JEE Advanced 2024 Paper 2 Question Paper with Answer Key and Solution PDF for May 26 is available for download. The exam was successfully conducted by the IITs from 2:30 PM to 5:30 PM. As per the student’s initial reactions, JEE Advanced 2024 Paper 2 Question Paper for May 26 was reported as Challenging. The Mathematics section in JEE Advanced 2024 May 26 Paper 2 Question Paper was reported as Tough, Physics as Moderate to Difficult, and Chemistry as Moderate.
Download JEE Advanced 2025 Question Paper with Solution PDF
JEE Advanced 2024 May 26 Paper 2 Question Paper with Answer Key PDF
Candidates can download the JEE Advanced 2024 Paper 2 Question Paper with Solution and Answer Key PDFs for May 26 using the link below.
| JEE Advanced 2024 Paper 2 Question Paper with Answer Key | Check Solution |
JEE Advanced 2024 May 26 Paper 2 Questions with Solutions
Mathematics
Question 1:
Considering only the principal values of the inverse trigonometric functions, the value of tan(sin-1(3/5) - 2 cos-1(2/√5)) is:
Question 2:
Let S = {(x, y) ∈ ℝ × ℝ : x ≥ 0, y ≥ 0, y2 ≤ 4x, y2 ≤ 12 - 2x, and 3y + √8 · x ≤ 5√8}. If the area of the region S is α√2, then α is equal to:
Question 3:
Let k ∈ ℝ. If limx→0+ (sin(sin(kx)) + cos x + x)2/x = e6, then the value of k is:
Question 4:
Let f : ℝ → ℝ be a function defined by:
f(x) = \( f(x) = \begin{cases} x^2 \sin\left( \frac{\pi}{x^2} \right), & \text{if } x \neq 0, \\ 0, & \text{if } x = 0. \end{cases} \)
Then which of the following statements is TRUE?
Question 5:
Let S be the set of all (α, β) ∈ ℝ × ℝ such that limx→∞ sin(x2) (ln x)α sin(1/x2) / (xαβ (ln(1 + x))β) = 0. Then which of the following is (are) correct?
Question 6:
A straight line drawn from the point P(1, 3, 2), parallel to the line (x-2)/1 = (y-4)/2 = (z-6)/1, intersects the plane L1: x - y + 3z = 6 at the point Q. Another straight line passing through Q and perpendicular to the plane L1 intersects the plane L2: 2x - y + z = -4 at the point R. Then which of the following statements is (are) TRUE?
Question 7:
Let A1, B1, C1 be three points in the xy-plane. Suppose that the lines A1C1 and B1C1 are tangents to the curve y2 = 8x at A1 and B1, respectively. If O = (0, 0) and C1 = (-4, 0), then which of the following statements is (are) TRUE?
Question 8:
Let f : ℝ → ℝ be a function such that f(x + y) = f(x) + f(y) for all x, y ∈ ℝ and g : ℝ → (0, ∞) be a function such that g(x + y) = g(x)g(y) for all x, y ∈ ℝ. If f(-3/5) = 12 and g(-1/3) = 2, then the value of f(1/4) + g(-2) - 8 · g(0) is .
Question 9:
A bag contains N balls out of which 3 balls are white, 6 balls are green, and the remaining balls are blue. Assume that the balls are identical otherwise. Three balls are drawn randomly one after the other without replacement. For i = 1, 2, 3, let Wi, Gi, and Bi denote the events that the ball drawn in the i-th draw is a white ball, green ball, and blue ball, respectively. If the probability P(W1 ∩ G2 ∩ B3) = 2/(5N) and the conditional probability P(B3 | W1 ∪ G2) = 2/9, then N equals .
