JEE Main 2026 Jan 24 Shift 2 question paper is available here with answer key and solutions. NTA conducted the first shift of the day on Jan 24, 2026, from 3 PM to 6 PM.
JEE Main 2026 24th Jan Shift 2 Question Paper with Solution PDF
Also Check:
- Download JEE Main 2026 Session 1 Question Paper for all Shifts
- Predict your rank using Collegedunia’s JEE Main Rank Predictor for free
Based on initial analysis, JEE Main Jan 24th Shift 2 was Easy to Medium, where Mathematics remains the lengthiest section. Students can access the JEE Main Jan 24 Shift 2 official questions with answer keys here.

The largest value of \(n\), for which \(40^n\) divides \(60!\), is
Consider the following three statements for the function \(f : (0,\infty) \rightarrow \mathbb{R}\) defined by
\[ f(x) = \left| \log_e x \right| - |x - 1| : \]
(I) \(f\) is differentiable at all \(x > 0\).
(II) \(f\) is increasing in \((0,1)\).
(III) \(f\) is decreasing in \((1,\infty)\).
Then,
Let \(P=[p_{ij}]\) and \(Q=[q_{ij}]\) be two square matrices of order \(3\) such that \(q_{ij}=2^{(i+j-1)}p_{ij}\) and \(\det(Q)=2^{10}\). Then the value of \(\det(\operatorname{adj}(\operatorname{adj} P))\) is
Let \(X=\{x\in\mathbb{N}:1\le x\le19\}\) and for some \(a,b\in\mathbb{R}\), \(Y=\{ax+b:x\in X\}\).
If the mean and variance of the elements of \(Y\) are \(30\) and \(750\) respectively, then the sum of all possible values of \(b\) is
Let the angles made with the positive \(x\)-axis by two straight lines drawn from the point \(P(2,3)\) and meeting the line \(x+y=6\) at a distance \(\sqrt{\frac{2}{3}}\) from the point \(P\) be \(\theta_1\) and \(\theta_2\). Then the value of \((\theta_1+\theta_2)\) is
Let \(a_1,a_2,a_3,a_4\) be an A.P. of four terms such that each term of the A.P. and its common difference are integers. If \(a_1+a_2+a_3+a_4=48\) and \(a_1^2a_2a_3a_4+1^4=361\), then the largest term of the A.P. is equal to
The letters of the word ``UDAYPUR'' are written in all possible ways with or without meaning and these words are arranged as in a dictionary. The rank of the word ``UDAYPUR'' is
The sum of all values of \(\alpha\), for which the shortest distance between the lines \(\dfrac{x+1}{\alpha}=\dfrac{y-2}{-1}=\dfrac{z-4}{-\alpha}\) and \(\dfrac{x}{\alpha}=\dfrac{y-1}{2}=\dfrac{z-1}{2\alpha}\) is \(\sqrt{2}\), is
If the domain of the function \(f(x)=\sin^{-1}\!\left(\dfrac{1}{x^2-2x-2}\right)\) is \((-\infty,\alpha)\cup[\beta,\gamma]\cup[\delta,\infty)\), then \(\alpha+\beta+\gamma+\delta\) is equal to
Let the length of the latus rectum of an ellipse \(\dfrac{x^2}{a^2}+\dfrac{y^2}{b^2}=1\) \((a>b)\) be \(30\).
If its eccentricity is the maximum value of the function \(f(t)=-\dfrac{3}{4}+2t-t^2\), then \((a^2+b^2)\) is equal to
Let \(\vec{a}=2\hat{i}-\hat{j}-\hat{k}\), \(\vec{b}=\hat{i}+3\hat{j}-\hat{k}\) and \(\vec{c}=2\hat{i}+\hat{j}+3\hat{k}\).
Let \(\vec{v}\) be the vector in the plane of \(\vec{a}\) and \(\vec{b}\), such that the length of its projection on the vector \(\vec{c}\) is \(\dfrac{1}{\sqrt{14}}\).
