JEE Main 2023 Jan 29 Shift 2 Question Paper is going to be updated here after the conclusion of the exam. NTA is going to conduct JEE Main 2023 Jan 29 Shift 2 from 3 PM to 6 PM for B.E./B.Tech paper. Candidates will be able to download the memory-based JEE Main 2023 Question Paper PDF with Solution and Answer Key for Jan 29 Shift 2 using the link below.
Related Links:
- Download JEE Main Previous Year Question Papers PDF with Solutions
- Download JEE Main 2025 Question Paper for all Shifts
JEE Main 2023 Question Paper Jan 29 Shift 2- Download PDF
| JEE Main 2023 29 Jan Shift 2 Question Paper with Solution PDF | Check Solution |

The statement \( B \Rightarrow ((\sim A) \lor B) \) is equivalent to:
Shortest distance between the lines \[ \frac{x-1}{2} = \frac{y+8}{-7} = \frac{z-4}{5} \quad and \quad \frac{x-1}{2} = \frac{y-2}{1} = \frac{z-6}{-3} \]
is:
If \( \vec{a} = \hat{i} + 2\hat{k}, \vec{b} = \hat{i} + \hat{j} + \hat{k}, \vec{c} = 7\hat{i} - 3\hat{j} + 4\hat{k} \), and \[ \vec{r} \times \vec{b} + \vec{b} \times \vec{c} = 0 \quad and \quad \vec{r} \cdot \vec{a} = 0, \]
then \( \vec{r} \cdot \vec{c} \) is equal to:
View Solution
Step 1: Simplify \( \vec{r} \times \vec{b} + \vec{b} \times \vec{c} = 0 \)
\[ \vec{r} \times \vec{b} = -\vec{b} \times \vec{c}. \]
This implies \( \vec{r} \) can be written as: \[ \vec{r} = \vec{c} + \lambda \vec{b}, \quad for some scalar \lambda. \]
Step 2: Use \( \vec{r} \cdot \vec{a} = 0 \)
Substitute \( \vec{r} = \vec{c} + \lambda \vec{b} \) into \( \vec{r} \cdot \vec{a} = 0 \): \[ (\vec{c} + \lambda \vec{b}) \cdot \vec{a} = 0. \]
Simplify: \[ \vec{c} \cdot \vec{a} + \lambda (\vec{b} \cdot \vec{a}) = 0. \]
Step 3: Compute dot products
\[ \vec{c} \cdot \vec{a} = (7)(1) + (-3)(0) + (4)(2) = 15, \] \[ \vec{b} \cdot \vec{a} = (1)(1) + (1)(0) + (1)(2) = 3. \]
Substitute: \[ 15 + 3\lambda = 0 \implies \lambda = -5. \]
Step 4: Find \( \vec{r} \)
\[ \vec{r} = \vec{c} + \lambda \vec{b} = (7\hat{i} - 3\hat{j} + 4\hat{k}) - 5(\hat{i} + \hat{j} + \hat{k}), \] \[ \vec{r} = 2\hat{i} - 8\hat{j} - \hat{k}. \]
Step 5: Compute \( \vec{r} \cdot \vec{c} \)
\[ \vec{r} \cdot \vec{c} = (2)(7) + (-8)(-3) + (-1)(4), \] \[ \vec{r} \cdot \vec{c} = 14 + 24 - 4 = 34. \] Quick Tip: To solve vector problems with constraints, express vectors in parametric form and solve for scalars systematically.
