JEE Main 2025 April 4 Chemistry Question Paper is available for download. NTA conducted JEE Main 2025 Shift 1 B.Tech Exam on 3rd April 2025 from 9:00 AM to 12:00 PM and for JEE Main 2025 B.Tech Shift 2 appearing candidates from 3:00 PM to 6:00 PM. The JEE Main 2025 3rd April B.Tech Question Paper was Moderate to Tough.
Also Check: JEE Main 2025 Question Paper with Solution PDF Download
JEE Main 2025 April 4 Shift 1 Chemistry Question Paper with Solutions
| JEE Main 2025 April 4 Shift 1 Chemistry Question Paper Pdf | Download PDF | View Solution |

JEE Main 2025 Chemistry Questions with Solutions
Question 1:
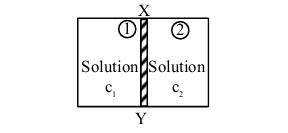
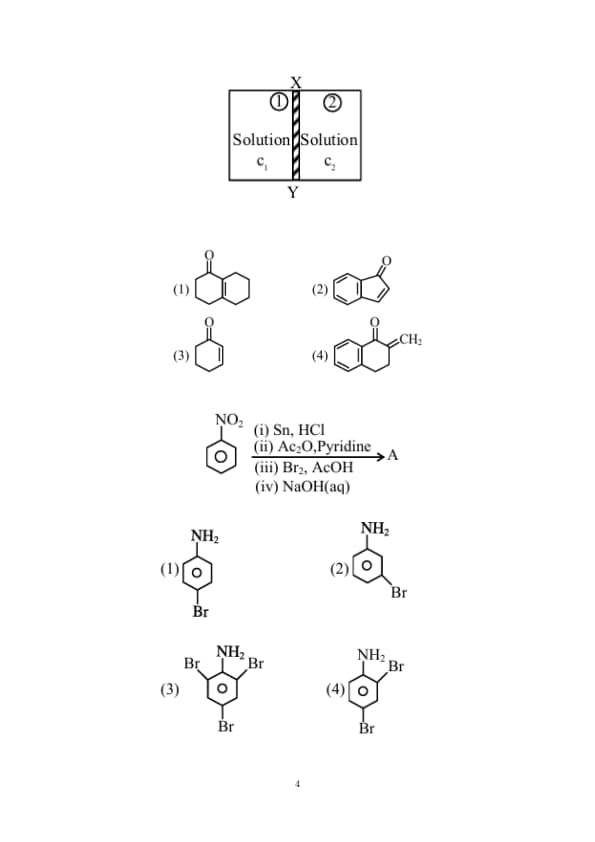
\(XY\) is the membrane / partition between two chambers 1 and 2 containing sugar solutions of concentration \(\mathrm{c}_{1}\) and \(\mathrm{c}_{2}\left(\mathrm{c}_{1}>\mathrm{c}_{2}\right) \mathrm{mol} \mathrm{L}^{-1}\). For the reverse osmosis to take place identify the correct condition
(Here \(\mathrm{p}_{1}\) and \(\mathrm{p}_{2}\) are pressures applied on chamber 1 and 2 )
Let us consider a reversible reaction at temperature, T . In this reaction, both \(\Delta \mathrm{H}\) and \(\Delta \mathrm{S}\) were observed to have positive values. If the equilibrium temperature is \(\mathrm{T}_{\mathrm{e}}\), then the reaction becomes spontaneous at:
Which of the following molecules(s) show/s paramagnetic behavior?
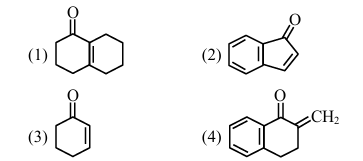
Aldol condensation is a popular and classical method to prepare \(\alpha, \beta\)-unsaturated carbonyl compounds. This reaction can be both intermolecular and intramolecular. Predict which one of the following is not a product of intramolecular aldol condensation?
One mole of an ideal gas expands isothermally and reversibly from \(10 \mathrm{dm}^{3}\) to \(20 \mathrm{dm}^{3}\) at \(300 \mathrm{~K} . \Delta \mathrm{U}\), q and work done in the process respectively are :
Given : \(\mathrm{R}=8.3 \mathrm{JK}^{-1}\) and \(\mathrm{mol}^{-1}\)
In \(10=2.3\)
\(\log 2=0.30\)
\(\log 3=0.48\)
Which one of the following complexes will have \(\Delta_{0}=0\) and \(\mu=5.96\) B.M.?
For \(\mathrm{A}_{2}+\mathrm{B}_{2} \rightleftharpoons 2 \mathrm{AB}\) \(\mathrm{E}_{\mathrm{a}}\) for forward and backward reaction are 180 and \(200 \mathrm{~kJ} \mathrm{~mol}^{-1}\) respectively. If catalyst lowers \(\mathrm{E}_{\mathrm{a}}\) for both reaction by \(100 \mathrm{~kJ} \mathrm{~mol}^{-1}\). Which of the following statement is correct?
Rate law for a reaction between \(A\) and \(B\) is given by \(\mathrm{R}=\mathrm{k}[\mathrm{A}]^{\mathrm{n}}[\mathrm{B}]^{\mathrm{m}}\). If concentration of A is doubled and concentration of B is halved from their initial value, the ratio of new rate of reaction to the initial rate of reaction \(\left(\frac{\mathrm{r}_{2}}{\mathrm{r}_{1}}\right)\) is
Number of stereoisomers possible for the complexes, \(\left[\mathrm{CrCl}_{3}(\mathrm{py})_{3}\right]\) and \(\left[\mathrm{CrCl}_{2}(\mathrm{ox})_{2}\right]^{3-}\) are respectively
(py = pyridine, ox = oxalate)
The major product (A) formed in the following reaction sequence is

