JEE Main 4 April Shift 1 question paper with solutions and answers pdf is available here for download. NTA conducted JEE Main 4 April Shift 1 exam from 9 AM to 12 PM. The question paper for JEE Main 4 April Shift 1 includes 90 questions equally divided into Physics, Chemistry and Maths. Candidates must attempt 75 questions in a 3-hour time duration.
Students who are appearing for JEE Main upcoming shifts can check the JEE Main April 4 Shift 1 Question Paper Pdf to understand the difficulty level of the exam. Jee Main 2025 April 3 Exam is reported to be easy to moderate. Physics and Chemistry had a lot of NCERT- based questions.
JEE Main 2025 is conducted in various cities across the country, with 15 destinations abroad. In JEE Main April 4 Shift 1 students are required to attempt 75 questions following a marking scheme of +4 for correct answers and -1 for incorrect ones. You can find JEE Main April 4 Shift 1 Answer key here.
The Memory-Based Question Paper for JEE Main April 4 Shift 1 for B.E/B. Tech Paper is available for download shortly. The second shift of JEE Main April 4 is scheduled from 3:00 pm to 6:00 pm.
JEE Main 2025 April 4 Shift 1 Question Paper with Solutions
| JEE Main 2025 April 4 Shift 1 Question Paper Pdf | Download Pdf | View Solutions |

JEE Main 2025 April 4 Shift 1 Questions with Solutions
Let \(f, g: (1, \infty) \rightarrow \mathbb{R}\) be defined as \(f(x) = \frac{2x + 3}{5x + 2}\) and \(g(x) = \frac{2 - 3x}{1 - x}\). If the range of the function \(fog: [2, 4] \rightarrow \mathbb{R}\) is \([\alpha, \beta]\), then \(\frac{1}{\beta - \alpha}\) is equal to
Consider the sets \(A = \{(x, y) \in \mathbb{R} \times \mathbb{R} : x^2 + y^2 = 25\}\), \(B = \{(x, y) \in \mathbb{R} \times \mathbb{R} : x^2 + 9y^2 = 144\}\), \(C = \{(x, y) \in \mathbb{Z} \times \mathbb{Z} : x^2 + y^2 \leq 4\}\), and \(D = A \cap B\). The total number of one-one functions from the set \(D\) to the set \(C\) is:
Let \(A = \{1, 6, 11, 16, \ldots\}\) and \(B = \{9, 16, 23, 30, \ldots\}\) be the sets consisting of the first 2025 terms of two arithmetic progressions. Then \(n(A \cup B)\) is
For an integer \(n \geq 2\), if the arithmetic mean of all coefficients in the binomial expansion of \((x + y)^{2n-3}\) is 16, then the distance of the point \(P(2n-1, n^2-4n)\) from the line \(x + y = 8\) is:
The probability of forming a 12 persons committee from 4 engineers, 2 doctors, and 10 professors containing at least 3 engineers and at least 1 doctor is:
Let the shortest distance between the lines \(\frac{x-3}{3} = \frac{y-\alpha}{-1} = \frac{z-3}{1}\) and \(\frac{x+3}{-3} = \frac{y+7}{2} = \frac{z-\beta}{4}\) be \(3\sqrt{30}\). Then the positive value of \(5\alpha + \beta\) is
If \(\lim_{x \to 1} \frac{(x-1)(6+\lambda \cos(x-1)) + \mu \sin(1-x)}{(x-1)^3} = -1\), where \(\lambda, \mu \in \mathbb{R}\), then \(\lambda + \mu\) is equal to
Let \(f: [0, \infty) \to \mathbb{R}\) be a differentiable function such that \(f(x) = 1 - 2x + \int_0^x e^{x-t} f(t) \, dt\) for all \(x \in [0, \infty)\). Then the area of the region bounded by \(y = f(x)\) and the coordinate axes is
Let \(A\) and \(B\) be two distinct points on the line \(L: \frac{x-6}{3} = \frac{y-7}{2} = \frac{z-7}{-2}\). Both \(A\) and \(B\) are at a distance \(2\sqrt{17}\) from the foot of perpendicular drawn from the point \((1, 2, 3)\) on the line \(L\). If \(O\) is the origin, then \(\overrightarrow{OA} \cdot \overrightarrow{OB}\) is equal to:
Let \(f: \mathbb{R} \to \mathbb{R}\) be a continuous function satisfying \(f(0) = 1\) and \(f(2x) - f(x) = x\) for all \(x \in \mathbb{R}\). If \(\lim_{n \to \infty} \left\{ f(x) - f\left( \frac{x}{2^n} \right) \right\} = G(x)\), then \(\sum_{r=1}^{10} G(r^2)\) is equal to
1 + 3 + \(5^2\) + 7 + \(9^2\) + \(\ldots\) upto 40 terms is equal to
In the expansion of \(\left( \sqrt{5} + \frac{1}{\sqrt{5}} \right)^n\), \(n \in \mathbb{N}\), if the ratio of \(15^{th}\) term from the beginning to the \(15^{th}\) term from the end is \(\frac{1}{6}\), then the value of \(^nC_3\) is:
Considering the principal values of the inverse trigonometric functions, \(\sin^{-1} \left( \frac{\sqrt{3}}{2} x + \frac{1}{2} \sqrt{1-x^2} \right)\), \(-\frac{1}{2} < x < \frac{1}{\sqrt{2}}\), is equal to
Consider two vectors \(\vec{u} = 3\hat{i} - \hat{j}\) and \(\vec{v} = 2\hat{i} + \hat{j} - \lambda \hat{k}\), \(\lambda > 0\). The angle between them is given by \(\cos^{-1} \left( \frac{\sqrt{5}}{2\sqrt{7}} \right)\). Let \(\vec{v} = \vec{v}_1 + \vec{v}_2\), where \(\vec{v}_1\) is parallel to \(\vec{u}\) and \(\vec{v}_2\) is perpendicular to \(\vec{u}\). Then the value \(|\vec{v}_1|^2 + |\vec{v}_2|^2\) is equal to
Let the three sides of a triangle are on the lines \(4x - 7y + 10 = 0\), \(x + y = 5\), and \(7x + 4y = 15\). Then the distance of its orthocenter from the orthocenter of the triangle formed by the lines \(x = 0\), \(y = 0\), and \(x + y = 1\) is
The value of \(\int_{-1}^{1} \frac{(1 + \sqrt{|x| - x})e^x + (\sqrt{|x| - x})e^{-x}}{e^x + e^{-x}} \, dx\) is equal to
The length of the latus-rectum of the ellipse, whose foci are \((2, 5)\) and \((2, -3)\) and eccentricity is \(\frac{4}{5}\), is
Consider the equation \(x^2 + 4x - n = 0\), where \(n \in [20, 100]\) is a natural number. Then the number of all distinct values of \(n\), for which the given equation has integral roots, is equal to
A box contains 10 pens of which 3 are defective. A sample of 2 pens is drawn at random and let \(X\) denote the number of defective pens. Then the variance of \(X\) is
If \(10 \sin^4 \theta + 15 \cos^4 \theta = 6\), then the value of \(\frac{27 \csc^6 \theta + 8 \sec^6 \theta}{16 \sec^8 \theta}\) is:
If the area of the region \(\{ (x, y) : |x - 5| \leq y \leq 4\sqrt{x} \}\) is \(A\), then \(3A\) is equal to
Let \(A = \begin{bmatrix} \cos \theta & 0 & -\sin \theta
0 & 1 & 0
\sin \theta & 0 & \cos \theta \end{bmatrix}\). If for some \(\theta \in (0, \pi)\), \(A^2 = A^T\), then the sum of the diagonal elements of the matrix \((A + I)^3 + (A - I)^3 - 6A\) is equal to
Let \(A = \{ z \in \mathbb{C} : |z - 2 - i| = 3 \}\), \(B = \{ z \in \mathbb{C} : Re(z - iz) = 2 \}\), and \(S = A \cap B\). Then \(\sum_{z \in S} |z|^2\) is equal to
Let \(C\) be the circle \(x^2 + (y - 1)^2 = 2\), \(E_1\) and \(E_2\) be two ellipses whose centres lie at the origin and major axes lie on the \(x\)-axis and \(y\)-axis respectively. Let the straight line \(x + y = 3\) touch the curves \(C\), \(E_1\), and \(E_2\) at \(P(x_1, y_1)\), \(Q(x_2, y_2)\), and \(R(x_3, y_3)\) respectively. Given that \(P\) is the mid-point of the line segment \(QR\) and \(PQ = \frac{2\sqrt{2}}{3}\), the value of \(9(x_1 y_1 + x_2 y_2 + x_3 y_3)\) is equal to
