IIT Kanpur conducted JEE Advanced 2025 Paper 1(English) on May 18, 2025, from 9:00 AM to 12:00 PM in 3 hours.
The JEE Advanced 2025 Paper was held in a CBT Mode and consisted of 54 questions, divided equally among Physics, Chemistry, and Mathematics (18 questions from each subject).
The IIT JEE Advanced 2025 Question Paper is available here for download and students can analyse JEE Advanced Result through JEE Advanced response sheet 2025.
The JEE Advanced 2025 Paper 1 Question Paper contains multiple-choice questions, numerical value answer-type questions, and matching-type questions, testing candidates' problem-solving skills and conceptual understanding.
Jee Advanced 2025 Paper 1 Solutions PDF (English)
| JEE Advanced 2025 Paper 1 Question Paper with Answer Key | Download | Check Solution |

Let \( \mathbb{R} \) denote the set of all real numbers. Let \( a_i, b_i \in \mathbb{R} \) for \( i \in \{1, 2, 3\} \).
Define the functions \( f: \mathbb{R} \to \mathbb{R},\ g: \mathbb{R} \to \mathbb{R},\ h: \mathbb{R} \to \mathbb{R} \) by:
\[ f(x) = a_1 + 10x + a_2x^2 + a_3x^3 + x^4,\quad g(x) = b_1 + 3x + b_2x^2 + b_3x^3 + x^4, \] \[ h(x) = f(x+1) - g(x+2) \]
If \( f(x) \ne g(x) \) for every \( x \in \mathbb{R} \), then the coefficient of \( x^3 \) in \( h(x) \) is:
View Solution
Three students \( S_1, S_2, \) and \( S_3 \) are given a problem to solve. Consider the following events:
\( U \): At least one of \( S_1, S_2, S_3 \) can solve the problem,
\( V \): \( S_1 \) can solve the problem, given that neither \( S_2 \) nor \( S_3 \) can solve the problem,
\( W \): \( S_2 \) can solve the problem and \( S_3 \) cannot solve the problem,
\( T \): \( S_3 \) can solve the problem.
For any event \( E \), let \( P(E) \) denote the probability of \( E \). If \[ P(U) = \frac{1}{2}, \quad P(V) = \frac{1}{10}, \quad and \quad P(W) = \frac{1}{12}, \]
then \( P(T) \) is equal to:
View Solution
Let \( \mathbb{R} \) denote the set of all real numbers. Define the function \( f: \mathbb{R} \to \mathbb{R} \) by \[ f(x) = \begin{cases} 2 - 2x^2 - x^2 \sin\left(\frac{1}{x}\right), & if x \ne 0,
2, & if x = 0. \end{cases} \]
Then which one of the following statements is TRUE?
View Solution
Consider the matrix \[ P = \begin{pmatrix} 2 & 0 & 0
0 & 2 & 0
0 & 0 & 3 \end{pmatrix}. \]
Let the transpose of a matrix \( X \) be denoted by \( X^T \). Then the number of \( 3 \times 3 \) invertible matrices \( Q \) with integer entries, such that \[ Q^{-1} = Q^T \quad and \quad PQ = QP, \]
is:
View Solution
Let \( L_1 \) be the line of intersection of the planes given by the equations \[ 2x + 3y + z = 4 \quad and \quad x + 2y + z = 5. \]
Let \( L_2 \) be the line passing through the point \( P(2, -1, 3) \) and parallel to \( L_1 \). Let \( M \) denote the plane given by the equation \[ 2x + y - 2z = 6. \]
Suppose that the line \( L_2 \) meets the plane \( M \) at the point \( Q \). Let \( R \) be the foot of the perpendicular drawn from \( P \) to the plane \( M \).
Then which of the following statements is (are) TRUE?
View Solution
Let \( \mathbb{N} \) denote the set of all natural numbers, and \( \mathbb{Z} \) denote the set of all integers. Consider the functions \( f : \mathbb{N} \to \mathbb{Z} \) and \( g : \mathbb{Z} \to \mathbb{N} \) defined by \[ f(n) = \begin{cases} \frac{n + 1}{2}, & if n is odd
\frac{4 - n}{2}, & if n is even \end{cases} \quad and \quad g(n) = \begin{cases} 3 + 2n, & if n \ge 0
-2n, & if n < 0 \end{cases} \]
Define \( (g \circ f)(n) = g(f(n)) \) for all \( n \in \mathbb{N} \), and \( (f \circ g)(n) = f(g(n)) \) for all \( n \in \mathbb{Z} \).
Then which of the following statements is (are) TRUE?
View Solution
Let \( \mathbb{R} \) denote the set of all real numbers. Let \( z_1 = 1 + 2i \) and \( z_2 = 3i \) be two complex numbers, where \( i = \sqrt{-1} \). Let \[ S = \{(x, y) \in \mathbb{R} \times \mathbb{R} : |x + iy - z_1| = 2|x + iy - z_2|\}. \]
Then which of the following statements is (are) TRUE?
