JEE Main 2023 12 April Shift 1 Question Paper is here. NTA conducted JEE Main 2023 April 12 Shift 1 from 9 AM to 12 PM. Candidates can download the official JEE Main 2023 Question Paper PDF with Solution and Answer Key for 12 April Shift 1 using the link below.
Also Check: JEE Main 2024 Question Paper with Solution PDF Download
JEE Main 2023 12 April Shift 1 Question Paper with Answer Key PDF
| JEE Main 2023 12th April Shift 1 Question Paper with Solution PDF | Check Solution |

Question 1:
The number of five-digit numbers, greater than 40000 and divisible by 5, which can be formed using the digits 0, 1, 3, 5, 7, and 9 without repetition, is equal to:
View Solution
Step 1: Determine the possibilities for the unit place.
Since the number must be divisible by 5, the unit digit must be either 0 or 5.
Step 2: Consider the case when the unit digit is 0.
The first digit must be greater than 4 (i.e., 5, 7, or 9). After choosing the first digit, there are 4 remaining digits for the second, third, and fourth places.
Thus, the number of such five-digit numbers is: \[ 3 \times 4 \times 3 \times 2 = 72 \]
Step 3: Consider the case when the unit digit is 5.
The first digit must be greater than 4 (i.e., 7 or 9). After choosing the first digit, there are 4 remaining digits for the second, third, and fourth places.
Thus, the number of such five-digit numbers is: \[ 2 \times 4 \times 3 \times 2 = 48 \]
Step 4: Total number of such five-digit numbers
Adding the results from Step 2 and Step 3: \[ 72 + 48 = 120 \] Quick Tip: To solve this type of problem, break it into cases based on the divisibility condition (here, the number must end in 0 or 5) and calculate the possibilities for each case separately.
Let \( \alpha, \beta \) be the roots of the quadratic equation \( x^2 + \sqrt{6}x + 3 = 0 \). Then, \( \frac{\alpha^{23} + \beta^{23} + \alpha^{14} + \beta^{14}}{\alpha^{15} + \beta^{15} + \alpha^{10} + \beta^{10}} \) is equal to:
View Solution
Let < an
where \( p_1, p_2, \dots, p_m \) are the first \( m \) prime numbers, then \( m \) is equal to:
View Solution
Let the lines \( l_1 : \frac{x + 5}{3} = \frac{y + 4}{1} = \frac{z - \alpha}{-2} \) and \( l_2 : 3x + 2y + z - 2 = 0, \, x - 3y + 2z - 13 = 0 \) be coplanar. If the point P(a, b, c) on \( l_1 \) is nearest to the point Q(-4, -3, 2), then \( |a| + |b| + |c| \) is equal to:
View Solution
Let \( P \left( \frac{2\sqrt{3}}{7}, \frac{6}{\sqrt{7}} \right), Q, R, \) and \( S \) be four points on the ellipse \( 9x^2 + 4y^2 = 36 \). Let PQ and RS be mutually perpendicular and pass through the origin. If \[ \frac{1}{(PQ)^2} + \frac{1}{(RS)^2} = \frac{p}{q} \]
where \( p \) and \( q \) are coprime, then \( p + q \) is equal to:
View Solution
Let \(a, b, c\) be three distinct real numbers, none equal to one. If the vectors \( a \hat{i} + \hat{j} + \hat{k}, \, \hat{i} + b\hat{j} + \hat{k}, \, a \hat{i} + \hat{j} + c \hat{k} \) are coplanar, then \( \frac{1}{1 - a} + \frac{1}{1 - b} + \frac{1}{1 - c} \) is equal to:
View Solution
If the local maximum value of the function \[ f(x) = \left( \frac{\sqrt{3e}}{2 \sin x} \right)^{\sin^2 x}, \quad x \in \left( 0, \frac{\pi}{2} \right) \]
is \( \frac{k}{e} \), then \[ \left( \frac{k}{e} \right)^8 + \frac{k^8}{e^5} + k^8 \]
is equal to:
View Solution
Let \( D \) be the domain of the function \( f(x) = \sin^{-1} \left( \log_{3x} \left( \frac{6 + 2 \log_3 x}{-5x} \right) \right) \). If the range of the function \( g: D \to \mathbb{R} \) defined by \( g(x) = x - \lfloor x \rfloor \), where \( \lfloor x \rfloor \) is the greatest integer function, is \( (\alpha, \beta) \), then \( \alpha^2 + \frac{5}{\beta} \) is equal to:
View Solution
Let \( y = y(x), \, y > 0 \), be a solution curve of the differential equation \( (1 + x^2) \, dy = y(x - y) \, dx \). If \( y(0) = 1 \) and \( y(2\sqrt{2}) = \beta \), then:
View Solution
Step 1: Rearrange the given differential equation.
