JEE Main 2026 Jan 21 Shift 2 question paper is available here with answer key and solutions. NTA conducted the JEE Main shift 2 from 3 PM to 6 PM.
Also Check:
- JEE Main 2026 April 2 Shift 1 Question Paper with Solutions
- Download JEE Main 2026 Session 2 Question Paper for all Shifts
JEE Main 2026 21st Jan Shift 2 Question Paper with Solution PDF
| JEE Main 2026 21st Jan Shift 2 Question Paper | Download PDF | View Solution |
Check JEE Main Jan 21st Shift 2 Answer Key
Based on initial analysis, JEE Main Jan 21 Shift 2 was Easy to Moderate, where Mathematics remained the lengthiest section. Students can access the JEE Main today paper shift 2 pdf with answer keys here.
Students can check the detailed paper analysis for JEE Main Jan 21st Shift 2 here.

Let \(A = \{2, 3, 5, 7, 9\}\). Let \(R\) be the relation on \(A\) defined by \(xRy\) if and only if \(2x \le 3y\). Let \(l\) be the number of elements in \(R\), and \(m\) be the minimum number of elements required to be added in \(R\) to make it a symmetric relation. Then \(l + m\) is equal to :
Let \(z\) be the complex number satisfying \(|z - 5| \le 3\) and having maximum positive principal argument. Then \(34 \left| \frac{5z - 12}{5iz + 16} \right|^2\) is equal to :
Let \(\alpha\) and \(\beta\) be the roots of the equation \(x^2 + 2ax + (3a + 10) = 0\) such that \(\alpha < 1 < \beta\). Then the set of all possible values of \(a\) is :
If the line \(ax + 4y = \sqrt{7}\), where \(a \in \mathbb{R}\), touches the ellipse \(3x^2 + 4y^2 = 1\) at the point \(P\) in the first quadrant, then one of the focal distances of \(P\) is :
Let \(A = \{x : |x^2 - 10| \le 6\}\) and \(B = \{x : |x - 2| > 1\}\). Then
Let \(f(x) = x^3 + x^2 f'(1) + 2x f''(2) + f'''(3), x \in \mathbb{R}\). Then the value of \(f'(5)\) is :
Let the line \(L_1\) be parallel to the vector \(-3\hat{i} + 2\hat{j} + 4\hat{k}\) and pass through the point \((2, 6, 7)\), and the line \(L_2\) be parallel to the vector \(2\hat{i} + \hat{j} + 3\hat{k}\) and pass through the point \((4, 3, 5)\). If the line \(L_3\) is parallel to the vector \(-3\hat{i} + 5\hat{j} + 16\hat{k}\) and intersects the lines \(L_1\) and \(L_2\) at the points \(C\) and \(D\), respectively, then \(|\vec{CD}|^2\) is equal to :
Let \(y = y(x)\) be the solution of the differential equation \(\sec x \frac{dy}{dx} - 2y = 2 + 3\sin x, x \in (-\frac{\pi}{2}, \frac{\pi}{2})\). If \(y(0) = -\frac{7}{4}\), then \(y(\frac{\pi}{6})\) is equal to :
If the area of the region \(\{(x, y) : 1 - 2x \le y \le 4 - x^2, x \ge 0, y \ge 0\}\) is \(\frac{\alpha}{\beta}\), \(\alpha, \beta \in \mathbb{N}\), \(\gcd(\alpha, \beta) = 1\), then the value of \((\alpha + \beta)\) is :
Let \(a_1, \frac{a_2}{2}, \frac{a_3}{2^2}, \dots, \frac{a_{10}}{2^9}\) be a G.P. of common ratio \(\frac{1}{\sqrt{2}}\). If \(a_1 + a_2 + \dots + a_{10} = 62\), then \(a_1\) is equal to :
Let \( f: \mathbb{R} \to \mathbb{R} \) be a twice differentiable function such that \( f''(x) > 0 \) for all \( x \in \mathbb{R} \) and \( f'(a-1) = 0 \), where \( a \) is a real number. Let \( g(x) = f(\tan^2 x - 2\tan x + a) \), \( 0 < x < \frac{\pi}{2} \).
Consider the following two statements :
(I) \( g \) is increasing in \( (0, \frac{\pi}{4}) \)
(II) \( g \) is decreasing in \( (\frac{\pi}{4}, \frac{\pi}{2}) \)
Then,
For the matrices \( A = \begin{bmatrix} 3 & -4
1 & -1 \end{bmatrix} \) and \( B = \begin{bmatrix} -29 & 49
-13 & 18 \end{bmatrix} \), if \( (A^{15} + B) \begin{bmatrix} x
y \end{bmatrix} = \begin{bmatrix} 0
0 \end{bmatrix} \), then among the following which one is true?
Let one end of a focal chord of the parabola \( y^2 = 16x \) be \( (16, 16) \). If \( P(\alpha, \beta) \) divides this focal chord internally in the ratio \( 5 : 2 \), then the minimum value of \( \alpha + \beta \) is equal to :