Question 10:
Let the function \( f : \mathbb{R} \to \mathbb{R} \) be defined by
f(x) = ½ × (sin x) / (eπx) × (x2023 + 2024x + 2025) / (x² - x + 3) + (2 / eπx) × (x2023 + 2024x + 2025) / (x² - x + 3).
Then the number of solutions of f(x) = 0 in ℝ is:
Question 11:
Let \( \vec{p} = 2\hat{i} + \hat{j} + 3\hat{k} \) and \( \vec{q} = \hat{i} - \hat{j} + \hat{k} \). If for some real numbers \( \alpha \), \( \beta \), \( \gamma \), we have:
\( 15\hat{i} + 10\hat{j} + 6\hat{k} = \alpha (2\vec{p} + \vec{q}) + \beta (\vec{p} - 2\vec{q}) + \gamma (\vec{p} \times \vec{q}) \),
then the value of \( \gamma \) is:
Question 12:
A normal with slope 1/√6 is drawn from the point (0, -α) to the parabola x² = -4ay, where a > 0. Let L be the line passing through (0, -α) and parallel to the directrix of the parabola. Suppose that L intersects the parabola at two points A and B. Let r denote the length of the latus rectum and s denote the square of the length of the line segment AB. If r : s = 1 : 16, then the value of 24a is:
Question 13:
Let the function \( f : [1, \infty) \to \mathbb{R} \) be defined by
f(t) =
- \((-1)^{n+1}2\), if \( t = 2n-1 \), \( n \in \mathbb{N} \),
- \(\frac{(2n+1-t)}{2} f(2n-1) + \frac{(t-(2n-1))}{2} f(2n+1)\), if \( 2n-1 < t < 2n+1 \), \( n \in \mathbb{N} \).
Define \( g(x) = \int_1^x f(t) \, dt \), \( x \in (1, \infty) \). Let \( \alpha \) denote the number of solutions of the equation \( g(x) = 0 \) in the interval \( (1, 8] \) and \( \beta = \lim_{x \to 1^+} \frac{g(x)}{x-1} \). Then the value of \( \alpha + \beta \) is ____.
Question 14:
If \( n(X) = \binom{m}{6} \), then the value of \( m \) is ____.
Question 15:
If the value of n(Y) + n(Z) is k2, then |k| is ____.
Question 16:
The value of \( 2 \int_0^{\frac{\pi}{2}} f(x)g(x) \, dx - \int_0^{\frac{\pi}{2}} g(x) \, dx \) is:
Question 17:
The value of \( \frac{16}{\pi^3} \int_0^{\frac{\pi}{2}} f(x)g(x) \, dx \) is:
Physics
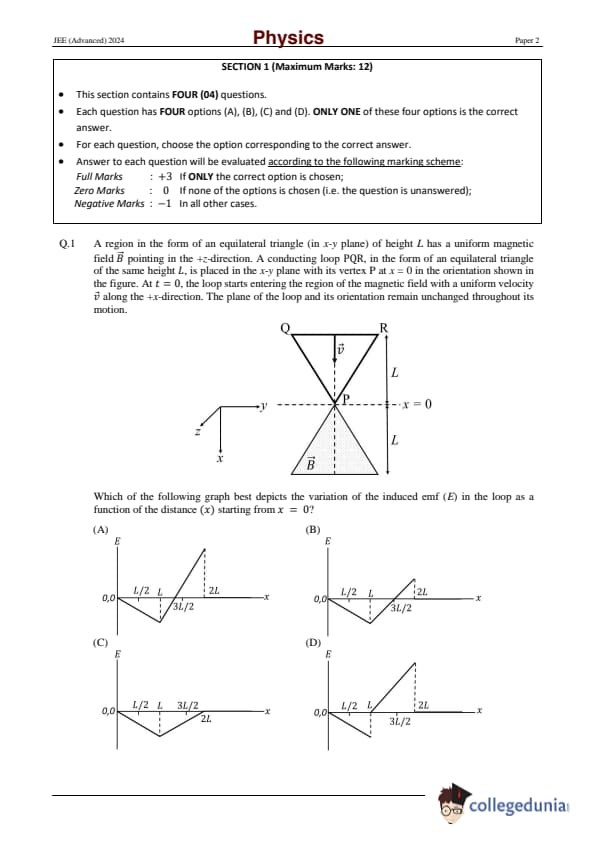
Question 1:
Consider a region in the form of an equilateral triangle (in x-y plane) of height L with a uniform magnetic field B pointing in the +z-direction. A conducting loop PQR, in the form of an equilateral triangle of the same height L, is placed in the x-y plane with its vertex P at x = 0 in the orientation shown in the figure. At t = 0, the loop starts entering the region of the magnetic field with a uniform velocity v along the +x-direction. The plane of the loop and its orientation remain unchanged throughout its motion. Which of the following graphs best depicts the variation of the induced emf (E) in the loop as a function of the distance (x) starting from x = 0?