The given equation is: \[ (1 + x^2) \, \frac{dy}{dx} = y(x - y) \]
We can express it as: \[ \frac{dy}{dx} = \frac{y(x - y)}{1 + x^2} \]
Step 2: Transform into a solvable form.
Rewrite the equation as: \[ \frac{dy}{dx} + y \left( \frac{-x}{1 + x^2} \right) = \left( \frac{-1}{1 + x^2} \right) y^2 \]
Multiply through by \( \frac{1}{y} \): \[ \frac{1}{y} \frac{dy}{dx} + \frac{-x}{(1 + x^2)y} = \frac{-1}{(1 + x^2)} \]
Step 3: Simplify the integrals.
Let \( \frac{1}{y} = t \), then we have the differential equation: \[ \frac{-1}{y^2} \frac{dy}{dx} = \frac{dt}{dx} \]
Integrating both sides: \[ \int \frac{1}{1 + x^2} \, dx = \int \frac{1}{y} \, dt \]
Thus, we obtain the general solution: \[ \sqrt{1 + x^2} = y \ln(e(x + \sqrt{1 + x^2})) \]
Step 4: Apply the boundary conditions.
We know that \( y(0) = 1 \), so we can use this to find the value of the constant.
Substitute \( x = 0 \) and \( y = 1 \) into the equation: \[ 1 = \sqrt{1 + 0^2} \ln(e(0 + \sqrt{1 + 0^2})) \]
This simplifies to \( 1 = \ln(e(1)) = 1 \), confirming the constant is correct.
Step 5: Find the value of \( \beta \).
Now, for \( y(2\sqrt{2}) = \beta \), we substitute \( x = 2\sqrt{2} \) into the solution to find \( \beta \): \[ \beta = \frac{3}{\ln(e(3 + 2\sqrt{2}))} \]
Thus, we obtain \( 3 = \ln(e(3 + 2\sqrt{2})) \), which leads to: \[ e^{3\beta - 1} = e^{(3 + 2\sqrt{2})} \] Quick Tip: When solving first-order differential equations, use the method of integrating factors. This helps in simplifying the equation to an easily solvable form.
Among the two statements
(Statement1): \( (p \Rightarrow q) \land (q \land (\sim q)) \) is a contradiction and
(Statement2): \( (p \land q) \lor (\sim p) \land (\sim q) \) is a tautology,
View Solution
Let \( \lambda \in \mathbb{Z}, \vec{a} = \lambda \hat{i} + \hat{j} - \hat{k} \) and \( \vec{b} = 3\hat{i} - \hat{j} + 2\hat{k} \). Let \( \vec{c} \) be a vector such that \[ (\vec{a} + \vec{b} + \vec{c}) \times \vec{c} = 0, \quad \vec{a} \cdot \vec{c} = -17 \quad and \quad \vec{b} \cdot \vec{c} = -20. \]
Then \( \left| \vec{c} \times (\lambda \hat{i} + \hat{j} + \hat{k}) \right|^2 \) is equal to:
View Solution
The sum of the coefficients of the first 50 terms in the binomial expansion of \((1-x)^{100}\), is equal to\
View Solution
The area of the region enclosed by the curve \( y = x^3 \) and its tangent at the point \( (-1, -1) \) is:
View Solution
Let \( A = \begin{bmatrix} 1 & \frac{1}{51}
0 & 1 \end{bmatrix} \). If \( B = \begin{bmatrix} 1 & 2
-1 & -1 \end{bmatrix} A \begin{bmatrix} -1 & -2
1 & 1 \end{bmatrix} \),
then the sum of all the elements of the matrix \[ \sum_{n=1}^{50} B^n \]
is equal to:
View Solution
Let the plane \( P : 4x - y + z = 10 \) be rotated by an angle \( \frac{\pi}{2} \) about its line of intersection with the plane \( x + y - z = 4 \). If \( \alpha \) is the distance of the point \( (2, 3, -4) \) from the new position of the plane P, then \( 35\alpha \) is:
View Solution
If \(\frac{1}{n+1}\) \({^{n}C_{n}}+\frac{1}{n}\) \({^{n}C_{n-1}}\) + \dots + \(\frac{1}{2}\) \({^{n}C_{1}}\) + \({^{n}C_{0}}\) = \(\frac{1023}{10}\) then n is equal to:
View Solution
Let \( C \) be the circle in the complex plane with centre \( z_0 = \frac{1}{2}(1 + 3i) \) and radius \( r = 1 \). Let \( z_1 = 1 + i \) and the complex number \( z_2 \) be outside the circle \( C \) such that \( |z_1 - z_0| = |z_2 - z_0| = 1 \). If \( z_0, z_1 \) and \( z_2 \) are collinear, then the smaller value of \( |z_2|^2 \) is equal to:
View Solution

View Solution
Two dice A and B are rolled, Let the numbers obtained on A and B be \( \alpha \) and \( \beta \) respectively. If the variance of \( \alpha - \beta \) is \( \frac{p}{q} \), where \( p \) and \( q \) are coprime, then the sum of the positive divisors of \( p \) is equal to:
View Solution
In a triangle ABC, if \( \cos A + 2 \cos B + \cos C = 2 \) and the lengths of the sides opposite to the angles A and C are 3 and 7 respectively, then \( \cos A - \cos C \) is equal to:
View Solution
We are given the equation: \[ \cos A + 2 \cos B + \cos C = 2 \]
Also, the lengths of the sides opposite to angles A and C are \( a = 3 \) and \( c = 7 \), respectively.
