JEE Main 2026 Jan 21 Shift 1 official NTA question paper is available here with answer key and solutions. NTA conducted the first shift of the day on Jan 21, 2026, from 09:00 AM to 12:00 PM.
JEE Main 2026 21st Jan Shift 1 Question Paper with Solution PDF
Also Check:
- JEE Main 2026 April 2 Shift 1 Question Paper with Solutions
- Download JEE Main 2026 Session 2 Question Paper for all Shifts
- Predict your rank using Collegedunia’s JEE Main Rank Predictor for free
Based on previous year analysis, JEE Main Jan 21 Shift 1 was Moderate to Difficult, where Mathematics remains the lengthiest section. Students can access the JEE Main Jan 21 Shift 1 official questions with answer keys here. Students can check the detailed JEE Main Jan 21st Shift 1 Paper Analysis here
If the coefficient of x in the expansion of \((ax^2+bx+c)(1-2x)^{26}\) is \(-56\) and the coefficients of \(x^2\) and \(x^3\) are both zero, then \(a+b+c\) is equal to:
If \(x^2+x+1=0\), then the value of \(\left(x + \frac{1}{x}\right)^2 + \left(x^2 + \frac{1}{x^2}\right)^2 + \left(x^3 + \frac{1}{x^3}\right)^2 + \dots + \left(x^{25} + \frac{1}{x^{25}}\right)^2\) is:
Let \(f: R \to (0, \infty)\) be a twice differentiable function such that \(f(3) = 18\), \(f'(3)=0\) and \(f''(3) = 4\). Then \(\lim_{x \to 1} \log_e \left[ \frac{f(2+x)}{f(3)} \right]^{\frac{18}{(x-1)^2}}\) is equal to:
Let O be the vertex of the parabola \(x^2=4y\) and Q be any point on it. Let the locus of the point P, which divides the line segment OQ internally in the ratio 2:3 be the conic C. Then the equation of the chord of C, which is bisected at the point (1, 2), is:
The value of \(\int_{-\pi/6}^{\pi/6} \frac{\pi+4x^{11}}{1-\sin(|x|+\pi/6)} dx\) is equal to:
The number of relations, defined on the set {a, b, c, d}, which are both reflexive and symmetric, is equal to:
Let \(a_1, a_2, a_3, \dots\) be a G.P. of increasing positive terms such that \(a_2 \cdot a_3 \cdot a_4=64\) and \(a_1 + a_3 + a_5 = \frac{813}{7}\). Then \(a_3 + a_5 + a_7\) is equal to:
The number of strictly increasing functions \(f\) from the set \{1, 2, 3, 4, 5, 6\ to the set \{1, 2, 3, ...., 9\ such that \(f(i) > i\) for \(1 \le i \le 6\), is equal to:
Let \(\vec{a} = -\hat{i} + 2\hat{j} + 2\hat{k}\), \(\vec{b} = 8\hat{i} + 7\hat{j} - 3\hat{k}\) and \(\vec{c}\) be a vector such that \(\vec{a} \times \vec{c} = \vec{b}\). If \(\vec{c} \cdot (\hat{i}+\hat{j}+\hat{k}) = 4\), then \(|\vec{a}+\vec{c}|^2\) is equal to:
Let PQ and MN be two straight lines touching the circle \(x^2+y^2-4x-6y-3=0\) at the points A and B respectively. Let O be the centre of the circle and \(\angle AOB = \pi/3\). Then the locus of the point of intersection of the lines PQ and MN is:
The area of the region, inside the ellipse \(x^2+4y^2=4\) and outside the region bounded by the curves \(y=x-1\) and \(y=1-x\), is:
Let the foci of a hyperbola coincide with the foci of the ellipse \(\frac{x^2}{36} + \frac{y^2}{16} = 1\). If the eccentricity of the hyperbola is 5, then the length of its latus rectum is:
The sum of all the roots of the equation \((x-1)^2 - 5|x-1| + 6 = 0\), is:
Let \((\alpha, \beta, \gamma)\) be the co-ordinates of the foot of the perpendicular drawn from the point (5, 4, 2) on the line \(\vec{r}=(-\hat{i}+3\hat{j}+\hat{k})+\lambda(2\hat{i}+3\hat{j}-\hat{k})\). Then the length of the projection of the vector \(\alpha\hat{i}+\beta\hat{j}+\gamma\hat{k}\) on the vector \(6\hat{i}+2\hat{j}+3\hat{k}\) is:
Let a point A lie between the parallel lines \(L_1\) and \(L_2\) such that its distances from \(L_1\) and \(L_2\) are 6 and 3 units, respectively. Then the area (in sq. units) of the equilateral triangle ABC, where the points B and C lie on the lines \(L_1\) and \(L_2\), respectively, is:
The value of \(cosec10^\circ - \sqrt{3} \sec10^\circ\) is equal to:
If the domain of the function \(f(x) = \cos^{-1}\left(\frac{2x-5}{11-3x}\right) + \sin^{-1}(2x^2-3x+1)\) is the interval \([\alpha, \beta]\), then \(\alpha + 2\beta\) is equal to:
Let \(y=y(x)\) be the solution curve of the differential equation \((1+x^2)dy+(y-\tan^{-1}x) dx=0\), \(y(0) = 1\). Then the value of \(y(1)\) is:
Let \(\vec{c}\) and \(\vec{d}\) be vectors such that \(|\vec{c}+\vec{d}| = \sqrt{29}\) and \(\vec{c} \times (2\hat{i}+3\hat{j}+4\hat{k}) = (2\hat{i}+3\hat{j}+4\hat{k}) \times \vec{d}\). If \(\lambda_1, \lambda_2 (\lambda_1 > \lambda_2)\) are the possible values of \((\vec{c}+\vec{d}) \cdot (-7\hat{i}+2\hat{j}+3\hat{k})\), then the equation \(K^2x^2+(K^2-5K+\lambda_1)xy+(3K+\lambda_2^2)y^2-8x+12y+\lambda_2 = 0\) represents a circle, for K equal to:
Let the mean and variance of 7 observations 2, 4, 10, x, 12, 14, y, where x > y, be 8 and 16 respectively. Two numbers are chosen from {1, 2, 3, x-4, y, 5} one after another without replacement, then the probability, that the smaller number among the two chosen numbers is less than 4, is:
Let \(f: R \to R\) be a twice differentiable function such that the quadratic equation \(f(x)m^2-2f'(x) m+f''(x) = 0\) in \(m\), has two equal roots for every \(x \in R\). If \(f(0)=1, f'(0) = 2\), and \((\alpha, \beta)\) is the largest interval in which the function \(g(x) = f(\log_e x - x)\) is increasing, then \(\alpha+\beta\) is equal to:
Let S={(m, n): m, n \(\in\) \{1, 2, 3, ....., 50\\. If the number of elements (m, n) in S such that \(6^m+9^n\) is a multiple of 5 is p and the number of elements (m, n) in S such that m + n is a square of a prime number is q, then p + q is equal to:
For some \(\alpha, \beta \in R\), let \(A = \begin{pmatrix} \alpha & 2
1 & 2 \end{pmatrix}\) and \(B = \begin{pmatrix} 1 & 1
\beta & 1 \end{pmatrix}\) be such that \(A^2-4A+2I-B^2-3B+I=O\). Then \((\det(adj(A^3-B^3)))^2\) is equal to:
Let \(a_1=1\) and for \(n \ge 1\), \(a_{n+1} = \frac{1}{2}a_n + \frac{n^2-2n-1}{2n^2(n+1)^2}\). Then \(\sum_{n=1}^\infty (a_n - \frac{2}{n^2})\) is equal to:
\(6 \int_{0}^{\pi} (\sin 3x + \sin 2x + \sin x)dx\) is equal to:
A point charge of \(10^{-8}\) C is placed at origin. The work done in moving a point charge 2 \(\mu\)C from point A(4, 4, 2) m to B(2, 2, 1) m is ________ J. (\(\frac{1}{4\pi\epsilon_0} = 9 \times 10^9\) in SI units)
An aluminium and steel rods having same lengths and cross-sections are joined to make total length of 120 cm at 30\(^\circ\)C. The coefficient of linear expansion of aluminium and steel are \(24 \times 10^{-6}\)/\(^\circ\)C and \(1.2 \times 10^{-5}\)/\(^\circ\)C, respectively. The length of this composite rod when its temperature is raised to 100\(^\circ\)C, is _________ cm.