Then \(|\vec{v}|\) is equal to
Let \(f\) be a function such that \(3f(x)+2f\!\left(\dfrac{m}{19x}\right)=5x\), \(x\ne0\), where \(m=\displaystyle\sum_{i=1}^{9} i^2\).
Then \(f(5)-f(2)\) is equal to
Let \(f(\alpha)\) denote the area of the region in the first quadrant bounded by \(x=0\), \(x=1\), \(y^2=x\) and \(y=|\alpha x-5|-|1-\alpha x|+\alpha^2\).
Then \((f(0)+f(1))\) is equal to
The smallest positive integral value of \(a\), for which all the roots of \(x^4-ax^2+9=0\) are real and distinct, is equal to
Let \(\vec{a}=2\hat{i}-5\hat{j}+5\hat{k}\) and \(\vec{b}=\hat{i}-\hat{j}+3\hat{k}\).
If \(\vec{c}\) is a vector such that \(2(\vec{a}\times\vec{c})+3(\vec{b}\times\vec{c})=\vec{0}\) and \((\vec{a}-\vec{b})\cdot\vec{c}=-97\), then \(|\vec{c}\times\hat{k}|^2\) is equal to
Let \([t]\) denote the greatest integer less than or equal to \(t\).
If the function \[ f(x)= \begin{cases} b^2\sin\!\left[\dfrac{\pi}{2}\left[\dfrac{\pi}{2}(\cos x+\sin x)\cos x\right]\right], & x<0
\dfrac{\sin x-\dfrac{1}{2}\sin 2x}{x^3}, & x>0
a, & x=0 \end{cases} \]
is continuous at \(x=0\), then \(a^2+b^2\) is equal to
Let \[ f(x)=\int \frac{7x^{10}+9x^8}{(1+x^2+2x^9)^2}\,dx,\quad x>0, \]
and \[ A= \begin{bmatrix} 0 & 0 & 1
\frac14 & f'(1) & 1
\alpha & 4 & 1 \end{bmatrix}. \]
If \(B=\operatorname{adj}(\operatorname{adj} A)\), then the value of \(\alpha\) for which \(\det(B)=1\) is
The value of \[ \left(\frac13+\frac47\right) +\left(\frac1{3^2}+\frac13\times\frac47+\frac4{7^2}\right) +\left(\frac1{3^3}+\frac1{3^2}\times\frac47+\frac13\times\frac4{7^2}+\frac4{7^3}\right) +\cdots up to infinite terms is
\]
Let \(y=y(x)\) be a differentiable function in the interval \((0,\infty)\) such that \(y(1)=2\), and \[ \lim_{t\to x}\left(\frac{t^2y(x)-x^2y(t)}{x-t}\right)=3 for each x>0. \]
Then \(2y(2)\) is equal to
Let the image of parabola \(x^2=4y\) in the line \(x-y=1\) be \((y+a)^2=b(x-c)\), where \(a,b,c\in\mathbb{N}\).
Then \(a+b+c\) is equal to
The number of elements in the set \(\{x\in[0,180^\circ]: \tan(x+100^\circ)=\tan(x+50^\circ)\tan x\tan(x-50^\circ)\}\) is
Let \(z=(1+i)(1+2i)(1+3i)\cdots(1+ni)\), where \(i=\sqrt{-1}\).
If \(|z|^2=44200\), then \(n\) is equal to
Let \((h,k)\) lie on the circle \(C:x^2+y^2=4\) and the point \((2h+1,\,3k+2)\) lie on an ellipse with eccentricity \(e\).
Then the value of \(\dfrac{5}{e^2}\) is equal to
If \(f(x)\) satisfies the relation \[ f(x)=e^x+\int_0^1 (y+x e^x)f(y)\,dy, \]
then \(e+f(0)\) is equal to
Let \(S\) be a set of \(5\) elements and \(P(S)\) denote the power set of \(S\).