We can use the identity: \[ \cos \left( \frac{A + C}{2} \right) = \sin \left( \frac{B}{2} \right) \]
which simplifies to: \[ \cos A - \cos C = 2 \sin \frac{B}{2} \cos \frac{B}{2} \]
Next, we use the given identity \( 2 \cos B / 2 \cos A \) and simplify the calculation to the sum of \( A + C \) and thus \( \cos A - \cos C \).
So, after calculating the entire expression: \[ \boxed{\cos A - \cos C = \frac{10}{7}} \] Quick Tip: For triangle trigonometry problems, use the relationship between the sides and angles, as well as the trigonometric identities for the sum and difference of angles. In this case, use the cosine and sine half-angle identities to simplify.
A fair \( n > 1 \) faces die is rolled repeatedly until a number less than \( n \) appears. If the mean of the number of tosses required is \( \frac{n}{9} \), then \( n \) is equal to:
View Solution
Let the digits \( a, b, c \) be in A.P. Nine-digit numbers are to be formed using each of these three digits thrice such that three consecutive digits are in A.P. at least once. How many such numbers can be formed?
View Solution
Let \( [x] \) be the greatest integer \( \leq x \). Then the number of points in the interval \( (-2, 1) \), where the function \( f(x) = |[x]| + \sqrt{x - [x]} \) is discontinuous is:
View Solution
Let the plane \( x + 3y - 2z + 6 = 0 \) meet the co-ordinate axes at the points A, B, C. If the orthocentre of the triangle ABC is \( (\alpha, \beta, \frac{6}{7}) \), then \( 98(\alpha + \beta)^2 \) is equal to:
View Solution
Let I(x) = \(\int \sqrt{\frac{x+7}{x}}\) \, dx and I(9) = 12 + 7 \(\log_e\) 7.
If I(1) = \(\alpha + 7 \log_e (1+2\sqrt{2})\), then \(\alpha^4\) is equal to:
View Solution

View Solution
Let the positive numbers \( a_1, a_2, a_3, a_4 \), and \( a_5 \) be in a G.P. Let their mean and variance be \( \frac{31}{10} \) and \( \frac{m}{n} \), respectively, where \( m \) and \( n \) are co-prime. If the mean of their reciprocals is \( \frac{31}{40} \) and \( a_3 + a_4 + a_5 = 14 \), then \( m + n \) is equal to:
View Solution
The number of relations, on the set \( \{1, 2, 3\} \) containing \( (1, 2) \) and \( (2, 3) \), which are reflexive and transitive but not symmetric, is:
View Solution
If \[ \int_{-0.15}^{0.15} \left| 100x^2 - 1 \right| \, dx = \frac{k}{3000}, then k is equal to: \]
View Solution
Two circles in the first quadrant of radii \( r_1 \) and \( r_2 \) touch the coordinate axes. Each of them cuts off an intercept of 2 units with the line \( x + y = 2 \). Then \( r_1^2 + r_2^2 - r_1r_2 \) is equal to:
View Solution
An ice cube has a bubble inside. When viewed from one side the apparent distance of the bubble is 12 cm. When viewed from the opposite side, the apparent distance of the bubble is observed as 4 cm. If the side of the ice cube is 24 cm, the refractive index of the ice cube is:
View Solution
Two satellites A and B move round the earth in the same orbit. The mass of A is twice the mass of B. The quantity which is same for the two satellites will be:
View Solution
Step 1: Understanding orbital motion of satellites:
The total energy \( E \) and kinetic energy \( K.E. \) of a satellite moving in a circular orbit around the Earth depend only on the radius of the orbit and the gravitational constant, not on the mass of the satellite.
