JEE Main 7 April Shift 1 question paper with solutions and answers pdf is available here for download. NTA conducted JEE Main 7 April Shift 1 exam from 9 AM to 12 PM. The question paper for JEE Main 7 April Shift 1 includes 90 questions equally divided into Physics, Chemistry and Maths. Candidates must attempt 75 questions in a 3-hour time duration.
Students who are appeared for JEE Main April 7 Shift 1 Exam can check memory based question paper and difficulty level of the exam on this page. JEE Main 2025 April 7 Shift 1 exam is reported to be easy to moderate. Physics and Chemistry had a lot of NCERT- based questions.
JEE Main 2025 is conducted in various cities across the country, with 15 destinations abroad. In JEE Main April 7 Shift 1 students are required to attempt 75 questions following a marking scheme of +4 for correct answers and -1 for incorrect ones. Check JEE Main April 7 Shift 1 Answer Key
JEE Main 2025 April 7 Shift 1 Question Paper with Solutions
| JEE Main 2025 April 7 Shift 1 Question Paper Pdf | Download | Check Solutions |

MATHEMATICS
SECTION-A
Question 1:
Evaluate the following limit: \[ \lim_{x \to 0^+} \frac{\tan\left(5x^{\frac{1}{3}}\right) \log\left(1 + 3x^2\right)}{\left(\tan^{-1}\left(3\sqrt{x}\right)\right)^2 \left(e^{5x^{\frac{4}{3}}} - 1\right)} \]
If the shortest distance between the lines \( \frac{x-1}{2} = \frac{y-2}{3} = \frac{z-3}{4} \) and \( \frac{x}{1} = \frac{y}{\alpha} = \frac{z-5}{1} \) is \( \frac{5}{\sqrt{6}} \), then the sum of all possible values of \( \alpha \) is:
Let \( x = -1 \) and \( x = 2 \) be the critical points of the function \( f(x) = x^3 + ax^2 + b \log|x| + 1 \), where \( x \neq 0 \). Let \( m \) and \( M \) be the absolute minimum and maximum values of \( f \) in the interval \( \left[-2, -\frac{1}{2}\right] \). Then, \( |M + m| \) is equal to:
The remainder when \( \left( (64)^{64} \right)^{64} \) is divided by 7 is equal to:
Let P be the parabola, whose focus is \( (-2, 1) \) and directrix is \( 2x + y + 2 = 0 \). Then the sum of the ordinates of the points on P, whose abscissa is -2, is:
Let \( y = y(x) \) be the solution curve of the differential equation \[ x(x^2 + e^x) \, dy + \left( e^x(x - 2) y - x^3 \right) \, dx = 0, \quad x > 0, \]
passing through the point \( (1, 0) \). Then \( y(2) \) is equal to:
From a group of 7 batsmen and 6 bowlers, 10 players are to be chosen for a team, which should include at least 4 batsmen and at least 4 bowlers. One batsman and one bowler who are captain and vice-captain respectively of the team should be included. Then the total number of ways such a selection can be made, is:
If for \( \theta \in \left[ -\frac{\pi}{3}, 0 \right] \), the points \[ (x, y) = \left( 3 \tan\left( \theta + \frac{\pi}{3} \right), 2 \tan\left( \theta + \frac{\pi}{6} \right) \right) \]
lie on \( xy + \alpha x + \beta y + \gamma = 0 \), then \( \alpha^2 + \beta^2 + \gamma^2 \) is equal to:
Let \( C_1 \) be the circle in the third quadrant of radius 3, that touches both coordinate axes. Let \( C_2 \) be the circle with center \( (1, 3) \) that touches \( C_1 \) externally at the point \( (\alpha, \beta) \). If \( (\beta - \alpha)^2 = \frac{m}{n} \), and \( \gcd(m, n) = 1 \), then \( m + n \) is equal to:
The integral \[ \int_0^\pi \frac{(x + 3) \sin x}{1 + 3 \cos^2 x} \, dx \]
is equal to:
Among the statements:
(S1): The set \( \{ z \in \mathbb{C} - \{-i\} : |z| = 1 and \frac{z - i}{z + i} is purely real \} \) contains exactly two elements.
(S2): The set \( \{ z \in \mathbb{C} - \{-1\} : |z| = 1 and \frac{z - 1}{z + 1} is purely imaginary \} \) contains infinitely many elements.