View Solution
Let the set of all relations \( R \) on the set \( \{a, b, c, d, e, f\} \), such that \( R \) is reflexive and symmetric, and \( R \) contains exactly 10 elements, be denoted by \( S \).
Then the number of elements in \( S \) is \underline{\hspace{2cm.
View Solution
For any two points \( M \) and \( N \) in the \( XY \)-plane, let \( \overrightarrow{MN} \) denote the vector from \( M \) to \( N \), and \( \vec{0} \) denote the zero vector. Let \( P, Q \), and \( R \) be three distinct points in the \( XY \)-plane. Let \( S \) be a point inside the triangle \( \Delta PQR \) such that \[ \overrightarrow{SP} + 5\overrightarrow{SQ} + 6\overrightarrow{SR} = \vec{0}. \]
Let \( E \) and \( F \) be the mid-points of the sides \( PR \) and \( QR \), respectively. Then the value of \[ \frac{length of the line segment EF}{length of the line segment ES} \]
is __________.
View Solution
Let \( S \) be the set of all seven-digit numbers that can be formed using the digits 0, 1 and 2. For example, 2210222 is in \( S \), but 0210222 is NOT in \( S \).
Then the number of elements \( x \) in \( S \) such that at least one of the digits 0 and 1 appears exactly twice in \( x \), is equal to __________.
% No options since it's a numerical answer
View Solution
Let \( \alpha \) and \( \beta \) be the real numbers such that \[ \lim_{x \to 0} \frac{1}{x^3} \left( \frac{\alpha}{2} \int_{0}^{x} \frac{1}{1 - t^2} \, dt + \beta x \cos x \right) = 2. \]
Then the value of \( \alpha + \beta \) is __________.
% Numerical Answer
View Solution
Let \( \mathbb{R} \) denote the set of all real numbers. Let \( f: \mathbb{R} \to \mathbb{R} \) be a function such that \( f(x) > 0 \) for all \( x \in \mathbb{R} \), and \( f(x + y) = f(x)f(y) \) for all \( x, y \in \mathbb{R} \).
Let the real numbers \( a_1, a_2, \ldots, a_{50} \) be in an arithmetic progression. If \( f(a_{31}) = 64f(a_{25}) \), and \[ \sum_{i=1}^{50} f(a_i) = 3(2^{25} + 1), \]
then the value of \[ \sum_{i=6}^{30} f(a_i) \]
is __________.
View Solution
For all \( x > 0 \), let \( y_1(x), y_2(x), y_3(x) \) be the functions satisfying \[ \frac{dy_1}{dx} - (\sin x)^2 y_1 = 0, \quad y_1(1) = 5, \] \[ \frac{dy_2}{dx} - (\cos x)^2 y_2 = 0, \quad y_2(1) = \frac{1}{3}, \] \[ \frac{dy_3}{dx} - \left(\frac{2 - x^3}{x^3}\right) y_3 = 0, \quad y_3(1) = \frac{3}{5e}, \]
respectively. Then \[ \lim_{x \to 0^+} \frac{y_1(x)y_2(x)y_3(x) + 2x}{e^{3x} \sin x} \]
is equal to __________.
View Solution
Consider the following frequency distribution:
\begin{tabular{|c|c c c c c c c c|
\hline
Value & 4 & 5 & 8 & 9 & 6 & 12 & 11
Frequency & 5 & \(f_1\) & \(f_2\) & 2 & 1 & 1 & 3
\hline
\end{tabular
Suppose that the sum of the frequencies is 19 and the median of this frequency distribution is 6.
For the given frequency distribution, let \( \alpha \) denote the mean deviation about the mean, \( \beta \) denote the mean deviation about the median, and \( \sigma^2 \) denote the variance.
Match each entry in List-I to the correct entry in List-II and choose the correct option.
\begin{multicols{2
List-I
[(P)] \( 7f_1 + 9f_2 \) is equal to
[(Q)] \( 19\alpha \) is equal to
[(R)] \( 19\beta \) is equal to
[(S)] \( 19\sigma^2 \) is equal to
\columnbreak
List-II
[(1)] 146
[(2)] 47
[(3)] 48
[(4)] 145
[(5)] 55
\end{multicols
Options:
View Solution
Let \( \mathbb{R} \) denote the set of all real numbers. For a real number \( x \), let \( \lfloor x \rfloor \) denote the greatest integer less than or equal to \( x \). Let \( n \) denote a natural number.