Let \( y^2 = 12x \) be the parabola with its vertex at \( O \). Let \( P \) be a point on the parabola and \( A \) be a point on the \( x \)-axis such that \( \angle OPA = 90^\circ \). Then the locus of the centroid of such triangles \( OPA \) is :
The positive integer \( n \), for which the solutions of the equation \( x(x+2) + (x+2)(x+4) + \dots + (x+2n-2)(x+2n) = \frac{8n}{3} \) are two consecutive even integers, is :
A random variable \( X \) takes values \( 0, 1, 2, 3 \) with probabilities \( \frac{2a+1}{30}, \frac{8a-1}{30}, \frac{4a+1}{30}, b \) respectively, where \( a, b \in \mathbb{R} \). Let \( \mu \) and \( \sigma \) respectively be the mean and standard deviation of \( X \) such that \( \sigma^2 + \mu^2 = 2 \). Then \( \frac{a}{b} \) is equal to :
Let the line \( L \) pass through the point \( (-3, 5, 2) \) and make equal angles with the positive coordinate axes. If the distance of \( L \) from the point \( (-2, r, 1) \) is \( \sqrt{\frac{14}{3}} \), then the sum of all possible values of \( r \) is :
The largest \( n \in \mathbb{N} \), for which \( 7^n \) divides \( 101! \), is :
For a triangle \( ABC \), let \( \vec{p} = \vec{BC} \), \( \vec{q} = \vec{CA} \) and \( \vec{r} = \vec{BA} \). If \( |\vec{p}| = 2\sqrt{3}, |\vec{q}| = 2 \) and \( \cos \theta = -\frac{1}{\sqrt{3}} \), where \( \theta \) is the angle between \( \vec{p} \) and \( \vec{q} \), then \( |\vec{p} \times (\vec{q} - 3\vec{r})|^2 + 3|\vec{r}|^2 \) is equal to :
If the system of equations
\( 3x + y + 4z = 3 \)
\( 2x + \alpha y - z = -3 \)
\( x + 2y + z = 4 \)
has no solution, then the value of \( \alpha \) is equal to :
Let the maximum value of \( (\sin^{-1}x)^2 + (\cos^{-1}x)^2 \) for \( x \in \left[ -\frac{\sqrt{3}}{2}, \frac{1}{\sqrt{2}} \right] \) be \( \frac{m}{n}\pi^2 \), where \( \gcd(m, n) = 1 \). Then \( m + n \) is equal to ___________.
If \( \left( \frac{1}{{}^{15}C_0} + \frac{1}{{}^{15}C_1} \right) \left( \frac{1}{{}^{15}C_1} + \frac{1}{{}^{15}C_2} \right) \dots \left( \frac{1}{{}^{15}C_{12}} + \frac{1}{{}^{15}C_{13}} \right) = \frac{\alpha^{13}}{{}^{14}C_0 {}^{14}C_1 \dots {}^{14}C_{12}} \), then \( 30\alpha \) is equal to ___________.
Let \( [ \cdot ] \) denote the greatest integer function and \( f(x) = \lim_{n \to \infty} \frac{1}{n^3} \sum_{k=1}^{n} \left[ \frac{k^2}{3^x} \right] \). Then \( 12 \sum_{j=1}^{\infty} f(j) \) is equal to ___________.
If \( P \) is a point on the circle \( x^2 + y^2 = 4 \), \( Q \) is a point on the straight line \( 5x + y + 2 = 0 \) and \( x - y + 1 = 0 \) is the perpendicular bisector of \( PQ \), then 13 times the sum of abscissa of all such points \( P \) is ___________.
If \( \int_0^1 4 \cot^{-1}(1 - 2x + 4x^2) dx = a \tan^{-1}(2) - b \ln(5) \), where \( a, b \in \mathbb{N} \), then \( (2a + b) \) is equal to ___________.
Consider two identical metallic spheres of radius \(R\) each having charge \(Q\) and mass \(m\). Their centers have an initial separation of \(4R\). Both the spheres are given an initial speed of \(u\) towards each other. The minimum value of \(u\), so that they can just touch each other is :
(Take \(k = \frac{1}{4 \pi \epsilon_0}\) and assume \(kQ^2 > Gm^2\) where \(G\) is the Gravitational constant)
Surface tension of two liquids (having same densities), \(T_1\) and \(T_2\) are measured using capillary rise method utilizing two tubes with inner radii of \(r_1\) and \(r_2\) where \(r_1 > r_2\). The measured liquid heights in these tubes are \(h_1\) and \(h_2\) respectively. [Ignore the weight of the liquid about the lowest point of meniscus]. The heights \(h_1\) and \(h_2\) and surface tensions \(T_1\) and \(T_2\) satisfy the relation :
The total length of potentiometer wire AB is \(50\) cm in the arrangement as shown in figure. If P is the point where the galvanometer shows zero reading then the length AP is ___________ cm.

