JEE Main 2025 22nd Jan Shift 1 Question Paper is now available for download with Solution PDF. NTA conducted the exam successfully on 22nd Jan 2025 from 09:00 AM to 12:00 Noon.
| JEE Main 2025 Jan 22 Shift 1 Question Paper with Solution | Download | Check Solution |
The JEE Main 2025 Question Paper for 22nd Jan Shift 1 was reported as Moderately tough. No question was reported as “out of syllabus”. The role of NCERT was crucial in solving Organic Chemistry questions.
Also Check
- Download JEE Main 2025 PYQ for all shifts
- Download JEE Main Last 10 Year Question Papers for all shifts

Check - JEE Main 2025 Jan 22 Shift 2 Question Paper Solutions Pdf
JEE Main 2025 Jan 22 Shift 1 Questions with Solutions
The number of non-empty equivalence relations on the set \(\{1,2,3\}\) is :
View Solution
Let \( f : \mathbb{R} \to \mathbb{R} \) be a twice differentiable function such that \( f(x + y) = f(x) f(y) \) for all \( x, y \in \mathbb{R} \). If \( f'(0) = 4a \) and \( f \) satisfies \( f''(x) - 3a f'(x) - f(x) = 0 \), where \( a > 0 \), then the area of the region \( R = \{(x, y) | 0 \leq y \leq f(ax), 0 \leq x \leq 2\} \) is :
View Solution
Let the triangle PQR be the image of the triangle with vertices \( (1, 3), (3, 1) \) and \( (2, 4) \) in the line \( x + 2y = 2 \). If the centroid of \( \Delta PQR \) is the point \( (\alpha, \beta) \), then \( 15(\alpha - \beta) \) is equal to :
View Solution
Let \( z_1, z_2, z_3 \) be three complex numbers on the circle \( |z| = 1 \) with \( \arg(z_1) = -\frac{\pi}{4}, \arg(z_2) = 0 \) and \( \arg(z_3) = \frac{\pi}{4} \). If \( |z_1 \overline{z_2} + z_2 \overline{z_3} + z_3 \overline{z_1}|^2 = \alpha + \beta \sqrt{2} \), where \( \alpha, \beta \in \mathbb{Z} \), then the value of \( \alpha^2 + \beta^2 \) is :
View Solution
Using the principal values of the inverse trigonometric functions the sum of the maximum and the minimum values of \(16((sec^{-1}x)^{2}+(cosec^{-1}x)^{2})\) is:
View Solution
A coin is tossed three times. Let X denote the number of times a tail follows a head. If \(\mu\) and \(\sigma^2\) denote the mean and variance of X, then the value of \(64(\mu + \sigma^2)\) is:
View Solution
Let \( a_1, a_2, a_3, \dots \) be a G.P. of increasing positive terms. If \( a_1 a_5 = 28 \) and \( a_2 + a_4 = 29 \), then the value of \( a_6 \) is equal to:
View Solution
Let \( L_1: \frac{x-1}{2} = \frac{y-2}{3} = \frac{z-3}{4} \) and \( L_2: \frac{x-2}{3} = \frac{y-4}{4} = \frac{z-5}{5} \) be two lines. Then which of the following points lies on the line of the shortest distance between \( L_1 \) and \( L_2 \)?
