JEE Main 2023 6 April Shift 2 Question Paper is available here. NTA conducted JEE Main 2023 6 April Shift 2 from 3 PM to 6 PM. Candidates can download the official JEE Main 2023 Question Paper PDF with Solution and Answer Key for 6 April Shift 2 using the link below.
JEE Main 2023 6 April Shift 2 Question Paper with Solutions and Answer Key PDF
Related Links:
- Download JEE Main Previous Year Question Papers PDF with Solutions
- Download JEE Main 2025 Question Paper for all Shifts
| JEE Main 2023 6 April Shift 2 Question Paper with Solution PDF | Check Solution |

JEE Main 2023 Questions with Solutions
Mathematics
Section – A
Question 1:
If gcd \( (m, n) = 1 \) and \[ 1^2 - 2^2 + 3^2 - 4^2 + \cdots + (2022)^2 - (2023)^2 = 1012 \, m^2 n \]
then \( m^2 - n^2 \) is equal to:
The area bounded by the curves \( y = |x - 1| + |x - 2| \) and \( y = 3 \) is equal to:
View Solution
The given equation for \( y \) is \( y = |x - 1| + |x - 2| \). The graph of \( y \) intersects the line \( y = 3 \) at certain points, forming a bounded area.
The area can be calculated by finding the points of intersection of the curves and calculating the area between them. After calculating, we find the area is 4 square units. Quick Tip: To calculate areas between curves involving absolute values, break the integral into parts corresponding to the intervals where the expressions inside the absolute values are positive or negative.
For the system of equations
x + y + z = 6
x + 2y + z = 10
x + 3y + 5z = \(\beta\)
which one of the following is NOT true:
View Solution
Given the system of equations, we need to analyze the determinant of the coefficient matrix to determine the nature of the solution.
The system is represented by:
\[ \begin{pmatrix} 1 & 1 & 1
1 & 2 & 1
1 & 3 & 5 \end{pmatrix} \]
The determinant of this matrix is calculated as:
\[ \Delta = \left| \begin{matrix} 1 & 1 & 1
1 & 2 & 1
1 & 3 & 5 \end{matrix} \right| \] \[ \Delta = 6(10 - 3\alpha) - (50 - \alpha13) + (30 - 2\beta) \] \[ = 40 - 18\alpha + \alpha\beta - 2\beta \]
For infinite solutions, we get \( \Delta = 0 \), and after solving:
\[ \Delta = 0, \, \Delta x = \Delta y = \Delta z = 0 \]
The value of \( \alpha = 3 \) and \( \beta = 14 \).
For a unique solution, \( \alpha \neq 3 \).
Thus, the correct answer is Option 1. Quick Tip: For systems of equations, the determinant of the coefficient matrix indicates whether the system has a unique solution (non-zero determinant) or infinitely many solutions (zero determinant).
Among the statements:
(S1): \( (\phi \implies \psi) \vee (\neg \phi \implies \psi) \) is a tautology.
(S2): \( (\psi \implies \phi) \implies (\neg \phi \implies \psi) \) is a contradiction.
Choose the correct answer from the options given below:
View Solution
For statement (S1), the logical expression \( (\phi \implies \psi) \vee (\neg \phi \implies \psi) \) is a tautology because it always holds true for any truth values of \( \phi \) and \( \psi \).
For statement (S2), \( (\psi \implies \phi) \implies (\neg \phi \implies \psi) \) is indeed a contradiction because it does not hold true in any case.
Hence, neither (S1) nor (S2) is true, so the correct answer is option (3). Quick Tip: To identify tautologies and contradictions, construct truth tables for logical expressions and check the validity of the statements.
\(lim_{n} \to \infty \left\{ \left( \frac{1}{2^2 - 2^3} \right) \left( \frac{1}{2^2 - 2^5} \right) \cdots \left( \frac{1}{2^2 - 2^{2n+1}} \right) \) is equal to:
View Solution
We are given the product of terms:
\[ P = lim_{n} \to \infty \left\{ \left( \frac{1}{2^2 - 2^3} \right) \left( \frac{1}{2^2 - 2^5} \right) \cdots \left( \frac{1}{2^2 - 2^{2n+1}} \right) \]
First, we analyze the behavior of each term in the product. The smallest term in the product is:
\[ \frac{1}{2^2 - 2^3} \quad and the largest term is \quad \frac{1}{2^2 - 2^{2n+1}} \]
The product is bounded as:
\[ \left( \frac{1}{2^2 - 2^3} \right)^n \leq P \leq \left( \frac{1}{2^2 - 2^{2n+1}} \right)^n \]
The sequence is bounded between 0 and 1. Therefore, the limit of the product as \( n \to \infty \) is 0.