Match each entry in List-I to the correct entry in List-II and choose the correct option.
\begin{multicols{2
List-I
[(P)] The minimum value of \( n \) for which the function
\[ f(x) = \left\lfloor \frac{10x^3 - 45x^2 + 60x + 35}{n} \right\rfloor \]
is continuous on the interval \( [1, 2] \), is
[(Q)] The minimum value of \( n \) for which
\[ g(x) = (2n^2 - 13n - 15)(x^3 + 3x), \]
\( x \in \mathbb{R} \), is an increasing function on \( \mathbb{R} \), is
[(R)] The smallest natural number \( n \) which is greater than 5, such that \( x = 3 \) is a point of local minima of
\[ h(x) = (x^2 - 9)^n (x^2 + 2x + 3), \]
is
[(S)] Number of \( x_0 \in \mathbb{R} \) such that
\[ l(x) = \sum_{k=0}^{4} \left( \sin |x - k| + \cos \left| x - k + \frac{1}{2} \right| \right) \]
is not differentiable at \( x_0 \), is
\columnbreak
List-II
[(1)] 8
[(2)] 9
[(3)] 5
[(4)] 6
[(5)] 10
\end{multicols
Options:
View Solution
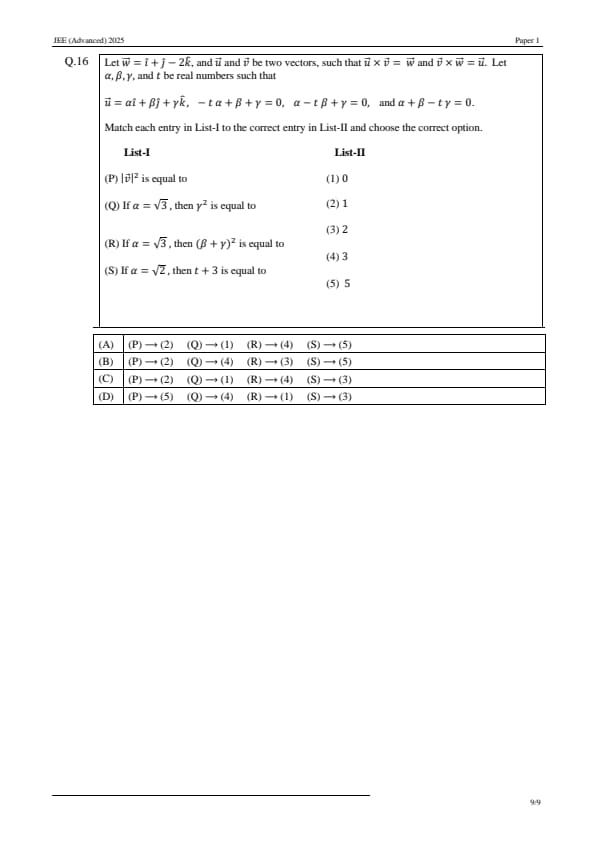
Let \( \vec{w} = \hat{i} + \hat{j} - 2\hat{k} \), and \( \vec{u} \) and \( \vec{v} \) be two vectors, such that \( \vec{u} \times \vec{v} = \vec{w} \) and \( \vec{v} \times \vec{w} = \vec{u} \). Let \( \alpha, \beta, \gamma \), and \( t \) be real numbers such that: \[ \vec{u} = \alpha \hat{i} + \beta \hat{j} + \gamma \hat{k}, \]
and the system of equations is: \[ -t\alpha + \beta + \gamma = 0 \quad \cdots (1) \] \[ \alpha - t\beta + \gamma = 0 \quad \cdots (2) \] \[ \alpha + \beta - t\gamma = 0 \quad \cdots (3) \]
Match each entry in List-I to the correct entry in List-II and choose the correct option.
\begin{multicols{2
List-I
[(P)] \( |\vec{v}|^2 \) is equal to
[(Q)] If \( \alpha = \sqrt{3} \), then \( \gamma^2 \) is equal to
[(R)] If \( \alpha = \sqrt{3} \), then \( (\beta + \gamma)^2 \) is equal to
[(S)] If \( \alpha = \sqrt{2} \), then \( t + 3 \) is equal to
\columnbreak
List-II
[(1)] 0
[(2)] 1
[(3)] 2
[(4)] 3
[(5)] 5
\end{multicols
View Solution
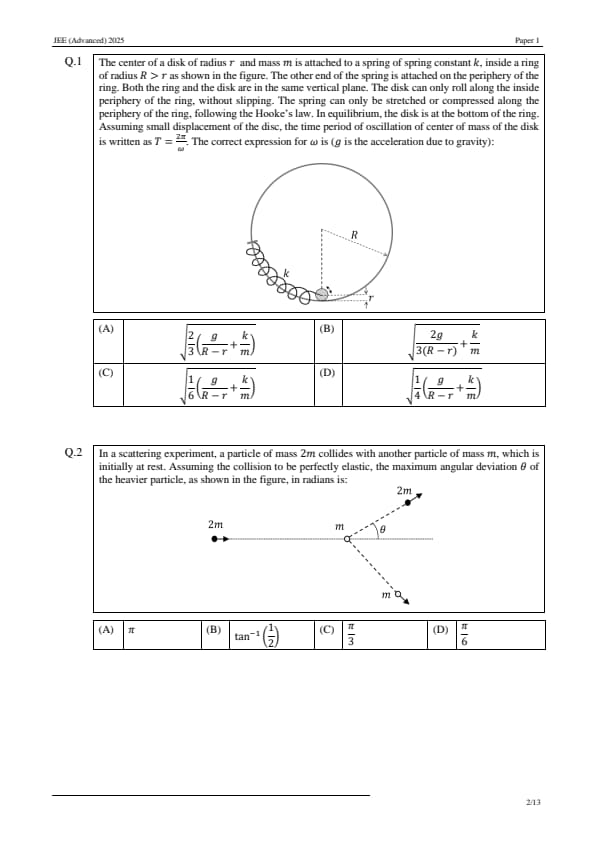
The center of a disk of radius \( r \) and mass \( m \) is attached to a spring of spring constant \( k \), inside a ring of radius \( R > r \) as shown in the figure. The other end of the spring is attached on the periphery of the ring. Both the ring and the disk are in the same vertical plane. The disk can only roll along the inside periphery of the ring, without slipping. The spring can only be stretched or compressed along the periphery of the ring, following Hooke’s law. In equilibrium, the disk is at the bottom of the ring. Assuming small displacement of the disc, the time period of oscillation of center of mass of the disk is written as \( T = \frac{2\pi}{\omega} \). The correct expression for \( \omega \) is ( \( g \) is the acceleration due to gravity):
View Solution
In a scattering experiment, a particle of mass \( 2m \) collides with another particle of mass \( m \), which is initially at rest. Assuming the collision to be perfectly elastic, the maximum angular deviation \( \theta \) of the heavier particle, as shown in the figure, in radians is:
View Solution
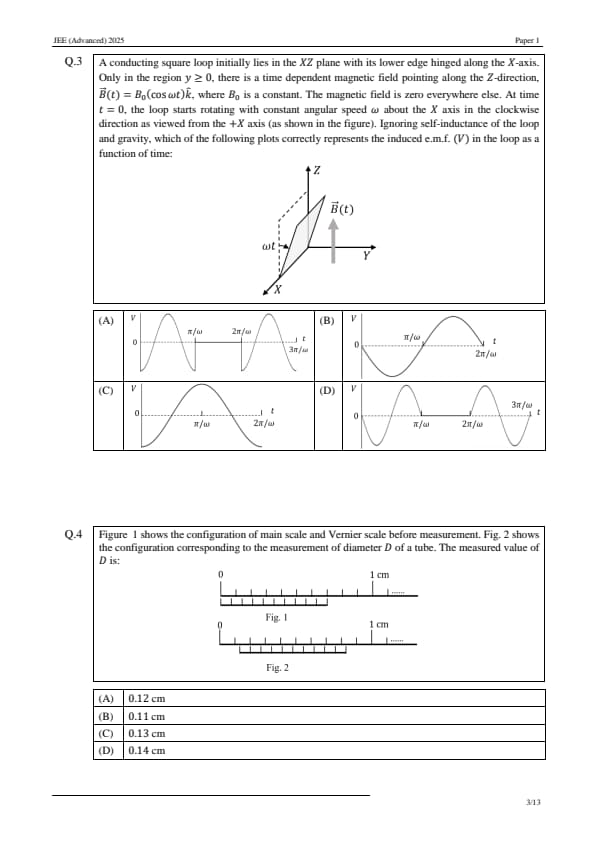