Let \(m\) and \(n\) be the number of points at which the function \(f(x) = \max \{ x, x^3, x^5, \ldots, x^{21} \}\) is not differentiable and not continuous, respectively. Then \(m + n\) is equal to
The mean free path and the average speed of oxygen molecules at 300 K and 1 atm are \(3 \times 10^{-7} \mathrm{~m}\) and \(600 \mathrm{~m} / \mathrm{s}\), respectively. Find the frequency of its collisions.
A small mirror of mass \(m\) is suspended by a massless thread of length \(l\). Then the small angle through which the thread will be deflected when a short pulse of laser of energy \(E\) falls normal on the mirror ( \(c=\) speed of light in vacuum and \(g=\) acceleration due to gravity).
Two liquids A and B have \(\theta_{\mathrm{A}}\) and \(\theta_{\mathrm{B}}\) as contact angles in a capillary tube. If \(K=\cos \theta_{\mathrm{A}} / \cos \theta_{\mathrm{B}}\), then identify the correct statement:
Which of the following are correct expression for torque acting on a body?
A. \(\ddot{\tau}=\ddot{\mathrm{r}} \times \ddot{\mathrm{L}}\)
B. \(\ddot{\tau}=\frac{\mathrm{d}}{\mathrm{dt}}(\ddot{\mathrm{r}} \times \ddot{\mathrm{p}})\)
C. \(\ddot{\tau}=\ddot{\mathrm{r}} \times \frac{\mathrm{d} \dot{\mathrm{p}}}{\mathrm{dt}}\)
D. \(\ddot{\tau}=\mathrm{I} \dot{\alpha}\)
E. \(\ddot{\tau}=\ddot{\mathrm{r}} \times \ddot{\mathrm{F}}\)
( \(\ddot{r}=\) position vector; \(\dot{\mathrm{p}}=\) linear momentum; \(\ddot{\mathrm{L}}=\) angular momentum; \(\ddot{\alpha}=\) angular acceleration; \(\mathrm{I}=\) moment of inertia; \(\ddot{\mathrm{F}}=\) force; \(\mathrm{t}=\) time \()\)
Choose the correct answer from the options given below:
In a Young's double slit experiment, the slits are separated by 0.2 mm. If the slits separation is increased to 0.4 mm, the percentage change of the fringe width is:
An alternating current is represented by the equation, \(\mathrm{i}=100 \sqrt{2} \sin (100 \pi \mathrm{t})\) ampere. The RMS value of current and the frequency of the given alternating current are
Consider the sound wave travelling in ideal gases of \(\mathrm{He}, \mathrm{CH}_{4}\), and \(\mathrm{CO}_{2}\). All the gases have the same ratio \(\frac{\mathrm{P}}{\rho}\), where P is the pressure and \(\rho\) is the density. The ratio of the speed of sound through the gases \(\mathrm{v}_{\mathrm{He}}: \mathrm{v}_{\mathrm{CH}_{4}}: \mathrm{v}_{\mathrm{CO}_{2}}\) is given by
In an electromagnetic system, the quantity representing the ratio of electric flux and magnetic flux has dimension of \(\mathrm{M}^{\mathrm{B}} \mathrm{L}^{\mathrm{O}} \mathrm{T}^{\mathrm{B}} \mathrm{A}^{\mathrm{S}}\), where value of 'Q' and 'R' are
When an object is placed 40 cm away from a spherical mirror an image of magnification \(\frac{1}{2}\) is produced. To obtain an image with magnification of \(\frac{1}{3}\), the object is to be moved:
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: In photoelectric effect, on increasing the intensity of incident light the stopping potential increases. Reason R: Increase in intensity of light increases the rate of photoelectrons emitted, provided the frequency of incident light is greater than threshold frequency.