Then, which of the following is correct?
The mean and standard deviation of 100 observations are 40 and 5.1, respectively. By mistake one observation is taken as 50 instead of 40. If the correct mean and the correct standard deviation are \( \mu \) and \( \sigma \) respectively, then \( 10(\mu + \sigma) \) is equal to:
Let \( x_1, x_2, x_3, x_4 \) be in a geometric progression. If 2, 7, 9, 5 are subtracted respectively from \( x_1, x_2, x_3, x_4 \), then the resulting numbers are in an arithmetic progression. Then the value of \( \frac{1}{24} (x_1 x_2 x_3 x_4) \) is:
Let the set of all values of \( p \in \mathbb{R} \), for which both the roots of the equation \( x^2 - (p + 2)x + (2p + 9) = 0 \) are negative real numbers, be the interval \( (\alpha, \beta) \). Then \( \beta - 2\alpha \) is equal to:
Let \( A \) be a \( 3 \times 3 \) matrix such that \[ | adj (adj A) | = 81. \]
If \[ S = \left\{ n \in \mathbb{Z}: \left| adj (adj A) \right|^{\frac{(n - 1)^2}{2}} = |A|^{(3n^2 - 5n - 4)} \right\}, \]
then the value of \[ \sum_{n \in S} |A| (n^2 + n) \]
is:
If the area of the region bounded by the curves \( y = 4 - \frac{x^2}{4} \) and \( y = \frac{x - 4}{2} \) is equal to \( \alpha \), then \( 6\alpha \) equals:
Let the system of equations be: \[ 2x + 3y + 5z = 9, \] \[ 7x + 3y - 2z = 8, \] \[ 12x + 3y - (4 + \lambda)z = 16 - \mu, \]
which has infinitely many solutions. Then the radius of the circle centered at \( (\lambda, \mu) \) and touching the line \( 4x = 3y \) is:
Let the line \( L \) pass through \( (1, 1, 1) \) and intersect the lines \[ \frac{x - 1}{2} = \frac{y + 1}{3} = \frac{z - 1}{4} \]
and \[ \frac{x - 3}{1} = \frac{y - 4}{2} = \frac{z}{1}. \]
Then, which of the following points lies on the line \( L \)?
Let the angle \( \theta, 0 < \theta < \frac{\pi}{2} \) between two unit vectors \( \hat{a} \) and \( \hat{b} \) be \( \sin^{-1} \left( \frac{\sqrt{65}}{9} \right) \). If the vector \( \vec{c} = 3\hat{a} + 6\hat{b} + 9(\hat{a} \times \hat{b}) \), then the value of \( 9(\vec{c} \cdot \hat{a}) - 3(\vec{c} \cdot \hat{b}) \) is:
Let \( ABC \) be the triangle such that the equations of lines \( AB \) and \( AC \) are: \[ 3y - x = 2 \quad and \quad x + y = 2, \]
respectively, and the points \( B \) and \( C \) lie on the x-axis. If \( P \) is the orthocentre of the triangle \( ABC \), then the area of the triangle \( PBC \) is equal to:
SECTION-B
Question 21:
The number of points of discontinuity of the function \[ f(x) = \left\lfloor \frac{x^2}{2} \right\rfloor - \left\lfloor \sqrt{x} \right\rfloor, \quad x \in [0, 4], \]
where \( \left\lfloor \cdot \right\rfloor \) denotes the greatest integer function, is:
The number of relations on the set \( A = \{1, 2, 3\} \) containing at most 6 elements including \( (1, 2) \), which are reflexive and transitive but not symmetric, is:
Consider the hyperbola \[ \frac{x^2}{a^2} - \frac{y^2}{b^2} = 1, \]
having one of its foci at \( P(-3, 0) \). If the latus rectum through its other focus subtends a right angle at \( P \), and \[ a^2b^2 = \alpha\sqrt{2} - \beta, \quad \alpha, \beta \in \mathbb{N}, \]
then find \( \alpha \) and \( \beta \).
The number of singular matrices of order 2, whose elements are from the set \( \{2, 3, 6, 9\} \) is:
For \( n \geq 2 \), let \( S_n \) denote the set of all subsets of \( \{1, 2, 3, \ldots, n\} \) with no two consecutive numbers. For example, \( \{1, 3, 5\} \in S_6 \), but \( \{1, 2, 4\} \notin S_6 \). Then, find \( n(S_5) \).