A conducting square loop initially lies in the \( XZ \) plane with its lower edge hinged along the \( X \)-axis. Only in the region \( y \geq 0 \), there is a time dependent magnetic field pointing along the \( Z \)-direction, \( \vec{B}(t) = B_0 (\cos \omega t) \hat{k} \), where \( B_0 \) is a constant. The magnetic field is zero everywhere else. At time \( t = 0 \), the loop starts rotating with constant angular speed \( \omega \) about the \( X \) axis in the clockwise direction as viewed from the \( +X \) axis (as shown in the figure). Ignoring self-inductance of the loop and gravity, which of the following plots correctly represents the induced e.m.f. (\( V \)) in the loop as a function of time:
Figure 1 shows the configuration of main scale and Vernier scale before measurement. Fig. 2 shows the configuration corresponding to the measurement of diameter \( D \) of a tube. The measured value of \( D \) is:
View Solution
A conducting square loop of side \( L \), mass \( M \), and resistance \( R \) is moving in the \( XY \) plane with its edges parallel to the \( X \) and \( Y \) axes. The region \( y \geq 0 \) has a uniform magnetic field, \( \vec{B} = B_0 \hat{k} \). The magnetic field is zero everywhere else. At time \( t = 0 \), the loop starts to enter the magnetic field with an initial velocity \( v_0 \hat{j} \, m/s \), as shown in the figure. Considering the quantity \( K = \frac{B_0^2 L^2}{RM} \) in appropriate units, ignoring self-inductance of the loop and gravity, which of the following statements is/are correct:
View Solution
Length, breadth and thickness of a strip having a uniform cross section are measured to be \( 10.5 \, cm, 0.05 \, mm \), and \( 6.0 \, \mum \), respectively. Which of the following option(s) give(s) the volume of the strip in \( cm^3 \) with correct significant figures:
View Solution
Consider a system of three connected strings, \( S_1, S_2 \) and \( S_3 \) with uniform linear mass densities \( \mu \, kg/m, 4\mu \, kg/m \) and \( 16\mu \, kg/m \), respectively, as shown in the figure. \( S_1 \) and \( S_2 \) are connected at point \( P \), whereas \( S_2 \) and \( S_3 \) are connected at the point \( Q \), and the other end of \( S_3 \) is connected to a wall. A wave generator \( O \) is connected to the free end of \( S_1 \). The wave from the generator is represented by \( y = y_0 \cos(\omega t - kx) \) cm, where \( y_0, \omega \) and \( k \) are constants of appropriate dimensions. Which of the following statements is/are correct:
View Solution
A person sitting inside an elevator performs a weighing experiment with an object of mass \( 50 \, kg \). Suppose that the variation of the height \( y \) (in m) of the elevator, from the ground, with time \( t \) (in s) is given by \[ y = 8\left[1 + \sin\left(\frac{2\pi t}{T}\right)\right], \]
where \( T = 40\pi \, s \). Taking acceleration due to gravity, \( g = 10 \, m/s^2 \), the maximum variation of the object's weight (in N) as observed in the experiment is ____.
View Solution