If \(\overrightarrow{\mathrm{L}}\) and \(\overrightarrow{\mathrm{P}}\) represent the angular momentum and linear momentum respectively of a particle of mass ' \(m\) ' having position vector \(\overrightarrow{\mathrm{r}}=\mathrm{a}(\hat{\mathrm{i}} \cos \omega \mathrm{t}+\hat{\mathrm{j}} \sin \omega \mathrm{t})\). The direction of force is
A body of mass \(m\) is suspended by two strings making angles \(\theta_{1}\) and \(\theta_{2}\) with the horizontal ceiling with tensions \(\mathrm{T}_{1}\) and \(\mathrm{T}_{2}\) simultaneously. \(\mathrm{T}_{1}\) and \(\mathrm{T}_{2}\) are related by \(\mathrm{T}_{1}=\sqrt{3} \mathrm{~T}_{2}\). the angles \(\theta_{1}\) and \(\theta_{2}\) are
Current passing through a wire as function of time is given as \(I(t)=0.02 \mathrm{t}+0.01 \mathrm{~A}\). The charge that will flow through the wire from \(t=1 \mathrm{~s}\) to \(\mathrm{t}=2 \mathrm{~s}\) is:
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: The kinetic energy needed to project a body of mass \(m\) from earth surface to infinity is \(\frac{1}{2} \mathrm{mgR}\), where R is the radius of earth. Reason R: The maximum potential energy of a body is zero when it is projected to infinity from earth surface.
The Boolean expression \(\mathrm{Y}=\mathrm{A} \overline{\mathrm{B}} \mathrm{C}+\overline{\mathrm{AC}}\) can be realised with which of the following gate configurations.
A. One 3-input AND gate, 3 NOT gates and one 2-input OR gate, One 2-input AND gate
B. One 3-input AND gate, 1 NOT gate, One 2-input NOR gate and one 2-input OR gate
C. 3-input OR gate, 3 NOT gates and one 2-input AND gate
Choose the correct answer from the options given below:
Two simple pendulums having lengths \(l_{1}\) and \(l_{2}\) with negligible string mass undergo angular displacements \(\theta_{1}\) and \(\theta_{2}\), from their mean positions, respectively. If the angular accelerations of both pendulums are same, then which expression is correct?
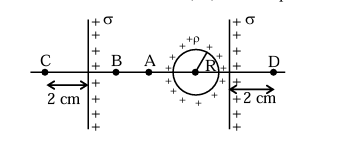
Two infinite identical charged sheets and a charged spherical body of charge density ' \(\rho\) ' are arranged as shown in figure. Then the correct relation between the electrical fields at \(\mathrm{A}, \mathrm{B}, \mathrm{C}\) and D points is:
Two small spherical balls of mass 10 g each with charges \(-2 \mu \mathrm{C}\) and \(2 \mu \mathrm{C}\), are attached to two ends of very light rigid rod of length 20 cm. The arrangement is now placed near an infinite nonconducting charge sheet with uniform charge density of \(100 \mu \mathrm{C} / \mathrm{m}^{2}\) such that length of rod makes an angle of \(30^{\circ}\) with electric field generated by charge sheet. Net torque acting on the rod is:
Considering the Bohr model of hydrogen like atoms, the ratio of the radius \(5^{\text {th }}\) orbit of the electron in \(\mathrm{Li}^{2+}\) and \(\mathrm{He}^{+}\)is
A circular ring and a solid sphere having same radius roll down on an inclined plane from rest without slipping. The ratio of their velocities when reached at the bottom of the plane is \(\sqrt{\frac{\mathrm{x}}{5}}\) where \(\mathrm{x}=\) _______.
