JEE Main 2023 Jan 24 Shift 2 Question Paper is available here. NTA successfully conducted JEE Main 2023 Jan 24 Shift 2 from 3 PM to 6 PM for B.E./B.Tech paper. According to experts, the questions of JEE Main 2023 Shift 2 were easier and less time-consuming as compared to those asked in JEE Main 2023 Shift 1 held on January 24, 2023. JEE Main 2023 Paper Analysis showed that even in JEE Main 2023 Jan 24 Shift 2 the mathematics section was more difficult as compared to the chemistry and physics sections. Candidates can download the JEE Main 2023 Question Paper PDF with Solution and Answer Key for Jan 24 Shift 2 using the links below.
Also Check: JEE Main 2025 Jan 24 Question Paper for Shift 2 and Shift 1 Here
Related Links:
- Download JEE Main Previous Year Question Papers PDF with Solutions
- Download JEE Main 2025 Question Paper for all Shifts
JEE Main 2023 Question Paper Jan 24 Shift 2- Download PDF
| JEE Main 2023 Jan 24 Shift 2 Question Paper with Solution PDF | Check Solution |

The electric potential at the centre of two concentric half rings of radii \( R_1 \) and \( R_2 \), having the same linear charge density \( \lambda \) is:
View Solution
Let \( \gamma_1 \) be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and \( \gamma_2 \) be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio \( \frac{\gamma_1}{\gamma_2} \) is:
View Solution
An \( \alpha \)-particle, a proton and an electron have the same kinetic energy. Which one of the following is correct in case of their De-Broglie wavelength:
View Solution
If the distance of the earth from Sun is \( 1.5 \times 10^6 \) km, then the distance of an imaginary planet from Sun, if its period of revolution is 2.83 years, is:
View Solution
Match List I with List II:
Choose the correct answer from the options given below:
View Solution
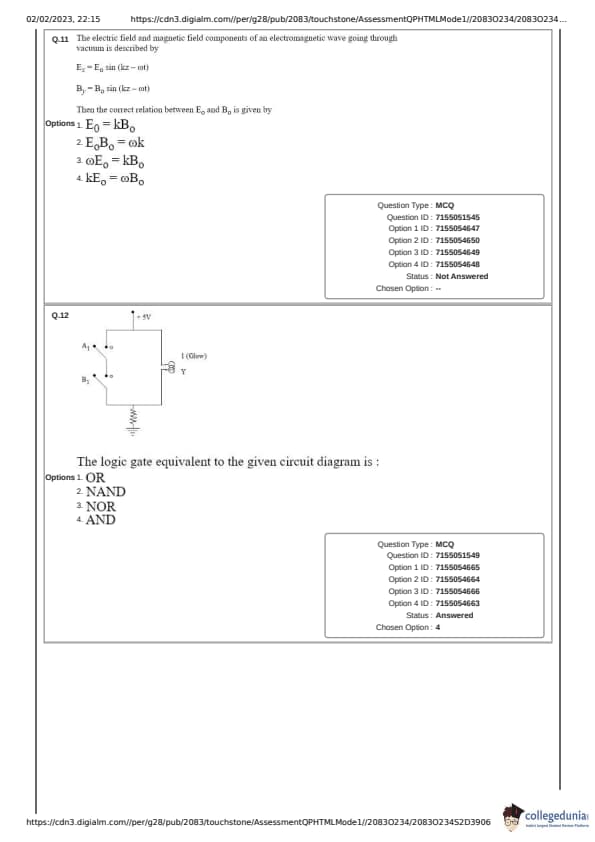
The logic gate equivalent to the given circuit diagram is:
View Solution
A long solenoid is formed by winding 70 turns cm\(^{-1}\). If 2.0 A current flows, then the magnetic field produced inside the solenoid is:
View Solution
Given below are two statements:
Statement I: Acceleration due to earth's gravity decreases as you go 'up' or 'down' from earth's surface.