A light wave described by E = 60[\(\sin(3 \times 10^{15})t + \sin(12 \times 10^{15})t\)] (in SI units) falls on a metal surface of work function 2.8 eV. The maximum kinetic energy of ejected photoelectron is (approximately) _________ eV. (h=\(6.6 \times 10^{-34}\) J.s. and e=\(1.6 \times 10^{-19}\) C)
A parallel plate capacitor has capacitance C, when there is vacuum within the parallel plates. A sheet having thickness \( \frac{1}{3}d \) of the separation between the plates and relative permittivity K is introduced between the plates. The new capacitance of the system is:
In an experiment the values of two spring constants were measured as \(k_1 = (10 \pm 0.2)\) N/m and \(k_2 = (20 \pm 0.3)\) N/m. If these springs are connected in parallel, then the percentage error in equivalent spring constant is:
Potential energy (V) versus distance (x) is given by the graph. Rank various regions as per the magnitudes of the force (F) acting on a particle from high to low.

Consider a modified Bernoulli equation. \[ P + \frac{A}{Bt^2} + \rho g(h + Bt) + \frac{1}{2} \rho v^2 = constant \]
If t has the dimension of time then the dimensions of A and B are __________ respectively.
The electric field in a plane electromagnetic wave is given by : \( E_y = 69 \sin[0.6 \times 10^3 x - 1.8 \times 10^{11} t] \) V/m. The expression for magnetic field associated with this electromagnetic wave is __________ T.
A uniform rod of mass m and length l suspended by means of two identical inextensible light strings as shown in figure. Tension in one string immediately after the other string is cut, is __________ (g = acceleration due to gravity).

A gas based geyser heats water flowing at the rate of 5.0 litres per minute from 27 °C to 87 °C. The rate of consumption of the gas is __________ g/s. (Take heat of combustion of gas = \(5.0 \times 10^4\) J/g, specific heat capacity of water = 4200 J/kg.°C).
A current carrying solenoid is placed vertically and a particle of mass m with charge Q is released from rest. The particle moves along the axis of solenoid. If g is acceleration due to gravity then the acceleration (a) of the charged particle will satisfy:
Water flows through a horizontal tube as shown in the figure. The difference in height between the water columns in vertical tubes is 5 cm and the area of cross-sections at A and B are 6 cm\(^2\) and 3 cm\(^2\) respectively. The rate of flow will be __________ cm\(^3\)/s. (take g = 10 m/s\(^2\)).

A 4 kg mass moves under the influence of a force \(\vec{F} = (4t^3\hat{i} - 3t^2\hat{j})\) N where t is the time in second. If mass starts from origin at t=0, the velocity and position after t = 2 s will be:
A conducting circular loop of area 1.0 m\(^2\) is placed perpendicular to a magnetic field which varies as B = sin(100t) Tesla. If the resistance of the loop is 100 \(\Omega\), then the average thermal energy dissipated in the loop in one period is __________ J.
Two strings (A, B) having linear densities \(\mu_A = 2 \times 10^{-4}\) kg/m and, \(\mu_B = 4 \times 10^{-4}\) kg/m and lengths \(L_A = 2.5\) m and \(L_B = 1.5\) m respectively are joined. Free ends of A and B are tied to two rigid supports C and D, respectively creating a tension of 500 N in the wire. Two identical pulses, sent from C and D ends, take time \(t_A\) and \(t_B\), respectively, to reach the joint. The ratio \(t_A/t_B\) is:
Initially a satellite of 100 kg is in a circular orbit of radius 1.5\(R_E\). This satellite can be moved to a circular orbit of radius 3\(R_E\) by supplying \(a \times 10^6\) J of energy. The value of a is __________ . (Take Radius of Earth \(R_E = 6 \times 10^6\) m and g = 10 m/s\(^2\)).