Let \( S = \{W_1, W_2, \ldots\} \) be the sample space associated with a random experiment. Let \( P(W_n) = \frac{P(W_n-_1)}{2} \), \( n \geq 2 \). Let \( A = \{2k + 3l; k, l \in \mathbb{N}\} \) and \( B = \{W_n; n \in A\} \). Then \( P(B) \) is equal to:
The value of the integral \[ \int_{1}^2 \frac{t^4 + 1}{t^6 + 1} \, dt \]
is:
Let \( K \) be the sum of the coefficients of the odd powers of \( x \) in the expansion of \( (1+x)^{99} \). Let \( a \) be the middle term in the expansion of \( \left(2 + \frac{1}{\sqrt{2}}\right)^{200} \). If \[ \frac{^{200}C_{99}K}{a} = \frac{2^\ell m}{n}, \]
where \( m \) and \( n \) are odd numbers, then the ordered pair \( (\ell, n) \) is equal to:
Let \( f \) and \( g \) be twice differentiable functions on \( \mathbb{R} \) such that \( f''(x) = g''(x) + 6x \), \( f'(1) = 4g'(1) - 3 = 9 \), and \( f(2) = 3g(2) = 12 \). Which of the following is NOT true?
The set of all values of \( t \in \mathbb{R} \), for which the matrix \[ \begin{bmatrix} e^t & e^{-t}(\sin t - 2\cos t) & e^{-t}(-2\sin t - \cos t)
e^t & e^{-t}(2\sin t + \cos t) & e^{-t}(\sin t - 2\cos t)
e^t & e^{-t}\cos t & e^{-t}\sin t \end{bmatrix} \]
is invertible, is:
The area of the region \( A = \{(x, y) : |\cos x - \sin x| \leq y \leq \sin x, 0 \leq x \leq \frac{\pi}{2}\} \) is:
The set of all values of \( \lambda \) for which the equation \( \cos^2(2x) - 2\sin x - 2\cos(2x) = \lambda \) holds is:
The letters of the word OUGHT are written in all possible ways and these words are arranged as in a dictionary, in a series. Then the serial number of the word TOUGH is:
The plane \( 2x - y + z = 4 \) intersects the line segment joining the points \( A(a, -2, 4) \) and \( B(2, b, -3) \) at the point \( C \) in the ratio 2:1, and the distance of \( C \) from the origin is \( \sqrt{5} \). If \( ab < 0 \), and \( P \) is the point \( (a - b, b, 2b - a) \), then \( CP^2 \) is equal to:
Let \( \vec{a} = 4\hat{i} + 3\hat{j} \), \( b = 3\hat{i} - 4\hat{j} + 5\hat{k} \), and \( c \) be a vector such that \( (\vec{a} \times \vec{b}) \cdot \vec{c} + 25 = 0 \), \( \vec{c} \cdot (\hat{i} + \hat{j} + \hat{k}) = 4 \), and the projection of \( \vec{c} \) on \( \vec{a} \) is 1. Then the projection of \( \vec{c} \) on \( \vec{b} \) equals:
View Solution
(A)Compute \( \vec{a} \times \vec{b} \):
\[ \vec{a} \times \vec{b} = \begin{vmatrix} \hat{i} & \hat{j} & \hat{k}
4 & 3 & 0
3 & -4 & 5 \end{vmatrix} = 15\hat{i} - 20\hat{j} - 25\hat{k}. \]
(B)Let \( \vec{c} = x\hat{i} + y\hat{j} + z\hat{k} \). Using the condition \( (\vec{a} \times \vec{b}) \cdot \vec{c} + 25 = 0 \):
\[ 15x - 20y - 25z + 25 = 0 \implies 3x - 4y - 5z = -5. \quad (1) \]
(C)Using \( \vec{c} \cdot (\hat{i} + \hat{j} + \hat{k}) = 4 \):
\[ x + y + z = 4. \quad (2) \]
(D)Using the projection condition \( \frac{\vec{c} \cdot \vec{a}}{|\vec{a}|} = 1 \):
\[ \frac{4x + 3y}{5} = 1 \implies 4x + 3y = 5. \quad (3) \]
(E)Solve equations (1), (2), and (3) to find \( \vec{c} = 2\hat{i} - \hat{j} + 3\hat{k} \).