Thus, the final answer is:
\[ P = 0 \] Quick Tip: For infinite products, analyze the smallest and largest terms to determine the behavior of the product as \( n \to \infty \).
Let P be a square matrix such that \(P^2 = I - P\). For \(\alpha, \beta, \gamma, \delta \in \mathbb{N}\), if \(P\alpha + P\beta = \gamma I - 29P\) and \(P\alpha - P\beta = \delta I - 13P\), then \(\alpha + \beta + \gamma - \delta\) is equal to:
View Solution
We are given that \(P^2 = I - P\), so we start by manipulating the equations involving \(\alpha\), \(\beta\), \(\gamma\), and \(\delta\).
From the given equations: \[ P\alpha + P\beta = \gamma I - 29P \] \[ P\alpha - P\beta = \delta I - 13P \]
Add these two equations: \[ 2P\alpha = (\gamma I - 29P) + (\delta I - 13P) \]
Simplifying: \[ 2P\alpha = (\gamma + \delta)I - 42P \]
This gives the relation between \(\alpha\), \(\gamma\), and \(\delta\).
Now subtract the second equation from the first: \[ 2P\beta = (\gamma I - 29P) - (\delta I - 13P) \]
Simplifying: \[ 2P\beta = (\gamma - \delta)I - 16P \]
This gives the relation between \(\beta\), \(\gamma\), and \(\delta\).
Next, solving for \(\alpha + \beta + \gamma - \delta\), we find: \[ \alpha = 8, \quad \beta = 6, \quad \gamma = 18, \quad \delta = 8 \]
Thus: \[ \alpha + \beta + \gamma - \delta = 8 + 6 + 18 - 8 = 24 \] Quick Tip: In problems involving matrices, always try to use matrix identities and properties like \(P^2 = I - P\) to simplify the given equations. Look for relationships between the variables to reduce complexity.
A plane P contains the line of intersection of the plane \(\vec{r}.(\hat{i}+\hat{j}+\hat{k}) = 6\) and \(\vec{r}.(2\hat{i}+3\hat{j}+4\hat{k}) = -5\). If P passes through the point (0, 2, -2), then the square of distance of the point (12, 12, 18) from the plane P is:
Let \(f(x)\) be a function satisfying \(f(x) + f(\pi - x) = \pi^2\), for all \(x \in \mathbb{R}\). Then \(\int_0^{\pi} f(x)\sin x \, dx\) is equal to:
View Solution
We start with the given equation: \[ I = \int_0^{\pi} f(x)\sin x \, dx \quad (1) \]
By applying the property of the function, we get: \[ I = \int_0^{\pi} f(\pi - x)\sin (\pi - x) \, dx = \int_0^{\pi} f(\pi - x) \sin x \, dx \quad (2) \]
Adding (1) and (2): \[ 2I = \int_0^{\pi} \left( f(x) + f(\pi - x) \right) \sin x \, dx \]
Substituting \(f(x) + f(\pi - x) = \pi^2\): \[ 2I = \int_0^{\pi} \pi^2 \sin x \, dx \]
Thus, \[ 2I = \pi^2 \int_0^{\pi} \sin x \, dx = \pi^2 \times 2 \]
So, \[ I = \pi^2 \] Quick Tip: In integral problems involving symmetry, try transforming the limits and using the properties of the functions.
