JEE Main 2025 April 8 Chemistry Question Paper is available for download. NTA conducted JEE Main 2025 Shift 1 B.Tech Exam on 3rd April 2025 from 9:00 AM to 12:00 PM and for JEE Main 2025 B.Tech Shift 2 appearing candidates from 3:00 PM to 6:00 PM. The JEE Main 2025 8th April B.Tech Question Paper was Moderate to Tough.
Also Check: JEE Main 2025 Question Paper with Solution PDF Download
JEE Main 2025 April 8 Shift 2 Chemistry Question Paper with Solutions
| JEE Main 2025 April 8 Shift 2 Chemistry Question Paper Pdf | Download PDF | View Solution |

Given below are two statements:
Statement I: \( H_2Se \) is more acidic than \( H_2Te \)
Statement II: \( H_2Se \) has higher bond enthalpy for dissociation than \( H_2Te \)
In the light of the above statements, choose the correct answer from the options given below.
The correct decreasing order of spin only magnetic moment values (BM) of \( Cu^+ \), \( Cu^{2+} \), \( Cr^{2+} \) and \( Cr^{3+} \) ions is:
Match the LIST-I with LIST-II
LIST-I (Reagent) LIST-II (Functional Group detected)
A. Sodium bicarbonate solution I. double bond/unsaturation
B. Neutral ferric chloride II. carboxylic acid
C. Ceric ammonium nitrate III. phenolic -OH
D. Alkaline KMnO\(_4\) IV. alcoholic -OH
Choose the correct answer from the options given below:
Given below are two statements:
Statement I: A homoleptic octahedral complex, formed using monodentate ligands, will not show stereoisomerism
Statement II: cis- and trans-platin are heteroleptic complexes of Pd.
In the light of the above statements, choose the correct answer from the options given below
What is the correct IUPAC name of the following compound?
A shows positive Lassaigne's test for N and its molar mass is 12(1)
B gives effervescence with aqueous NaHCO(3)
C gives fruity smell.
Identify A, B, and C from the following.
On combustion 0.210 g of an organic compound containing C, H and O gave 0.127 g \( H_2O \) and 0.307 g \( CO_2 \). The percentages of hydrogen and oxygen in the given organic compound respectively are:
HA \( (aq) \rightleftharpoons H^+ (aq) + A^- (aq) \)
The freezing point depression of a 0.1 m aqueous solution of a monobasic weak acid HA is 0.20 °C. The dissociation constant for the acid is
Given: \( K_f(H_2O) = 1.8 \, K kg mol^{-1} \), molality ≡ molarity
Match the LIST-I with LIST-II
LIST-I LIST-II
A. Carbocation I. Species that can supply a pair of electrons.
B. C-Free radical II. Species that can receive a pair of electrons.
C. Nucleophile III. sp\(^2\) hybridized carbon with empty p-orbital.
D. Electrophile IV. sp\(^2\)/sp\(^3\) hybridized carbon with one unpaired electron.
Choose the correct answer from the options given below:
In a first order decomposition reaction, the time taken for the decomposition of reactant to one fourth and one eighth of its initial concentration are \( t_1 \) and \( t_2 \) (s), respectively. The ratio \( t_1 / t_2 \) will be:
Match the LIST-I with LIST-II
LIST-I (Complex/Species) LIST-II (Shape & magnetic moment)
A. \([Ni(CO)_4]\) I. Tetrahedral, 2.8 BM
B. \([Ni(CN)_4]^{2-}\) II. Square planar, 0 BM
C. \([NiCl_4]^{2-}\) III. Tetrahedral, 0 BM
D. \([MnBr_4]^{2-}\) IV. Tetrahedral, 5.9 BM
Choose the correct answer from the options given below:
Which one of the following reactions will not lead to the desired ether formation in major proportion?