Two slabs with square cross section of different materials \((1,2)\) with equal sides \((l)\) and thickness \(\mathrm{d}_{1}\) and \(\mathrm{d}_{2}\) such that \(\mathrm{d}_{2}=2 \mathrm{~d}_{1}\) and \(l>\mathrm{d}_{2}\). Considering lower edges of these slabs are fixed to the floor, we apply equal shearing force on the narrow faces. The angle of deformation is \(\theta_{2}=2 \theta_{1}\). If the shear moduli of material 1 is \(4 \times 10^{9} \mathrm{~N} / \mathrm{m}^{2}\), then shear moduli of material 2 is \(\mathrm{x} \times 10^{9} \mathrm{~N} / \mathrm{m}^{2}\), where value of x is _______ .
Distance between object and its image (magnified by \(-\frac{1}{3}\) ) is 30 cm. The focal length of the mirror used is \(\left(\frac{\mathrm{x}}{4}\right) \mathrm{cm}\), where magnitude of value of x is _______ .
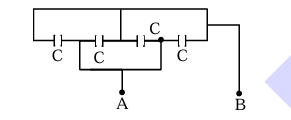
Four capacitors each of capacitance \(16\,\mu F\) are connected as shown in the figure. The capacitance between points A and B is: _________ (in \(\mu F\)).
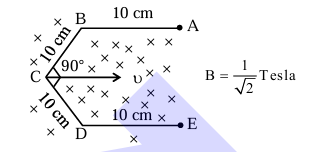
Conductor wire ABCDE with each arm 10 cm in length is placed in magnetic field of \(\frac{1}{\sqrt{2}}\) Tesla, perpendicular to its plane. When conductor is pulled towards right with constant velocity of \(10 \mathrm{~cm} / \mathrm{s}\), induced emf between points A and E is _______ mV.
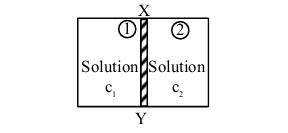
\(XY\) is the membrane / partition between two chambers 1 and 2 containing sugar solutions of concentration \(\mathrm{c}_{1}\) and \(\mathrm{c}_{2}\left(\mathrm{c}_{1}>\mathrm{c}_{2}\right) \mathrm{mol} \mathrm{L}^{-1}\). For the reverse osmosis to take place identify the correct condition
(Here \(\mathrm{p}_{1}\) and \(\mathrm{p}_{2}\) are pressures applied on chamber 1 and 2 )
Let us consider a reversible reaction at temperature, T . In this reaction, both \(\Delta \mathrm{H}\) and \(\Delta \mathrm{S}\) were observed to have positive values. If the equilibrium temperature is \(\mathrm{T}_{\mathrm{e}}\), then the reaction becomes spontaneous at:
Which of the following molecules(s) show/s paramagnetic behavior?
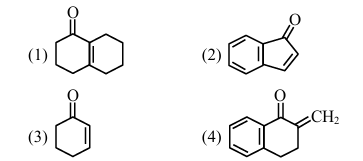
Aldol condensation is a popular and classical method to prepare \(\alpha, \beta\)-unsaturated carbonyl compounds. This reaction can be both intermolecular and intramolecular. Predict which one of the following is not a product of intramolecular aldol condensation?