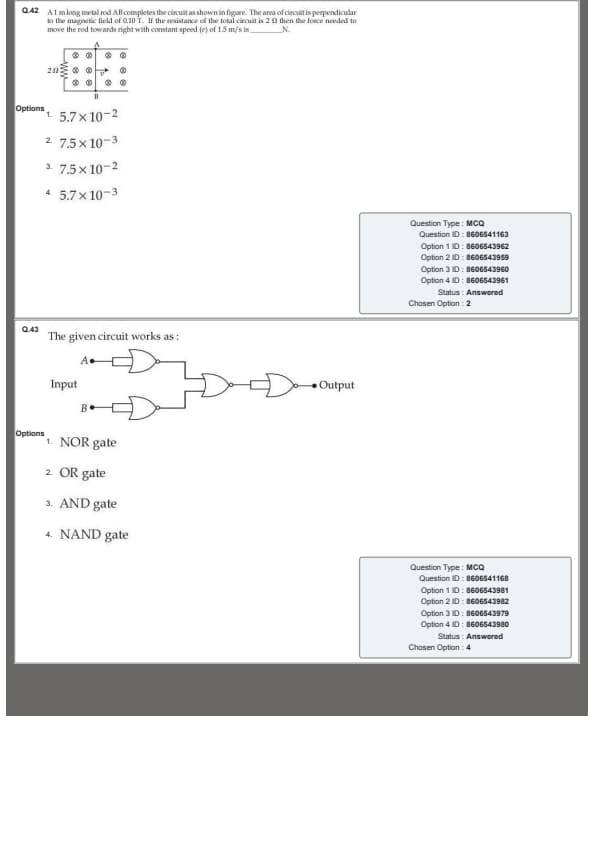
A 1 m long metal rod AB completes the circuit as shown in figure. The area of circuit is perpendicular to the magnetic field of 0.10 T. If the resistance of the total circuit is 2 \(\Omega\) then the force needed to move the rod towards right with constant speed (v) of 1.5 m/s is __________ N.

The given circuit works as:

In a double slit experiment the distance between the slits is 0.1 cm and the screen is placed at 50 cm from the slits plane. When one slit is covered with a transparent sheet having thickness t and refractive index n(=1.5), the central fringe shifts by 0.2 cm. The value of t is __________ cm.
If an alpha particle with energy 7.7 MeV is bombarded on a thin gold foil, the closest distance from nucleus it can reach is ______ m.
(Atomic number of gold = 79 and \(\frac{1}{4\pi\epsilon_0}=9\times10^{9}\) SI units)
Two identical thin rods of mass M kg and length L m are connected as shown in figure. Moment of inertia of the combined rod system about an axis passing through point P and perpendicular to the plane of the rods is \(\frac{x}{12} ML^2\) kg m\(^2\). The value of x is ______.

The heat generated in 1 minute between points A and B in the given circuit, when a battery of 9 V with internal resistance of 1 \(\Omega\) is connected across these points is ______ J.

In a microscope, the objective has a focal length \(f_o=2\) cm and the eye-piece has a focal length \(f_e=4\) cm. The tube length is 32 cm. The magnification produced by this microscope for normal adjustment is ______.
A collimated beam of light of diameter 2 mm is propagating along the x-axis. The beam is required to be expanded into a collimated beam of diameter 14 mm using a system of two convex lenses. If the first lens has focal length 40 mm, then the focal length of the second lens is ______ mm.
10 mole of oxygen is heated at constant volume from 30 \(^{\circ}\)C to 40 \(^{\circ}\)C. The change in the internal energy of the gas is ______ cal.
(The molecular specific heat of oxygen at constant pressure, \(C_p=7\) cal/mol \(^{\circ}\)C and \(R=2\) cal/mol \(^{\circ}\)C.)
Consider the following reactions.
