the composite string is \(v_0\).
(B) With an antinode at \(O\), the minimum frequency of vibration of the composite string is \(2v_0\).
(C) When the composite string vibrates at the minimum frequency with a node at \(O\), it has \(6\) nodes, including the end nodes.
(D) No vibration mode with an antinode at \(O\) is possible for the composite string.

Question 7:
A glass beaker has a solid, plano-convex base of refractive index \(1.60\), as shown in the figure. The radius of curvature of the convex surface (SPU) is \(9 \, \mathrm{cm}\), while the planar surface (STU) acts as a mirror. This beaker is filled with a liquid of refractive index \(n\) up to the level QPR. If the image of a point object \(O\) at a height of \(h\) (OT in the figure) is formed onto itself, then, which of the following option(s) is (are) correct?
(A) For \(n = 1.42, h = 50 \, \mathrm{cm}\).
(B) For \(n = 1.35, h = 36 \, \mathrm{cm}\).
(C) For \(n = 1.45, h = 65 \, \mathrm{cm}\).
(D) For \(n = 1.48, h = 85 \, \mathrm{cm}\).

(B) For \(n = 1.35, h = 36 \, \mathrm{cm}\). View Solution
Question 8:
The specific heat capacity of a substance is temperature dependent and is given by the formula \(C = kT\), where \(k\) is a constant of suitable dimensions in SI units, and \(T\) is the absolute temperature. If the heat required to raise the temperature of \(1 \, \mathrm{kg}\) of the substance from \(-73^\circ \mathrm{C}\) to \(27^\circ \mathrm{C}\) is \(nk\), the value of \(n\) is: [Given: \(0 \, \mathrm{K} = -273^\circ \mathrm{C}\).]
Question 9:
A disc of mass \(M\) and radius \(R\) is free to rotate about its vertical axis as shown in the figure. A battery-operated motor of negligible mass is fixed to this disc at a point on its circumference. Another disc of the same mass \(M\) and radius \(R/2\) is fixed to the motor’s thin shaft. Initially, both the discs are at rest. The motor is switched on so that the smaller disc rotates at a uniform angular speed \(\omega\). If the angular speed at which the large disc rotates is \(\omega/n\), then the value of \(n\) is __.

Question 10:
A point source \(S\) emits unpolarised light uniformly in all directions. At two points \(A\) and \(B\), the ratio \(r = \frac{I_A}{I_B}\) of the intensities of light is \(2\). If a set of two polaroids having \(45^\circ\) angle between their pass-axes is placed just before point \(B\), then the new value of \(r\) will be __.
Question 11:
A source (S) of sound has frequency \(240 \, \mathrm{Hz}\). When the observer (O) and the source move towards each other at a speed \(v\) with respect to the ground (as shown in Case 1 in the figure), the observer measures the frequency of the sound to be \(288 \, \mathrm{Hz}\). However, when the observer and the source move away from each other at the same speed \(v\) with respect to the ground (as shown in Case 2 in the figure), the observer measures the frequency of the sound to be \(n \, \mathrm{Hz}\). The value of \(n\) is __. \includegraphics[width=10cm]{P11.png
Question 12:
Two large, identical water tanks, 1 and 2, kept on the top of a building of height \(H\), are filled with water up to height \(h\) in each tank. Both the tanks contain an identical hole of small radius on their sides, close to their bottom. A pipe of the same internal radius as that of the hole is connected to tank 2, and the pipe ends at the ground level. When the water flows from the tanks 1 and 2 through the holes, the times taken to empty the tanks are \(t_1\) and \(t_2\), respectively. If \(H = \frac{16}{9} h\), then the ratio \(t_1/t_2\) is __.
Question 13:
A thin uniform rod of length \(L\) and certain mass is kept on a frictionless horizontal table with a massless string of length \(L\) fixed to one end (top view is shown in the figure). The other end of the string is pivoted to a point \(O\). If a horizontal impulse \(P\) is imparted to the rod at a distance \(x = L/n\) from the mid-point of the rod (see figure), then the rod and string revolve together around the point \(O\), with the rod remaining aligned with the string. In such a case, the value of \(n\) is __.