One mole of an ideal gas expands isothermally and reversibly from \(10 \mathrm{dm}^{3}\) to \(20 \mathrm{dm}^{3}\) at \(300 \mathrm{~K} . \Delta \mathrm{U}\), q and work done in the process respectively are :
Given : \(\mathrm{R}=8.3 \mathrm{JK}^{-1}\) and \(\mathrm{mol}^{-1}\)
In \(10=2.3\)
\(\log 2=0.30\)
\(\log 3=0.48\)
Which one of the following complexes will have \(\Delta_{0}=0\) and \(\mu=5.96\) B.M.?
For \(\mathrm{A}_{2}+\mathrm{B}_{2} \rightleftharpoons 2 \mathrm{AB}\) \(\mathrm{E}_{\mathrm{a}}\) for forward and backward reaction are 180 and \(200 \mathrm{~kJ} \mathrm{~mol}^{-1}\) respectively. If catalyst lowers \(\mathrm{E}_{\mathrm{a}}\) for both reaction by \(100 \mathrm{~kJ} \mathrm{~mol}^{-1}\). Which of the following statement is correct?
Rate law for a reaction between \(A\) and \(B\) is given by \(\mathrm{R}=\mathrm{k}[\mathrm{A}]^{\mathrm{n}}[\mathrm{B}]^{\mathrm{m}}\). If concentration of A is doubled and concentration of B is halved from their initial value, the ratio of new rate of reaction to the initial rate of reaction \(\left(\frac{\mathrm{r}_{2}}{\mathrm{r}_{1}}\right)\) is
Number of stereoisomers possible for the complexes, \(\left[\mathrm{CrCl}_{3}(\mathrm{py})_{3}\right]\) and \(\left[\mathrm{CrCl}_{2}(\mathrm{ox})_{2}\right]^{3-}\) are respectively
(py = pyridine, ox = oxalate)
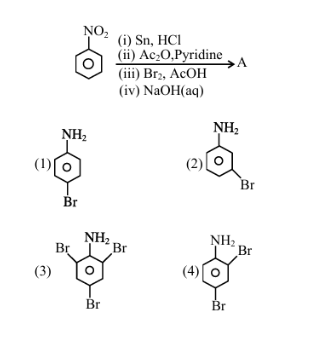
The major product (A) formed in the following reaction sequence is
On charging the lead storage battery, the oxidation state of lead changes from \(\mathrm{x}_{1}\) to \(\mathrm{y}_{1}\) at the anode and from \(\mathrm{x}_{2}\) to \(\mathrm{y}_{2}\) at the cathode. The values of \(\mathrm{x}_{1}, \mathrm{y}_{1}, \mathrm{x}_{2}, \mathrm{y}_{2}\) are respectively:
Given below are two statements:
Statement I: Nitrogen forms oxides with +1 to +5 oxidation states due to the formation of \(\mathrm{p} \pi-\mathrm{p} \pi\) bond with oxygen.
Statement II: Nitrogen does not form halides with +5 oxidation state due to the absence of d-orbital in it.
In the light of the above statements, choose the correct answer from the options given below:
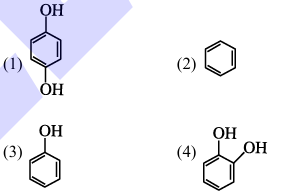
Benzene is treated with oleum to produce compound (X) which when further heated with molten sodium hydroxide followed by acidification produces compound (Y).The compound Y is treated with zinc metal to produce compound (Z). Identify the structure of compound (Z) from the following option.
Identify the pair of reactants that upon reaction, with elimination of HCl will give rise to the dipeptide Gly-Ala.