View Solution
The product of all solutions of the equation \( e^{5(\log_e x)^2 + 3} = x^8, x > 0 \), is :
View Solution
If \( \sum_{r=1}^n T_r = \frac{(2n-1)(2n+1)(2n+3)(2n+5)}{64} \), then \( \lim_{n \to \infty} \sum_{r=1}^n \frac{1}{T_r} \) is equal to :
View Solution
From all the English alphabets, five letters are chosen and are arranged in alphabetical order. The total number of ways, in which the middle letter is ‘M’, is :
View Solution
Let \( x = x(y) \) be the solution of the differential equation \( y^2 dx + \left( x - \frac{1}{y} \right) dy = 0 \). If \( x(1) = 1 \), then \( x\left( \frac{1}{2} \right) \) is :
View Solution
Let the parabola \(y = x^2 + px - 3\) meet the coordinate axes at the points P, Q and R. If the circle C with centre at (-1, -1) passes through the points P, Q and R, then the area of \(\triangle PQR\) is:
View Solution
A circle C of radius 2 lies in the second quadrant and touches both the coordinate axes. Let \( r \) be the radius of a circle that has centre at the point \( (2, 5) \) and intersects the circle C at exactly two points. If the set of all possible values of \( r \) is the interval \( (\alpha, \beta) \), then \( 3\beta - 2\alpha \) is equal to:
View Solution
Let for \( f(x) = 7\tan^8 x + 7\tan^6 x - 3\tan^4 x - 3\tan^2 x \), \( I_1 = \int_0^{\frac{\pi}{4}} f(x)dx \) and \( I_2 = \int_0^{\frac{\pi}{4}} x f(x)dx \). Then \( 7I_1 + 12I_2 \) is equal to:
View Solution
Let \( f(x) \) be a real differentiable function such that \( f(0) = 1 \) and \( f(x + y) = f(x)f'(y) + f'(x)f(y) \) for all \( x, y \in \mathbb{R} \). Then \( \sum_{n=1}^{100} \log_e f(n) \) is equal to :
View Solution
Let \( A = \{1, 2, 3, \dots, 10\} \) and \( B = \left\{ \frac{m}{n} : m, n \in A, m < n and \gcd(m, n) = 1 \right\} \).
Then \( n(B) \) is equal to :
View Solution
The area of the region, inside the circle \((x-2\sqrt{3})^2 + y^2 = 12\) and outside the parabola \(y^2 = 2\sqrt{3}x\) is:
View Solution
Two balls are selected at random one by one without replacement from a bag containing 4 white and 6 black balls. If the probability that the first selected ball is black, given that the second selected ball is also black, is \(\frac{m}{n}\), where gcd(m, n) = 1, then m + n is equal to:
View Solution
Let the foci of a hyperbola be \( (1, 14) \) and \( (1, -12) \). If it passes through the point \( (1, 6) \), then the length of its latus-rectum is:
View Solution
Let the function, \( f(x) = \begin{cases} -3ax^2 - 2, & x < 1
a^2 + bx, & x \geq 1 \end{cases} \)
Be differentiable for all \( x \in \mathbb{R} \), where \( a > 1 \), \( b \in \mathbb{R} \).
If the area of the region enclosed by \( y = f(x) \) and the line \( y = -20 \) is \( \alpha + \beta\sqrt{3} \), where \( \alpha, \beta \in \mathbb{Z} \), then the value of \( \alpha + \beta \) is:
View Solution
If \( \sum_{r=0}^5 \frac{{}^{11}C_{2r+1}}{2r+2} = \frac{m}{n} \), gcd(m, n) = 1, then \( m - n \) is equal to:
View Solution
Let \( A \) be a square matrix of order 3 such that \( det(A) = -2 \) and \( det(3 \cdot adj(-6 \cdot adj(3A))) = 2^{m+n} \cdot 3^{mn} \), where \( m > n \). Then \( 4m + 2n \) is equal to:
View Solution
Let \( L_1 : \frac{x - 1}{3} = \frac{y - 1}{-1} = \frac{z + 1}{0} \) and \( L_2 : \frac{x - 2}{2} = \frac{y}{0} = \frac{z + 4}{\alpha} \), where \( \alpha \in \mathbb{R} \), be two lines which intersect at the point \( B \).
If \( P \) is the foot of the perpendicular from the point \( A(1, 1, -1) \) on \( L_2 \), then the value of \( 26 \alpha (PB)^2 \) is:
View Solution
Let \( \vec{c} \) be the projection vector of \( \mathbf{b} = \lambda \hat{i} + 4 \hat{k}, \lambda > 0 \), on the vector \( \vec{a} = \hat{i} + 2 \hat{j} + 2 \hat{k} \). If \( |\vec{a} + \vec{c}| = 7 \), then the area of the parallelogram formed by the vectors \( \vec{b} \) and \( \vec{c} \) is:
View Solution
Given below are two statements:
Statement I : In a vernier callipers, one vernier scale division is always smaller than one main scale division.
Statement II : The vernier constant is given by one main scale division multiplied by the number of vernier scale divisions.
In light of the above statements, choose the correct answer from the options given below.