PHYSICS
SECTION-A
Question 26:
Two harmonic waves moving in the same direction superimpose to form a wave \( x = a \cos(1.5t) \cos(50.5t) \) where \( t \) is in seconds. Find the period with which they beat (close to the nearest integer):
Two plane polarized light waves combine at a certain point whose electric field components are \( E_1 = E_0 \sin(\omega t) \) \( E_2 = E_0 \sin(\omega t + \frac{\pi}{3}) \)
Find the amplitude of the resultant wave.
A wire of resistance \( R \) is bent into a triangular pyramid as shown in the figure, with each segment having the same length. The resistance between points \( A \) and \( B \) is \( \frac{R}{n} \). The value of \( n \) is:
Uniform magnetic fields of different strengths \( B_1 \) and \( B_2 \), both normal to the plane of the paper, exist as shown in the figure. A charged particle of mass \( m \) and charge \( q \), at the interface at an instant, moves into region 2 with velocity \( v \) and returns to the interface. It continues to move into region 1 and finally reaches the interface. What is the displacement of the particle during this movement along the interface?
Consider the velocity of the particle to be normal to the magnetic field and \( B_2 > B_1 \).
If \( \epsilon_0 \) denotes the permittivity of free space and \( \Phi_E \) is the flux of the electric field through the area bounded by the closed surface, then the dimension of \( \epsilon_0 \frac{d\Phi_E}{dt} \) are that of:
A rod of length \( 5L \) is bent at a right angle, keeping one side length as \( 2L \).
The position of the centre of mass of the system (Consider \( L = 10 \) cm):
The percentage increase in magnetic field \( B \) when space within a current-carrying solenoid is filled with magnesium (magnetic susceptibility \( \chi_{mg} = 1.2 \times 10^{-5} \)) is:
A lens having refractive index 1.6 has focal length of 12 cm, when it is in air. Find the focal length of the lens when it is placed in water.
(Take refractive index of water as 1.28)
An AC current is represented as: \[ i = 5\sqrt{2} + 10 \cos\left(650\pi t + \frac{\pi}{6}\right) Amp \]
The RMS value of the current is:
Two thin convex lenses of focal lengths 30 cm and 10 cm are placed coaxially, 10 cm apart. The power of this combination is:
In the following circuit, the reading of the ammeter will be: (Take Zener breakdown voltage = 4 V)
Two projectiles are fired from the ground with the same initial speeds from the same point at angles \( (45^\circ + \alpha) \) and \( (45^\circ - \alpha) \) with the horizontal direction. The ratio of their times of flights is:
Match the List-I with List-II.
Choose the correct answer from the options given below:
A cubic block of mass \( m \) is sliding down on an inclined plane at \( 60^\circ \) with an acceleration of \( \frac{g}{2} \), the value of coefficient of kinetic friction is:
In a hydrogen-like ion, the energy difference between the 2nd excitation energy state and ground is 108.8 eV. The atomic number of the ion is:
For a hydrogen atom, the ratio of the largest wavelength of the Lyman series to that of the Balmer series is:
A particle of charge \( q \), mass \( m \), and kinetic energy \( E \) enters in a magnetic field perpendicular to its velocity and undergoes a circular arc of radius \( r \). Which of the following curves represents the variation of \( r \) with \( E \)?
An object of mass 1000 g experiences a time-dependent force \( \vec{F} = (2t \hat{i} + 3t^2 \hat{j}) \, N \). The power generated by the force at time \( t \) is:
Two wires A and B are made of the same material, having the ratio of lengths \( \frac{L_A}{L_B} = \frac{1}{3} \) and their diameters ratio \( \frac{d_A}{d_B} = 2 \). If both the wires are stretched using the same force, what would be the ratio of their respective elongations?