A cube of unit volume contains \( 35 \times 10^7 \) photons of frequency \( 10^{15} \, Hz \). If the energy of all the photons is viewed as the average energy being contained in the electromagnetic waves within the same volume, then the amplitude of the magnetic field is \( \alpha \times 10^{-9} \, T \). Taking permeability of free space \( \mu_0 = 4\pi \times 10^{-7} \, Tm/A \), Planck’s constant \( h = 6 \times 10^{-34} \, Js \) and \( \pi = \frac{22}{7} \), the value of \( \alpha \) is ____
View Solution
Two identical plates \( P \) and \( Q \), radiating as perfect black bodies, are kept in vacuum at constant absolute temperatures \( T_P \) and \( T_Q \), respectively, with \( T_Q < T_P \), as shown in Fig. 1. The radiated power transferred per unit area from \( P \) to \( Q \) is \( W_0 \). Subsequently, two more plates, identical to \( P \) and \( Q \), are introduced between \( P \) and \( Q \), as shown in Fig. 2. Assume that heat transfer takes place only between adjacent plates. If the power transferred per unit area in the direction from \( P \) to \( Q \) (Fig. 2) in the steady state is \( W_S \), then the ratio \( \dfrac{W_0}{W_S} \) is ____.
View Solution
A solid glass sphere of refractive index \( n = \sqrt{3} \) and radius \( R \) contains a spherical air cavity of radius \( \dfrac{R}{2} \), as shown in the figure. A very thin glass layer is present at the point \( O \) so that the air cavity (refractive index \( n = 1 \)) remains inside the glass sphere. An unpolarized, unidirectional and monochromatic light source \( S \) emits a light ray from a point inside the glass sphere towards the periphery of the glass sphere. If the light is reflected from the point \( O \) and is fully polarized, then the angle of incidence at the inner surface of the glass sphere is \( \theta \). The value of \( \sin \theta \) is ____
View Solution
A single slit diffraction experiment is performed to determine the slit width using the equation, \( \dfrac{b d}{D} = m \lambda \), where \( b \) is the slit width, \( D \) the shortest distance between the slit and the screen, \( d \) the distance between the \( m^{th} \) diffraction maximum and the central maximum, and \( \lambda \) is the wavelength. \( D \) and \( d \) are measured with scales of least count of 1 cm and 1 mm, respectively. The values of \( \lambda \) and \( m \) are known precisely to be \( 600 \, nm \) and 3, respectively. The absolute error (in \( \mum \)) in the value of \( b \) estimated using the diffraction maximum that occurs for \( m = 3 \) with \( d = 5 \, mm \) and \( D = 1 \, m \) is ___.
View Solution
Consider an electron in the \( n = 3 \) orbit of a hydrogen-like atom with atomic number \( Z \). At absolute temperature \( T \), a neutron having thermal energy \( k_B T \) has the same de Broglie wavelength as that of this electron. If this temperature is given by \[ T = \frac{Z^2 h^2}{\alpha \pi^2 a_0^2 m_N k_B} \]
(where \( h \) is Planck’s constant, \( k_B \) is Boltzmann’s constant, \( m_N \) is the mass of the neutron, and \( a_0 \) is the Bohr radius), then the value of \( \alpha \) is ___.