Given below are the pairs of group 13 elements showing their relation in terms of atomic radius. \((\mathrm{B}<\mathrm{Al}),(\mathrm{Al}<\mathrm{Ga}),(\mathrm{Ga}<\mathrm{In})\) and \((\mathrm{In}<\mathrm{Tl})\) Identify the elements present in the incorrect pair and in that pair find out the element (X) that has higher ionic radius \(\left(\mathrm{M}^{3+}\right)\) than the other one. The atomic number of the element (X) is
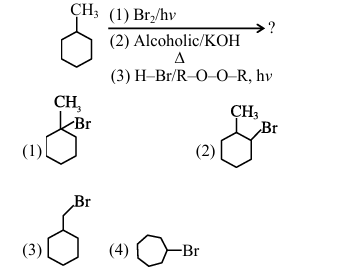
An organic compound (X) with molecular formula \(\mathrm{C}_{3} \mathrm{H}_{6} \mathrm{O}\) is not readily oxidised. On reduction it gives \(\left(\mathrm{C}_{3} \mathrm{H}_{8} \mathrm{O}(\mathrm{Y})\right.\) which reacts with HBr to give a bromide (Z) which is converted to Grignard reagent. This Grignard reagent on reaction with (X) followed by hydrolysis give 2,3-dimethylbutan-2-ol. Compounds (X), (Y) and (Z) respectively are:
Given below are two statements.
In the light of the above statements, choose the correct answer from the options given below:

Pair of transition metal ions having the same number of unpaired electrons is :
Which one of the following about an electron occupying the 1 s orbital in a hydrogen atom is incorrect ? (Bohr's radius is represented by \(\mathrm{a}_{0}\) )
In Dumas' method for estimation of nitrogen 1 g of an organic compound gave 150 mL of nitrogen collected at 300 K temperature and 900 mm Hg pressure. The percentage composition of nitrogen in the compound is _______ % (nearest integer).
(Aqueous tension at \(300 \mathrm{~K}=15 \mathrm{~mm} \mathrm{Hg}\) )
\(\mathrm{KMnO}_{4}\) acts as an oxidising agent in acidic medium. ' X ' is the difference between the oxidation states of Mn in reactant and product. ' Y ' is the number of ' d ' electrons present in the brown red precipitate formed at the end of the acetate ion test with neutral ferric chloride. The value of \(\mathrm{X}+\mathrm{Y}\) is _______ .
Fortification of food with iron is done using \(\mathrm{FeSO}_{4} .7 \mathrm{H}_{2} \mathrm{O}\). The mass in grams of the \(\mathrm{FeSO}_{4} .7 \mathrm{H}_{2} \mathrm{O}\) required to achieve 12 ppm of iron in 150 kg of wheat is _______ (Nearest integer).
(Given : Molar mass of \(\mathrm{Fe}, \mathrm{S}\) and O respectively are 56,32 and \(16 \mathrm{~g} \mathrm{~mol}^{-1}\) )
The pH of a 0.01 M weak acid \(\mathrm{HX}\left(\mathrm{K}_{\mathrm{a}}=4 \times 10^{-10}\right)\) is found to be 5 . Now the acid solution is diluted with excess of water so that the pH of the solution changes to 6 . The new concentration of the diluted weak acid is given as \(\mathrm{x} \times 10^{-4} \mathrm{M}\). The value of x is _______ (nearest integer).
The total number of hydrogen bonds of a DNA-double Helix strand whose one strand has the following sequence of bases is _______ .
\(5^{\prime}-\mathrm{G}-\mathrm{G}-\mathrm{C}-\mathrm{A}-\mathrm{A}-\mathrm{A}-\mathrm{T}-\mathrm{C}-\mathrm{G}-\mathrm{G}-\mathrm{C}-\mathrm{T}-\mathrm{A}-3^{\prime}\)
JEE Main April 4 Shift 1 Question Paper With Video Solutions
The JEE Main 2025 April 4 Question Paper with Video Solution is available here after the examination. Students can match their responses with the JEE Main 2025 April 4 Shift 1 Answer key with Solutions. Students who are appearing for JEE Main upcoming shifts can check the JEE Main April 4 Shift 1 Question Paper and video solution to understand the difficulty level of the exam.