(F) Compute the projection of \( \vec{c} \) on \( \vec{b} \):
\[ Projection = \frac{\vec{c} \cdot \vec{b}}{|\vec{b}|}. \]
Simplify:
\[ Projection = \frac{25}{\sqrt{50}} = \frac{5}{\sqrt{2}}. \] Quick Tip: To solve vector problems, combine dot and cross product conditions systematically and solve equations step-by-step.
If the lines \( \frac{x-1}{1} = \frac{y-2}{2} = \frac{z+3}{1} \) and \( \frac{x-a}{2} = \frac{y+2}{3} = \frac{z-3}{1} \) intersect at the point \( P \), then the distance of the point \( P \) from the plane \( z = a \) is:
The value of the integral \[ \int_{1/2}^2 \frac{\tan^{-1} x}{x} \, dx \]
is equal to:
If the tangent at a point \( P \) on the parabola \( y^2 = 3x \) is parallel to the line \( x + 2y = 1 \), and the tangents at the points \( Q \) and \( R \) on the ellipse \( \frac{x^2}{4} + \frac{y^2}{1} = 1 \) are perpendicular to the line \( x - y = 2 \), then the area of the triangle \( PQR \) is:
Let \( y = y(x) \) be the solution of the differential equation \[ x \log_e x \frac{dy}{dx} + y = x^2 \log_e x, \quad (x > 1). \]
If \( y(2) = 2 \), then \( y(e) \) is equal to:
The number of 3-digit numbers that are divisible by either 3 or 4 but not divisible by 48 is:
Let \( R \) be a relation defined on \( \mathbb{N} \) as \( a R b \) if \( 2a + 3b \) is a multiple of 5. Then \( R \) is:
Consider a function \( f : \mathbb{N} \to \mathbb{R} \) satisfying \( f(1) + 2f(2) + 3f(3) + \dots + x f(x) = x(x + 1)f(x) \) for \( x \geq 2 \), with \( f(1) = 1 \). Then \( f(2022) \) is equal to:
The total number of 4-digit numbers whose greatest common divisor with 54 is 2, is:
A triangle is formed by the tangents at the point (2, 2) on the curves \( y^2 = 2x \) and \( x^2 + y^2 = 4x \), and the line \( x + y + 2 = 0 \). If \( r \) is the radius of its circumcircle, then \( r^2 \) is equal to:
A circle with center \( (2, 3) \) and radius 4 intersects the line \( x + y = 3 \) at points \( P \) and \( Q \). If the tangents at \( P \) and \( Q \) intersect at \( S(\alpha, \beta) \), then \( 4\alpha - 7\beta \) is equal to:
Let \( a_1 = b_1 = 1 \) and \( a_n = a_{n-1} + (n-1) \), \( b_n = b_{n-1} + a_{n-1} \), \( \forall n \geq 2 \). If \( S = \sum_{n=1}^\infty \frac{a_n}{2^n} \) and \( T = \sum_{n=1}^\infty \frac{b_n}{2^n} \), then \( 2^7(2S - T) \) is equal to:
If the equation of the normal to the curve \( y = \frac{x - a}{(x + b)(x - 2)} \) at the point \( (1, -3) \) is \( x - 4y = 13 \), then the value of \( a + b \) is:
Let \( A \) be a symmetric matrix such that \( |A| = 2 \) and \[ \begin{bmatrix} 3 & -2