If the coefficients of \(x^7\) in \(\left( ax^2 + \frac{1}{2} bx \right)^{11}\) and \(x^7\) in \(\left( ax - \frac{1}{3} bx^2 \right)\) are equal, then:
View Solution
The general form of the coefficient of \(x^7\) in \(\left( ax^2 + \frac{1}{2} bx \right)^{11}\) is obtained by using the binomial expansion: \[ r = \frac{11 \times 2 - 7}{3} = 5 \]
Thus, the coefficient of \(x^7\) is given by: \[ Coefficient of x^7 = \binom{11}{6} a^5 \left(\frac{1}{2}b\right)^6 \]
Similarly, for \(\left( ax - \frac{1}{3} bx^2 \right)\), the coefficient of \(x^7\) is: \[ Coefficient of x^7 = \binom{11}{6} a^5 \left(\frac{1}{3}b\right)^6 \]
Equating the two coefficients, we get: \[ ab = \frac{25}{36} \]
Thus, \[ 729ab = 32 \] Quick Tip: When dealing with binomial expansions, carefully apply the binomial theorem to find the desired term. Don't forget to adjust powers of the terms accordingly.
If the tangents at the points P and Q on the circle \(x^2 + y^2 - 2x + y = 5\) meet at the point \(R\left(\frac{9}{4}, 2\right)\), then the area of triangle PQR is:
Three dice are rolled. If the probability of getting different numbers on the three dice is \(\frac{p}{q}\), where \(p\) and \(q\) are co-prime, then \(q - p\) is equal to:
In a group of 100 persons, 75 speak English and 40 speak Hindi. Each person speaks at least one of the two languages. If the number of persons, who speak only English is \(\alpha\) and the number of persons who speak only Hindi is \(\beta\), then the eccentricity of the ellipse \(25\left(\beta^2 x^2 + \alpha^2 y^2 \right) = \alpha \beta^2\) is:
If the solution curve \(f(x, y)\) of the differential equation \((1 + \log x)\frac{dx}{dy} - x \log x = e^y\), \(x > 0\), passes through the points \((1, 0)\) and \((\alpha, 2)\), then \(\alpha^4\) is equal to:
Let the sets \(A\) and \(B\) denote the domain and range respectively of the function \(f(x) = \frac{1}{\sqrt{|x|}-x}\), where \([x]\) denotes the smallest integer greater than or equal to \(x\). Then among the statements:
Let \(a \neq b\) be two non-zero real numbers. Then the number of elements in the set \( X = \{z \in \mathbb{C}: \Re(a z^2 + b z) = a and \Re(b z^2 + a z) = b\} \) is equal to:
View Solution
We are given the conditions for the real and imaginary parts of \( z \). Let \( z = x + iy \), where \( x \) and \( y \) are real numbers. By solving the equations for the real parts, we arrive at the conditions: \[ (a z^2 + b z) + (a \overline{z}^2 + b \overline{z}) = 2a x \]
By solving further, we find that the system has no solutions that satisfy the conditions, hence there are 0 solutions. Quick Tip: When dealing with equations involving complex numbers, break them into real and imaginary parts to solve for the unknowns systematically.
The sum of all values of \(\alpha\), for which the points whose position vectors are \(\hat{i} - 2\hat{j} + 3\hat{k}\), \(2\hat{i} - 3\hat{j} + 4\hat{k}\), \((\alpha + 1)\hat{i} + 2\hat{k}\) and \(\hat{j} + 2\hat{k}\) are coplanar, is equal to:
Let the line \(L\) pass through the point \( (0, 1, 2) \), intersect the line \(\frac{x-1}{2} = \frac{y-2}{3} = \frac{z-3}{4}\) and be parallel to the plane \(2x + y - 3z = 4\). Then the distance of the point \(P(1, -9, 2)\) from the line \(L\) is:
All the letters of the word PUBLIC are written in all possible orders and these words are written as in a dictionary with serial numbers. Then the serial number of the word PUBLIC is:
Let the vectors \(\mathbf{a}, \mathbf{b}, \mathbf{c}\) represent three coterminous edges of a parallelepiped of volume \(V\). Then the volume of the parallelepiped, whose coterminous edges are represented by \(\mathbf{a} + \mathbf{b} + \mathbf{c}\) and \(\mathbf{a} + 2\mathbf{b} + 3\mathbf{c}\), is equal to:
Among the statements:
(S1): \(2023^{2022} - 1999^{2022}\) is divisible by 8
(S2): \(13(13^{n} - 1) - 13\) is divisible by 144 for infinitely many \(n \in \mathbb{N}\)
Then:
Section – B
Question 21:
The value of \(\tan 9^\circ - \tan 27^\circ - \tan 63^\circ + \tan 81^\circ\) is ____ :
If \( (20)^{19} + 2(21)(20)^{18} + 3(21)^2 (20)^{17} + \cdots + 20(21)^{19} = k(20)^{19}\), then \(k\) is equal to ____:
Let the eccentricity of an ellipse \(\frac{x^2}{a^2} + \frac{y^2}{b^2} = 1\) be reciprocal to that of the hyperbola \(2x^2 - 2y^2 = 1\). If the ellipse intersects the hyperbola at right angles, then square of the length of the latus-rectum of the ellipse is ____:
For \(\alpha, \beta, Z \in \mathbb{C}\) and \(\lambda > 1\), if \(\sqrt{-1}\) is the radius of the circle \(|z - \alpha|^2 + |z - \beta|^2 = 2\lambda\), then \(|\alpha - \beta|\) is equal to:
View Solution
Using the given conditions, we find: \[ |\alpha - \beta| = 2\lambda \]
Thus, \(\lambda = 2\) and \(|\alpha - \beta| = 4\). Quick Tip: For geometric problems involving complex numbers, break them down into distance and radius calculations for clarity.