(iso-Bu = isobutyl, sec-Bu = sec-butyl, nPr = n-propyl, tBu = tert-butyl, Et = ethyl)
Correct statements for an element with atomic number 9 are
A. There can be 5 electrons for which \( m_s = +\frac{1}{2} \) and 4 electrons for which \( m_s = -\frac{1}{2} \)
B. There is only one electron in \( p_z \) orbital.
C. The last electron goes to orbital with \( n = 2 \) and \( l = 1 \).
D. The sum of angular nodes of all the atomic orbitals is 1.
Choose the correct answer from the options given below:
The number of species from the following that are involved in \( sp^3d^2 \) hybridization is \[ [Co(NH_3)_6]^{3+}, SF_6, [CrF_6]^{3-}, [CoF_6]^{3-}, [Mn(CN)_6]^{3-} \] and \[ [MnCl_6]^{3-} \]
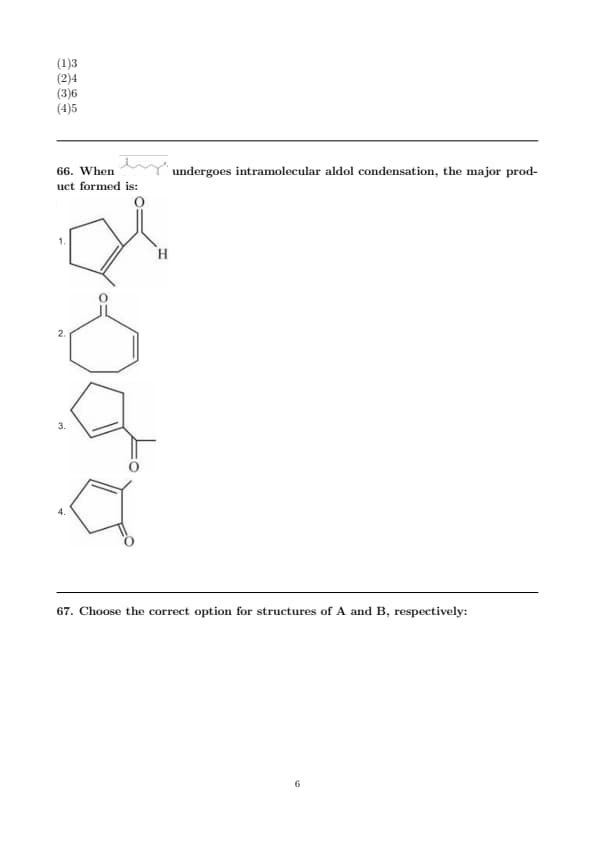
When undergoes intramolecular aldol condensation, the major product formed is:
Choose the correct option for structures of A and B, respectively:
Choose the correct set of reagents for the following conversion:
Which of the following binary mixture does not show the behavior of minimum boiling azeotropes?
The atomic number of the element from the following with lowest \( 1^{st} \) ionization enthalpy is:
20 mL of sodium iodide solution gave 4.74 g silver iodide when treated with excess of silver nitrate solution. The molarity of the sodium iodide solution is _____ M. (Nearest Integer value)
(Given : Na = 23, I = 127, Ag = 108, N = 14, O = 16 g mol\(^{-1}\))
The equilibrium constant for decomposition of \( H_2O \) (g)
\[ H_2O(g) \rightleftharpoons H_2(g) + \frac{1}{2} O_2(g) \quad (\Delta G^\circ = 92.34 \, kJ mol^{-1}) \]
is \( 8.0 \times 10^{-3} \) at 2300 K and total pressure at equilibrium is 1 bar. Under this condition, the degree of dissociation (\( \alpha \)) of water is _____ \(\times 10^{-2}\) (nearest integer value).
[Assume \( \alpha \) is negligible with respect to 1]
Resonance in X\(_2\)Y can be represented as
The enthalpy of formation of X\(_2\)Y is 80 kJ mol\(^{-1}\), and the magnitude of resonance energy of X\(_2\)Y is:
The energy of an electron in first Bohr orbit of H-atom is \(-13.6\) eV. The magnitude of energy value of electron in the first excited state of Be\(^{3+}\) is _____ eV (nearest integer value)
Consider the following half cell reaction
\[ Cr_2O_7^{2-} (aq) + 6e^- + 14H^+ (aq) \longrightarrow 2Cr^{3+} (aq) + 7H_2O(1) \]
The reaction was conducted with the ratio of \[\frac{[Cr^{3+}]^2}{[Cr_2O_7^{2-}]} = 10^{-6}\]
The pH value at which the EMF of the half cell will become zero is _____ (nearest integer value)
[Given : standard half cell reduction potential \[E^\circ_{Cr_2O_7^{2-}, H^+/Cr^{3+}} = 1.33V, \quad \frac{2.303RT}{F} = 0.059V\]


















Comments