Chemistry
Question 1:
A closed vessel contains 10 g of an ideal gas \( X \) at 300 K, which exerts 2 atm pressure. At the same temperature, 80 g of another ideal gas \( Y \) is added to it, and the pressure becomes 6 atm. The ratio of root mean square velocities of \( X \) and \( Y \) at 300 K is:
[(A)] \( 2\sqrt{2} : \sqrt{3} \)
[(B)] \( 2\sqrt{2} : 1 \)
[(C)] \( 1 : 2 \)
[(D)] \( 2 : 1 \)
Question 2:
At room temperature, disproportionation of an aqueous solution of \( \text{in situ} \) generated nitrous acid (\( \text{HNO}_2 \)) gives the species:
[(A)] \( \text{H}_3\text{O}^+, \text{NO}_3^- \text{ and NO} \)
[(B)] \( \text{H}_3\text{O}^+, \text{NO}_3^- \text{ and NO}_2 \)
[(C)] \( \text{H}_3\text{O}^+, \text{NO}^- \text{ and NO}_2 \)
[(D)] \( \text{H}_3\text{O}^+, \text{NO}_3^- \text{ and N}_2\text{O} \)
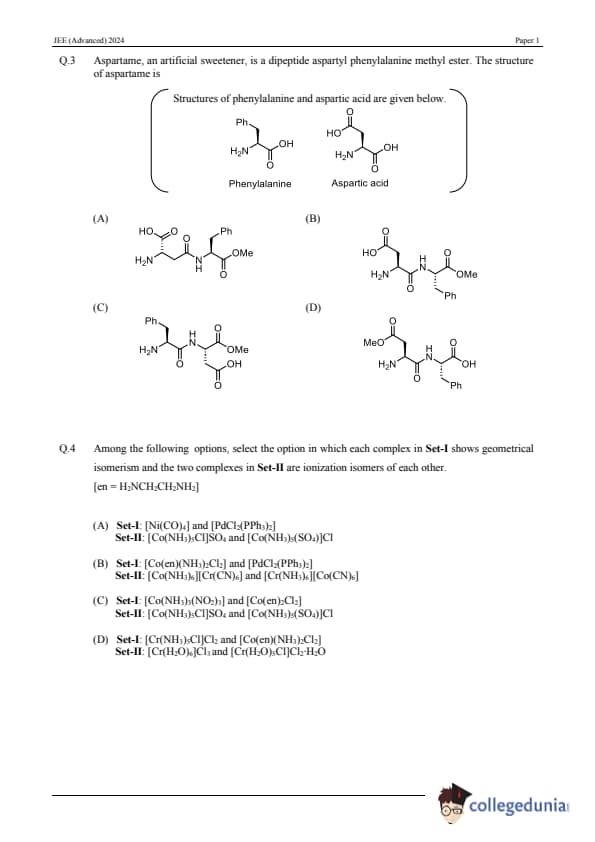
Question 3:
Aspartame, an artificial sweetener, is a dipeptide aspartyl phenylalanine methyl ester. The structure of aspartame is: 
Question 4:
Among the following options, select the option in which each complex in Set-I shows geometrical isomerism, and the two complexes in Set-II are ionization isomers of each other. \[ \text{[en = H}_2\text{NCH}_2\text{CH}_2\text{NH}_2] \]