Let \(E\) be the event of choosing an ordered pair \((A,B)\) from \(P(S)\times P(S)\) such that \(A\cap B=\varnothing\).
If the probability of the event \(E\) is \(\dfrac{3^p}{2^q}\), where \(p,q\in\mathbb{N}\), then \(p+q\) is equal to
The binding energy for the following nuclear reactions are expressed in MeV.
\[ {}^{3}_{2}He + {}^{1}_{0}n \rightarrow {}^{4}_{2}He + 20 MeV \]
\[ {}^{4}_{2}He + {}^{1}_{0}n \rightarrow {}^{5}_{2}He - 0.9 MeV \]
If \( X_3, X_4, X_5 \) denote the stability of \( {}^{3}_{2}He, {}^{4}_{2}He \) and \( {}^{5}_{2}He \), respectively, then the correct order is:
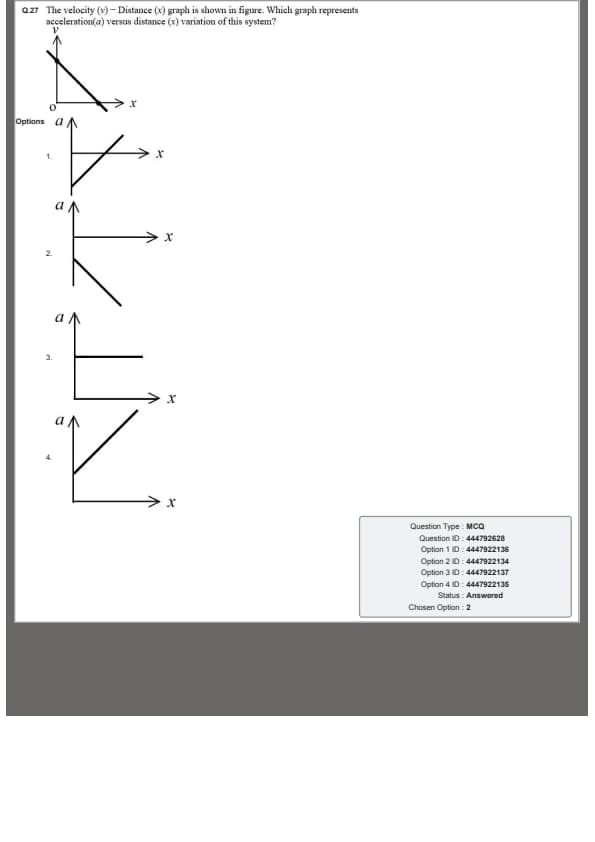
The velocity (\(v\)) – distance (\(x\)) graph is shown in the figure. Which graph represents acceleration (\(a\)) versus distance (\(x\)) variation of this system?
A regular hexagon is formed by six wires each of resistance \( r \,\Omega \) and the corners are joined to the centre by wires of same resistance. If the current enters at one corner and leaves at the opposite corner, the equivalent resistance of the hexagon between the two opposite corners will be
Distance between an object and three times magnified real image is 40 cm. The focal length of the mirror used is ____ cm.
In case of vertical circular motion of a particle by a thread of length \( r \), if the tension in the thread is zero at an angle \(30^\circ\) as shown in the figure, the velocity at the bottom point (A) of the vertical circular path is ( \( g \) = gravitational acceleration ).
The fifth harmonic of a closed organ pipe is found to be in unison with the first harmonic of an open pipe. The ratio of lengths of closed pipe to that of the open pipe is \( \frac{5}{x} \). The value of \( x \) is ____.
Three parallel plate capacitors each with area \(A\) and separation \(d\) are filled with two dielectric (\(k_1\) and \(k_2\)) in the following fashion. (\(k_1 > k_2\)) Which of the following is true?