On charging the lead storage battery, the oxidation state of lead changes from \(\mathrm{x}_{1}\) to \(\mathrm{y}_{1}\) at the anode and from \(\mathrm{x}_{2}\) to \(\mathrm{y}_{2}\) at the cathode. The values of \(\mathrm{x}_{1}, \mathrm{y}_{1}, \mathrm{x}_{2}, \mathrm{y}_{2}\) are respectively:
Given below are two statements:
Statement I: Nitrogen forms oxides with +1 to +5 oxidation states due to the formation of \(\mathrm{p} \pi-\mathrm{p} \pi\) bond with oxygen.
Statement II: Nitrogen does not form halides with +5 oxidation state due to the absence of d-orbital in it.
In the light of the above statements, choose the correct answer from the options given below:
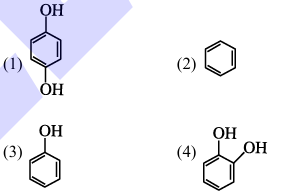
Benzene is treated with oleum to produce compound (X) which when further heated with molten sodium hydroxide followed by acidification produces compound (Y).The compound Y is treated with zinc metal to produce compound (Z). Identify the structure of compound (Z) from the following option.
Identify the pair of reactants that upon reaction, with elimination of HCl will give rise to the dipeptide Gly-Ala.
Given below are the pairs of group 13 elements showing their relation in terms of atomic radius. \((\mathrm{B}<\mathrm{Al}),(\mathrm{Al}<\mathrm{Ga}),(\mathrm{Ga}<\mathrm{In})\) and \((\mathrm{In}<\mathrm{Tl})\) Identify the elements present in the incorrect pair and in that pair find out the element (X) that has higher ionic radius \(\left(\mathrm{M}^{3+}\right)\) than the other one. The atomic number of the element (X) is
An organic compound (X) with molecular formula \(\mathrm{C}_{3} \mathrm{H}_{6} \mathrm{O}\) is not readily oxidised. On reduction it gives \(\left(\mathrm{C}_{3} \mathrm{H}_{8} \mathrm{O}(\mathrm{Y})\right.\) which reacts with HBr to give a bromide (Z) which is converted to Grignard reagent. This Grignard reagent on reaction with (X) followed by hydrolysis give 2,3-dimethylbutan-2-ol. Compounds (X), (Y) and (Z) respectively are:
Predict the major product of the following reaction sequence:
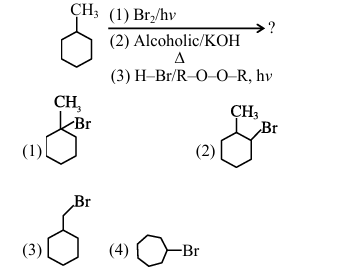
Given below are two statements.

In the light of the above statements, choose the correct answer from the options given below:
Pair of transition metal ions having the same number of unpaired electrons is :
Which one of the following about an electron occupying the 1 s orbital in a hydrogen atom is incorrect ? (Bohr's radius is represented by \(\mathrm{a}_{0}\) )
In Dumas' method for estimation of nitrogen 1 g of an organic compound gave 150 mL of nitrogen collected at 300 K temperature and 900 mm Hg pressure. The percentage composition of nitrogen in the compound is _______ % (nearest integer).
(Aqueous tension at \(300 \mathrm{~K}=15 \mathrm{~mm} \mathrm{Hg}\) )
\(\mathrm{KMnO}_{4}\) acts as an oxidising agent in acidic medium. ' X ' is the difference between the oxidation states of Mn in reactant and product. ' Y ' is the number of ' d ' electrons present in the brown red precipitate formed at the end of the acetate ion test with neutral ferric chloride. The value of \(\mathrm{X}+\mathrm{Y}\) is _______ .
Fortification of food with iron is done using \(\mathrm{FeSO}_{4} .7 \mathrm{H}_{2} \mathrm{O}\). The mass in grams of the \(\mathrm{FeSO}_{4} .7 \mathrm{H}_{2} \mathrm{O}\) required to achieve 12 ppm of iron in 150 kg of wheat is _______ (Nearest integer).
(Given : Molar mass of \(\mathrm{Fe}, \mathrm{S}\) and O respectively are 56,32 and \(16 \mathrm{~g} \mathrm{~mol}^{-1}\) )
The pH of a 0.01 M weak acid \(\mathrm{HX}\left(\mathrm{K}_{\mathrm{a}}=4 \times 10^{-10}\right)\) is found to be 5 . Now the acid solution is diluted with excess of water so that the pH of the solution changes to 6 . The new concentration of the diluted weak acid is given as \(\mathrm{x} \times 10^{-4} \mathrm{M}\). The value of x is _______ (nearest integer).
The total number of hydrogen bonds of a DNA-double Helix strand whose one strand has the following sequence of bases is _______ .
\(5^{\prime}-\mathrm{G}-\mathrm{G}-\mathrm{C}-\mathrm{A}-\mathrm{A}-\mathrm{A}-\mathrm{T}-\mathrm{C}-\mathrm{G}-\mathrm{G}-\mathrm{C}-\mathrm{T}-\mathrm{A}-3^{\prime}\)















Comments