JEE Main Session 2 Difficulty Level Analysis
| JEE Mains 2025 Session 2 exam dates | Paper | Exam Analysis |
|---|---|---|
| April 2, 2025 | Paper 1 (B.E/BTech) |
|
| April 3, 2025 | Paper 1 (B.E/BTech) |
|
| April 4, 2025 | Paper 1 (B.E/BTech) |
|
| April 7, 2025 | Paper 1 (B.E/BTech) | TBA |
| April 8, 2025 | Paper 1 (B.E/BTech) | TBA |
| April 9, 2025 | Paper 2A (BArch) and Paper 2B (BPlanning) | TBA |
Related Links:
- JEE Main 2025 Question Paper pdf with solutions
- JEE Main Previous Years Question Paper with Solution PDF
JEE Main 2025 April 4 Shift 1 Answer Key: Coaching Institute PDF
| Coaching Institutes | Question Paper with Solutions PDF |
|---|---|
| Aakash BYJUs | Download Now |
| Allen Institute | Download Now |
| Resonance | Download Now |
| Vedantu | Download Now |
| Sri Chaitanya | Download Now |
JEE Main Marking Scheme 2025
For JEE Main Paper 1(B.E/BTech), students are required to attempt 75 questions following a marking scheme of +4 for correct answers and -1 for incorrect ones. The marking scheme is the same across Physics, Chemistry, and Mathematics sections totaling to 300 marks.
| Paper | Sections | Questions | Marks | Marking Scheme |
|---|---|---|---|---|
| Paper 1 | Physics, Chemistry, Mathematics | 75 Questions (25 Physics, 25 Chemistry, 25 Mathematics) | 300 total |
|
Is there any negative marking in JEE Main Paper 1?
- If you choose the wrong answer in both Section A and Section B, you will lose 1 mark because of negative marking.
- In Section B, instead of choosing an option, you have to type the correct whole number using the on screen number pad. If your answer is a decimal, round it off to the nearest whole number before entering it.
Once you calculate your expected marks, you can review the JEE Main Question papers to improve your preparation.
You can check:
- JEE Main Previous Years Question Paper with Solution
- JEE Main 2024 Question Paper with Solution PDF
- JEE Main 2023 Question Paper with Solution PDF
- JEE Main 2022 Question Paper with Solution PDF
What to carry at the JEE Main Test Centre ?
Every aspirant must check the JEE Main exam day guidelines before the examination. The JEE Main exam instructions comprise of all the dos and don’ts to be followed for a hassle-free exam process. JEE Mains rules and regulations for the exam are provided below:
| What to Carry | What NOT to Carry |
|---|---|
| JEE Main 2025 Admit Card (Mandatory) | Electronic gadgets (Mobile phones, smartwatches, calculators, Bluetooth devices) |
| Valid ID Proof (Aadhaar Card, PAN Card, Voter ID, Passport, etc.) | Written/Printed Study Materials (Books, Notes, Papers) |
| A simple transparent ballpoint pen (for rough work) | Any type of rough paper (NTA will provide sheets) |
| A passport-size photograph (same as on the admit card) | Smart devices (Earphones, Digital Watches, Fitness Bands) |
| PwD Certificate (if applicable) | Geometry boxes, Log tables, or Any extra stationery items |
| A transparent water bottle (Optional) | Wallets, Purses, or Bags |
How to attempt JEE Main 2025 MCQ’s with Maximum Accuracy?
For accuracy in JEE Main MCQ’s concepts need to be well understood. On average, based on past JEE exams, almost 60 % of the incorrect answers are because of reading the question wrong or marking the wrong answers.
For numerical value questions, if the question requires you to round it off to the nearest integer, accuracy is important. It can cost 4 marks per question which can have a significant impact on your rank.
In revising an answer, many students choose to trade their initial answer for another incorrect alternative. Based on the past year trends, first solutions are accurate most of the time, a figure close to 70%. One should second-guess only on a sound reason for doubt about error.







Comments