Step 2: Speed of the satellite:
The speed of the satellite \( v \) in orbit is given by: \[ v = \sqrt{\frac{GM_p}{R}} \]
Since the radius of orbit and gravitational constant are the same for both satellites, their speed will be the same. Quick Tip: In orbital motion problems, remember that speed is independent of the mass of the satellite, while potential energy and kinetic energy depend on the mass.
The amplitude of 15 sin (1000 \( \pi t \)) is modulated by 10 sin (4\( \pi t \)) signal. The amplitude modulated signal contains frequencies of:
(1) 500 Hz.
(2) 2 Hz.
(3) 250 Hz.
(4) 498 Hz.
(5) 502 Hz.
View Solution
In an n-p-n common emitter (CE) transistor, the collector current changes from 5 mA to 16 mA for the change in base current from 100 μA and 200 μA, respectively. The current gain of the transistor is:
View Solution
If the r.m.s. speed of chlorine molecules is 490 m/s at 27°C, the r.m.s. speed of argon molecules at the same temperature will be (Atomic mass of argon = 39.9u, molecular mass of chlorine = 70.9u):
View Solution
The r.m.s. speed of gas molecules is related to the temperature and molecular mass by the formula:
\[ V_{rms} = \sqrt{\frac{3RT}{M}} \]
Where:
\( R \) is the gas constant.
\( T \) is the temperature.
\( M \) is the molar mass of the gas.
Since both gases are at the same temperature, we can use the ratio of their r.m.s. speeds:
\[ \frac{\nu_{Ar}}{\nu_{Cl}} = \sqrt{\frac{M_{Cl}}{M_{Ar}}} \]
Substitute the given values:
\( M_{Cl} = 70.9 \, u \)
\( M_{Ar} = 39.9 \, u \)
\( \nu_{Cl} = 490 \, m/s \)
\[ \frac{\nu_{Ar}}{490} = \sqrt{\frac{70.9}{39.9}} = 1.33 \]
Thus,
\[ \nu_{Ar} = 1.33 \times 490 = 651.7 \, m/s \]
Thus, the r.m.s. speed of argon molecules is \( \boxed{651.7} \, m/s \). Quick Tip: The r.m.s. speed of gas molecules is inversely proportional to the square root of their molar mass. Use this relation to compare the speeds of different gases at the same temperature.
A proton and an \( \alpha \)-particle are accelerated from rest by 2V and 4V potentials, respectively. The ratio of their de-Broglie wavelength is:
View Solution
Given below are two statements:
Statement I: The diamagnetic property depends on temperature.
Statement II: The included magnetic dipole moment in a diamagnetic sample is always opposite to the magnetizing field.
In light of the given statement, choose the correct answer from the options below:
View Solution
A wire of resistance 160 \( \Omega \) is melted and drawn into a wire of one-fourth of its length. The new resistance of the wire will be:
View Solution
Match List I with List II:
Choose the correct answer from the options given below:
Three forces \( F_1 = 10 \, N, F_2 = 8 \, N, F_3 = 6 \, N \) are acting on a particle of mass 5 kg. The forces \( F_2 \) and \( F_3 \) are applied perpendicular so that particle remains at rest. If the force \( F_1 \) is removed, then the acceleration of the particle is:
View Solution
A body cools from 80°C to 60°C in 5 minutes. The temperature of the surrounding is 20°C. The time it takes to cool from 60°C to 40°C is:
View Solution
An engine operating between the boiling and freezing points of water will have:
(1) efficiency more than 27%.
(2) efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
(3) efficiency equal to 27%.
(4) efficiency less than 27%.
View Solution
Given below are two statements:
Statement I: A truck and a car moving with the same kinetic energy are brought to rest by applying brakes which provide equal retarding forces. Both come to rest in equal distance.
Statement II: A car moving towards east takes a turn and moves towards north, the speed remains unchanged. The acceleration of the car is zero.
In light of the given statements, choose the most appropriate answer from the options given below:
View Solution
A particle is executing Simple Harmonic Motion (SHM). The ratio of potential energy and kinetic energy of the particle when its displacement is half of its amplitude will be:
View Solution
A ball is thrown vertically upward with an initial velocity of 150 m/s. The ratio of velocity after 3 s and 5 s is \( \frac{x+1}{x} \). The value of \( x \) is:
Take \( g = 10 \, m/s^2 \).