View Solution
Question 5:
Among the following, the correct statement(s) for electrons in an atom is(are): [(A)] Uncertainty principle rules out the existence of definite paths for electrons. [(B)] The energy of an electron in \(2s\) orbital of an atom is lower than the energy of an electron that is infinitely far away from the nucleus. [(C)] According to Bohr's model, the most negative energy value for an electron is given by \(n = 1\), which corresponds to the most stable orbit. [(D)] According to Bohr's model, the magnitude of velocity of electrons increases with increases in values of \(n\).
Question 6:
Reaction of iso-propylbenzene with \( O_2 \) followed by treatment with \( H_3O^+ \) forms phenol and a by-product \( P \). Reaction of \( P \) with 3 equivalents of \( Cl_2 \) gives compound \( Q \). Treatment of \( Q \) with \( Ca(OH)_2 \) produces compound \( R \) and calcium salt \( S \). The correct statement(s) regarding \( P \), \( Q \), \( R \), and \( S \) is(are): Options:
Question 7:
The option(s) in which at least three molecules follow the Octet Rule is(are):
(A) \( CO_2, C_2H_4, NO \) and \( HCl \)
(D) \( CO_2, BCl_3, O_3 \) and \( C_2H_4 \)
View SolutionQuestion 8:
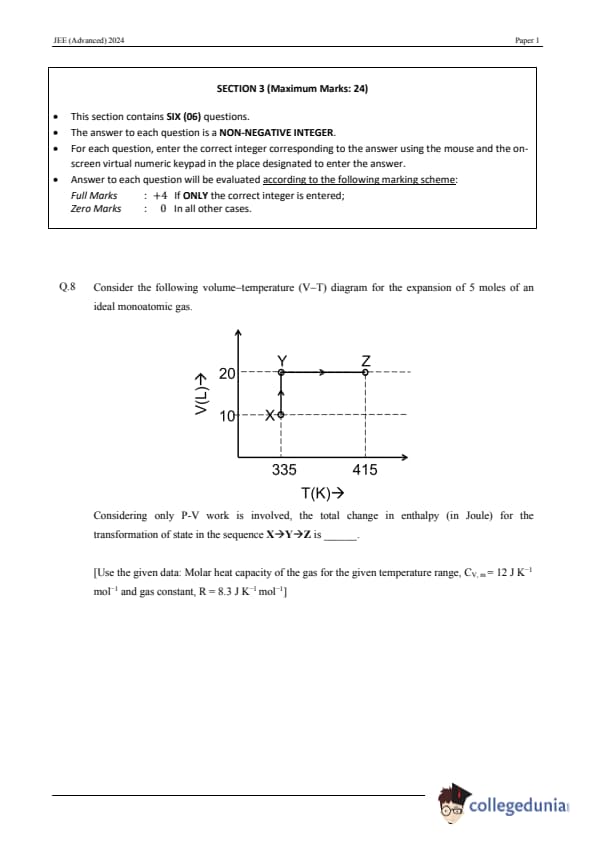
Consider the following volume–temperature (V–T) diagram for the expansion of 5 moles of an ideal monoatomic gas. \includegraphics[width=0.6\textwidth]{C8.png Considering only P-V work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence \( X \rightarrow Y \rightarrow Z \) is ______. [Use the given data: Molar heat capacity of the gas for the given temperature range, \( C_{V,m} = 12 \) J \( K^{-1} \) mol\(^{-1} \) and gas constant, \( R = 8.3 \) J \( K^{-1} \) mol\(^{-1} \)].
Question 9:
Consider the following reaction, \[ 2H_2 (g) + 2NO (g) \rightarrow N_2 (g) + 2H_2O (g) \] which follows the mechanism given below: \includegraphics[width=10cm]{C9.png The order of the reaction is ______.