2 & 1 \end{bmatrix} A = \begin{bmatrix} 1 & 2
2 & 7 \end{bmatrix}. \]
If the sum of the diagonal elements of \( A \) is \( s \), then \( \frac{\beta s}{\alpha^2} \) is equal to:
Let \( \{a_k\} \) and \( \{b_k\}, k \in \mathbb{N} \), be two G.P.s with common ratios \( r_1 \) and \( r_2 \), respectively, such that \( a_1 = b_1 = 4 \) and \( r_1 < r_2 \). Let \( c_k = a_k + b_k, k \in \mathbb{N} \). If \( c_2 = 5 \) and \( c_3 = 13 \), then \( \Sigma_{k=1}^4 c_k - (12a_6 + 8b_4) \) is equal to:
Let \( X = \{11, 12, 13, \dots, 41\} \) and \( Y = \{61, 62, 63, \dots, 91\} \) be two sets of observations. If \( \bar{x} \) and \( \bar{y} \) are their respective means and \( \sigma^2 \) is the variance of all observations in \( X \cup Y \), then \( |\bar{x} + \bar{y} - \sigma^2| \) is equal to:
Let \( \alpha = 8 - 14i \), \( A = \{z \in \mathbb{C} : |z^2 - \alpha^2| = |z^2 - \overline{\alpha}^2| \} \), and \( B = \{z \in \mathbb{C} : |z + 3i| = 4 \} \). Then \( \sum_{z \in A \cap B} (\operatorname{Re} z - \operatorname{Im} z) \) is equal to:
Let \( \alpha_1, \alpha_2, \dots, \alpha_7 \) be the roots of the equation \( x^7 + 3x^5 - 13x^3 - 15x = 0 \) and \( |\alpha_1| \geq |\alpha_2| \geq \dots \geq |\alpha_7| \). Then \( \alpha_1 \alpha_2 - \alpha_3 \alpha_4 + \alpha_5 \alpha_6 \) is equal to:
Substance A has atomic mass number 16 and half-life of 1 day. Another substance B has atomic mass number 32 and half-life of ½ day. If both A and B simultaneously start undergoing radioactivity at the same time with initial mass 320 g each, how many total atoms of A and B combined would be left after 2 days?
At 300 K, the rms speed of oxygen molecules is \(\frac{\alpha+5}{\alpha}\) times to that of its average speed in the gas. Then, the value of \(\alpha\) will be (used \(\pi = \frac{22}{7}\)):
The ratio of de-Broglie wavelength of an \(\alpha\)-particle and a proton accelerated from rest by the same potential is \(\sqrt{m}\). The value of \(m\) is:
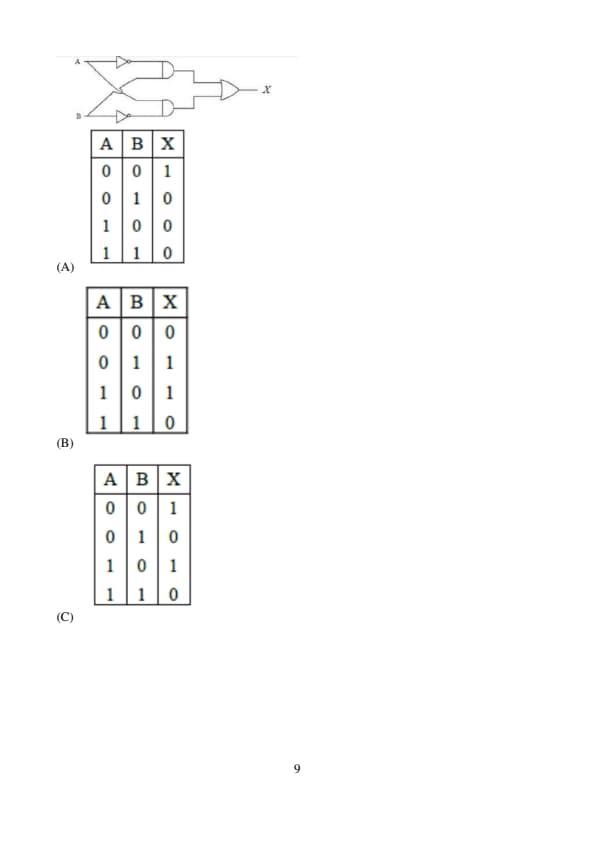
For the given logic gates combination, the correct truth table will be:
The time taken by an object to slide down a 45° rough inclined plane is \(n\) times as it takes to slide down a perfectly smooth 45° inclined plane. The coefficient of kinetic friction between the object and the inclined plane is:
Force acts for 20 s on a body of mass 20 kg, starting from rest, after which the force ceases, and then the body describes 50 m in the next 10 s. The value of force will be:
A fully loaded Boeing aircraft has a mass of \(5.4 \times 10^5 \, kg\). Its total wing area is \(500 \, m^2\). It is in level flight with a speed of \(1080 \, km/h\). If the density of air (\(\rho\)) is \(1.2 \, kg/m^3\), the fractional increase in the speed of the air on the upper surface of the wing relative to the lower surface in percentage will be (\(g = 10 \, m/s^2\)):
Identify the correct statements from the following:
An object moves at a constant speed along a circular path in a horizontal plane with the center at the origin. When the object is at \(x = +2 \, m\), its velocity is \(-4\hat{j} \, m/s\). The object's velocity (\(v\)) and acceleration (\(a\)) at \(x = -2 \, m\) will be:
A point charge \(2 \times 10^{-2} \, C\) is moved from \(P\) to \(S\) in a uniform electric field of \(30 \, N/C\) directed along the positive x-axis. If the coordinates of \(P\) and \(S\) are \((1, 2, 0) \, m\) and \((0, 0, 0) \, m\), respectively, the work done by the electric field will be:
The modulation index for an A.M. wave having maximum and minimum peak-to-peak voltages of 14 mV and 6 mV respectively is:
The electric current in a circular coil of four turns produces a magnetic induction of 32 T at its center. The coil is unwound and rewound into a circular coil of single turn. The magnetic induction at the center of the coil by the same current will be:
With the help of a potentiometer, we can determine the value of the emf of a given cell. The sensitivity of the potentiometer is:
A scientist is observing bacteria through a compound microscope. For better analysis and to improve its resolving power, he should:
Given below are two statements:
Statement I: Electromagnetic waves are not deflected by electric and magnetic fields.
Statement II: The amplitude of the electric field and the magnetic field in electromagnetic waves are related as \(E_0 = \sqrt{\mu_0 / \epsilon_0} \, B_0\).
Choose the correct answer:
Heat energy of \(184 \, kJ\) is given to ice of mass \(600 \, g\) at \(-12^\circ C\). Specific heat of ice is \(2222.3 \, J/kg/^\circ C\) and latent heat of ice is \(336 \, kJ/kg\).
For the given figures, choose the correct option:
The time period of a satellite of Earth is 24 hours. If the separation between the Earth and the satellite is decreased to one-fourth of the previous value, then its new time period will become:
The equation of a circle is given by \(x^2 + y^2 = a^2\), where \(a\) is the radius. If the equation is modified to change the origin other than \((0, 0)\), then find out the correct dimensions of \(A\) and \(B\) in a new equation:
\[ (x - At)^2 + \left(y - \frac{t}{B}\right)^2 = a^2. \]
The dimensions of \(t\) are given as \([T^{-1}]\).