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: If an electric dipole of dipole moment \( 30 \times 10^{-5} \) Cm is enclosed by a closed surface, the net flux coming out of the surface will be zero.
Reason R: Electric dipole consists of two equal and opposite charges.
In the light of above statements, choose the correct answer from the options given below:
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: EM waves used for optical communication have longer wavelengths than that of microwave, employed in Radar technology.
Reason R: Infrared EM waves are more energetic than microwaves.
In the light of given statements, choose the correct answer from the options given below:
View Solution
A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. The number of spectral lines emitted will be:
View Solution
The ratio of escape velocity of a planet to the escape velocity of Earth will be:
Given: Mass of the planet is 16 times the mass of Earth and radius of the planet is 4 times the radius of Earth.
View Solution
Given below are two statements:
Statement I: When the frequency of an a.c. source in a series LCR circuit increases, the current in the circuit first increases, attains a maximum value and then decreases.
Statement II: In a series LCR circuit, the value of the power factor at resonance is one.
In light of the given statements, choose the most appropriate answer from the options given below:
View Solution
For a certain organ pipe, the first three resonance frequencies are in the ratio of 1:3:5 respectively. If the frequency of the fifth harmonic is 405 Hz and the speed of sound in air is 324 m/s\(^{-1}\), the length of the organ pipe is _______ m.
View Solution
For a rolling spherical shell, the ratio of rotational kinetic energy and total kinetic energy is \( \frac{x}{5} \). The value of \( x \) is:
View Solution
A compass needle oscillates 20 times per minute at a place where the dip is 30° and 30 times per minute where the dip is 60°. The ratio of total magnetic field due to the earth at two places respectively is \( \frac{4}{\sqrt{x}} \). The value of \( x \) is:
View Solution
A conducting circular loop is placed in a uniform magnetic field of 0.4 T with its plane perpendicular to the field. Somehow, the radius of the loop starts expanding at a constant rate of 1 mm/s. The magnitude of induced emf in the loop at an instant when the radius of the loop is 2 cm will be ___________, \(\mu V\).
View Solution
To maintain a speed of 80 km/h by a bus of mass 500 kg on a plane rough road for 4 km distance, the work done by the engine of the bus will be ________ KJ.
[The coefficient of friction between tyre of bus and road is 0.04].
View Solution
A common example of alpha decay is \[ ^{238}_{92} U \rightarrow ^{234}_{90} Th + ^{4}_{2} He + Q. \]
Given: \[ ^{238}_{92} U = 238.05060 \, u, \quad ^{234}_{90} Th = 234.04360 \, u, \quad ^{4}_{2} He = 4.00260 \, u, \quad 1u = 931.5 \, MeV/c^2. \]
The energy released \( Q \) during the alpha decay of \( ^{238}_{92} U \) is ______ MeV.
View Solution
Energy released \( Q \) is given by the equation: \[ Q = (\Delta m)_{amu} \times 931.5 \, MeV. \]
Here, the mass defect \( \Delta m \) is: \[ \Delta m = m_u - m_{Th} - m_{He} = 238.05060 \, u - 234.04360 \, u - 4.00260 \, u = 0.0044 \, u. \]
Thus, \[ Q = 0.0044 \times 931.5 \, MeV = 4.0986 \, MeV. \]
Therefore, the energy released during the alpha decay of \( ^{238}_{92} U \) is 4.0986 MeV. Quick Tip: To calculate the energy released during a nuclear decay, find the mass defect and multiply it by \( 931.5 \, MeV \) (the energy equivalent of 1 atomic mass unit).
The current flowing through a conductor connected across a source is 2A and 1.2 A at 0°C and 100°C respectively. The current flowing through the conductor at 50°C will be _______ \( \times 10^2 \) mA.
View Solution
Two convex lenses of focal length 20 cm each are placed coaxially with a separation of 60 cm between them. The image of the distant object formed by the combination is at _______ cm from the first lens.
View Solution
Glycerine of density \( 1.25 \times 10^3 \, kg/m^{-3} \) is flowing through the conical section of pipe. The area of cross-section of the pipe at its ends is 10 cm\(^2\) and 5 cm\(^2\) and pressure drop across its length is 3 Nm\(^{-2}\). The rate of flow of glycerine through the pipe is \( x \times 10^{-5} \, m^3 \, s^{-1} \). The value of \( x \) is _______ .
