
Education Journalist | Study Abroad Strategy Lead
An alcohol is an organic compound with a hydroxyl (OH) functional group on an aliphatic carbon atom. Because OH is the functional group of all alcohols, we often represent alcohols by the general formula ROH, where R is an alkyl group. The simplest derivatives of benzene is Phenol which is the common name as well as the accepted IUPAC name. And by naming the two alkyl or aryl groups that are linked to the oxygen atom, we get the common names of ethers.
| Table of Content |
Key Terms: Organic chemistry, Alcohol, Phenol, functional groups, compound, ether, organic compounds, oxygen, atom, hydroxyl group, alkyl group
Classification of Alcohol, Phenol and Ether
[Click Here for Sample Questions]
The classification of the compounds is needed to make their study more systematic and simple. Alcohol, phenol, ether are classes of organic compounds having the functional group -OH.
- Alcohol- Alcohol is formed when a hydroxyl (-OH) group is bound to a saturated carbon atom .
- Phenol- Phenol is formed when a hydrogen atom present in a benzene molecule is replaced by the functional group -OH.
- Ether- Ether is formed when two alkyl or aryl groups are connected to an oxygen atom.
The classification of Alcohol and Phenol is similar whereas it is different in the case of ethers.
Also Read:
Classification of Alcohol:
[Click Here for Sample Questions]

Classification of Alcohol
On the basis of the total number of hydroxyl groups attached to the carbon atom, alcohols are classified into three categories-
- Monohydric alcohols: Alcohols that contain one -OH group are called Monohydric Alcohol. For example, CH3CH2-OH and CH3CH2CH2-OH
- Dihydric alcohols: Alcohols that contain two -OH groups are called Dihydric Alcohol. For example, 1,2- Ethanediol and 1,3- Propanediol
- Trihydric alcohols: Alcohols that contain three -OH groups are called Trihydric Alcohol. For example 1,2,3-Propantriol and 1,2,3 -Butantriol.
On the basis of carbon atoms directly attached to the -OH group, the Monohydric Alcohols are classified into three types:
- Primary alcohol: In primary alcohol, one carbon atom is directly attached to the -OH group.
- Secondary alcohol: In secondary alcohol, two carbon atoms are directly attached to the -OH group.
- Tertiary alcohol: In tertiary alcohol, three carbon atoms are directly attached to the -OH group.
Nomenclature of Alcohol-
[Click Here for Sample Questions]
The common name of an alcohol is formed from the common name of the alkyl group by adding alcohol to it. The IUPAC name of alcohol is alkanols. Generally, they are represented in reactions by the general formula R-OH.
Rules of IUPAC Nomenclature of Alcohol:
According to the International Union of Pure and Applied Chemistry (IUPAC), certain rules are decided for naming alcohols:
- The longest chain of the carbon atoms containing the -OH group is taken as the parent compound. The numbering of the chain is done from the end nearest to the -OH group.
- The number that indicates the position of the -OH group is prefixed to the name of the parent hydrocarbon, and the suffix -ane of the parent alkane is replaced by the suffix -onal. In case of the cyclic alcohols, the carbon atom having the -OH group is designated as c-1, but 1 is not used while naming the alcohol.
- If there are more than one -OH group in the same molecule then the suffixes like -diol and -triol are used.
IUPAC names of some alcohols:
1- 
IUPAC Name- Ethan 1- ol or Ethanol
2- 
IUPAC Name- Propan 1- ol or Propanol
3- 
IUPAC Name- Propan-2-ol
4- 
IUPAC Name- Butan-1-ol
5- 
IUPAC Name- Butan-2-ol
6- 
IUPAC Name- Pentan-3-ol
Common and IUPAC Names of some Alcohols:

Common and IUPAC Names of some Alcohols
Physical Properties of Alcohol
[Click Here for Sample Questions]
- The Boiling Point of Alcohols
Alcohols have higher boiling points in comparison to other hydrocarbons which have equal molecular masses. The reason behind this is the presence of intermolecular hydrogen bonding between hydroxyl groups of alcohol molecules.
- Solubility of Alcohols
The solubility of alcohol decreases with the increase in the size of the alkyl group. This is due to the water present in the hydroxyl group which results in the formation of intermolecular hydrogen bonding.
- The Acidity of Alcohols
Alcohols react with active metals such as sodium, potassium etc. and form alkoxide. That is why the alcohols are acidic in nature. The acidic nature of alcohol is due to the polarity of –OH bond. The acidity of alcohol decreases when an electron-donating group is attached to the hydroxyl group.
Chemical Properties of Alcohol
[Click Here for Sample Questions]
- Oxidation of Alcohol
Alcohols undergo oxidation in the presence of an oxidizing agent to produce aldehydes and ketones which on further oxidation give carboxylic acids.