Keeping the significant figures in view, the sum of the physical quantities \(52.01\) m, \(153.2\) m and \(0.123\) m is :
Two cars \(A\) and \(B\) each of mass \(10^3\) kg are moving on parallel tracks separated by a distance of \(10\) m, in same direction with speeds \(72\) km/h and \(36\) km/h. The magnitude of angular momentum of car \(A\) with respect to car \(B\) is ___________ J.s.
Two known resistances of \(R \Omega\) and \(2R \Omega\) and one unknown resistance \(X \Omega\) are connected in a circuit as shown in the figure. If the equivalent resistance between points \(A\) and \(B\) in the circuit is \(X \Omega\), then the value of \(X\) is ___________ \(\Omega\).

An infinitely long straight wire carrying current \(I\) is bent in a planar shape as shown in the diagram. The radius of the circular part is \(r\). The magnetic field at the centre \(O\) of the circular loop is :

The energy of an electron in an orbit of the Bohr's atom is \(-0.04 E_g\) where \(E_g\) is the ground state energy. If \(L\) is the angular momentum of the electron in this orbit and \(h\) is the Planck's constant, then \(\frac{2 \pi L}{h}\) is ___________ :
As shown in the diagram, when the incident ray is parallel to base of the prism, the emergent ray grazes along the second surface. If refractive index of the material of prism is \(\sqrt{2}\), the angle \(\theta\) of prism is :

Given below are two statements :
Statement I : In a Young's double slit experiment, the angular separation of fringes will increase as the screen is moved away from the plane of the slits
Statement II : In a Young's double slit experiment, the angular separation of fringes will increase when monochromatic source is replaced by another monochromatic source of higher wavelength
In the light of the above statements, choose the correct answer from the options given below :
The kinetic energy of a simple harmonic oscillator is oscillating with angular frequency of 176 rad/s. The frequency of this simple harmonic oscillator is \hspace{1cm} Hz. [Take \(\pi = \frac{22}{7}\)]
A body of mass 2 kg is moving along x-direction such that its displacement as function of time is given by \(x(t) = \alpha t^2 + \beta t + \gamma\) m, where \(\alpha = 1\) m/s\(^2\), \(\beta = 1\) m/s and \(\gamma = 1\) m. The work done on the body during the time interval \(t = 2\) s to \(t = 3\) s, is ___________ J.
A large drum having radius \(R\) is spinning around its axis with angular velocity \(\omega\), as shown in figure. The minimum value of \(\omega\) so that a body of mass \(M\) remains stuck to the inner wall of the drum, taking the coefficient of friction between the drum surface and mass \(M\) as \(\mu\), is :