View Solution
64 identical drops each charged up to a potential of 10 mV are combined to form a bigger drop. The potential of the bigger drop will be _______ mV.
View Solution
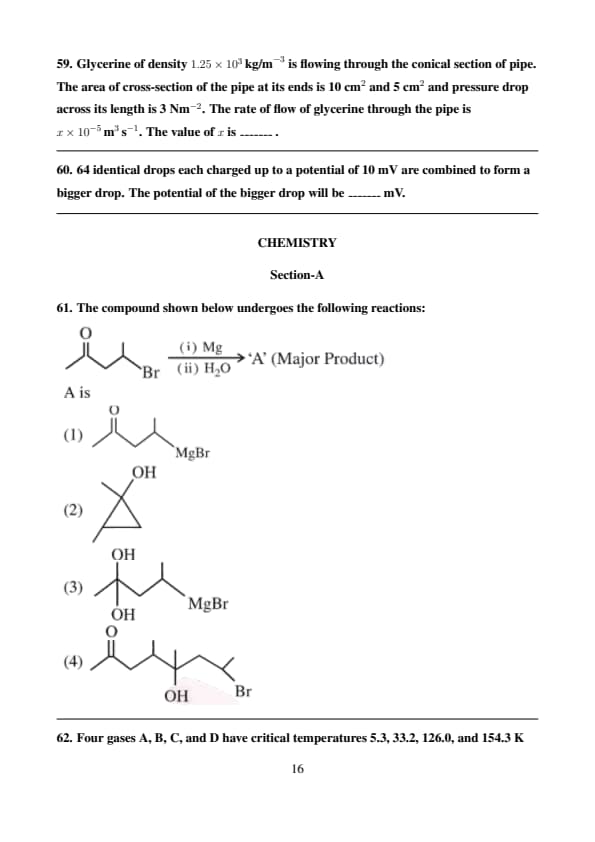
The compound shown below undergoes the following reactions:
View Solution
The reaction shown involves the conversion of an alkyl bromide to an alcohol in two steps.
1. First, the reaction with magnesium (\( Mg \)) in dry ether will convert the alkyl bromide to a Grignard reagent:
\[ C_6H_5Br + Mg \xrightarrow{ether} C_6H_5MgBr \]
This results in the formation of the Grignard reagent, which is an organomagnesium compound.
2. In the second step, the Grignard reagent reacts with water (\( H_2O \)):
\[ C_6H_5MgBr + H_2O \rightarrow C_6H_5OH + MgBr \]
The final product is phenol (C6H5OH), formed by the addition of a hydroxyl group (\( OH \)) to the carbon atom that was originally bonded to the bromine atom.
Thus, the major product is the phenol, which is option (4).
Therefore, the correct answer is \( \boxed{4} \). Quick Tip: When an alkyl halide reacts with magnesium in ether, it forms a Grignard reagent. This reacts with water to produce an alcohol as the major product.
Four gases A, B, C, and D have critical temperatures 5.3, 33.2, 126.0, and 154.3 K respectively.
For their adsorption on a fixed amount of charcoal, the correct order is:
View Solution
Given below are two statements:
Assertion A: 5f electrons can participate in bonding to a far greater extent than 4f electrons.
Reason R: 5f orbitals are not as buried as 4f orbitals.
In light of the above statements, choose the correct answer from the options given below:
View Solution
The incorrect statement regarding the reaction given below is:
View Solution
Match List I with List II
Choose the correct answer from the options given below:
Match List I with List II
Choose the correct answer from the options given below:
The density of alkali metals is in the order:
View Solution
Given below are two statements:
\text{Statements: \quad \mathrm{SbCl\(_5\) \text{ is more covalent than \mathrm{SbCl\(_3\)
\text{Statements: \quad \text{The higher oxides of halogens also tend to be more stable than the lower ones.
In light of the above statements, choose the most appropriate answer from the options given below:
View Solution
A metal chloride contains 55.0% of chlorine by weight. 100 mL vapours of the metal chloride at STP weigh 0.57 g. The molecular formula of the metal chloride is: \[ (Given: Atomic mass of chlorine is 35.5 u) \]
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: In the Ellingham diagram, a sharp change in the slope of the line is observed for \( Mg \rightarrow MgO \) at \( \sim 1120^\circ C \).
Reason R: There is a large change of entropy associated with the change of state.