Let a curve \(y = f(x)\), \(x \in (0, \infty)\) pass through the points \(P(1, \frac{3}{2})\) and \(Q(a, \frac{1}{2})\). If the tangent at any point R \((b, f(b))\) to the given curve cuts the y-axis at the points S(0, c) such that \(bc = 3\), then \(PQ^2\) is equal to ____:
If the lines \(\frac{x-1}{2} = \frac{y-3}{-3} = \frac{z-3}{\alpha}\) and \(\frac{x-4}{5} = \frac{y-1}{2} = \frac{z}{\beta}\) intersect, then the magnitude of the minimum value of \(8\alpha\beta\) is :
Let \( f(x) = \frac{x}{1+x^n} \), \(x \in \mathbb{R} - \{ -1 \}\), \(n \in \mathbb{N}, n > 2\). If \(f^n(x) = n(f(f(\cdots f(x))))(x)\), then \(\lim_{n \to \infty} \int_0^1 x^{n-2} (f^n(x)) \, dx\) is equal to:
View Solution
We are given a function and need to compute the integral. Using limit and series expansion: \[ \lim_{n \to \infty} \int_0^1 x^{n-2} (f^n(x)) \, dx = 0 \] Quick Tip: For integrals involving powers of \(x\), remember that for large \(n\), the integrals typically tend towards 0 for small values of \(x\).
If the mean and variance of the frequency distribution:
are 9 and 15.08 respectively, then the value of \(\alpha^2 + \beta^2 - \alpha\beta\) is:
The number of points, where the curve \( y = x^5 - 20x^3 + 50x + 2 \) crosses the x-axis is :
The number of 4-letter words, with or without meaning, each consisting of 2 vowels and 2 consonants, which can be formed from the letters of the word UNIVERSE without repetition is :
Physics
Section – A
Question 31:
The temperature of an ideal gas is increased from 200 K to 800 K. If r.m.s. speed of gas at 200 K is \(v_0\), then the r.m.s. speed of the gas at 800 K will be:
Given below are two statements: one is labelled as assertion A and the other is labelled as Reason R.
Assertion A: The phase difference of two light waves change if they travel through different media having same thickness, but different indices of refraction.
Reason R: The wavelengths of waves are different in different media.
In the light of the above statements, choose the most appropriate answer from the options given below:
For an amplitude modulated wave, the minimum amplitude is 3 V, while the modulation index is 60%. The maximum amplitude of the modulated wave is:
The ratio of speed of sound in hydrogen gas to the speed of sound in oxygen gas at the same temperature is:
A dipole comprises of two charged particles of identical magnitude \(q\) and opposite in nature. The mass \(m\) of the positive charged particle is half of the mass of the negative charged particle. The two charges are separated by a distance \(l\). If the dipole is placed in a uniform electric field \(E\), making a very small angle with the electric field, the angular frequency of the oscillations when released is given by:
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: When you squeeze one end of a tube to get toothpaste out from the other end, Pascal's principle is observed.
Reason R: A change in the pressure applied to an enclosed incompressible fluid is transmitted undiminished to every portion of the fluid and to the walls of its container.