Question 2:
A particle of mass m is under the influence of the gravitational field of a body of mass M (M >> m). The particle is moving in a circular orbit of radius r0 with time period T0 around the mass M. Then, the particle is subjected to an additional central force, corresponding to the potential energy V(r) = (αm)/r3, where α is a positive constant of suitable dimensions and r is the distance from the center of the orbit. If the particle moves in the same circular orbit of radius r0 in the combined gravitational potential due to M and Vc(r), but with a new time period T1, then
(T12 - T02)/T12 is given by:
Question 3:
A metal target with atomic number Z = 46 is bombarded with a high-energy electron beam. The emission of X-rays from the target is analyzed. The ratio r of the wavelengths of the Kα-line and the cutoff is found to be r = 2. If the same electron beam bombards another metal target with Z = 41, the value of r will be:
Question 4:
A thin stiff insulated metal wire is bent into a circular loop with its two ends extending tangentially from the same point of the loop. The wire loop has mass m and radius r and is in a uniform vertical magnetic field B0. When a current I is passed through the loop, the loop turns about the line PQ by an angle θ. The angle θ is given by:

Question 5:
A small electric dipole p0, having a moment of inertia I about its center, is kept at a distance r from the center of a spherical shell of radius R. The surface charge density σ is uniformly distributed on the spherical shell. The dipole is initially oriented at a small angle θ as shown in the figure. While staying at a distance r, the dipole is free to rotate about its center. If released from rest, then which of the following statement(s) is(are) correct?

Question 6:
A table tennis ball has radius 3/2 × 10-2 m and mass 22/7 × 10-3 kg. It is slowly pushed down into a swimming pool to a depth d = 0.7 m below the water surface and then released from rest. It emerges from the water surface at speed v, without getting wet, and rises to a height H. Which of the following option(s) is(are) correct?
(Given: π = 22/7, g = 10 m/s2, density of water = 1 × 103 kg/m3, viscosity of water = 1 × 10-3 Pa·s.)
Question 7:
A positive, singly ionized atom of mass number AM is accelerated from rest by the voltage 192 V. Thereafter, it enters a rectangular region of width w with magnetic field B0 = 0.1 T. The ion finally hits a detector at the distance x below its starting trajectory. Which of the following option(s) is(are) correct?
(Given: Mass of neutron/proton = 5/3 × 10-27 kg, charge of the electron = 1.6 × 10-19 C).


Question 8:
The dimensions of a cone are measured using a scale with a least count of 2 mm. The diameter of the base and the height are both measured to be 20.0 cm. The maximum percentage error in the determination of the volume is ____.
Question 9:
A ball is thrown from the location (x0, y0) = (0, 0) of a horizontal playground with an initial speed v0 at an angle θ0 from the +x-direction. The ball is to be hit by a stone, which is thrown at the same time from the location (x1, y1) = (L, 0). The stone is thrown at an angle (180° - θ1) from the +x-direction with a suitable initial speed. For a fixed v0, when (θ0, θ1) = (45°, 45°), the stone hits the ball after time T1, and when (θ0, θ1) = (60°, 30°), it hits the ball after time T2. In such a case, (T1 / T2)2 is ____.
Question 10:
A charge is kept at the central point P of a cylindrical region. The two edges subtend a half-angle θ at P, as shown in the figure. When θ = 30°, then the electric flux through the curved surface of the cylinder is Φ. If θ = 60°, then the electric flux through the curved surface becomes Φ / √n, where the value of n is ____.