View Solution
List-I shows four configurations, each consisting of a pair of ideal electric dipoles. Each dipole has a dipole moment of magnitude \( p \), oriented as marked by arrows in the figures. In all the configurations the dipoles are fixed such that they are at a distance \( 2r \) apart along the \( x \)-direction. The midpoint of the line joining the two dipoles is \( X \). The possible resultant electric fields \( \vec{E} \) at \( X \) are given in List-II.
Choose the option that describes the correct match between the entries in List-I to those in List-II.
View Solution
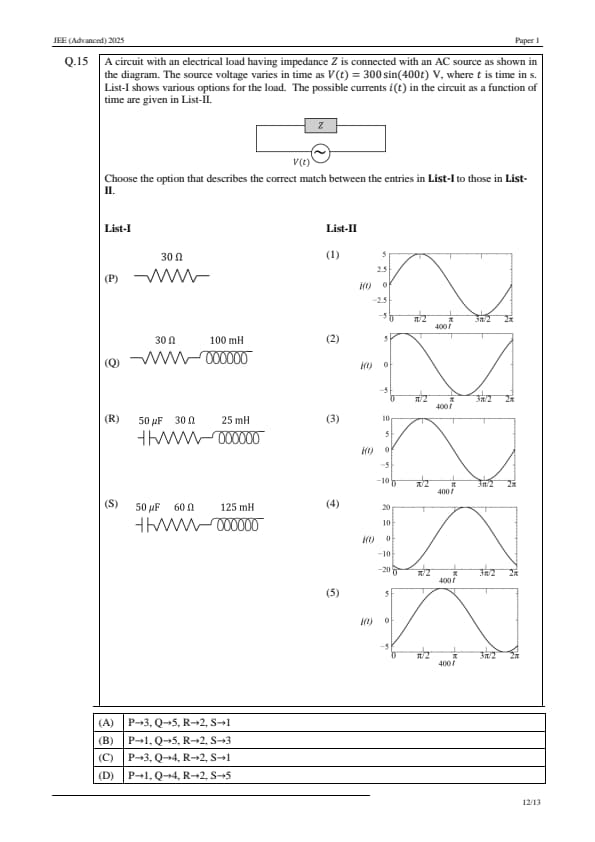
A circuit with an electrical load having impedance \( Z \) is connected with an AC source as shown in the diagram. The source voltage varies in time as \( V(t) = 300 \sin(400t) \, V \), where \( t \) is time in seconds. List-I shows various options for the load. The possible currents \( i(t) \) in the circuit as a function of time are given in List-II.
Choose the option that describes the correct match between the entries in List-I to those in List-II.
View Solution
List-I shows various functional dependencies of energy \( E \) on the atomic number \( Z \). Energies associated with certain phenomena are given in List-II.
Choose the option that describes the correct match between the entries in List-I to those in List-II.
View Solution
The heating of \( NH_4NO_2 \) at 60--70\(^\circ\)C and \( NH_4NO_3 \) at 200--250\(^\circ\)C is associated with the formation of nitrogen containing compounds X and Y, respectively. X and Y, respectively, are:
View Solution
The correct order of the wavelength maxima of the absorption band in the ultraviolet-visible region for the given complexes is:
View Solution
One of the products formed from the reaction of permanganate ion with iodide ion in neutral aqueous medium is:
View Solution
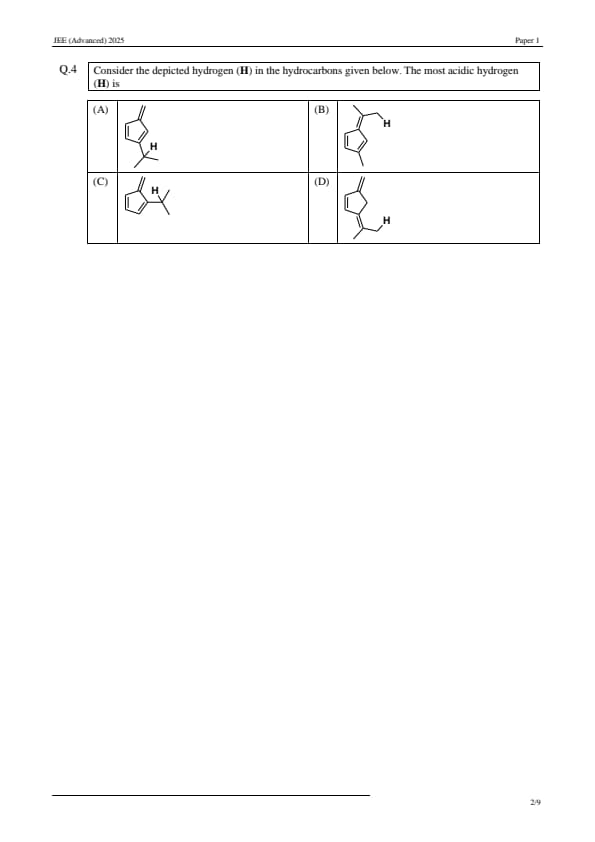
Consider the depicted hydrogen (H) in the hydrocarbons given below. The most acidic hydrogen (H) is:
View Solution
Regarding the molecular orbital (MO) energy levels for homonuclear diatomic molecules, the \textbf{INCORRECT} statement(s) is (are):
View Solution
The pair(s) of diamagnetic ions is (are):
View Solution
For the reaction sequence given below, the correct statement(s) is (are):
(In the options, X is any atom other than carbon and hydrogen, and it is different in P, Q, and R.)