Statement II: Acceleration due to earth's gravity is the same at a height \( h \) and depth \( d \) from earth's surface, if \( h = d \).
In the light of above statements, choose the most appropriate answer from the options given below:
View Solution
A metallic rod of length \( L \) is rotated with an angular speed of \( \omega \) normal to a uniform magnetic field \( B \) about an axis passing through one end of the rod as shown in the figure. The induced emf will be:
View Solution
When a beam of white light is allowed to pass through a convex lens parallel to the principal axis, the different colours of light converge at different points on the principal axis after refraction. This is called:
View Solution
The frequency (\( v \)) of an oscillating liquid drop may depend upon radius (\( r \)) of the drop, density (\( \rho \)) of liquid and the surface tension (\( s \)) of the liquid as: \[ v = r^a \rho^b s^c \]
The values of \( a \), \( b \), and \( c \) respectively are:
View Solution
A body of mass 200g is tied to a spring of spring constant 12.5 N/m, while the other end of the spring is fixed at point O. If the body moves about O in a circular path on a smooth horizontal surface with constant angular speed 5 rad/s, then the ratio of extension in the spring to its natural length will be:
View Solution
A cell of emf 90 V is connected across a series combination of two resistors each of 100\(\, \Omega\) resistance. A voltmeter of resistance 400\(\, \Omega\) is used to measure the potential difference across each resistor. The reading of the voltmeter will be:
View Solution
The electric field and magnetic field components of an electromagnetic wave going through vacuum is described by: \[ E_x = E_0 \sin (kz - \omega t) \] \[ B_y = B_0 \sin (kz - \omega t) \]
Then the correct relation between \( E_0 \) and \( B_0 \) is given by:
View Solution
The velocity-time graph of a body moving in a straight line is shown in the figure.
The ratio of displacement to distance travelled by the body in time 0 to 10s is:
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Steel is used in the construction of buildings and bridges.
Reason R: Steel is more elastic and its elastic limit is high.
In the light of above statements, choose the most appropriate answer from the options given below:
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: A pendulum clock when taken to Mount Everest becomes fast.
Reason R: The value of \( g \) (acceleration due to gravity) is less at Mount Everest than its value on the surface of the Earth.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
A photon is emitted in transition from \( n = 4 \) to \( n = 1 \) level in hydrogen atom. The corresponding wavelength for this transition is (given, \( h = 4 \times 10^{-15} \, eV \cdot s \)):
View Solution
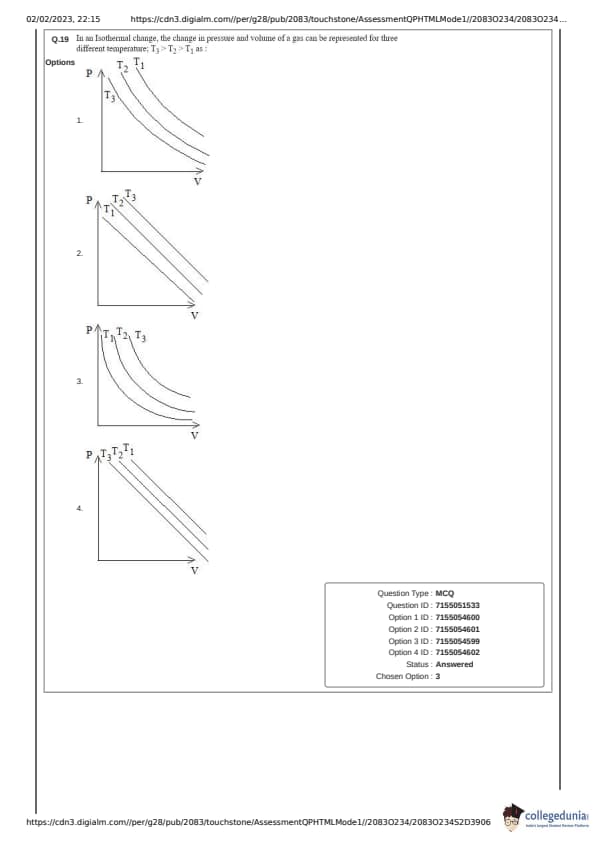
In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperatures; \( T_3 > T_2 > T_1 \) as:
If two vectors \( \mathbf{P} = \hat{i} + 2\hat{j} + m\hat{k} \) and \( \mathbf{Q} = 4\hat{i} - 2\hat{j} + \hat{k} \) are perpendicular to each other, then the value of \( m \) will be:
View Solution
A uniform solid cylinder with radius \( R \) and length \( L \) has moment of inertia \( I_1 \), about the axis of the cylinder. A concentric solid cylinder of radius \( R' = \frac{R}{2} \) and length \( L' = \frac{L}{2} \) is carved out of the original cylinder. If \( I_2 \) is the moment of inertia of the carved-out portion of the cylinder, then \( \frac{I_1}{I_2} \) is:
View Solution
A mass \( m \) attached to the free end of a spring executes SHM with a period of 1s. If the mass is increased by 3 kg, the period of oscillation increases by one second, the value of mass \( m \) is:
View Solution
The energy released per fission of nucleus of \(^{240}X\) is 200 MeV. The energy released if all the atoms in 120g of pure \(^{240}X\) undergo fission is:
View Solution
A parallel plate capacitor with air between the plates has a capacitance of 15 pF. The separation between the plates becomes twice and the space between them is filled with a material of dielectric constant 3.5. Then the capacitance becomes:
View Solution
A body of mass 1 kg begins to move under the action of a time dependent force \( \mathbf{F} = \hat{i} + 3t \hat{j} \) N. where \( \hat{i} \) and \( \hat{j} \) are the unit vectors along x and y axis. The power developed by above force, at the time \( t = 2 \) s will be:
View Solution
If a copper wire is stretched to increase its length by 20%. The percentage increase in resistance of the wire is:
View Solution
A single turn current loop in the shape of a right angle triangle with sides 5 cm, 12 cm, 13 cm is carrying a current of 2A. The loop is in a uniform magnetic field of magnitude 0.75 T whose direction is parallel to the current in the 13 cm side of the loop. The magnitude of the magnetic force on the 5 cm side will be:
View Solution
Three identical resistors with resistance \( R = 12 \, \Omega \) and two identical inductors with self inductance \( L = 5 \, mH \) are connected to an ideal battery with emf of 12 V as shown in figure. The current through the battery long after the switch has been closed will be:
View Solution
A convex lens of refractive index 1.5 and focal length 18 cm in air is immersed in water. The change in focal length of the lens will be:
(Given refractive index of water \( = \frac{4}{3} \))
View Solution
A spherical ball of radius 1mm and density 10.5 g/cc is dropped in glycerine of coefficient of viscosity 9.8 poise and density 1.5 g/cc. Viscous force on the ball when it attains constant velocity is 3696 \( \times 10^{-7} \) N. The value of \( x \) is:
View Solution
Which one amongst the following are good oxidizing agents?
A. \( Sm^{2+} \) B. \( Ce^{2+} \)
C. \( Ce^{4+} \) D. \( Tb^{4+} \)
Choose the most appropriate answer from the options given below:
What is the number of unpaired electrons(s) in the highest occupied molecular orbital of the following species: \( N_2, N_2^+, O_2, O_2^? \)
View Solution
Which of the following cannot be explained by crystal field theory?
(1) The order of spectrochemical series
(2) Magnetic properties of transition metal complexes
(3) Colour of metal complexes
(4) Stability of metal complexes
View Solution
A student has studied the decomposition of a gas \( AB_3 \) at 25°C. He obtained the following data. \[ p (mm Hg) 50, 100, 200, 400 \quad Relative t_{1/2} \quad 4, 2, 1, 0.5 \]
The order of the reaction is:
View Solution
The number of s-electrons present in an ion with 55 protons in its unipositive state is:
View Solution
In which of the following reactions the hydrogen peroxide acts as a reducing agent?