Two charges \( q_1 \) and \( q_2 \) are separated by a distance of 30 cm. A third charge \( q_3 \) initially at C as shown in the figure, is moved along the circular path of radius 40 cm from C to D. If the difference in potential energy due to the movement of \( q_3 \) from C to D is given by \( \frac{q_3 K}{4 \pi \epsilon_0} \), the value of \( K \) is:
SECTION-B
Question 46:
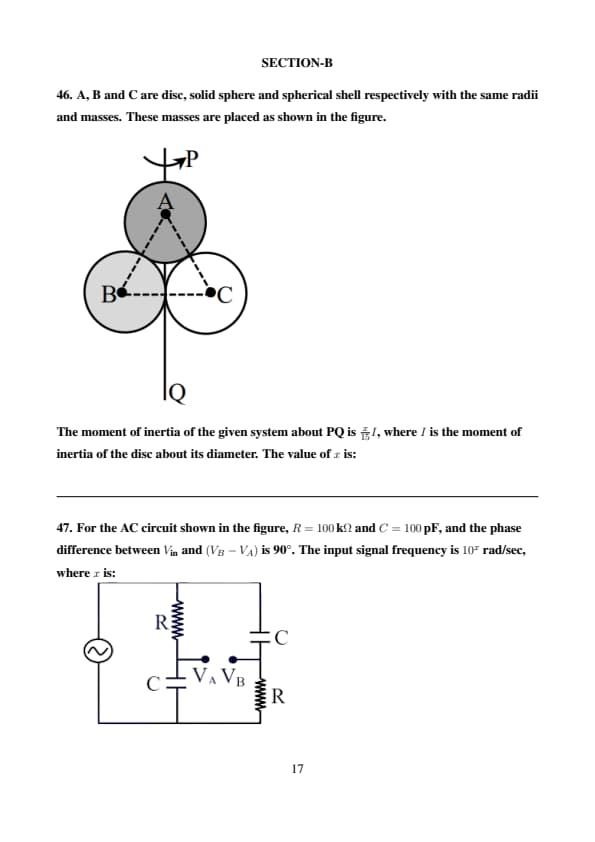
A, B and C are disc, solid sphere and spherical shell respectively with the same radii and masses. These masses are placed as shown in the figure.
The moment of inertia of the given system about PQ is \( \frac{x}{15} I \), where \( I \) is the moment of inertia of the disc about its diameter. The value of \( x \) is:
For the AC circuit shown in the figure, \( R = 100 \, k\Omega \) and \( C = 100 \, pF \), and the phase difference between \( V_{in} \) and \( (V_B - V_A) \) is 90°. The input signal frequency is \( 10^x \) rad/sec, where \( x \) is:
A container contains a liquid with refractive index of 1.2 up to a height of 60 cm and another liquid having refractive index 1.6 is added to height \( H \) above the first liquid. If viewed from above, the apparent shift in the position of the bottom of the container is 40 cm. The value of \( H \) is ___ cm.
A wire of length 10 cm and diameter 0.5 mm is used in a bulb. The temperature of the wire is 1727°C and power radiated by the wire is 94.2 W. Its emissivity is \( \frac{x}{8} \), where \( x = \ldots \)
An ideal gas has undergone through the cyclic process as shown in the figure. Work done by the gas in the entire cycle is _____ \( \times 10^{-1} \) J. (Take \( \pi = 3.14 \))
CHEMISTRY
SECTION-A
Question 51:
Given below are two statements:
Statement I: Ozonolysis followed by treatment with Zn, H\(_2\)O of cis-2-butene gives ethanal.
Statement II: The product obtained by ozonolysis followed by treatment with Zn, H\(_2\)O of 3, 6-dimethyloct-4-ene has no chiral carbon atom.
In the light of the above statements, choose the correct answer from the options given below.
Which of the following amine(s) show(s) positive carbamylamine test?
Reaction A(g) → 2B(g) + C(g) is a first-order reaction. It was started with pure A.
The following table shows the pressure of the system at different times:
Which of the following options is incorrect?
Total enthalpy change for freezing of 1 mol water at 10°C to ice at -10°C is ______
(Given: \( \Delta_{fus}H = x \, kJ/mol \), \( C_p[H_2O(l)] = y \, J mol^{-1} K^{-1} \), and \( C_p[H_2O(s)] = z \, J mol^{-1} K^{-1} \))
An aqueous solution of HCl with pH 1.0 is diluted by adding equal volume of water (ignoring dissociation of water). The pH of HCl solution would be: \[ (Given log 2 = 0.30) \]
Given below are two statements:
Statement I: Dimethyl ether is completely soluble in water. However, diethyl ether is soluble in water to a very small extent.