Two identical circular loops \(P\) and \(Q\) each of radius \(r\) are lying in parallel planes such that they have common axis. The current through \(P\) and \(Q\) are \(I\) and \(4I\) respectively in clockwise direction as seen from \(O\). The net magnetic field at \(O\) is:
10 mole of an ideal gas is undergoing the process shown in the figure. The heat involved in the process from \(P_1\) to \(P_2\) is \( \alpha \) Joule \((P_1 = 21.7 Pa,\; P_2 = 30 Pa,\; C_v = 21 J/K·mol,\; R = 8.3 J/mol·K)\). The value of \( \alpha \) is ____.
In a vernier callipers, 50 vernier scale divisions are equal to 48 main scale divisions. If one main scale division = 0.05 mm, then the least count of the vernier callipers is ____ mm.
A flexible chain of mass \(m\) hangs between two fixed points at the same level. The inclination of the chain with the horizontal at the two points of support is \(30^\circ\). Considering the equilibrium of each half of the chain, the tension of the chain at the lowest point is ____.
Identify the correct truth table of the given logic circuit.
A moving coil galvanometer of resistance \(100\,\Omega\) shows a full scale deflection for a current of \(1\,mA\). The value of resistance required to convert this galvanometer into an ammeter, showing full scale deflection for a current of \(5\,mA\), is ____ \(\Omega\).
A point source is kept at the center of a spherically enclosed detector. If the volume of the detector is increased by 8 times, the intensity will
Five persons \(P_1, P_2, P_3, P_4\) and \(P_5\) recorded object distance (\(u\)) and image distance (\(v\)) using same convex lens having power \(+5\) D as (25,96), (30,62), (35,37), (45,35) and (50,32) respectively. Identify correct statement.
In the Young's double slit experiment the intensity produced by each one of the individual slits is \(I_0\). The distance between two slits is \(2\,mm\). The distance of screen from slits is \(10\,m\). The wavelength of light is \(6000\,\AA\). The intensity of light on the screen in front of one of the slits is ____.
A cubical block of density \( \rho_b = 600\,kg/m^3 \) floats in a liquid of density \( \rho_l = 900\,kg/m^3 \). If the height of block is \(H = 8.0\,cm\), then height of the submerged part is ____ cm.
The reading of the ammeter (\(A\)) in steady state in the following circuit (assuming negligible internal resistance of the ammeter) is ____ A.
A thin uniform rod (\(X\)) of mass \(M\) and length \(L\) is pivoted at a height \( \left(\dfrac{L}{3}\right) \) as shown in the figure. The rod is allowed to fall from a vertical position and lie horizontally on the table. The angular velocity of this rod when it hits the table top is ____. (\(g\) = gravitational acceleration)
When a light of a given wavelength falls on a metallic surface the stopping potential for photoelectrons is \(3.2\ V\). If a second light having wavelength twice of the first light is used, the stopping potential drops to \(0.7\ V\). The wavelength of the first light is ____ m.
A soap bubble of surface tension \(0.04\,N/m\) is blown to a diameter of \(7\,cm\). If \((15000 - x)\,\muJ\) of work is done in blowing it further to make its diameter \(14\,cm\) \((\pi = 22/7)\), then the value of \(x\) is ____.
A uniform solid cylinder of length \(L\) and radius \(R\) has moment of inertia about its axis equal to \(I_1\). A small co-centric cylinder of length \(L/2\) and radius \(R/3\) carved from it has moment of inertia about its axis equal to \(I_2\). The ratio \(I_1/I_2\) is ____.
In a meter bridge experiment to determine the value of unknown resistance, first the resistances \(2\,\Omega\) and \(3\,\Omega\) are connected in the left and right gaps of the bridge and the null point is obtained at a distance \(l\) cm from the left end. Now, when an unknown resistance \(x\,\Omega\) is connected in parallel to \(3\,\Omega\), the null point is shifted by \(10\,cm\) to the right. The value of \(x\) is ____ \(\Omega\).