A capacitor \(C\) is first charged fully with potential difference of \(V_0\) and disconnected from the battery. The charged capacitor is connected across an inductor having inductance \(L\). In \(t\) s 25% of the initial energy in the capacitor is transferred to the inductor. The value of \(t\) is ___________ s.
A spherical body of radius \(r\) and density \(\sigma\) falls freely through a viscous liquid having density \(\rho\) and viscosity \(\eta\) and attains a terminal velocity \(v_0\). Estimated maximum error in the quantity \(\eta\) is : (Ignore errors associated with \(\sigma\), \(\rho\) and \(g\), gravitational acceleration)
A river of width 200 m is flowing from west to east with a speed of 18 km/h. A boat, moving with speed of 36 km/h in still water, is made to travel one-round trip (bank to bank of the river). Minimum time taken by the boat for this journey and also the displacement along the river bank are \hspace{1cm} and \hspace{1cm} respectively.
The r.m.s. speed of oxygen molecules at 47 \(^\circ\)C is equal to that of the hydrogen molecules kept at ___________ \(^\circ\)C. (Mass of oxygen molecule/mass of hydrogen molecule = 32/2)
The charge stored by the capacitor C in the given circuit in the steady state is \hspace{1cm} \(\mu\)C.

A battery with EMF \(E\) and internal resistance \(r\) is connected across a resistance \(R\). The power consumption in \(R\) will be maximum when :
The pulley shown in figure is made using a thin rim and two rods of length equal to diameter of the rim. The rim and each rod have a mass of \(M\). Two blocks of mass of \(M\) and \(m\) are attached to two ends of a light string passing over the pulley, which is hinged to rotate freely in vertical plane about its center. The magnitudes of the acceleration experienced by the blocks is ___________ (assume no slipping of string on pulley).