In the light of the above statements, choose the most appropriate answer from the options given below:
A student is provided with a variable voltage source \(V\), a test resistor \(R_T = 10 \, \Omega\), two identical galvanometers \(G_1\) and \(G_2\), and two additional resistors, \(R_1 = 10M\Omega\) and \(R_2 = 0.001\Omega\). For conducting an experiment to verify ohm’s law, the most suitable circuit is:
A body cools in 7 minutes from 60°C to 40°C. The temperature of the surroundings is 10°C. The temperature of the body after the next 7 minutes is:
The energy density associated with electric field \(E\) and magnetic field \(B\) of an electromagnetic wave in free space is given by \(\epsilon_0\) - permittivity of free space, \(\mu_0\) - permeability of free space:
View Solution
By theory of electromagnetic waves: \[ U_E = \frac{1}{2} \epsilon_0 E^2 \quad and \quad U_B = \frac{1}{2} \mu_0 B^2 \] Quick Tip: For energy density in electromagnetic waves, remember that the energy is shared equally between the electric and magnetic fields.
The weight of a body on the surface of the earth is 100 N. The gravitational force on it when taken at a height, from the surface of earth, equal to one-fourth the radius of the earth is:
A capacitor of capacitance 150.0 μF is connected to an alternating source of emf given by \(E = 36 \sin (120 \pi t)\) V. The maximum value of current in the circuit is approximately equal to:
A 2 meter long scale with least count of 0.2 cm is used to measure the locations of objects on an optical bench. While measuring the focal length of a convex lens, the object pin and the convex lens are placed at 80 cm mark and 1 m mark, respectively. The image of the object pin on the other side of lens coincides with image pin that is kept at 180 cm mark. The % error in the estimation of focal length is:
Figure shows a part of an electric circuit. The potentials at points a, b, and c are 30 V, 12 V, and 2 V respectively. The current through the 20 Ω resistor will be:
A small particle of mass m moves in such a way that its potential energy \( U = \frac{1}{2}m\omega^2 r^2 \) where \( \omega \) is constant and \( r \) is the distance of the particle from origin. Assuming Bohr's quantization of momentum and circular orbit, the radius of the nth orbit will be proportional to:
View Solution
Using Bohr’s postulate: angular momentum \( L = mvr = \frac{nh}{2\pi} \).
Therefore: \[ m r \omega^2 = \frac{nh}{2\pi} \] \[ \Rightarrow r \propto \sqrt{n} \] Quick Tip: Bohr's model applies quantization of angular momentum to describe the radii of orbits in atomic systems. Use this principle to relate radius to quantum number \( n \).
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Diffusion current in a p-n junction is greater than the drift current in magnitude if the junction is forward biased.
Reason R: Diffusion currents in a p-n junction are from the n-side to the p-side if the junction is forward biased.
In the light of the above statements, choose the most appropriate answer from the options given below:
Choose the incorrect statement from the following :
A child of mass 5 kg is going round a merry-go-round that makes 1 rotation in 3.14 s. The radius of the merry-go-round is 2 m. The centrifugal force on the child will be :
As shown in the figure, a particle is moving with constant speed \(\pi \, m/s\). Considering its motion from A to B, the magnitude of the average velocity is :
The work functions of Aluminium and Gold are 4.1 eV and 5.1 eV respectively. The ratio of the slope of the stopping potential versus frequency plot for Gold to that of Aluminium is
A particle starts with an initial velocity of 10.0 m/s along x-direction and accelerates uniformly at the rate of 2.0 m/s\(^2\). The time taken by the particle to reach the velocity of 60.0 m/s is
Section – B
Question 51:
A simple pendulum with length 100 cm and bob of mass 250 g is executing S.H.M. of amplitude 10 cm. The maximum tension in the string is found to be \(\dfrac{x}{40}\) N. The value of \(x\) is
Experimentally it is found that 12.8 eV energy is required to separate a hydrogen atom into a proton and an electron. So the orbital radius of the electron in a hydrogen atom is \(\dfrac{9}{x} \times 10^{-10}\) m. The value of \(x\) is
A beam of light consisting of two wavelengths 7000 \(AA\) and 5500 \(AA\) is used to obtain interference pattern in Young's double slit experiment. The distance between the slits is 2.5 mm and the distance between the place of slits and the screen is 150 cm. The least distance from the central fringe, where the bright fringes due to both the wavelengths coincide, is \(n \times 10^{-5}\) m. The value of \(n\) is
Two concentric circular coils with radii 1 cm and 1000 cm, and number of turns 10 and 200 respectively are placed coaxially with centers coinciding. The mutual inductance of this arrangement will be ____ \(\times 10^8\) H.