Statement II: Sodium metal can be used to dry diethyl ether and not ethyl alcohol.
In the light of the given statements, choose the correct answer from the options given below:
Which of the following statements are correct, if the threshold frequency of caesium is \( 5.16 \times 10^{14} \, Hz \)?
Which of the following is the correct IUPAC name of the given organic compound (X)?
The structure of compound \( X \) is as follows: \[ H_3C - CH_3 - CH = CH - H - Br \]
At the sea level, the dry air mass percentage composition is given as nitrogen gas : 70.0, oxygen gas : 27.0, and argon gas : 3.0. If the total pressure is 1.15 atm, then calculate the ratio of the following respectively:
(i) Partial pressure of nitrogen gas to partial pressure of oxygen gas
(ii) Partial pressure of oxygen gas to partial pressure of argon gas
(Given: Molar mass of N, O, and Ar are 14, 16, and 40 g mol\(^{-1}\) respectively)
Given below are two statements:
Statement I: Mohr's salt is composed of only three types of ions—ferrous, ammonium, and sulphate.
Statement II: If the molar conductance at infinite dilution of ferrous, ammonium, and sulphate ions are \( x_1 \), \( x_2 \), and \( x_3 \) \( S cm^2 \, mol^{-1} \), respectively, then the molar conductance for Mohr's salt solution at infinite dilution would be given by \( x_1 + x_2 + 2x_3 \).
The number of valence electrons present in the metal among Cr, Co, Fe, and Ni which has the lowest enthalpy of atomisation is
When a salt is treated with sodium hydroxide solution, it gives gas X. On passing gas X through reagent Y, a brown coloured precipitate is formed. X and Y respectively, are:
The group 14 elements A and B have the first ionisation enthalpy values of 708 and 715 kJ mol\(^{-1}\) respectively. The above values are lowest among their group members. The nature of their ions A\(^{2+}\) and B\(^{4+}\) respectively is:
The first transition series metal 'M' has the highest enthalpy of atomisation in its series. One of its aquated ion (M\(^{n+}\)) exists in green colour. The nature of the oxide formed by the above M\(^{n-}\) ion is:
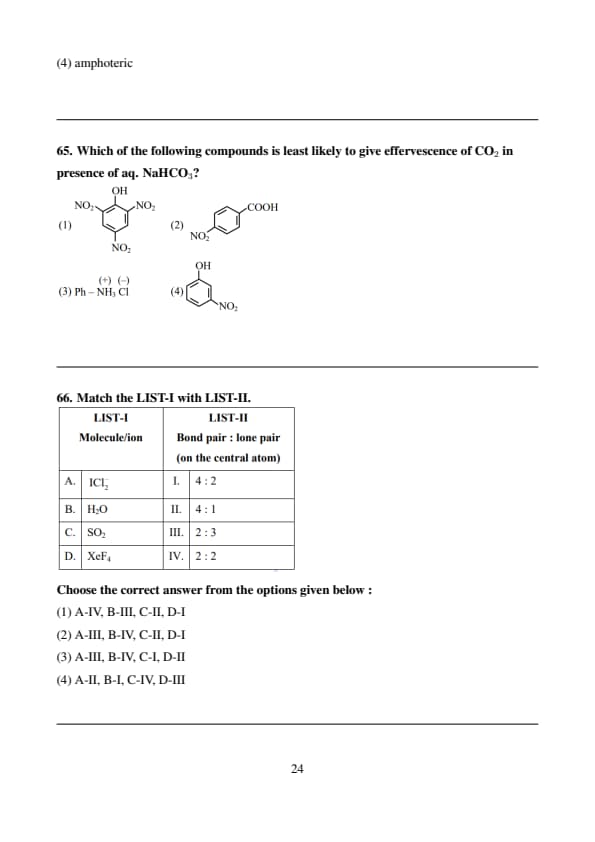
Which of the following compounds is least likely to give effervescence of CO\(_2\) in presence of aq. NaHCO\(_3\)?
Match the LIST-I with LIST-II.