A square loop of area \(25 \, cm^2\) has a resistance of \(10 \, \Omega\). The loop is placed in a uniform magnetic field of \(40.0 \, T\). The plane of the loop is perpendicular to the magnetic field. The work done in pulling the loop out of the magnetic field slowly and uniformly in 1.0 second will be:
When two resistances \(R_1\) and \(R_2\) are connected in series and introduced into the left gap of a meter bridge and a resistance of 10 \(\Omega\) is introduced into the right gap, a null point is found at 60 cm from the left side. When \(R_1\) and \(R_2\) are connected in parallel and introduced into the left gap, a resistance of 3 \(\Omega\) is introduced into the right gap to get the null point at 40 cm from the left end. The product of \(R_1 R_2\) is ______ \(\Omega^2\):
A particle of mass 100 g is projected at time \(t = 0\) with a speed of \(20 \, ms^{-1}\) at an angle \(45^\circ\) to the horizontal. The magnitude of the angular momentum of the particle about the starting point at time \(t = 2 \, s\) is found to be \(\sqrt{K} \, kgm^2/s\). The value of \(K\) is:
In an experiment measuring the refractive index of a glass slab using a traveling microscope, the real thickness of the slab is measured as \(5.25 \, mm\) and the apparent thickness as \(5.00 \, mm\). The estimated uncertainty in the measurement of refractive index is \(x \cdot 10^{-3}\), where \(x\) is:
For a charged spherical ball, the electrostatic potential inside the ball varies with \(r\) as \(V = 2ar^2 + b\). The volume charge density inside the ball is \(-\lambda \alpha \varepsilon_0\). The value of \(\lambda\) is:
A car is moving on a circular path of radius 600 m such that the magnitudes of the tangential acceleration and centripetal acceleration are equal. The time taken by the car to complete the first quarter of the revolution, if it is moving with an initial speed of 54 km/hr, is \(t(1-e^{-\pi/2}) \, s\). The value of \(t\) is:
An inductor of inductance \(2 \, \mu H\) is connected in series with a resistance, a variable capacitor, and an AC source of frequency 7 kHz. The value of capacitance for which maximum current is drawn into the circuit is \(1/x \, F\), where the value of \(x\) is:
A metal block of base area \(0.20 \, m^2\) is placed on a table. A liquid film of thickness \(0.25 \, mm\) is inserted between the block and the table. The block is pushed by a horizontal force of \(0.1 \, N\) and moves with a constant speed. If the viscosity of the liquid is \(5.0 \times 10^{-3} \, Pl\), the speed of the block is ______ \(\times 10^{-3} \, m/s\).
A particle of mass \(250 \, g\) executes simple harmonic motion under a periodic force \(F = -25x \, N\). The particle attains a maximum speed of \(4 \, m/s\) during its oscillation. The amplitude of the motion is ______ cm.
Unpolarised light is incident on the boundary between two dielectric media, whose dielectric constants are \(2.8\) (medium-1) and \(6.8\) (medium-2), respectively. To satisfy the condition such that the reflected and refracted rays are perpendicular to each other, the angle of incidence should be \(\tan^{-1}(\sqrt{\mu_2/\mu_1})\). The value of \(\theta\) is:
A null point is found at \(200 \, cm\) in a potentiometer when the cell in the secondary circuit is shunted by \(5 \, \Omega\). When a resistance of \(15 \, \Omega\) is used for shunting, the null point moves to \(300 \, cm\). The internal resistance of the cell is:
Given below are two statements:
Statement I: The decrease in first ionization enthalpy from B to Al is much larger than that from Al to Ga.
Statement II: The d orbitals in Ga are completely filled.
In the light of the above statements, choose the most appropriate answer from the options given below
Correct order of spin-only magnetic moment of the following complex ions is:
(Given At. No. Fe: 26, Co: 27)
Match List-I and List-II:
Choose the correct answer from the options given below:
The set of correct statements is:
(i) Manganese exhibits +7 oxidation state in its oxide.
(ii) Ruthenium and Osmium exhibit +8 oxidation in their oxides.
(iii) Sc shows +4 oxidation state which is oxidizing in nature.
(iv) Cr shows oxidising nature in +6 oxidation state.
Match List-I and List-II:
Options:
An indicator ‘X’ is used for studying the effect of variation in concentration of iodide on the rate of reaction of iodide ion with \(H_2O_2\) at room temperature. The indicator ‘X’ forms blue colored complex with compound ‘A’ present in the solution. The indicator ‘X’ and compound ‘A’ respectively are:
A doctor prescribed the drug Equanil to a patient. The patient was likely to have symptoms of which disease?