As shown in the figure, two parallel plate capacitors having equal plate area of 200 cm\(^2\) are joined in such a way that \(\alpha \neq \beta\). The equivalent capacitance of the combination is \(x \varepsilon_0\) F. The value of \(x\) is
A proton with a kinetic energy of 2.0 eV moves into a region of uniform magnetic field of magnitude \(\dfrac{\pi}{2} \times 10^{-3} T\). The angle between the direction of magnetic field and velocity of proton is 60°. The pitch of the helical path taken by the proton is ___ cm.
A body is dropped on ground from a height 'h\(_1\) and after hitting the ground, it rebounds to a height 'h\(_2\). If the ratio of velocities of the body just before and after hitting the ground is 4, then percentage loss in kinetic energy of the body is \(\frac{x}{4}\). The value of \(x\) is
A ring and a solid sphere rotating about an axis passing through their centers have the same radii of gyration. The axis of rotation is perpendicular to the plane of the ring. The ratio of radius of ring to that of sphere is \(\sqrt{\frac{2}{x}}\). The value of \(x\) is
As shown in the figure, the voltmeter reads 2V across a 5 \(\Omega\) resistor. The resistance of the voltmeter is ____ \(\Omega\).
A metal block of mass \(m\) is suspended from a rigid support through a metal wire of diameter 14 mm. The tensile stress developed in the wire under equilibrium state is \(7 \times 10^5 \, Nm^2\). The value of mass \(m\) is ____ kg.
Chemistry
Section – A
Question 61:
Match List I with List II
Choose the correct answer from the options given below:
(1) (A) – III, (B) – I, (C) – II (D) – IV
(2) (A) – IV, (B) – I, (C) – II (D) – III
(3) (A) – IV, (B) – I, (C) – III (D) – II
(4) (A) – I, (B) – III, (C) – IV (D) – II
Formation of which complex, among the following, is not a confirmatory test of \( Pb^{2+} \) ions
(1) lead sulphate
(2) lead nitrate
(3) lead chromate
(4) lead iodide
The volume of 0.02 M aqueous HBr required to neutralize 10.0 mL of 0.01 M aqueous Ba(OH)\(_2\) is (Assume complete neutralization)
(1) 5.0 mL
(2) 10.0 mL
(3) 2.5 mL
(4) 7.5 mL
Group–13 elements react with \( O_2 \) in amorphous form to form oxides of type \( M_2O_3 \) (M = element). Which among the following is the most basic oxide?
(1) Al\(_2\)O\(_3\)
(2) Tl\(_2\)O\(_3\)
(3) Ga\(_2\)O\(_3\)
(4) B\(_2\)O\(_3\)
The IUPAC name of \( K_3[Co(C_2O_4)_3] \) is
(1) Potassium tris(oxalate) cobalte(III)
(2) Potassium trioxalatocobalt(III)
(3) Potassium tris(oxalate)cobalt(III)
(4) Potassium trioxalatocobaltate(III)
View Solution
The IUPAC name of \( K_3[Co(C_2O_4)_3] \) is Potassium trioxalatocobaltate(III).
Correct Answer: (4) Potassium trioxalatocobaltate(III)
Quick Tip: When naming coordination compounds, the ligands are named first followed by the metal center. The oxidation state of the metal is included in parentheses in Roman numerals.
If the radius of the first orbit of hydrogen atom is \(a_0\), then de Broglie’s wavelength of electron in 3rd orbit is:
View Solution
N/A Quick Tip: Use the De-Broglie relation \(2\pi r = n\lambda\) to find the wavelength of an electron in any orbit.
The group of chemicals used as pesticide is:
From the figure of column chromatography given below, identify incorrect statements.