View Solution
A line charge of length \( \frac{a}{2} \) is kept at the center of an edge BC of a cube ABCDEFGH having edge length \( a \). If the density of the line is \( \lambda C \) per unit length, then the total electric flux through all the faces of the cube will be :
(Take \( \varepsilon_0 \) as the free space permittivity)
View Solution
Sliding contact of a potentiometer is in the middle of the potentiometer wire having resistance \( R_p = 1 \, \Omega \) as shown in the figure. An external resistance of \( R_e = 2 \, \Omega \) is connected via the sliding contact.
The current \( i \) is :
View Solution
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : If Young’s double slit experiment is performed in an optically denser medium than air, then the consecutive fringes come closer.
Reason (R) : The speed of light reduces in an optically denser medium than air while its frequency does not change.
In the light of the above statements, choose the most appropriate answer from the options given below :
View Solution
Two spherical bodies of same materials having radii 0.2 m and 0.8 m are placed in same atmosphere. The temperature of the smaller body is 800 K and temperature of bigger body is 400 K. If the energy radiate from the smaller body is E, the energy radiated from the bigger body is (assume, effect of the surrounding to be negligible):
View Solution
An amount of ice of mass \( 10^{-3} \) kg and temperature \( -10^\circ C \) is transformed to vapor of temperature \( 110^\circ C \) by applying heat. The total amount of work required for this conversion is,
(Take, specific heat of ice = 2100 J kg\(^{-1}\) K\(^{-1}\),
specific heat of water = 4180 J kg\(^{-1}\) K\(^{-1}\),
specific heat of steam = 1920 J kg\(^{-1}\) K\(^{-1}\),
Latent heat of ice = \( 3.35 \times 10^5 \) J kg\(^{-1}\),
Latent heat of steam = \( 2.25 \times 10^6 \) J kg\(^{-1}\))
View Solution
An electron in the ground state of the hydrogen atom has the orbital radius of \( 5.3 \times 10^{-11} \, m \) while that for the electron in the third excited state is \( 8.48 \times 10^{-10} \, m \). The ratio of the de Broglie wavelengths of the electron in the ground state to that in the excited state is:
View Solution
In the diagram given below, there are three lenses formed. Considering negligible thickness of each of them as compared to \( R_1 \) and \( R_2 \), i.e., the radii of curvature for upper and lower surfaces of the glass lens, the power of the combination is:
View Solution
An electron is made to enters symmetrically between two parallel and equally but oppositely charged metal plates, each of 10 cm length. The electron emerges out of the field region with a horizontal component of velocity \(10^6\) m/s. If the magnitude of the electric field between the plates is 9.1 V/cm, then the vertical component of velocity of electron is (mass of electron = \(9.1 \times 10^{-31}\) kg and charge of electron = \(1.6 \times 10^{-19}\) C):
View Solution
Which of the following resistivity (\(\rho\)) vs temperature (T) curves is most suitable to be used in wire-bound standard resistors?
A closed organ and an open organ tube filled by two different gases having the same bulk modulus but different densities \( \rho_1 \) and \( \rho_2 \), respectively. The frequency of the 9th harmonic of the closed tube is identical with the 4th harmonic of the open tube. If the length of the closed tube is 10 cm and the density ratio of the gases is \( \rho_1 : \rho_2 = 1 : 16 \), then the length of the open tube is:
View Solution
A uniform circular disc of radius \( R \) and mass \( M \) is rotating about an axis perpendicular to its plane and passing through its center. A small circular part of radius \( R/2 \) is removed from the original disc as shown in the figure. Find the moment of inertia of the remaining part of the original disc about the axis as given above.