View Solution
The metal which is extracted by oxidation and subsequent reduction from its ore is:
View Solution
Given below are two statements: Statement I and Statement II.
Statement I: \( H_2N- C_6H_5 \) under Clemensen reduction conditions will give \( HOOC-C_6H_5 \).
Statement II: \( Cl- C_6H_5 \) under Wolff-Kishner reduction condition will give \( HOOC-C_6H_5 \).
In light of the above statements, choose the correct answer from the options given below:
View Solution
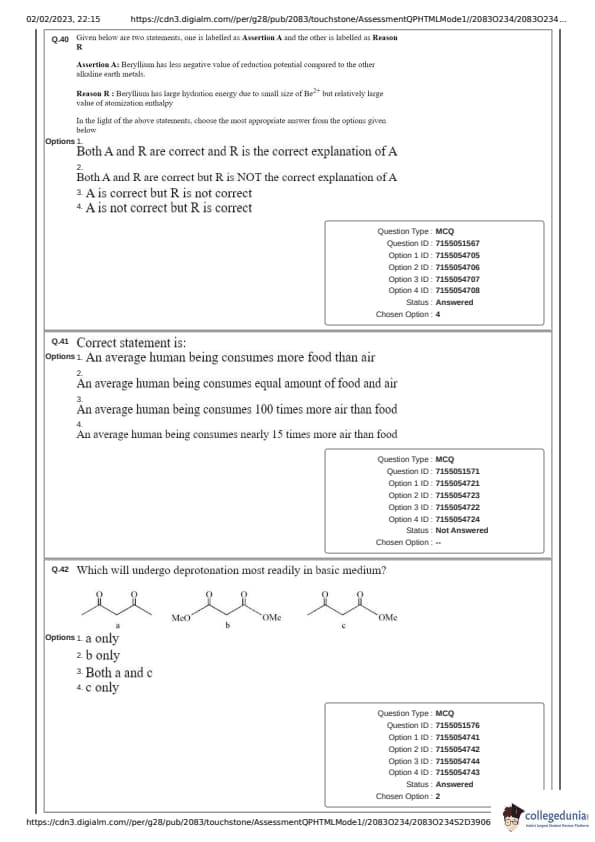
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Beryllium has less negative value of reduction potential compared to the other alkaline earth metals.
Reason R: Beryllium has large hydration energy due to small size of \( Be^{2+} \) but relatively large value of atomization enthalpy.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Match List I with List II
Choose the correct answer from the options given below:
View Solution
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Benzene is more stable than hypothetical cyclohexatriene.
Reason R: The delocalized \( \pi \)-electron cloud is attracted strongly by nuclei of carbon atoms.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
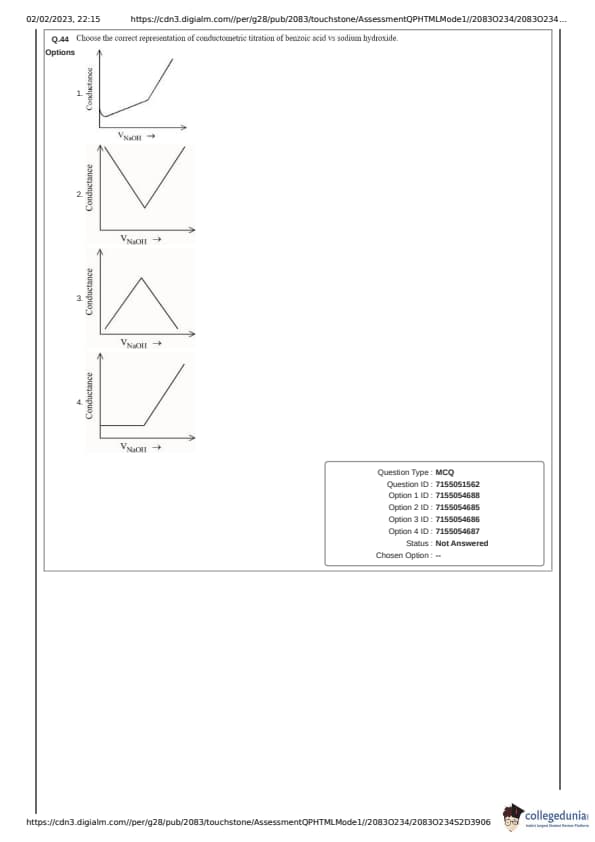
Choose the correct representation of conductometric titration of benzoic acid vs sodium hydroxide.