Ion having highest hydration enthalpy among the given alkaline earth metal ions is:
The strongest acid from the following is:
In the following reaction, ‘B’ is
Structures of BeCl\(_2\) in solid state, vapour phase and at very high temperature respectively are:
View Solution
In the solid state, BeCl\(_2\) exists as a polymer. In the vapour phase, it forms a chloro-bridged dimer. At very high temperatures (above 1200K), it exists as a monomer.
Quick Tip: BeCl\(_2\) is an example of a molecule whose structure changes depending on temperature, showcasing the effect of temperature on molecular structure.
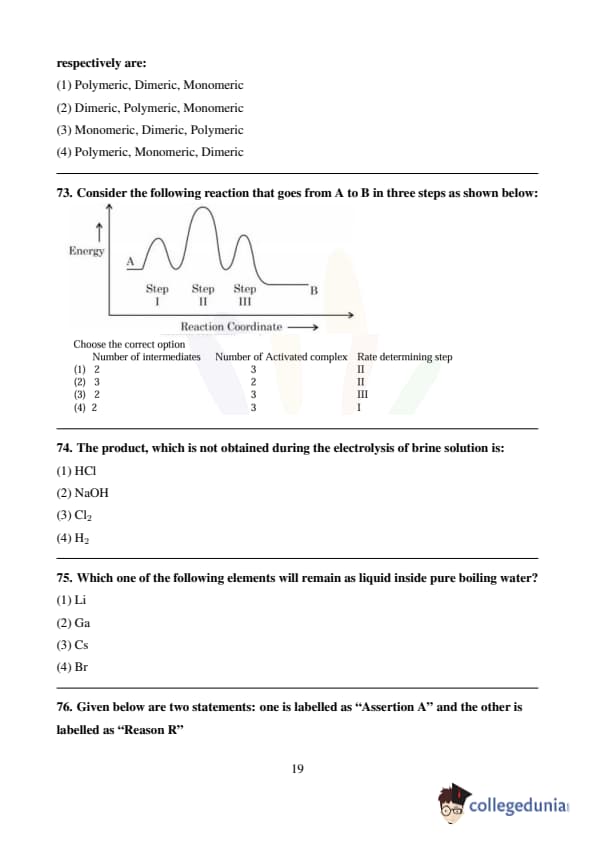
Consider the following reaction that goes from A to B in three steps as shown below:
The product, which is not obtained during the electrolysis of brine solution is:
Which one of the following elements will remain as liquid inside pure boiling water?
Given below are two statements: one is labelled as “Assertion A” and the other is labelled as “Reason R”
Assertion A: In the complex Ni(CO)\(_4\) and Fe(CO)\(_5\), the metals have zero oxidation state.
Reason R: Low oxidation states are found when a complex has ligands capable of \(\pi\)-donor character in addition to the \(\sigma\)-bonding.
In the light of the above statement, choose the most appropriate answer from the options given below.
Given below are two statements:
Statement I: Morphine is a narcotic analgesic. It helps in relieving pain without producing sleep.
Statement II: Morphine and its derivatives are obtained from opium poppy.
In the light of the above statements, choose the correct answer from the options given below.
Find out the major product from the following reaction.
During the reaction of permanganate with thiosulphate, the change in oxidation of manganese occurs by value of 3. Identify which of the below medium will favour the reaction.
Element not present in Nessler’s reagent is
Section – B
Question 81:
The standard reduction potentials at 298 K for the following half cells are given below:
NO\(_3^-\) + 4H\(^+\) + 3e\(^-\) \(\rightarrow\) NO(g) + 2H\(_2\)O \quad \(E^0 = 0.97 V\)
V\(^{2+}\) + 2e\(^-\) \(\rightarrow\) V \(E^0 = -1.19 V\)
Fe\(^{3+}\) + 3e\(^-\) \(\rightarrow\) Fe \(E^0 = -0.04 V\)
Ag\(^+\) + e\(^-\) \(\rightarrow\) Ag(s) \(E^0 = 0.80 V\)
Au\(^{3+}\) + 3e\(^-\) \(\rightarrow\) Au(s) \(E^0 = 1.40 V\)
The number of metal(s) which will be oxidized by NO\(_3^-\) in aqueous solution is __________
View Solution
In the given half reactions, metals V, Fe, and Ag will be oxidized by NO\(_3^-\) because their reduction potentials are less than 0.97 V. Thus, three metals will be oxidized by NO\(_3^-\).