View Solution
A small point of mass \(m\) is placed at a distance \(2R\) from the center \(O\) of a big uniform solid sphere of mass \(M\) and radius \(R\). The gravitational force on \(m\) due to \(M\) is \(F_1\). A spherical part of radius \(R/3\) is removed from the big sphere as shown in the figure, and the gravitational force on \(m\) due to the remaining part of \(M\) is found to be \(F_2\). The value of the ratio \( F_1 : F_2 \) is:
View Solution
The work functions of cesium (Cs) and lithium (Li) metals are 1.9 eV and 2.5 eV, respectively. If we incident a light of wavelength 550 nm on these two metal surfaces, then photo-electric effect is possible for the case of:
View Solution
If \( B \) is magnetic field and \( \mu_0 \) is permeability of free space, then the dimensions of \( \frac{B}{\mu_0} \) is:
View Solution
A bob of mass \(m\) is suspended at a point \(O\) by a light string of length \(l\) and left to perform vertical motion (circular) as shown in the figure. Initially, by applying horizontal velocity \(v_0\) at the point ‘A’, the string becomes slack when the bob reaches at the point ‘D’. The ratio of the kinetic energy of the bob at the points B and C is:
View Solution
Given below are two statements:
Statement-I: The equivalent emf of two nonideal batteries connected in parallel is smaller than either of the two emfs.
Statement-II: The equivalent internal resistance of two nonideal batteries connected in parallel is smaller than the internal resistance of either of the two batteries.
In light of the above statements, choose the correct answer from the options given below.
View Solution
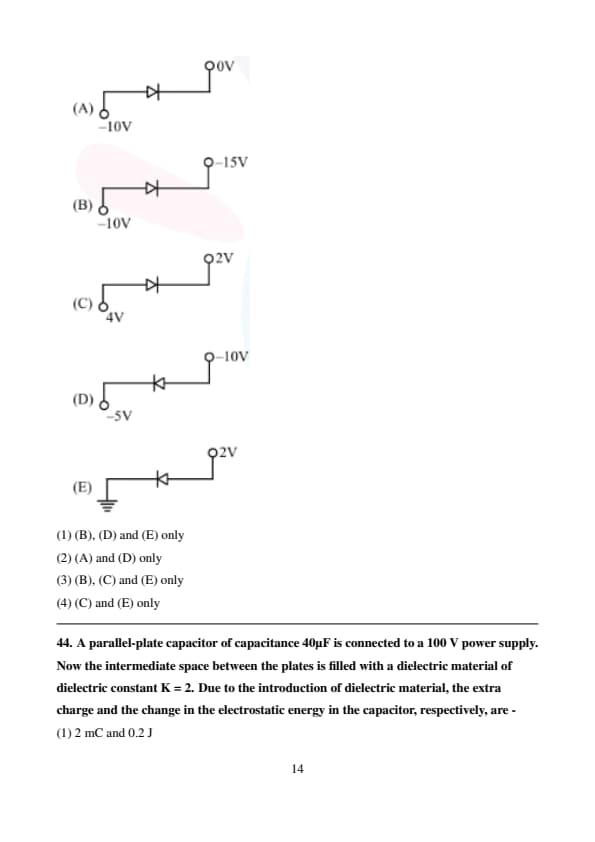
Which of the following circuits represents a forward biased diode?
View Solution
A parallel-plate capacitor of capacitance 40µF is connected to a 100 V power supply. Now the intermediate space between the plates is filled with a dielectric material of dielectric constant K = 2. Due to the introduction of dielectric material, the extra charge and the change in the electrostatic energy in the capacitor, respectively, are -
View Solution
Given is a thin convex lens of glass (refractive index \( \mu \)) and each side having radius of curvature \( R \). One side is polished for complete reflection. At what distance from the lens, an object placed on the optic axis so that the image gets formed on the object itself.
Two soap bubbles of radius 2 cm and 4 cm, respectively, are in contact with each other. The radius of curvature of the common surface, in cm, is ______.
View Solution
The driver sitting inside a parked car is watching vehicles approaching from behind with the help of his side view mirror, which is a convex mirror with radius of curvature \( R = 2 \, m \). Another car approaches him from behind with a uniform speed of 90 km/hr. When the car is at a distance of 24 m from him, the magnitude of the acceleration of the image of the side view mirror is \( a \). The value of \( 100a \) is ______ m/s\(^2\).