View Solution
In an electrochemical cell, dichromate ions in aqueous acidic medium are reduced to \( Cr^{3+} \). The current (in amperes) that flows through the cell for 48.25 minutes to produce 1 mole of \( Cr^{3+} \) is ____.
\textbf{Use:} 1 Faraday \( = 96500 \, C mol^{-1} \)
View Solution
At 25\(^\circ\)C, the concentration of H\(^+\) ions in \( 1.00 \times 10^{-3} \, M \) aqueous solution of a weak monobasic acid having acid dissociation constant \( K_a = 4.00 \times 10^{-11} \) is \( X \times 10^{-7} \, M \). The value of \( X \) is ____.
\textbf{Use:} Ionic product of water \( K_w = 1.00 \times 10^{-14} \) at 25\(^\circ\)C
View Solution
Molar volume (\( V_m \)) of a van der Waals gas can be calculated by expressing the van der Waals equation as a cubic equation with \( V_m \) as the variable. The ratio (in mol dm\(^{-3}\)) of the coefficient of \( V_m^2 \) to the coefficient of \( V_m \) for a gas having van der Waals constants \( a = 6.0 \, dm^6 \, atm mol^{-2} \) and \( b = 0.060 \, dm^3 \, mol^{-1} \) at 300 K and 300 atm is ____.
Use: Universal gas constant \( R = 0.082 \, dm^3 \, atm mol^{-1} \, K^{-1} \)
View Solution
Considering ideal gas behavior, the expansion work done (in kJ) when 144 g of water is electrolyzed completely under constant pressure at 300 K is ____.
\textbf{Use:} Universal gas constant \( R = 8.3 \, J K^{-1} mol^{-1} \); Atomic mass (in amu): H = 1, O = 16
View Solution
The monomer (X) involved in the synthesis of Nylon 6,6 gives positive carbylamine test. If 10 moles of X are analyzed using Dumas method, the amount (in grams) of nitrogen gas evolved is ____.
\textbf{Use:} Atomic mass of N (in amu) = 14
View Solution
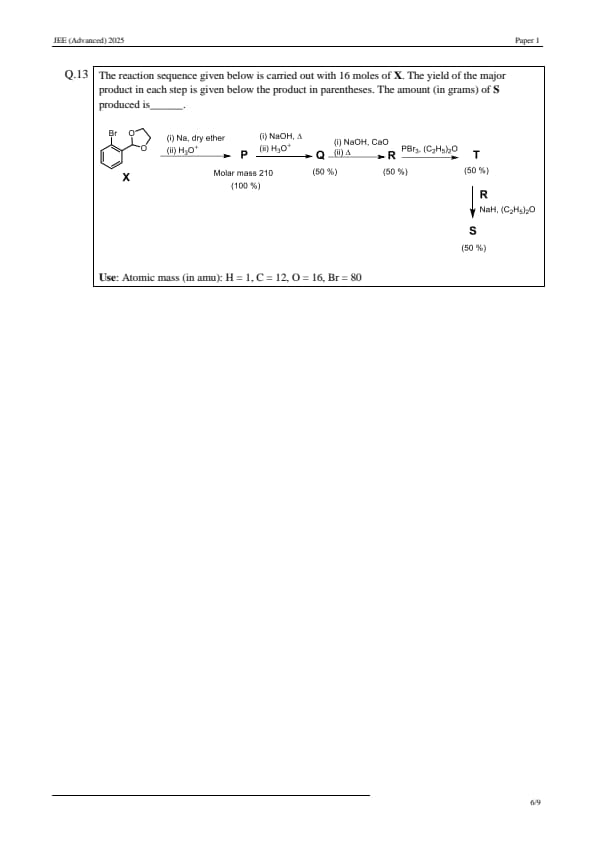
The reaction sequence given below is carried out with 16 moles of X. The yield of the major product in each step is given below the product in parentheses. The amount (in grams) of S produced is ____.