In light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II
Choose the correct answer from the options given below:
For lead storage battery pick the correct statements:
\text{A. During charging of battery, PbSO\(_4\) \text{ on anode is converted into PbO\(_2\)
\text{B. During charging of battery, PbSO\(_4\) \text{ on cathode is converted into PbO\(_2\)
\text{C. Lead storage battery, consists of grid of lead packed with PbO\(_2\) \text{ as anode
\text{D. Lead storage battery has \(\sim 38%\) solution of sulphuric acid as an electrolyte
Choose the correct answer from the options given below:
View Solution
View Solution
The reaction involves ozonolysis of 2-hexene, where ozone reacts with the double bond, resulting in the formation of two carbonyl compounds. The products formed from this reaction are:
Acetic acid (CH\(_3\)COOH) from the oxidation of the terminal carbon of the hexene.
Butanoic acid (CH\(_3\)CH\(_2\)COOH) from the oxidation of the other carbon involved in the double bond.
Thus, the correct products are Butanoic acid and acetic acid. Quick Tip: In ozonolysis of alkenes, the double bond undergoes cleavage, leading to the formation of two carbonyl compounds, typically acids or aldehydes depending on the substrate.
Correct statements for the given reaction are:
A. Compound 'B' is aromatic
B. The completion of the above reaction is very slow
C. 'A' shows tautomerism
D. The bond lengths C-C in compound 'B' are found to be same
Choose the correct answer from the options given below:
The bond order and magnetic property of acetylide ion are same as that of:
View Solution
In the given reaction cycle
Given below are two statements:
Statement I: Boron is extremely hard indicating its high lattice energy.
Statement II: Boron has the highest melting and boiling point compared to its other group members.
View Solution
View Solution
The reaction shown is an example of a Wittig reaction, which forms a cyclic compound through the reaction of the acetylide ion (\(C_2^{2-}\)) with an electrophile (\(OE^{-}\)). The acetylide ion undergoes a [2+2] cycloaddition with the electrophile to form a cyclic intermediate, which upon heating gives a cyclic product containing a carbonyl group.
The acetylide ion (\(C_2^{2-}\)) reacts with the electrophile in a manner that results in the formation of a five-membered cyclic compound, A, containing a carbonyl group. This is consistent with the structure shown in option (3).
Quick Tip: The Wittig reaction typically involves the reaction of an acetylide ion with an electrophile to form a new carbon-carbon bond, which often results in the formation of a cyclic structure. Heating can help drive this reaction to completion.
Match List I with List II
The major product \(P\) formed in the following sequence of reactions is:
View Solution
One mole of an ideal gas at 350K is in a 2.0 L vessel of thermally conducting walls, which are in contact with the surroundings. It undergoes isothermal reversible expansion from 2.0L to 3.0L against a constant pressure of 4 atm. The change in entropy of the surroundings \(\Delta S\) is ______ J K\(^{-1}\) (Nearest integer).
Given \(R = 8.314\) J K\(^{-1}\) Mol\(^{-1}\).
View Solution
The mass of NH\(_3\) produced when 131.8 kg of cyclohexanecarbaldehyde undergoes Tollens test is ______ kg. (Nearest Integer)
Given: Molar Mass of C = 12 g/mol, N = 14 g/mol, O = 16 g/mol.
View Solution
The chemical equation for the reaction is:
\[ Cyclohexanecarbaldehyde + 2 [Ag(NH_3)_2] OH \rightarrow Cyclohexanecarboxamide + 3 NH_3 + 2 Ag + H_2O \]
We are given the mass of cyclohexanecarbaldehyde:
\[ Mass of cyclohexanecarbaldehyde = 131.8 \, kg \]
The molecular weight of cyclohexanecarbaldehyde:
\[ Molecular weight = 12 \times 6 + 14 + 16 = 112 \, g/mol \]
From the stoichiometry of the reaction, we know that 1 mole of cyclohexanecarbaldehyde produces 3 moles of NH\(_3\). The number of moles of cyclohexanecarbaldehyde is:
\[ Moles of cyclohexanecarbaldehyde = \frac{131.8 \times 1000}{112} = 1176.8 \, mol \]
Since 1 mole of cyclohexanecarbaldehyde produces 3 moles of NH\(_3\), the moles of NH\(_3\) produced are:
\[ Moles of NH_3 = 3 \times 1176.8 = 3530.4 \, mol \]
The molar mass of NH\(_3\) is:
\[ Molar mass of NH_3 = 17 \, g/mol \]
The mass of NH\(_3\) produced is:
\[ Mass of NH_3 = 3530.4 \times 17 = 60,023.2 \, g = 60 \, kg \]
Thus, the mass of NH\(_3\) produced is:
\[ \boxed{60 \, kg} \] Quick Tip: In reactions like Tollens' test, always pay attention to the stoichiometry between the reactants and products to correctly calculate the mass of the desired product.