View Solution
Three conductors of same length having thermal conductivity \(k_1\), \(k_2\), and \(k_3\) are connected as shown in figure. Area of cross sections of 1st and 2nd conductor are same and for 3rd conductor it is double of the 1st conductor. The temperatures are given in the figure. In steady state condition, the value of θ is ______ °C. (Given: \(k_1\) = 60 Js⁻¹m⁻¹K⁻¹,\(k_2\) = 120 Js⁻¹m⁻¹K⁻¹, \(k_3\) = 135 Js⁻¹m⁻¹K⁻¹)
View Solution
The position vectors of two 1 kg particles, (A) and (B), are given by \[ \vec{r}_A = (\alpha_1 t \hat{i} + \alpha_2 t^2 \hat{j} + \alpha_3 t^3 \hat{k}) \, m \]
and \[ \vec{r}_B = (\beta_1 t \hat{i} + \beta_2 t^2 \hat{j} + \beta_3 t^3 \hat{k}) \, m, respectively; \] \[ (\alpha_1 = 1 \, m/s, \, \alpha_2 = 3 \, m/s^2, \, \alpha_3 = 2 \, m/s^3, \, \beta_1 = 2 \, m/s, \, \beta_2 = -1 \, m/s^2, \, \beta_3 = 4 \, m/s^3), \]
where \( t \) is time, and \( n \) and \( p \) are constants. At \( t = 1 \, s, \, |\vec{V}_A| = |\vec{V}_B| \) and velocities \( \vec{V}_A \) and \( \vec{V}_B \) are orthogonal to each other. At \( t = 1 \, s \), the magnitude of angular momentum of particle (A) with respect to the position of particle (B) is \( \sqrt{L} \, kgm^2s^{-1} \). The value of \( L \) is ______.
View Solution
A particle is projected at an angle of \( 30^\circ \) from horizontal at a speed of 60 m/s. The height traversed by the particle in the first second is \( h_0 \) and height traversed in the last second, before it reaches the maximum height, is \( h_1 \). The ratio \( \frac{h_0}{h_1} \) is ______.
[Take \( g = 10 \, m/s^2 \)]
View Solution
A solution of aluminium chloride is electrolyzed for 30 minutes using a current of 2A. The amount of the aluminium deposited at the cathode is ____ .
[Given: molar mass of aluminium and chlorine are 27 g mol\(^{-1}\) and 35.5 g mol\(^{-1}\) respectively, Faraday constant = 96500 C mol\(^{-1}\)]
View Solution
Which of the following statement is not true for radioactive decay?
View Solution
How many different stereoisomers are possible for the given molecule?
View Solution
Which of the following electronegativity order is incorrect?
View Solution
Lanthanoid ions with \(4f^7\) configuration are:
(A) Eu\(^{2+}\) (B) Gd\(^{3+}\) (C) Eu\(^{3+}\) (D) Tb\(^{3+}\) (E) Sm\(^{2+}\)
Choose the correct answer from the options given below:
View Solution
Match List-I with List-II:
View Solution
Which of the following acids is a vitamin?
View Solution
A liquid when kept inside a thermally insulated closed vessel at \(25^{\circ}C\) was mechanically stirred from outside. What will be the correct option for the following thermodynamic parameters?
View Solution
Radius of the first excited state of Helium ion is given as:
\(a_0\) = radius of first stationary state of hydrogen atom.
View Solution
Given below are two statements:
Statement I: CH\(_3\)-O-CH\(_2\)-Cl will undergo \( S_N1 \) reaction though it is a primary halide.
Statement II: CH_3-C(-CH_3)(-CH_3)-CH_2-Cl
will not undergo \( S_N2 \) reaction very easily though it is a primary halide.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Given below are two statements:
Statement I: One mole of propyne reacts with excess of sodium to liberate half a mole of H₂ gas.
Statement II: Four g of propyne reacts with NaNH₂ to liberate NH₃ gas which occupies 224 mL at STP.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
A vessel at 1000 K contains \( CO_2 \) with a pressure of 0.5 atm. Some of \( CO_2 \) is converted into \( CO \) on addition of graphite. If total pressure at equilibrium is 0.8 atm, then \( K_p \) is:
View Solution
The IUPAC name of the following compound is:
View Solution
Which of the following electrolyte can be used to obtain \( H_2S_2O_8 \) by the process of electrolysis?
View Solution
The compounds which give positive Fehling's test are:
Choose the CORRECT answer from the options given below:
View Solution
In which of the following complexes the CFSE, \(\Delta_0\) will be equal to zero?
View Solution
Arrange the following solutions in order of their increasing boiling points.