Question 11:
Two equilateral-triangular prisms P1 and P2 are kept with their sides parallel to each other, in vacuum, as shown in the figure. A light ray enters prism P1 at an angle of incidence θ such that the outgoing ray undergoes minimum deviation in prism P2. If the respective refractive indices of P1 and P2 are (√3/√2) and √3, then θ = sin-1(√3/√2 sin(π/β)), where the value of β is _____.

Question 12:
An infinitely long thin wire, having a uniform charge density per unit length of 5 nC/m, is passing through a spherical shell of radius 1 m, as shown in the figure. A 10 nC charge is distributed uniformly over the spherical shell. If the configuration of the charges remains static, the magnitude of the potential difference between points P and R, in Volt, is _____.
(Given: In SI units 1/4πε0 = 9 × 109, ln 2 = 0.7). Ignore the area pierced by the wire.

Question 13:
A spherical soap bubble inside an air chamber at pressure P0 = 105 Pa has a certain radius so that the excess pressure inside the bubble is ΔP = 144 Pa. Now, the chamber pressure is reduced to 8P0/27 so that the bubble radius and its excess pressure change. In this process, all the temperatures remain unchanged. Assume air to be an ideal gas and the excess pressure ΔP in both the cases to be much smaller than the chamber pressure. The new excess pressure ΔP in Pa is _____.
Question 14:
The 8th bright fringe above the point O oscillates with time between two extreme positions. The separation between these two extreme positions, in micrometer (μm), is _____.
Question 15:
The maximum speed in μm/s at which the 8th bright fringe will move is _____.
Question 16:
If the collision occurs at time t0 = 0, the value of vcm / (aω) will be _____.
Question 17:
If the collision occurs at time t0 = π/(2ω), then the value of 4b2/a2 will be _____.
Chemistry
Question 1:
According to Bohr's model, the highest kinetic energy is associated with the electron in the:
Question 2:
In a metal-deficient oxide sample, MxY2O4 (M and Y are metals), M is present in both +2 and +3 oxidation states and Y is in +3 oxidation state. If the fraction of M2+ ions present in M is 1/3, the value of x is _____.
Question 3:
In the following reaction sequence, the major product Q is:


Question 4:
The species formed on fluorination of phosphorus pentachloride in a polar organic solvent are:
Question 5:
An aqueous solution of hydrazine (N2H4) is electrochemically oxidized by O2, thereby releasing chemical energy in the form of electrical energy. One of the products generated from the electrochemical reaction is N2(g). Choose the correct statement(s) about the above process:
Question 6:
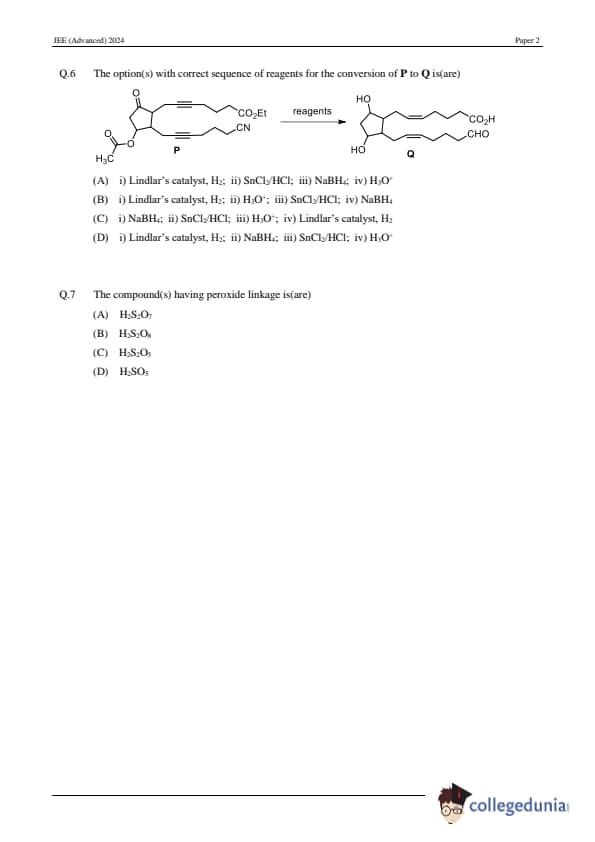
The option(s) with the correct sequence of reagents for the conversion of P to Q is (are):
Question 7:
The compound(s) having peroxide linkage is (are):
Question 8:
To form a complete monolayer of acetic acid on 1 g of charcoal, 100 mL of 0.5 M acetic acid was used. Some of the acetic acid remained unadsorbed. To neutralize the unadsorbed acetic acid, 40 mL of 1 M NaOH solution was required. If each molecule of acetic acid occupies P × 10-23 m2 surface area on charcoal, the value of P is _____.
[Use given data: Surface area of charcoal = 1.5 × 102 m2g-1; Avogadro's number (NA) = 6.0 × 1023 mol-1]
Question 9:
Vessel-1 contains w2 g of a non-volatile solute X dissolved in w1 g of water. Vessel-2 contains w2 g of another non-volatile solute Y dissolved in w1 g of water. Both the vessels are at the same temperature and pressure. The molar mass of X is 80% of that of Y. The van’t Hoff factor for X is 1.2 times that of Y for their respective concentrations. The elevation of boiling point for solution in Vessel-1 is _____ % of the solution in Vessel-2.
Question 10:
For a double-strand DNA, one strand is given below:


The amount of energy required to split the double-strand DNA into two single strands is _____ kcal mol-1. [Given: Average energy per H-bond for A-T base pair = 1 kcal mol-1, G-C base pair = 1.5 kcal mol-1, and A-U base pair = 1.25 kcal mol-1. Ignore electrostatic repulsion between the phosphate groups]
Question 11:
A sample initially contains only U-238 isotope of uranium. With time, some of the U-238 radioactively decays into Pb-206 while the rest remains undisintegrated. When the age of the sample is P × 108 years, the ratio of the mass of Pb-206 to that of U-238 in the sample is found to be 7. The value of P is:
[Given: Half-life of U-238 = 4.5 × 109 years; loge2 = 0.693]
Question 12:
Among [Co(CN)4]4-, [Co(CO)3(NO)], XeF4, [PCl4]+, [PdCl4]2-, [ICl4]-, [Cu(CN)4]3-, and P4, the total number of species with tetrahedral geometry is:
Question 13:
An organic compound P with molecular formula C6H6O3 gives a ferric chloride test and does not have an intramolecular hydrogen bond. The compound P reacts with 3 equivalents of NH2OH to produce oxime Q. Treatment of P with excess methyl iodide in the presence of KOH produces compound R as the major product. Reaction of R with excess iso-butylmagnesium bromide followed by treatment with H3O+ gives compound S as the major product. The total number of methyl (-CH3) groups in compound S is _____.
Question 14:
Sum of the number of oxygen atoms in S and T is _____.
Question 15:
The molecular weight of U is:
Questions 16 and 17:
When potassium iodide is added to an aqueous solution of potassium ferricyanide, a reversible reaction is observed in which a complex P is formed. In a strong acidic medium, the equilibrium shifts completely towards P. Addition of zinc chloride to P in a slightly acidic medium results in a sparingly soluble complex Q.
Question 16:
The number of moles of potassium iodide required to produce two moles of P is:
Question 17:
The number of zinc ions present in the molecular formula of Q is:
























JEE Advanced 2024 Paper 2 Answer Key: Coaching Institute PDF
| Coaching Institutes | Answer Key with Solutions PDF |
|---|---|
| Aakash BYJUs | Download PDF |
| Reliable Institute | To be updated |
| Resonance | To be updated |
| Allen | To be updated |
| Narayana | To be updated |
| Motion | Mathematics |
| Physics | |
| Chemistry |
Also Check:









Comments