Oxidation of Alcohol
- Dehydration of Alcohol
Alcohols undergo dehydration when they are treated with protic acids, to form alkenes.

Dehydration of Alcohol
Classification of Phenol:
On the basis of the total number of hydroxyl groups attached to the carbon atom, phenols are classified into three categories-
- Monohydric phenols: Phenols that contain one -OH group are called Monohydric Phenols. For example- Phenol and 2- Methyl Phenol
- Dihydric phenols: Phenols that contain two -OH groups are called Dihydric Phenols. They can be further divided as ortho-, meta- or para- derivative. For example- Quinoil and Catechol
- Trihydric phenols: Phenols that contain three -OH groups are called Trihydric Phenols. For Example- Hydroxyquinol and Pyrogallol.

Classification of Phenol
Nomenclature of Phenols
[Click Here for Sample Questions]
The hydroxy derivative of benzene is phenol. It is both its common name and IUPAC name. As the structure of phenol has a benzene ring in its substituted compounds, the terms ortho, meta and para are often used in the common names.
Phenols are further divided into three classes which are monohydric phenols, dihydric phenols and trihydric phenols. Certain rules are decided for the IUPAC nomenclature of Phenols.
Rules of IUPAC Nomenclature of Phenol
- Check the position of the hydroxyl group which is attached to the benzene ring.
- If more than one hydroxyl group is attached to the benzene rings then, the prefixes such as di, tri and tetra are used to denote the number of similar hydroxyl groups attached.
- In the case of substituted phenols, numbering of the positions of the other functional groups is done with respect to the position of the hydroxyl group attached. For example, if a methyl group is attached at the fourth carbon atom with respect to the hydroxyl group, the compound is named as, 4-Methyl phenol.
- On the basis of the position of the substituted functional group with respect to the hydroxyl group, terms like ortho (if functional group is attached to the adjacent carbon atoms), para (if functional group is attached to the third carbon atom) and meta (if functional group is attached to the second carbon atom) are also used for the nomenclature of phenols.
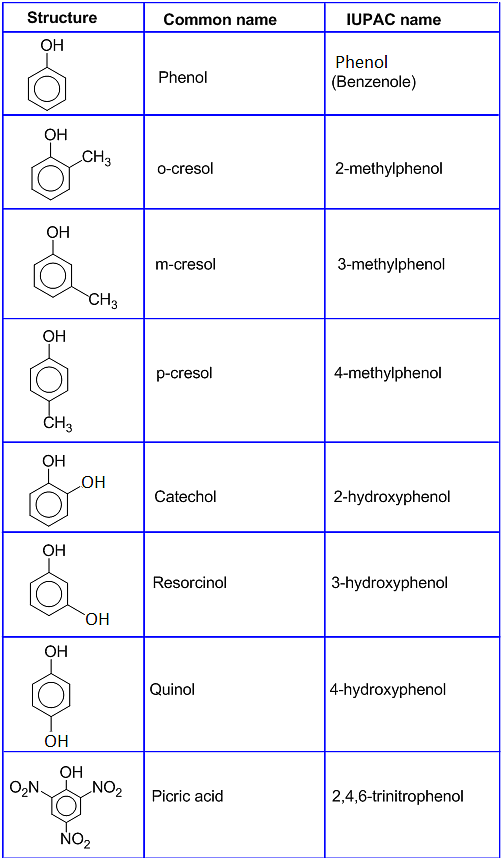
IUPAC names of some Phenols
Physical Properties of Phenol-
[Click Here for Sample Questions]
- Boiling Point of Phenols
Phenols have higher boiling point in comparison to other hydrocarbons which have equal molecular masses. The reason behind this is the presence of intermolecular hydrogen bonding between hydroxyl groups of phenols.
- Solubility of Phenols
The solubility of phenol is due to the presence of the hydroxyl group.The hydroxyl group in phenol is responsible for the formation of bonding with hydrogen atoms of water. The solubility of phenol decreases due to increase in the size of the aryl group.
- Acidity of Phenols
Phenols can react with the active metals such as sodium, potassium, etc. to form the corresponding phenoxide. These reactions of phenols are responsible for its acidic nature.
Classification of Ether
[Click Here for Sample Questions]