In an oligopeptide named Alanylglycylphenylalanylisoleucine, the number of \( sp^2 \) hybridised carbons is __________.
View Solution
In the given structure, we need to identify the carbons that are \( sp^2 \) hybridised.
The following carbons are \( sp^2 \) hybridised:
1. The carbonyl carbon of the peptide bond between alanine and glycine.
2. The carbonyl carbon of the peptide bond between phenylalanine and alanine.
3. The carbonyl carbon of the carboxyl group at the C-terminal of isoleucine.
4. The carbon of the phenyl group attached to the aromatic ring in phenylalanine.
Hence, counting all the \( sp^2 \) hybridised carbons in the structure:
One from each of the peptide bonds (two bonds).
One from the carboxyl group (terminal carbon).
One from the aromatic ring in phenylalanine (C6H5 group).
Thus, the number of \( sp^2 \) hybridised carbons is 10. Quick Tip: To identify \( sp^2 \) hybridised carbons, focus on carbonyl groups in peptide bonds and carboxyl groups, as well as carbon atoms involved in double bonds, such as those in aromatic rings.
An analyst wants to convert 1L HCl of pH = 1 to a solution of HCl of pH 2. The volume of water needed to do this dilution is ______ mL. (Nearest Integer)
View Solution
Given: \[ M_1 = 10 (pH = 1, concentration of HCl) \] \[ M_2 = 10^{-2} (pH = 2, concentration of HCl) \] \[ V_1 = 1L (initial volume of HCl) \]
Using the dilution formula: \[ (M_1 \times V_1) = (M_2 \times V_2) \] \[ 10 \times 1 = 10^{-2} \times V_2 \] \[ V_2 = 10L = 10000 mL \]
Thus, water added = \(10000 - 1000 = 9000 mL\) Quick Tip: For dilution, the product of concentration and volume remains constant. You can solve it using the dilution equation.
Three organic compounds A, B, and C were allowed to run in thin layer chromatography using hexane and gave the following result. The Rf value of the most polar compound is ________ x \(10^{-2}\)
View Solution
80 mole percent of MgCl\(_2\) is dissociated in aqueous solution. The vapour pressure of 1.0 molal aqueous solution of MgCl\(_2\) at 38°C is ______ mm Hg. (Nearest integer)
View Solution
The value of \( x \) in compound \( D \) is
At 600K, the root mean square (rms) speed of gas X (molar mass = 40) is equal to the most probable speed of gas Y at 90K. The molar mass of gas Y is ______ g/mol (Nearest integer).
View Solution
The reaction \( 2NO + Br_2 \to 2NOBr \) takes place through the mechanism given below: \[ NO + Br_2 \leftrightarrow NOB_2 \quad (fast) \] \[ NOB_2 + NO \to 2NOBr \quad (slow) \]
The overall order of the reaction is _______.
View Solution
Values of work function (\(W_0\)) for a few metals are given below.
The number of metals which will show the photoelectric effect when light of wavelength 400 nm falls on it is
View Solution






















Also Check:
JEE Main 12 April 2023 Shift 1 Question Paper with Answer Key: Coaching Institute PDF
| Coaching Institutes | Question Paper with Answer Key PDF |
|---|---|
| Aakash BYJUs | Check Here |
| Reliable Institute | Physics Chemistry Mathematics |
| Resonance | Physics Chemistry Mathematics |
| Vedantu | Check Here |
| Narayana College | Physics Chemistry Mathematics |
JEE Main 2023 Paper Analysis 12 April Shift 1
JEE Main 2023 Paper Analysis for the exam scheduled on 12 April Shift 1 is available here. Candidates can check subject-wise paper analysis for the exam scheduled on 12 April Shift 1 here along with the topics with the highest weightage.
Also Check:
JEE Main 2023 Question Paper Session 2 (April)
JEE Main 2023 Question Paper Session 1 (January)
JEE Main aspirants can practice and check their exam prep level by attempting the question papers from the January Session. The table below shows JEE Main 2023 Question Paper PDF for Session 1 to practice.
JEE Main Previous Year Question Paper
| JEE Main 2022 Question Paper | JEE Main 2021 Question Paper | JEE Main 2020 Question Paper |
| JEE Main 2019 Question Paper | JEE Main 2018 Question Paper | JEE Main 2017 Question Paper |








Comments