(i) \(10^{-4}\) M NaCl
(ii) \(10^{-4}\) M Urea
(iii) \(10^{-3}\) M NaCl
(iv) \(10^{-2}\) M NaCl
View Solution
The products formed in the following reaction sequence are:
From the magnetic behaviour of [NiCl4]²⁻ (paramagnetic) and [Ni(CO)4] (diamagnetic), choose the correct geometry and oxidation state.
View Solution
The incorrect statements regarding geometrical isomerism are:
(A) Propene shows geometrical isomerism.
(B) Trans isomer has identical atoms/groups on the opposite sides of the double bond.
(C) Cis-but-2-ene has higher dipole moment than trans-but-2-ene.
(D) 2-methylbut-2-ene shows two geometrical isomers.
(E) Trans-isomer has lower melting point than cis isomer.
View Solution
Some CO\(_2\) gas was kept in a sealed container at a pressure of 1 atm and at 273 K. This entire amount of CO\(_2\) gas was later passed through an aqueous solution of Ca(OH)\(_2\). The excess unreacted Ca(OH)\(_2\) was later neutralized with 0.1 M of 40 mL HCl. If the volume of the sealed container of CO\(_2\) was \(x\), then \(x\) is ______ cm\(^3\) (nearest integer).
[Given: The entire amount of CO\(_2\) reacted with exactly half the initial amount of Ca(OH)\(_2\) present in the aqueous solution.]
View Solution
In Carius method for estimation of halogens, 180 mg of an organic compound produced 143.5 mg of AgCl. The percentage composition of chlorine in the compound is _______ %.
[Given: Molar mass in g mol\(^{-1}\) of Ag = 108, Cl = 35.5]
View Solution
The number of molecules/ions that show linear geometry among the following is ______.
SO₂, BeCl₂, CO₂, N₃⁻, NO₂, F₂O, XeF₂, NO₂⁺, I₃⁻, O₃
View Solution
The molecule A changes into its isomeric form B by following a first order kinetics at a temperature of 1000 K. If the energy barrier with respect to reactant energy for such isomeric transformation is 191.48 kJ mol\(^{-1}\) and the frequency factor is \(10^{20}\), the time required for 50% molecules of A to become B is ______ picoseconds (nearest integer). [R = 8.314 J K\(^{-1}\) mol\(^{-1}\)]
View Solution
Consider the following sequence of reactions :
Molar mass of the product formed (A) is ______ g mol\(^{-1}\).
View Solution






















Also Check: Good Score in JEE Main 2025
JEE Main 22nd Jan Shift 1 Question Paper Analysis- Check Difficulty Level and Good Score
The question paper consisted of a total of 75 questions, divided equally across the three subjects:
- The Mathematics section in JEE Main 2025 22nd Jan Shift 1 Question Paper was reported as Tough and Lengthy with 9 questions coming majorly from topics like Integration, Co-ordinate Geometry, etc.
- Physics was reported as Moderate with 8 questions coming majorly from topics like Modern Physics, Thermodynamics, etc.
- Chemistry was reported as Easy to Moderate with 7 questions coming majorly from topics like Organic Chemistry, Physical Chemistry, etc.
Each section included a mix of multiple-choice questions (MCQs) and numerical value-based questions, offering a balanced challenge for students.
Check: JEE Main 22nd Jan Shift 2 Question Paper
JEE Main 2025:Shift-Wise Analysis
| Session | Shift | Difficulty Trend (Physics, Chemistry, Mathematics) |
Overall Difficulty |
|---|---|---|---|
| January | 22nd January Shift 1 |
|
Moderate |
| January | 22nd January Shift 2 |
|
Moderate |
| January | 23rd January Shift 1 |
|
Moderate |
| January | 23rd January Shift 2 |
|
Moderate |
| January | 24th January Shift 1 |
|
Moderate |
| January | 24th January Shift 2 |
|
Moderate |
| January | 28th January Shift 1 |
|
Tough |
| January | 28th January Shift 2 |
|
Tough |
| January | 29th January Shift 1 |
|
Moderate to Tough |
| January | 29th January Shift 2 |
|
Moderate to Tough |



.png?h=35&w=35&mode=stretch)





Comments