Classification of Ether
On the basis of alkyl or aryl groups attached to the oxygen atom, ethers are classified into the following two types:
- Symmetrical ether: Symmetrical ethers are also known as the simple ether. They have the same alkyl or the aryl group attached to both sides of the oxygen atoms. For Example- CH3OCH3, C2H5OC2H5, etc.
- Unsymmetrical ether: Unsymmetrical ethers are also known as the mixed either. They have different alkyl or the aryl group attached to both sides of the oxygen atoms. For Example- CH3OC2H5, C2H5OC6H5, etc.
Nomenclature of Ethers-
[Click Here for Sample Questions]
Ethers are the organic compounds which have two same or different alkyl or aryl groups bonded to an oxygen atom. They are commonly represented as- R-O-R or R1–O–R2. The ether functional group does not have a characteristic IUPAC nomenclature suffix. Certain rules are decided for the IUPAC nomenclature of Ethers.
Rules of IUPAC Nomenclature of Ethers:
- Commonly naming of different alkyl/aryl groups attached to the either side of the oxygen atom is done in the alphabetical order and finally the word ether is added to it. For example, CH3OC6H5 is named as Methyl phenyl ether.
- Ethers in which the oxygen atoms are attached to the same group are named with prefixes such as ‘di’ added before the alkyl/aryl groups. For example, CH3OCH3 is named as dimethyl ether.
- A substituent group containing more carbon atoms is chosen as the parent hydrocarbon and the other substituent group attached to the oxygen atom is named with a prefix ‘oxy’. For example, CH3OC2H5 is named as 1-methoxy ethane.
Common and IUPAC Names of some Ethers:

Common and IUPAC Names of some Ethers
Physical Properties of Ether-:
- An ether molecule possesses a net dipole moment which can be attributed to the polarity of C-O bonds.
- The boilng point of ethers can be compared to the alkanes. However, it is much lower compared to that of alcohols of comparable molecular mass. This is despite the fact of the polarity of C-O bond.
- The miscibility of ethers with water is similar to those of alcohols. As it is miscible in water, it can be attributed to the fact that like alcohols, the oxygen atom of ether can form hydrogen bonds as well with a water molecule.
Read More:-
Sample Questions
Ques: Name the following compound.(2 marks)

Ans: This compound has a three-carbon chain, so the parent alkane name is propane. Because the compound contains three hydroxyl groups, the suffix must be -triol. Therefore, the name is 1, 2, 3-propanetriol.
The common name of this compound is glycerol is also called glycerin and it is an important by-product in the manufacture of soaps.
Ques: Draw the Structure for each alcohol.
a) 3-hexanol
b) 3, 3-dimethyl-2-butanol
c) cyclocutanol (3 marks)
Ans: (a)

3-hexanol
(b)

3, 3-dimethyl-2-butanol
(c)

Cyclocutanol
Ques: How can ethanol be converted to ethene? (All India 011)
Ans:

Ques: Draw the structural formula of 2-methylpropan – 2- ol molecule. (Delhi 2012)?
Ans:

Ques: Give a chemical test to differentiate between 2-pentanol and 3-pentanol. (Comptt. Delhi 2013)
Ans: 2-pentanol gives lodoform test with yellow ppt. of lodoform whereas, 3-pentanol does not give this test.
Ques: Write the chemical reaction in order to explain Kolbe’s reaction. (Comptt. Delhi 2013)
Ans: In Kolbe’s reaction, Phenol reacts with CO2 in the presence of sodium hydroxide at 4 – 7 Atm and 390 – 410 K giving salicyclic acid.

Ques: Write the equation of the nitration of anisole. (Comptt. Delhi 2016)
Ans:

Ques: Explain the mechanism of acid catalysed hydration of an alkene to form corresponding alcohol. (All India 2012)
Ans: In acid catalysed hydration, alkenes react with water in the presence of acid as catalyst to form alcohols.

Mechanism, it involves three steps:
(i) Protonation of alkenes to form carbocation by electrophilic attack of H3O+.

For Latest Updates on Upcoming Board Exams, Click Here: https://t.me/class_10_12_board_updates
Check-Out:






Comments