An electromagnetic wave of frequency 100 MHz propagates through a medium of conductivity, \(\sigma = 10\) mho/m. The ratio of maximum conduction current density to maximum displacement current density is \hspace{1cm.
\(\left[ Take \frac{1{4\pi\epsilon_0} = 9 \times 10^9 Nm^2/C^2 \right]\)
A diatomic gas (\(\gamma = 1.4\)) does 100 J of work when it is expanded isobarically. Then the heat given to the gas is ___________ J.
In a Young's double slit experiment set up, the two slits are kept 0.4 mm apart and screen is placed at 1 m from slits. If a thin transparent sheet of thickness 20 \(\mu\)m is introduced in front of one of the slits then center bright fringe shifts by 20 mm on the screen. The refractive index of transparent sheet is given by \(\frac{\alpha}{10}\), where \(\alpha\) is ___________.
A particle having electric charge \(3 \times 10^{-19}\) C and mass \(6 \times 10^{-27}\) kg is accelerated by applying an electric potential of 1.21 V. Wavelength of the matter wave associated with the particle is \(\alpha \times 10^{-12}\) m. The value of \(\alpha\) is \hspace{1cm. (Take Planck's constant \(= 6.6 \times 10^{-34\) J.s)
The terminal velocity of a metallic ball of radius 6 mm in a viscous fluid is 20 cm/s. The terminal velocity of another ball of same material and having radius 3 mm in the same fluid will be \hspace{1cm} cm/s.
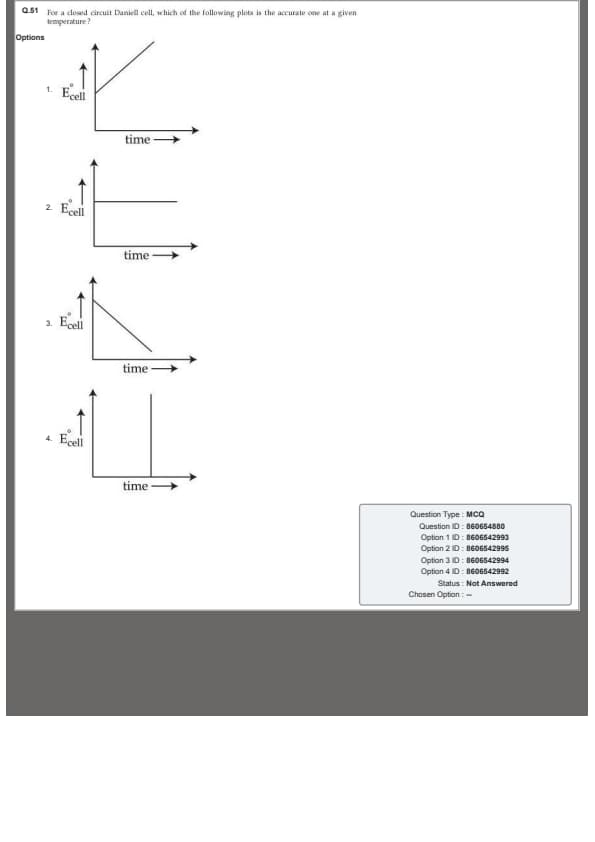
For a closed circuit Daniell cell, which of the following plots is the accurate one at a given temperature ?
Given below are four compounds :
(a) n-propyl chloride
(b) iso-propyl chloride
(c) sec-butyl chloride
(d) neo-pentyl chloride
Percentage of carbon in the one which exhibits optical isomerism is :
Given below are two statements :
Statement I : Crystal Field Stabilization Energy (CFSE) of \( [Cr(H_2O)_6]^{2+} \) is greater than that of \( [Mn(H_2O)_6]^{2+} \).
Statement II : Potassium ferricyanide has a greater spin-only magnetic moment than sodium ferrocyanide.
In the light of the above statements, choose the correct answer from the options given below :
The correct statements are :
A. Activation energy for enzyme catalysed hydrolysis of sucrose is lower than that of acid catalysed hydrolysis.
B. During denaturation, secondary and tertiary structures of a protein are destroyed but primary structure remains intact.
C. Nucleotides are joined together by glycosidic linkage between \( C_1 \) and \( C_4 \) carbons of the pentose sugar.
D. Quaternary structure of proteins represents overall folding of the polypeptide chain.
Choose the correct answer from the options given below :
By usual analysis, 1.00 g of compound (X) gave 1.79 g of magnesium pyrophosphate. The percentage of phosphorus in compound (X) is : \hspace{1cm} (nearest integer)
(Given, molar mass in g mol\(^{-1}\) ; O = 16, Mg = 24, P = 31)
Aqueous HCl reacts with \( MnO_2(s) \) to form \( MnCl_2(aq) \), \( Cl_2(g) \) and \( H_2O(l) \). What is the weight (in g) of \( Cl_2 \) liberated when 8.7 g of \( MnO_2(s) \) is reacted with excess aqueous HCl solution ?
(Given Molar mass in g mol\(^{-1}\) : Mn = 55, Cl = 35.5, O = 16, H = 1)
Match List - I with List - II.

Choose the correct answer from the options given below :
Given below are two statements :
Statement I : The correct order in terms of bond dissociation enthalpy is \( Cl_2 > Br_2 > F_2 > I_2 \).
Statement II : The correct trend in the covalent character of the metal halides is \( SnCl_2 > SnCl_4 \), \( PbCl_2 > PbCl_4 \) and \( UF_4 > UF_6 \).
In the light of the above statements, choose the correct answer from the options given below :
Consider the following spectral lines for atomic hydrogen :
A. First line of Paschen series
B. Second line of Balmer series
C. Third line of Paschen series
D. Fourth line of Bracket series
The correct arrangement of the above lines in ascending order of energy is :
Decomposition of A is a first order reaction at T(K) and is given by \( A(g) \rightarrow B(g) + C(g) \).
In a closed 1 L vessel, 1 bar A(g) is allowed to decompose at T(K). After 100 minutes, the total pressure was 1.5 bar. What is the rate constant (in \( min^{-1} \)) of the reaction ? (\( \log 2 = 0.3 \))
The correct order of the rate of the reaction for the following reaction with respect to nucleophiles is :
\( CH_3Br + Nu^\ominus \rightarrow CH_3Nu + Br^\ominus \)
Consider the following data :
\( \Delta_f H^\ominus (methane, g) = -X kJ mol^{-1} \)
Enthalpy of sublimation of graphite \( = Y kJ mol^{-1} \)
Dissociation enthalpy of \( H_2 = Z kJ mol^{-1} \)
The bond enthalpy of C-H bond is given by :
Given below are two statements :
Statement I : Compound (X), shown below, dissolves in \( NaHCO_3 \) solution and has two chiral carbon atoms.
Statement II : Compound (Y), shown below, has two carbons with \( sp^3 \) hybridization, one carbon with \( sp^2 \) and one carbon with \( sp \) hybridization.![]()
In the light of the above statements, choose the correct answer from the options given below :
Consider the following sequence of reactions. The number of bromine atom(s) in the final product (P) will be :