Choose the correct answer from the options given below :
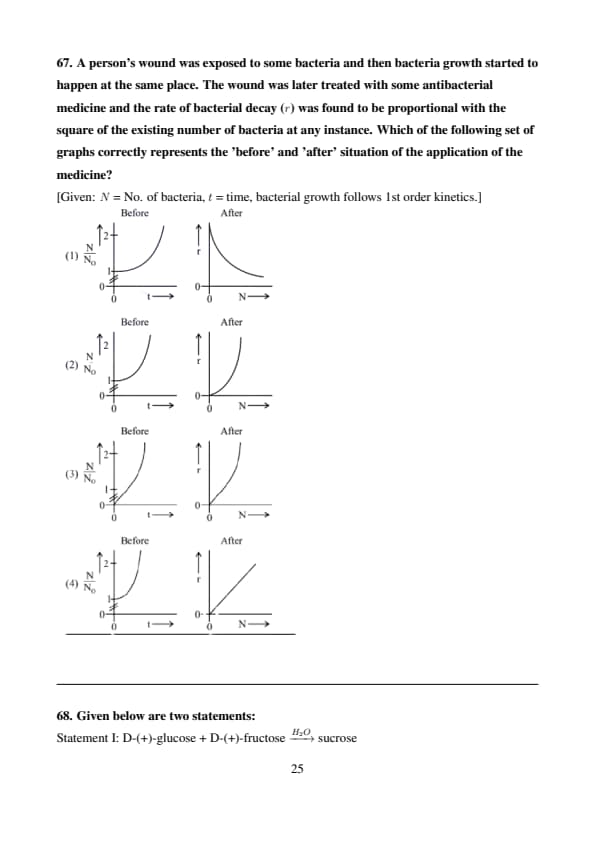
A person's wound was exposed to some bacteria and then bacteria growth started to happen at the same place. The wound was later treated with some antibacterial medicine and the rate of bacterial decay (\(r\)) was found to be proportional with the square of the existing number of bacteria at any instance. Which of the following set of graphs correctly represents the 'before' and 'after' situation of the application of the medicine?
[Given: \( N \) = No. of bacteria, \( t \) = time, bacterial growth follows 1st order kinetics.]
Given below are two statements:
Statement I: D-(+)-glucose + D-(+)-fructose \(\xrightarrow{H_2O}\) sucrose
sucrose \(\xrightarrow{Hydrolysis}\) D-(+)-glucose + D-(+)-fructose
Statement II: Invert sugar is formed during sucrose hydrolysis.
In the light of the above statements, choose the correct answer from the options given below -
An octahedral complex having molecular composition Co.5NH\(_3\).Cl.SO\(_4\) has two isomers A and B. The solution of A gives a white precipitate with AgNO\(_3\) solution and the solution of B gives a white precipitate with BaCl\(_2\) solution. The type of isomerism exhibited by the complex is,
The reactions which cannot be applied to prepare an alkene by elimination, are
Choose the correct answer from the options given below:
SECTION-B
Question 71:
An organic compound weighing 500 mg, produced 220 mg of CO\(_2\) on complete combustion. The percentage composition of carbon in the compound is ............... % (nearest integer).
(Given molar mass in g mol\(^-\) of C: 12, O: 16)
Thyroxine, the hormone has the given structure.
The percentage of iodine in thyroxine is ............... % (nearest integer).
(Given molar mass in g mol\(^-\) C:12, H:1, O:16, N:14, I:127)
1 Faraday electricity was passed through Cu\(^{2+}\) (1.5 M, 1 L)/Cu and 0.1 Faraday was passed through Ag\(^+\) (0.2 M, 1 L) electrolytic cells. After this, the two cells were connected as shown below to make an electrochemical cell. The emf of the cell thus formed at 298 K is:
Given: \[ E^\circ_{Cu^{2+}/Cu} = 0.34 \, V \] \[ E^\circ_{Ag^+/ Ag} = 0.8 \, V \] \[ \frac{2.303RT}{F} = 0.06 \, V \]
The percentage dissociation of a salt (MX\(_3\)) solution at a given temperature (van't Hoff factor \(i = 2\)) is .................. % (Nearest integer)
The number of paramagnetic complexes among \([FeF_6]^{3-}, [Fe(CN)_6]^{3-}, [Mn(CN)_6]^{3-}, [Co(C_2O_4)_3]^{3-}, [MnCl_6]^{3-}\) and \([CoF_6]^{3-}\), which involved \(d^2sp^3\) hybridization is ...............

