\(PbCl_2 + K_2CrO_4 \rightarrow A + 2KCl\) (Hot solution)
\(A + NaOH \rightarrow B + Na_2CrO_4\)
\(PbSO_4 + 4CH_3COONH_4 \rightarrow (NH_4)_2SO_4 + X\)
In the above reactions, A, B and X are respectively:
From the following, the least stable structure is:
(Assume standard resonance structures)
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual gases after cooling to 273 K occupy 224 mL. When the system is treated with KOH solution, the volume decreases to 64 mL. The formula of the hydrocarbon is:
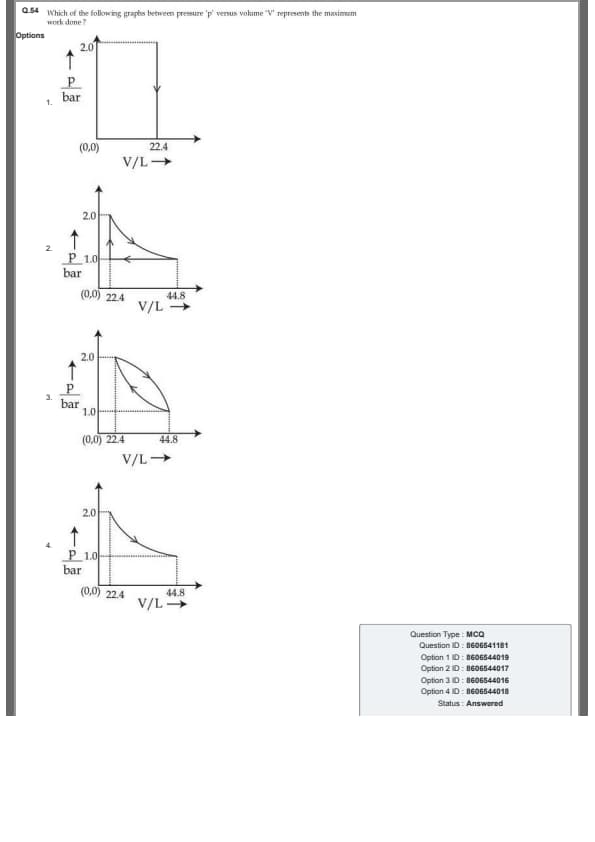
Which of the following graphs between pressure 'p' versus volume 'V' represents the maximum work done?
Which of the following represents the correct trend for the mentioned property?
A. F \(>\) P \(>\) S \(>\) B \quad -- First Ionization Energy
B. Cl \(>\) F \(>\) S \(>\) P \quad -- Electron Affinity
C. K \(>\) Al \(>\) Mg \(>\) B \quad -- Metallic character
D. K₂O \(>\) Na₂O \(>\) MgO \(>\) Al₂O₃ \quad -- Basic character
Choose the correct answer from the options given below:
MnO₄²⁻, in acidic medium, disproportionates to:
In Carius method, 0.75 g of an organic compound gave 1.2 g of barium sulphate, find percentage of sulphur (molar mass 32 g mol⁻¹). Molar mass of barium sulphate is 233 g mol⁻¹.
Identify correct statements from the following:
A. Propanal and propanone are functional isomers.
B. Ethoxyethane and methoxypropane are metamers.
C. But-2-ene shows optical isomerism.
D. But-1-ene and but-2-ene are functional isomers.
E. Pentane and 2, 2-dimethyl propane are chain isomers.
Choose the correct answer from the options given below:
Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and PQ₂. When 1 g of PQ is dissolved in 50 g of solvent 'A', ΔT_b was 1.176 K while when 1 g of PQ₂ is dissolved in 50 g of solvent 'A', ΔT_b was 0.689 K. (K_b of 'A' = 5 K kg mol⁻¹). The molar masses of elements P and Q (in g mol⁻¹) respectively, are:
A hydrocarbon 'P' (C₄H₈) on reaction with HCl gives an optically active compound 'Q' (C₄H₉Cl) which on reaction with one mole of ammonia gives compound 'R' (C₄H₁₁N). 'R' on diazotization followed by hydrolysis gives 'S'. Identify P, Q, R and S.
An organic compound (P) on treatment with aqueous ammonia under hot condition forms compound (Q) which on heating with Br₂ and KOH forms compound (R) having molecular formula C₆H₇N. Names of P, Q and R respectively are.
Given below are two statements:
Statement I: When an electric discharge is passed through gaseous hydrogen, the hydrogen molecules dissociate and the energetically excited hydrogen atoms produce electromagnetic radiation of discrete frequencies.
Statement II: The frequency of second line of Balmer series obtained from He⁺ is equal to that of first line of Lyman series obtained from hydrogen atom.
In the light of the above statements, choose the correct answer from the options given below:
Given below are two statements:
Statement I: Among [Cu(NH₃)₄]²⁺, [Ni(en)₃]²⁺, [Ni(NH₃)₆]²⁺ and [Mn(H₂O)₆]²⁺, [Mn(H₂O)₆]²⁺ has the maximum number of unpaired electrons.
Statement II: The number of pairs among {[NiCl₄]²⁻, [Ni(CO)₄]}, {[NiCl₄]²⁻, [Ni(CN)₄]²⁻} and {[Ni(CO)₄], [Ni(CN)₄]²⁻} that contain only diamagnetic species is two.