Find out the major product for the following reaction:
\(\rightarrow\) Major Product
The one giving maximum number of isomeric alkenes on dehydrohalogenation reaction is (excluding rearrangement):
When a hydrocarbon A undergoes combustion in the presence of air, it requires 9.5 equivalents of oxygen and produces 3 equivalents of water. What is the molecular formula of A?
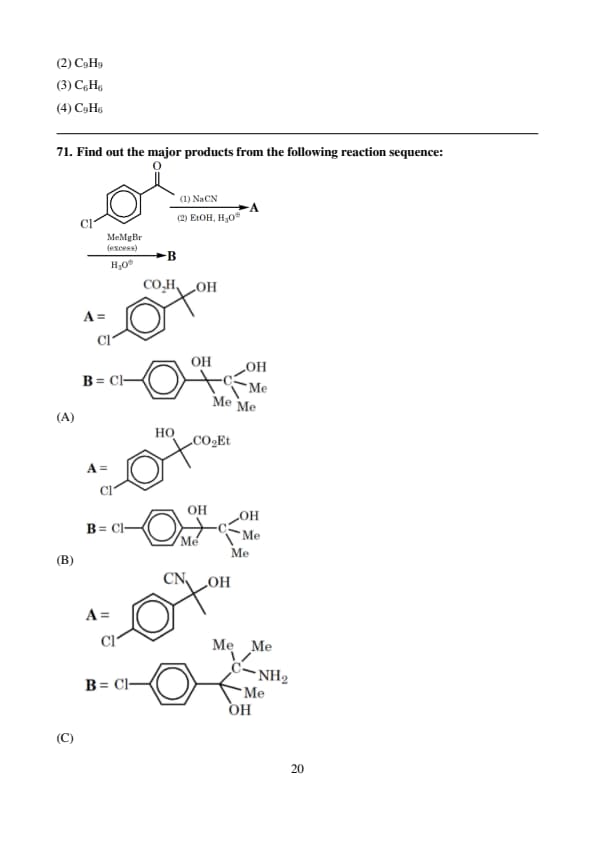
Find out the major products from the following reaction sequence:
According to MO theory, the bond orders for \(O_2^-\), CO, and NO+, respectively, are:
A solution of \(CrO_3\) in amyl alcohol has a __________ colour:
The concentration of dissolved oxygen in water for growth of fish should be more than X ppm, and biochemical oxygen demand in clean water should be less than Y ppm. X and Y in ppm are respectively:
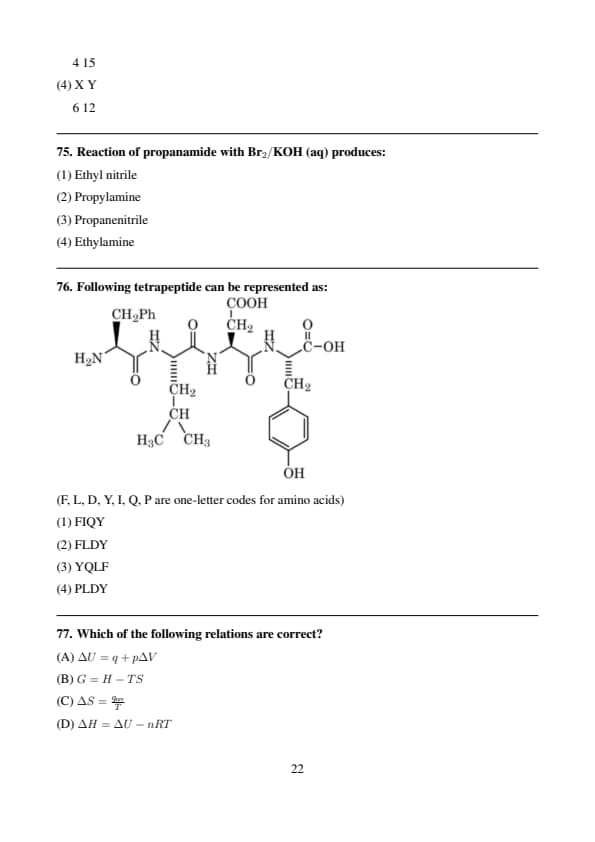
Reaction of propanamide with \(Br_2/KOH\) (aq) produces:
Following tetrapeptide can be represented as:
(F, L, D, Y, I, Q, P are one-letter codes for amino acids)
Which of the following relations are correct?