JEE Main April 7 Shift 1 Question Paper With Video Solutions
The JEE Main 2025 April 7 Question Paper with Video Solution is available here after the examination. Students can match their responses with the JEE Main 2025 April 7 Shift 1 Answer key with Solutions. Students who are appearing for JEE Main upcoming shifts can check the JEE Main April 7 Shift 1 Question Paper and video solution to understand the difficulty level of the exam.
JEE Main Session 2 Difficulty Level Analysis
| JEE Mains 2025 Session 2 exam dates | Paper | Exam Analysis |
|---|---|---|
| April 2, 2025 | Paper 1 (B.E/BTech) |
|
| April 3, 2025 | Paper 1 (B.E/BTech) |
|
| April 4, 2025 | Paper 1 (B.E/BTech) |
|
| April 7, 2025 | Paper 1 (B.E/BTech) | TBA |
| April 8, 2025 | Paper 1 (B.E/BTech) | TBA |
| April 9, 2025 | Paper 2A (BArch) and Paper 2B (BPlanning) | TBA |
Related Links:
- JEE Main 2025 Question Paper pdf with solutions
- JEE Main Previous Years Question Paper with Solution PDF
JEE Main 2025 April 7 Shift 1 Answer Key: Coaching Institute PDF
| Coaching Institutes | Question Paper with Solutions PDF |
|---|---|
| Aakash BYJUs | Download |
| Allen Institute | Download |
| Resonance | Download |
| Vedantu | Download |
| Sri Chaitanya | Download |
JEE Main Marking Scheme 2025
For JEE Main Paper 1(B.E/BTech), students are required to attempt 75 questions following a marking scheme of +4 for correct answers and -1 for incorrect ones. The marking scheme is the same across Physics, Chemistry, and Mathematics sections totaling to 300 marks.
| Paper | Sections | Questions | Marks | Marking Scheme |
|---|---|---|---|---|
| Paper 1 | Physics, Chemistry, Mathematics | 75 Questions (25 Physics, 25 Chemistry, 25 Mathematics) | 300 total |
|
Is there any negative marking in JEE Main Paper 1?
- If you choose the wrong answer in both Section A and Section B, you will lose 1 mark because of negative marking.
- In Section B, instead of choosing an option, you have to type the correct whole number using the on screen number pad. If your answer is a decimal, round it off to the nearest whole number before entering it.
Once you calculate your expected marks, you can review the JEE Main Question papers to improve your preparation.
You can check:
- JEE Main Previous Years Question Paper with Solution
- JEE Main 2024 Question Paper with Solution PDF
- JEE Main 2023 Question Paper with Solution PDF
- JEE Main 2022 Question Paper with Solution PDF
What to carry at the JEE Main Test Centre ?
Every aspirant must check the JEE Main exam day guidelines before the examination. The JEE Main exam instructions comprise of all the dos and don’ts to be followed for a hassle-free exam process. JEE Mains rules and regulations for the exam are provided below:
| What to Carry | What NOT to Carry |
|---|---|
| JEE Main 2025 Admit Card (Mandatory) | Electronic gadgets (Mobile phones, smartwatches, calculators, Bluetooth devices) |
| Valid ID Proof (Aadhaar Card, PAN Card, Voter ID, Passport, etc.) | Written/Printed Study Materials (Books, Notes, Papers) |
| A simple transparent ballpoint pen (for rough work) | Any type of rough paper (NTA will provide sheets) |
| A passport-size photograph (same as on the admit card) | Smart devices (Earphones, Digital Watches, Fitness Bands) |
| PwD Certificate (if applicable) | Geometry boxes, Log tables, or Any extra stationery items |
| A transparent water bottle (Optional) | Wallets, Purses, or Bags |
How to attempt JEE Main 2025 MCQ’s with Maximum Accuracy?
For accuracy in JEE Main MCQ’s concepts need to be well understood. On average, based on past JEE exams, almost 60 % of the incorrect answers are because of reading the question wrong or marking the wrong answers.
For numerical value questions, if the question requires you to round it off to the nearest integer, accuracy is important. It can cost 4 marks per question which can have a significant impact on your rank.
In revising an answer, many students choose to trade their initial answer for another incorrect alternative. Based on the past year trends, first solutions are accurate most of the time, a figure close to 70%. One should second-guess only on a sound reason for doubt about error.








Comments