Question 10:
Complete reaction of acetaldehyde with excess formaldehyde, upon heating with conc. NaOH solution, gives P and Q. Compound P does not give Tollens’ test, whereas Q on acidification gives positive Tollens’ test. Treatment of P with excess cyclohexanone in the presence of catalytic amount of p-toluenesulfonic acid (PTSA) gives product R. Sum of the number of methylene groups (-CH₂-) and oxygen atoms in R is ______.
Question 11:
Among V(CO)₆, Cr(CO)₅, Cu(CO)₃, Mn(CO)₅, Fe(CO)₅, [Co(CO)₃]³⁻, [Cr(CO)₄]⁴⁻, and Ir(CO)₃, the total number of species isoelectronic with Ni(CO)₄ is ______. [Given, atomic number: V = 23, Cr = 24, Mn = 25, Fe = 26, Co = 27, Ni = 28, Cu = 29, Ir = 77]
Question 12:
In the following reaction sequence, the major product P is formed. \includegraphics[width=10cm]{C12.png Glycerol reacts completely with excess P in the presence of an acid catalyst to form Q. Reaction of Q with excess NaOH followed by the treatment with CaCl₂ yields Ca-soap R, quantitatively. Starting with one mole of Q, the amount of R produced in grams is ______. [Given, atomic weight: H = 1, C = 12, N = 14, O = 16, Na = 23, Cl = 35, Ca = 40]
Question 13:
Among the following complexes, the total number of diamagnetic species is ______. \[ [\text{Mn(NH}_3)_6]^{3+}, [\text{MnCl}_6]^{3-}, [\text{FeF}_6]^{3-}, [\text{CoF}_6]^{3-}, [\text{Fe(NH}_3)_6]^{3+}, \text{and } [\text{Co(en)}_3]^{3+} \] [Given, atomic number: Mn = 25, Fe = 26, Co = 27; en = H₂NCH₂CH₂NH₂]
Question 14:
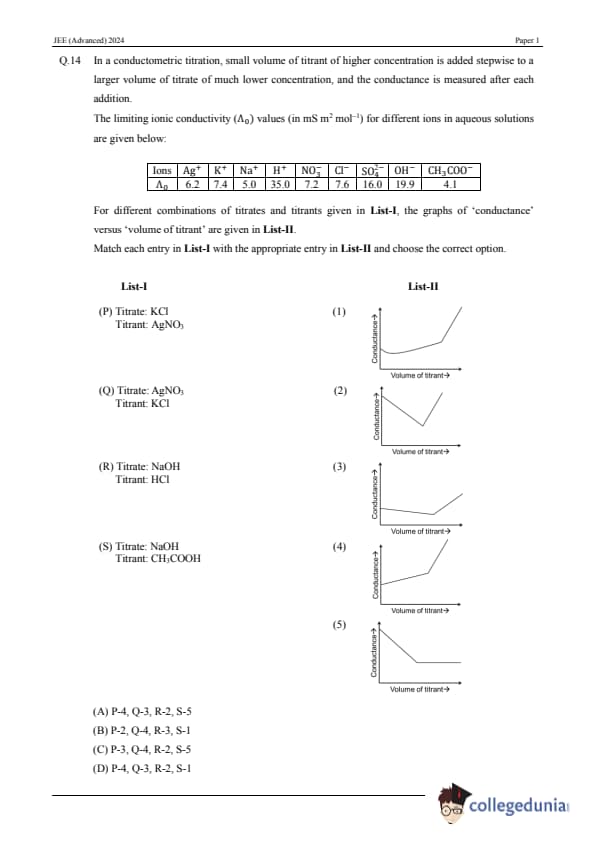
In a conductometric titration, a small volume of titrant of higher concentration is added stepwise to a larger volume of titrate of much lower concentration, and the conductance is measured after each addition. The limiting ionic conductivity (\(\Lambda_0\)) values (in mS m² mol\(^{-1}\)) for different ions in aqueous solutions are given below:

For different combinations of titrates and titrants given in List-I, the graphs of ‘conductance’ versus ‘volume of titrant’ are given in List-II. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
Question 15:
Based on VSEPR model, match the xenon compounds given in List-I with the corresponding geometries and the number of lone pairs on xenon given in List-II and choose the correct option.

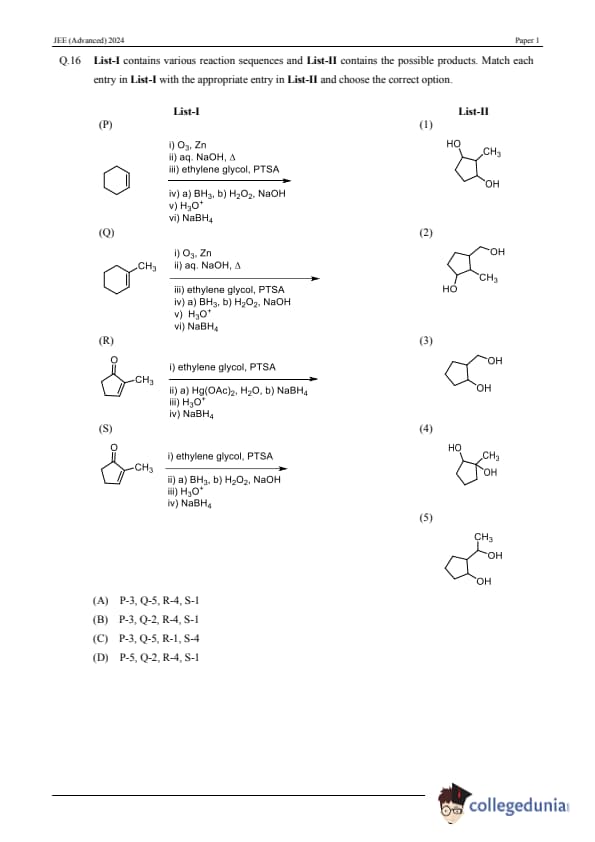
Question 16:
List-I contains various reaction sequences and List-II contains the possible products. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
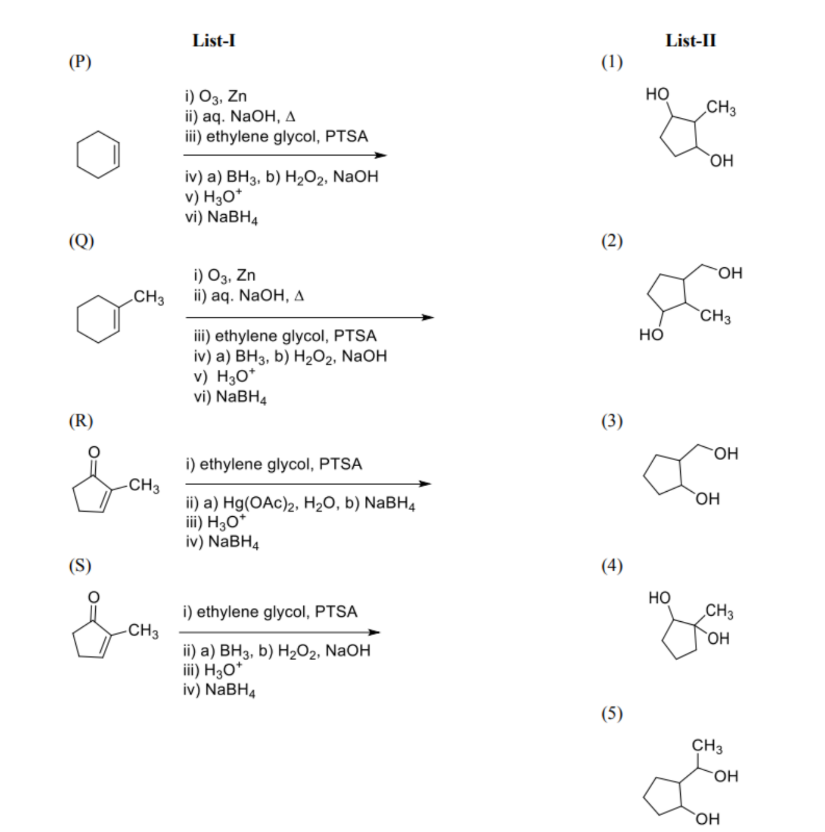
Question 17:
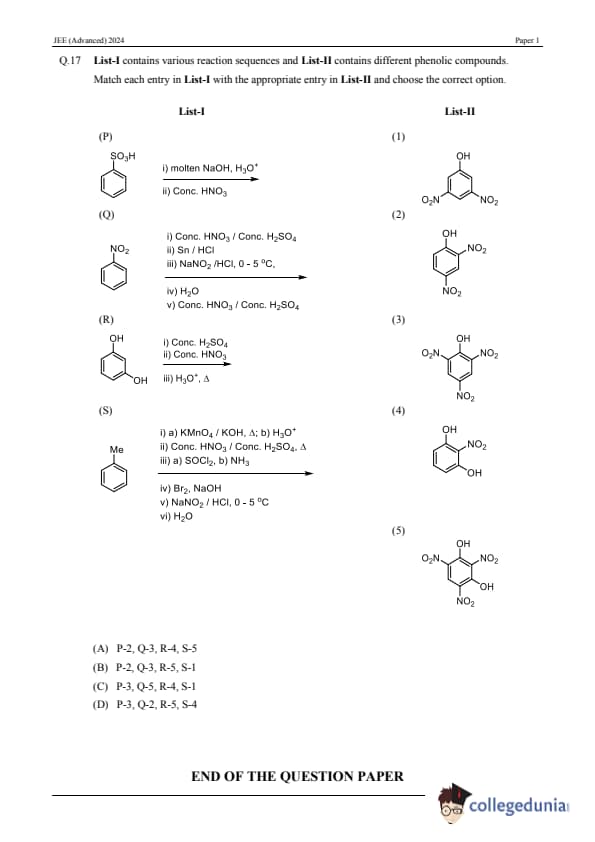
List-I contains various reaction sequences and List-II contains different phenolic compounds. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.

























| Coaching Institutes | Answer Key with Solutions PDF |
|---|---|
| Aakash BYJUs | Download PDF |
| Reliable Institute | To be updated |
| Resonance | To be updated |
| Allen | To be updated |
| Narayana | To be updated |
| Motion | Mathematics |
| Physics | |
| Chemistry |
Also Check:







Comments