\textbf{Use:} Atomic mass (in amu): H = 1, C = 12, O = 16, Br = 80
View Solution
The correct match of the group reagents in List-I for precipitating the metal ion given in List-II from solutions is:

View Solution
The major products obtained from the reactions in List-II are the reactants for the named reactions mentioned in List-I. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
View Solution
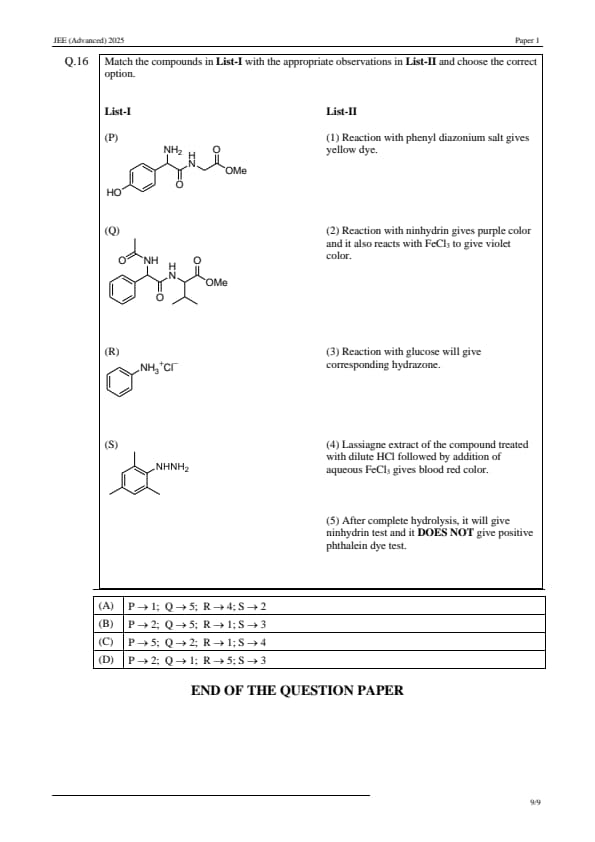
Match the compounds in List-I with the appropriate observations in List-II and choose the correct option.
View Solution
































































JEE Advanced 2025 Chapter-Wise Weightage
The JEE Advanced 2025 chapter-wise weightage is based on the past year trends and JEE Advanced question papers.
The JEE Advanced 2025 Question Paper is expected to follow a pattern similar to previous years, with in-depth conceptual knowledge and analytical reasoning.
Physics Chapter-Wise Weightage
| Chapter | Weightage (%) |
|---|---|
| Mechanics (including Rotational Motion, Laws of Motion, etc.) | 25–30% |
| Electrostatics & Capacitance | 7–9% |
| Current Electricity | 6–8% |
| Magnetism & Moving Charges | 5–7% |
| Electromagnetic Induction & AC | 5–6% |
| Heat and Thermodynamics | 6–8% |
| Kinetic Theory of Gases | 2–3% |
| Waves and Sound | 4–5% |
| Optics (Ray + Wave) | 8–10% |
| Modern Physics (Atoms, Nuclei, Semiconductors) | 6–8% |
| Units & Dimensions, Error Analysis | 2–3% |
Chemistry Chapter-Wise Weightage
| Chapter | Weightage (%) |
|---|---|
| Mole Concept & Stoichiometry | 4–5% |
| Atomic Structure & Periodic Table | 4–6% |
| Chemical Bonding & Molecular Structure | 6–8% |
| States of Matter | 3–5% |
| Thermodynamics & Thermochemistry | 5–7% |
| Equilibrium (Chemical + Ionic) | 5–7% |
| Redox Reactions & Electrochemistry | 5–6% |
| Chemical Kinetics | 3–4% |
| Coordination Compounds | 6–8% |
| The p-block, d- and f-block Elements | 5–7% |
| Hydrocarbons & General Organic Chemistry | 6–8% |
| Alcohols, Phenols & Ethers | 4–5% |
| Aldehydes, Ketones & Carboxylic Acids | 4–6% |
| Haloalkanes & Haloarenes | 3–5% |
| Amines & Biomolecules | 3–5% |
| Environmental Chemistry & Chemistry in Everyday Life | 1–2% |
Mathematics Chapter-Wise Weightage
| Chapter | Weightage (%) |
|---|---|
| Sets, Relations, Functions | 3–4% |
| Complex Numbers & Quadratic Equations | 4–5% |
| Permutations & Combinations | 3–4% |
| Binomial Theorem & Sequences | 3–4% |
| Matrices and Determinants | 5–6% |
| Probability | 4–5% |
| Trigonometry | 5–6% |
| Limits, Continuity, and Differentiability | 6–8% |
| Application of Derivatives | 5–6% |
| Integrals & Area under Curve | 7–9% |
| Differential Equations | 4–5% |
| Coordinate Geometry (Straight Line, Circle, Parabola, etc.) | 10–12% |
| Vectors and 3D Geometry | 6–8% |
JEE Advanced 2025 Important Topics
The JEE Advanced Question Paper 2025 will examine students' conceptual knowledge in Physics, Chemistry, and Mathematics.
Preparing these Important topics in the last minute can improve a candidate's score.
Subject-Wise Important Topics for JEE Advanced
| Subject | Important Topics |
| Physics |
|
| Chemistry |
|
| Mathematics |
|













Comments