The correct order of reactivity of the following benzyl halides towards reaction with KCN is :

Given below are some of the statements about Mn and \( Mn_2O_7 \). Identify the correct statements.
A. Mn forms the oxide \( Mn_2O_7 \), in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn.
C. \( Mn_2O_7 \) is an ionic oxide.
D. The structure of \( Mn_2O_7 \) consists of one bridged oxygen.
Choose the correct answer from the options given below :
On heating a mixture of common salt and \( K_2Cr_2O_7 \) in equal amount along with concentrated \( H_2SO_4 \) in a test tube, a gas is evolved. Formula of the gas evolved and oxidation state of the central metal atom in the gas respectively are :
Given below are two statements :
Statement I : The correct order in terms of atomic/ionic radii is \( Al > Mg > Mg^{2+} > Al^{3+} \).
Statement II : The correct order in terms of the magnitude of electron gain enthalpy is \( Cl > Br > S > O \).
In the light of the above statements, choose the correct answer from the options given below :
Match List - I with List - II.

Choose the correct answer from the options given below :
The correct increasing order of C-H(A), C-O(B), C=O(C) and C\(\equiv\)N(D) bonds in terms of covalent bond length is :
The first and second ionization constants of \(H_{2}X\) are \(2.5 \times 10^{-8}\) and \(1.0 \times 10^{-13}\) respectively. The concentration of \(X^{2-}\) in \(0.1\) M \(H_{2}X\) solution is \(\_\_\_\_\_\_ \times 10^{-13}\) M. (Nearest Integer)
The osmotic pressure of a living cell is 12 atm at 300 K. The strength of sodium chloride solution that is isotonic with the living cell at this temperature is \(\_\_\_\_\_\_\) g L\(^{-1}\). (Nearest integer)
Given: R = 0.08 L atm K\(^{-1}\) mol\(^{-1}\)
Assume complete dissociation of NaCl
(Given : Molar mass of Na and Cl are 23 and 35.5 g mol\(^{-1}\) respectively.)
Identify the metal ions among \(Co^{2+}, Ni^{2+}, Fe^{2+}, V^{3+}\) and \(Ti^{2+}\) having a spin-only magnetic moment value more than 3.0 BM. The sum of unpaired electrons present in the high spin octahedral complexes formed by those metal ions is \(\_\_\_\_\_\_\).
A substance 'X' (1.5 g) dissolved in 150 g of a solvent 'Y' (molar mass = 300 g mol\(^{-1}\)) led to an elevation of the boiling point by 0.5 K. The relative lowering in the vapour pressure of the solvent 'Y' is \(\_\_\_\_\_\_ \times 10^{-2}\). (nearest integer)
[Given : \(K_{b}\) of the solvent = 5.0 K kg mol\(^{-1}\)]
Assume the solution to be dilute and no association or dissociation of X takes place in solution.
MX is a sparingly soluble salt that follows the given solubility equilibrium at 298 K.
MX(s) \(\rightleftharpoons M^{+}(aq) + X^{-}(aq)\); \(K_{sp} = 10^{-10}\)
If the standard reduction potential for \(M^{+}(aq) + e^{-} \rightarrow M(s)\) is \((E^{\circ}_{M^{+}/M}) = 0.79\) V, then the value of the standard reduction potential for the metal/metal insoluble salt electrode \(E^{\circ}_{X^{-}/MX(s)/M}\) is \(\_\_\_\_\_\_\) mV. (nearest integer)
[Given : \(\frac{2.303 RT}{F} = 0.059\) V]
















































Comments