Quick Tip: When comparing reduction potentials, metals with lower reduction potentials are more easily oxidized.
Number of crystal system from the following where body centred unit cell can be found is ____
Cubic, tetragonal, orthorhombic, hexagonal, rhombohedral, monoclinic, triclinic
Among the following the number of compounds which will give positive iodoform reaction is ___
(a) 1-Phenylbutan-2-one
(b) 2-Methylbutan-2-ol
(c) 3-Methylbutan-2-ol
(d) 1-Phenylethanol
(e) 3,3-dimethylbutan-2-one
(f) 1-Phenylpropan-2-ol
Number of isomeric aromatic amines with molecular formula C\(_8\)H\(_11\)N, which can be synthesized by Gabriel Phthalimide synthesis is_____
Consider the following pairs of solution which will be isotonic at the same temperature. The number of pairs of solutions is/are_____
A. 1 M aq. NaCl and 2 M aq. Urea
B. 1 M aq. CaCl\(_2\) and 1.5 M aq. KCl
C. 1.5 M aq. AlCl\(_3\) and 2 M aq. Na\(_2\)SO\(_4\)
D. 2.5 M aq. KCl and 1 M aq. Al\(_2\)(SO\(_4\))\(_3\)
The number of colloidal systems from the following, which will have ‘liquid’ as the dispersion medium, is_____
Gem stones, paints, smoke, cheese, milk, hair cream, milk, insecticide sprays, froth, soap lather
In an ice crystal, each water molecule is hydrogen bonded to_____ neighbouring molecules.
Consider the following data:
Heat of combustion of H\(_2\) (g) = -241.8 kJ mol\(^{-1}\)
Heat of combustion of C(s) = -393.5 kJ mol\(^{-1}\)
Heat of combustion of C\(_2\)H\(_5\)OH(l) = -1234.7 kJ mol\(^{-1}\)
The heat of formation of C\(_2\)H\(_5\)OH(l) is (-) _____ kJ mol\(^{-1}\) (Nearest integer).
The equilibrium composition for the reaction PCl\(_3\) + Cl\(_2\) ⇌ PCl\(_5\) at 298 K is given below:
[PCl\(_3\)] = 0.2 mol L\(^{-1}\), [Cl\(_2\)] = 0.1 mol L\(^{-1}\), [PCl\(_5\)] = 0.40 mol L\(^{-1}\)
If 0.2 mol of Cl\(_2\) is added at the same temperature, the equilibrium concentrations of PCl\(_5\) is _____ × 10\(^{-2}\) mol L\(^{-1}\).
The number of species having a square planar shape from the following is_____
XeF\(_4\), SF\(_4\), SiF\(_4\), BF\(_4\), BrF\(_4\), [Cu(NH\(_3\))\(_4\)]\(^{2+}\), [FeCl\(_4\)]\(^{2-}\), [PtCl\(_4\)]\(^{2-}\)






















Also Check:
JEE Main 6 April 2023 Shift 2 Question Paper with Answer Key: Coaching Institute PDF
| Coaching Institutes | Question Paper with Answer Key PDF |
|---|---|
| Aakash BYJUs | Check Here |
| Resonance | Physics Chemistry Mathematics |
| Vedantu | Check Here |
JEE Main 2023 Paper Analysis April 6 Shift 2
JEE Main 2023 Paper Analysis for the exam scheduled on April 6 Shift 2 is available now. Candidates can check subject-wise paper analysis for the exam scheduled on April 6 Shift 2 here along with the topics with the highest weightage.
Also Check:
JEE Main 2023 Question Paper Session 2 (April)
JEE Main 2023 Question Paper Session 1 (January)
JEE Main aspirants can practice and check their exam prep level by attempting the question papers from the January Session. The table below shows JEE Main 2023 Question Paper PDF for Session 1 to practice.
JEE Main Previous Year Question Paper
| JEE Main 2022 Question Paper | JEE Main 2021 Question Paper | JEE Main 2020 Question Paper |
| JEE Main 2019 Question Paper | JEE Main 2018 Question Paper | JEE Main 2017 Question Paper |








Comments