The major component of which of the following ore is sulphide based mineral?
Given below are two statements:
Statement I: Nickel is being used as the catalyst for producing syn gas and edible fats.
Statement II: Silicon forms both electron-rich and electron-deficient hydrides.
Choose the most appropriate answer from the options given below:
Match List I with List II:
Choose the correct answer from the options given below:
On heating, LiNO\(_3\) gives how many compounds among the following? Li\(_2\)O, N\(_2\), O\(_2\), LiNO\(_2\), NO\(_2\)
At 298 K:
\[ N_2 + 3H_2 \rightleftharpoons 2NH_3, \, K_1 = 4 \times 10^5 \] \[ N_2 + O_2 \rightleftharpoons 2NO, \, K_2 = 1.6 \times 10^{12} \] \[ H_2 + \frac{1}{2}O_2 \rightleftharpoons H_2O, \, K_3 = 1.0 \times 10^{13} \]
Based on the above equilibria, the equilibrium constant of the reaction:
\[ 2NH_3 + \frac{5}{2}O_2 \rightleftharpoons 2NO + 3H_2O \]
is \( ….. \times 10^{-33} \) (nearest integer).
For conversion of compound A \(\rightarrow\) B, the rate constant of the reaction was found to be \(4.6 \times 10^{-5}\) L mol\(^{-1}\) s\(^{-1}\). The order of the reaction is _____
Total number of acidic oxides among \(N_2O_3, NO, N_2O, Cl_2O_7, SO_2, CO, CaO, Na_2O\) and \(NO_2\) is _____
When 0.01 mol of an organic compound containing 60% carbon was burnt completely, 4.4 g of CO\(_2\) was produced. The molar mass of the compound is _____ g mol\(^{-1}\) (nearest integer).
The denticity of the ligand present in Fehling's reagent is _____
A metal M forms hexagonal close-packed structure. The total number of voids in 0.02 mol of it is \(_____\times 10^{21}\) (Nearest integer). (Given \(N_A = 6.02 \times 10^{23}\))
Assume that the radius of the first Bohr orbit of hydrogen atom is 0.6 Å. The radius of the third Bohr orbit of He\(^{+}\) is …. picometer (Nearest integer).
The equilibrium constant for the reaction:
\(Zn(s) + Sn^{2+}(aq) \rightleftharpoons Zn^{2+}(aq) + Sn(s)\)
is \(1 \times 10^{20}\) at 298 K. The magnitude of standard electrode potential of \(Sn^{2+}/Sn\) if \(E^\circ_{Zn^{2+}/Zn} = -0.76 \, V\) is \(\ldots \times 10^{-2} \, V\) (Nearest integer).
The volume of HCl containing 73 g L\(^{-1}\), required to completely neutralize NaOH obtained by reacting 0.69 g of metallic sodium with water, is \(\ldots\) mL (Nearest integer).






















Also Check:
JEE Main 2023 Jan 29 Shift 2 Question Paper by Coaching Institute
| Coaching Institutes | Question Paper with Answer Key PDF |
|---|---|
| Aakash BYJUs | Check Here |
| Vedantu | Check Here |
JEE Main 2023 Paper Analysis Jan 29 Shift 2
JEE Main 2023 Paper Analysis for the exam scheduled on January 29 Shift 2 is updated here. Candidates can check subject-wise paper analysis for the exam scheduled on January 29 Shift 2 here along with the topics with the highest weightage.
Also Check:











Comments