View Solution
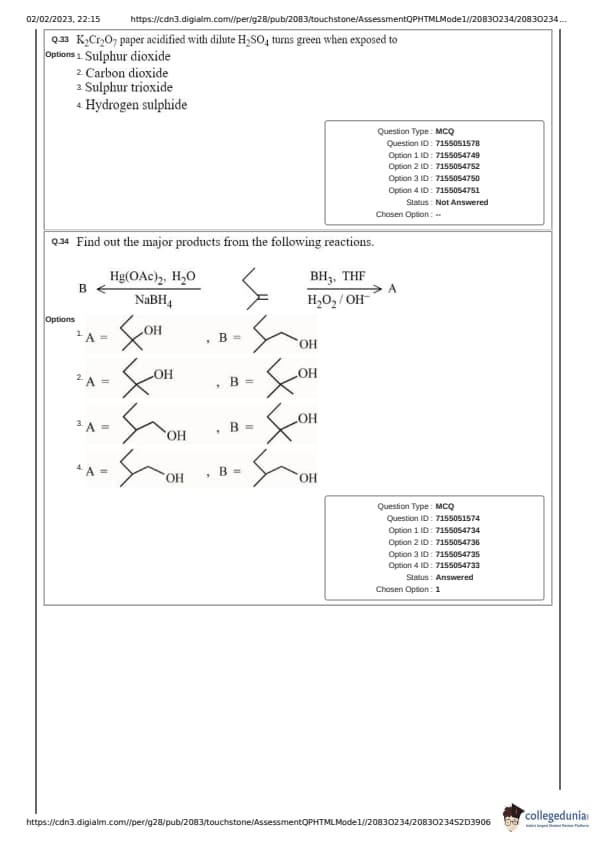
Find out the major products from the following reactions.
\[ B \xrightarrow{H_2O, NaBH_4} A \]
\[ B \xrightarrow{BH_3, THF} A \]
\[ B \xrightarrow{H_2O, OH^-} B \]
View Solution
Correct statement is:
(1) An average human being consumes more food than air
(2) An average human being consumes nearly 15 times more air than food
(3) An average human being consumes equal amount of food and air
(4) An average human being consumes 100 times more air than food
View Solution
Given below are two statements:
Statement I: Pure Aniline and other Arylamines are usually colourless.
Statement II: Arylamines get coloured on storage due to atmospheric reduction.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Which will undergo deprotonation most readily in basic medium?
View Solution
Choose the correct colour of the product for the following reaction:
View Solution
Identify the correct statements about alkali metals.