In the light of the above statements, choose the correct answer from the options given below:
Given below are two statements:
Statement I: The number of pairs among [SiO₂, CO₂], [SnO, SnO₂], [PbO, PbO₂] and [GeO, GeO₂], which contain oxides that are both amphoteric is 2.
Statement II: BF₃ is an electron deficient molecule, can act as a Lewis acid, forms adduct with NH₃ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below:
14.0 g of calcium metal is allowed to react with excess HCl at 1.0 atm pressure and 273 K. Which of the following statements is incorrect?
\([Given: Molar mass of Ca = 40, Cl = 35.5, H = 1 g mol^{-1}] \)
For the reaction, \(N_{2}O_{4} \rightleftharpoons 2NO_{2}\) graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is 5.40 kJ \(mol^{-1}\).
B. As \(\Delta G\) in graph is positive, \(N_{2}O_{4}\) will not dissociate into \(NO_{2}\) at all.
C. Reverse reaction will go to completion.
D. When 1 mole of \(N_{2}O_{4}\) changes into equilibrium mixture, value of \(\Delta G = -0.84 kJ mol^{-1}\).
E. When 2 mole of \(NO_{2}\) changes into equilibrium mixture, \(\Delta G\) for equilibrium mixture is \(-6.24 kJ mol^{-1}\).

Choose the correct answer from the following.
Identify the correct statements. (Note: Based on options, the question asks to identify INCORRECT statements).
A. Arginine and Tryptophan are essential amino acids.
B. Glycine does not have chiral centre.
C. Proline is a six membered cyclic ring amino acid.
D. Histidine does not contain heterocyclic ring in its structure.
E. Cysteine has characteristic feature of side chain as \(MeS-CH_2-CH_2-\).
An organic compound "P" of molecular formula \(C_7H_{12}O\) (likely \(C_7H_{12}O_2\) or similar based on options), gives positive Iodoform test but negative Tollen's test. When "P" is treated with dilute acid, it produces "Q". "Q" gives positive Tollen's test and also Iodoform test. The structure of "P" is:
Given below are two statements:
Statement I: The number of species among
\(SF_4, NH_4^+, Ni(CO)_4, XeF_4, [PtCl_{4}]^{2-}, SeF_4, [Ni(CN)_4]^{2-}\) that have tetrahedral geometry is 3.
Statement II: In the set \(NO, BeH_2, BF_3, AlCl_3\), all the molecules have incomplete octet around central atom.
In the light of the above statements, choose the correct answer from the options given below:
Pre-exponential factors of two different reactions of same order are identical. Let activation energy of first reaction exceed the activation energy of second reaction by 20 kJ mol\(^{-1}\). If \(k_1\) and \(k_2\) are the rate constants of first and second reaction respectively at 300 K, then \(\ln \frac{k_{2}}{k_{1}}\) will be ____. (nearest integer) [\(R=8.3 J K^{-1} mol^{-1}\)]

One mole each of \(A_2(g)\) and \(B_2(g)\) are taken in a 1 L closed flask and allowed to establish the equilibrium at 500 K: \(A_{2}(g)+B_{2}(g) \rightleftharpoons 2AB(g)\).
The value of x (missing enthalpy of \(B_2\) or related parameter) is ___. (Nearest integer)
Consider the reactions:
1. \(NaCl+K_{2}Cr_{2}O_{7}+H_{2}SO_{4} \rightarrow A + ...\)
2. \(A + NaOH \rightarrow B + ...\)
3. \(B + H_{2}SO_{4}+H_{2}O_{2} \rightarrow C + ...\)
In the product 'C', X is the number of \(O_{2}^{2-}\) units, Y is the total number of oxygen atoms, and Z is the oxidation state of Cr. The value of \(X+Y+Z\) is ___.
The pH and conductance of a weak acid (HX) was found to be 5 and \(4\times10^{-5}\) S, respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of 1 cm\(^2\) were at a distance of 15 cm apart. The value of the limiting molar conductivity (\(\Lambda_m^\circ\)) is ____ S cm\(^2\) mol\(^{-1}\) (nearest integer) (Given: degree of dissociation \(\alpha \ll 1\))

























































Comments