A. The order of standard reduction potential \( M^+ / M \) for alkali metal ions is Na \(>\) K \(>\) Rb \(>\) Li.
B. \( CsI \) is highly soluble in water.
C. Lithium carbonate is highly stable to heat.
D. Potassium dissolved in concentrated liquid ammonia is blue in colour and paramagnetic.
E. All the alkali metal hydrides are ionic solids.
Choose the correct answer from the options given below:
View Solution
The hybridization and magnetic behaviour of cobalt ion in \( [Co(NH_3)_6]^{3+} \) complex is:
View Solution
K_2\text{Cr_2\text{O_7 \text{ paper acidified with dilute H_2\text{SO_4 \text{ turns green when exposed to:
View Solution
The number of statement(s) which are the characteristics of physisorption is____
View Solution
Sum of \(\pi\)-bonds present in peroxodisulphuric acid and pyrosulphuric acid is:
View Solution
Maximum number of isomeric monochloro derivatives that can be obtained from 2,2,5,5-tetramethylhexane by chlorination is:
View Solution
Total number of tripeptides possible by mixing of valine and proline is:
View Solution
The number of units, which are used to express concentration of solutions from the following is: _____
Mass percent, Mole, Mole fraction, Molarity, ppm, Molarity.
View Solution
The number of statement(s), which are correct with respect to the compression of carbon dioxide from point (a) in the Andrews isotherm from the following is:
A. Carbon dioxide remains as a gas upto point (b)
B. Liquid carbon dioxide appears at point (c)
C. Liquid and gaseous carbon dioxide coexist between points (b) and (c)
D. As the volume decreases from (b) to (c), the amount of liquid decreases
View Solution
Let V.P. of pure B be \( P_0 \). When \( X_A = 0.7 \) and \( X_B = 0.3 \), the Total pressure is 350 mm Hg. If \( X_A = 0.2 \) and \( X_B = 0.8 \), the total pressure becomes 410 mm Hg. Calculate the vapor pressure of A and B.
View Solution
One mole of an ideal monatomic gas is subjected to changes as shown in the graph. The magnitude of the work done (by the system or on the system) is:
View Solution
If the pKa of lactic acid is 5, then the pH of 0.005 M calcium lactate solution at 25°C is:
View Solution
Following figure shows the spectrum of an ideal black body at four different temperatures. The number of correct statement(s) from the following is:
View Solution
Let the six numbers \( a_1, a_2, a_3, a_4, a_5, a_6 \) be in A.P., and \( a_1 + a_3 = 10 \). If the mean of these six numbers is \( \frac{19}{2} \) and their variance is \( \sigma^2 \), then \( 8\sigma^2 \) is equal to:
View Solution
Let \( f(x) \) be a function such that \( f(x + y) = f(x)f(y) \) for all \( x, y \in \mathbb{N} \). If \( f(1) = 3 \) and \( \sum_{k=1}^{n} f(k) = 3279 \), then the value of \( n \) is:
View Solution
The number of real solutions of the equation \( 3(x^2 + \frac{1}{x^2}) - 2(x+\frac{1}{x}) \), is:
View Solution
If \( f(x) = \frac{2^{2x}}{2^{2x} + 2} \), \( x \in \mathbb{R} \), then \( f\left(\frac{1}{2023}\right) + f\left(\frac{2}{2023}\right) + \ldots + f\left(\frac{2022}{2023}\right) \) is equal to:
View Solution
If \( f(x) = x^3 - x^2 f'(1) + x f''(2) - f'''(3) \), \( x \in \mathbb{R} \), then:
View Solution
The number of integers, greater than 7000 that can be formed, using the digits 3, 5, 6, 7, 8 without repetition, is:
View Solution
If the system of equations \[ x + 2y + 3z = 3 \quad \cdots (i) \] \[ 4x + 3y - 4z = 4 \quad \cdots (ii) \] \[ 8x + 4y - z = 9 + \mu \quad \cdots (iii) \]
has infinitely many solutions, then the ordered pair \( (\lambda, \mu) \) is equal to:
View Solution
The value of \[ \left( \frac{1 + \sin \frac{2\pi}{9} + i \cos \frac{2\pi}{9}}{1 + \sin \frac{2\pi}{9} - i \cos \frac{2\pi}{9}} \right)^3 \]
is:
View Solution
The equations of the sides AB and AC of a triangle ABC are: \[ (\lambda + 1)x + \lambda y = 4 \quad and \quad \lambda x + (1 - \lambda)y + \lambda = 0, \]
respectively. Its vertex A is on the y-axis and its orthocentre is \( (1, 2) \). The length of the tangent from the point C to the part of the parabola \( y^2 = 6x \) in the first quadrant is:
View Solution
The set of all values of \( a \) for which \[ \lim_{x \to a} \left([\![x - 5]\!] - [\![2x + 2]\!]\right) = 0, \]
where \( [\![x]\!] \) denotes the greatest integer less than or equal to \( x \), is equal to:
View Solution
If \[ {}({}^{30}C_1)^2+ 2({}^{30}C_2)^2 + 3({}^{30}C_2)^2 + \ldots + 30({}^{30}C_{30})^2 = \frac{\alpha \cdot 60!}{(30!)^2}, \]
then \( \alpha \) is equal to:
View Solution
Let the plane containing the line of intersection of the planes \[ P_1: x + (\lambda + 4)y + z = 1 \quad and \quad P_2: 2x + y + z = 2 \]
pass through the points \( (0, 1, 0) \) and \( (1, 0, 1) \). Then the distance of the point \( (2\lambda, \lambda, -\lambda) \) from the plane \( P_2 \) is:
View Solution
Let \( \vec{\alpha} = 4\hat{i} + 3\hat{j} + 5\hat{k} \) and \( \vec{\beta} = \hat{i} + 2\hat{j} - 4\hat{k} \). Let \( \vec{\beta}_1 \) be parallel to \( \vec{\alpha} \) and \( \vec{\beta}_2 \) be perpendicular to \( \vec{\alpha} \). If \( \vec{\beta} = \vec{\beta}_1 + \vec{\beta}_2 \), then the value of \( 5\vec{\beta}_2 \cdot (\hat{i} + \hat{j} + \hat{k}) \) is:
View Solution
The locus of the midpoints of the chords of the circle \( C_1: (x - 4)^2 + (y - 5)^2 = 4 \), which subtend an angle \( \theta_1 \) at the centre of the circle \( C_1 \), is a circle of radius \( r_1 \). If \( \theta_1 = \frac{\pi}{3} \), \( \theta_3 = \frac{2\pi}{3} \), and \( r_1^2 = r_2^2 + r_3^2 \), then \( \theta_2 \) is equal to:
View Solution
If the foot of the perpendicular drawn from \( (1, 9, 7) \) to the line passing through the point \( (3, 2, 1) \) and parallel to the planes \( x + 2y + z = 0 \) and \( 3y - z = 3 \) is \( (\alpha, \beta, \gamma) \), then \( \alpha + \beta + \gamma \) is equal to:
View Solution
Let \( y = y(x) \) be the solution of the differential equation \[ (x^2 - 3y^2)dx + 3xy \, dy = 0, \quad y(1) = 1. \]
Then \( 6y^2(e) \) is equal to:
View Solution
Let \( p \) and \( q \) be two statements. Then \( \sim(p \land (p \to \sim q)) \) is equivalent to:
View Solution
The number of square matrices of order 5 with entries from the set \( \{0, 1\} \), such that the sum of all the elements in each row is 1 and the sum of all the elements in each column is also 1, is:
View Solution
The value of the integral: \[ \int_{\frac{3\sqrt{2}}{4}}^{\frac{3\sqrt{3}}{4}} \frac{48}{\sqrt{9 - 4x^2}} \, dx \quad is equal to: \]
View Solution
Let \( A \) be a \( 3 \times 3 \) matrix such that \( |adj(adj(A))| = 12^4 \). Then \( |A^{-1}adj(A)| \) is equal to:
View Solution
The urns \( A \), \( B \), and \( C \) contain \( 4 \) red, \( 6 \) black; \( 5 \) red, \( 5 \) black, and \( \lambda \) red; \( 4 \) black balls respectively. One of the urns is selected at random, and a ball is drawn. If the ball drawn is red and the probability that it is drawn from urn \( C \) is \( 0.4 \), then the square of the length of the side of the largest equilateral triangle, inscribed in the parabola \( y^2 = \lambda x \) with one vertex at the vertex of the parabola, is:
View Solution
If the area of the region bounded by the curves \( y^2 - 2y = -x \) and \( x + y = 0 \) is \( A \), then \( 8A \) is equal to:
View Solution
If \[ \frac{1^3 + 2^3 + 3^3 + \ldots (up to \( n \) terms)}{1 \cdot 3 + 2 \cdot 5 + 3 \cdot 7 + \ldots (up to \( n \) terms)} = \frac{9}{5}, \]
then the value of \( n \) is:
View Solution
Let \( f \) be a differentiable function defined on \( \left(0, \frac{\pi}{2}\right) \) such that \( f(x) > 0 \) and \[ f(x) + \int_0^x f(t)\sqrt{1 - (\log_e f(t))^2} \, dt = e, \quad \forall x \in \left[0, \frac{\pi}{2}\right]. \]
Then \( \left(6 \log_e f\left(\frac{\pi}{6}\right)\right)^2 \) is equal to:
View Solution
The minimum number of elements that must be added to the relation \( R = \{(a, b), (b, c), (b, d)\} \) on the set \( \{a, b, c, d\} \) so that it is an equivalence relation, is:
View Solution
Let \( \mathbf{a} = \mathbf{i} + 2\mathbf{j} + \lambda \mathbf{k}, \, \mathbf{b} = 3\mathbf{i} - 5\mathbf{j} - \lambda \mathbf{k}, \, \mathbf{a} \cdot \mathbf{c} = 7, \, 2\mathbf{b} \cdot \mathbf{c} + 43 = 0, \, \mathbf{a} \times \mathbf{c} = \mathbf{b} \times \mathbf{c} \). Then \( |\mathbf{a} \cdot \mathbf{b}| \) is equal to:
View Solution
Let the sum of the coefficients of the first three terms in the expansion of \[ \left(x - \frac{3}{x^2}\right)^n, \quad x \neq 0, \, n \in \mathbb{N}, \]
be \( 376 \). Then the coefficient of \( x^4 \) is:
View Solution
If the shortest distance between the lines \[ \frac{x + \sqrt{6}}{2} = \frac{y - \sqrt{6}}{4} = \frac{z}{5}, \quad \frac{x - \lambda}{3} = \frac{y - 2\sqrt{6}}{4} = \frac{z + 2\sqrt{6}}{5} \]
is \( 6 \), then the square of the sum of all possible values of \( \lambda \) is:
View Solution
Let \( S = \{ \theta \in [0, 2\pi) : \tan(\cos \theta) + \tan(\sin \theta) = 0 \} \). Then \( \sum_{\theta \in S} \sin^2 \left(\theta + \frac{\pi}{4}\right) \) is equal to:
View Solution
The equations of the sides \( AB \), \( BC \), and \( CA \) of a triangle \( \Delta ABC \) are: \[ 2x + y = 0, \quad x + py = 21a \, (a \neq 0), \quad x - y = 3, \]
and \( P(2, a) \) is the centroid of \( \Delta ABC \). Then \( (BC)^2 \) is equal to:
View Solution



























Also Check:
JEE Main 2023 Jan 24 Shift 2 Question Paper by Coaching Institute
| Coaching Institutes | Question Paper with Answer Key PDF |
|---|---|
| Aakash BYJUs | Check Here |
| Vedantu | Check Here |
JEE Main 2023 Paper Analysis Jan 24 Shift 2
JEE Main 2023 Paper Analysis for the January 24 Shift 2 exam is updated here. Candidates can use the link provided below to check the subject-wise paper analysis for JEE Main 2023 Jan 24 Shift 2 along with the topics with the highest weightage.
| JEE Main 2023 Paper Analysis Jan 24 Shift 2 | Check Here |








Comments