When \(300\,J\) of heat is given to an ideal gas with \(C_p = \dfrac{7}{2}R\), its temperature rises from \(20^\circC\) to \(50^\circC\) keeping its volume constant. The mass of the gas is (approximately) ____ g. \((R = 8.314\,J/mol·K)\)
A point charge \(q = 1\,\muC\) is located at a distance \(2\,cm\) from one end of a thin insulating wire of length \(10\,cm\) having a charge \(Q = 24\,\muC\), distributed uniformly along its length, as shown in the figure. Force between \(q\) and wire is ____ N.
(Use: \(\dfrac{1}{4\pi\varepsilon_0} = 9 \times 10^9\,N·m^2/C^2\))
In the group analysis of cations, Ba\(^{2+}\) \& Ca\(^{2+}\) are precipitated respectively as
Given below are two statements:
Statement I: The dipole moment of R–CN is greater than R–NC and R–NC can undergo hydrolysis under acidic medium to produce R–COOH.
Statement II: R–CN hydrolyses under acidic medium to produce a compound which on treatment with SOCl\(_2\), followed by the addition of NH\(_3\) gives another compound (X). This compound (X) on treatment with NaOCl/NaOH gives a product, that on treatment with CHCl\(_3\)/KOH/\(\Delta\) produces R–NC.
In the light of the above statements, choose the correct answer from the options given below.
``X'' is an oxoanion of the lightest element of group 17 (in the periodic table). The metal is in +6 oxidation state in ``X''. The color of the potassium salt of X is
Choose the INCORRECT statement
Two liquids A and B form an ideal solution at temperature T K. At T K, the vapour pressures of pure A and pure B are 55 and 15 kPa respectively. What is the mole fraction of A in solution of A and B in equilibrium with a vapour in which the mole fraction of A is 0.8?
The number of possible tripeptides formed involving alanine (ala), glycine (gly) and valine (val), where no amino acid has been used more than once is
One mole of Cl\(_2\)(g) was passed into 2 L of cold 2 M KOH solution. After the reaction, the concentrations of Cl\(^-\), ClO\(^-\) and OH\(^-\) are respectively (assume volume remains constant)
Given below are two statements regarding conformations of n-butane. Choose the correct option.
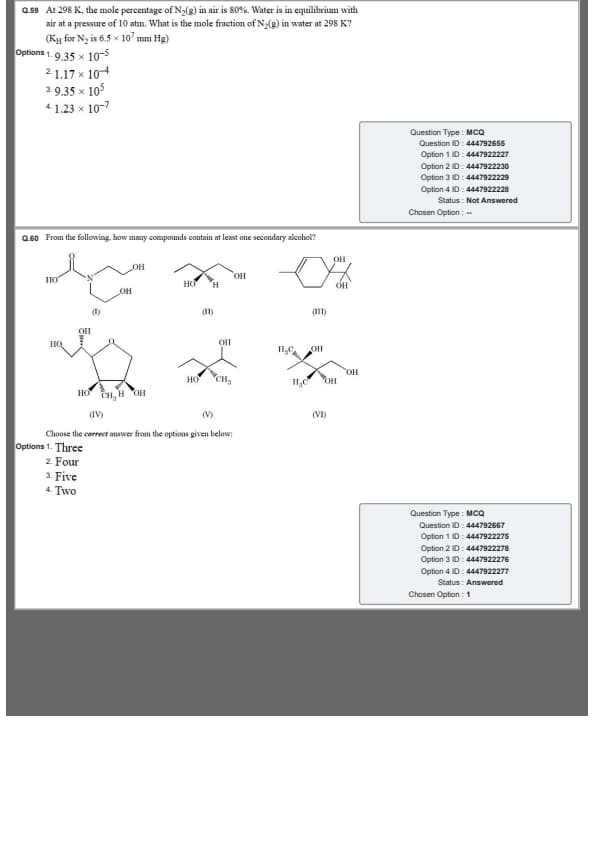
At 298 K, the mole percentage of N\(_2\)(g) in air is 80%. Water is in equilibrium with air at a pressure of 10 atm. What is the mole fraction of N\(_2\)(g) in water at 298 K? (\(K_H\) for N\(_2\) = \(6.5 \times 10^7\) mm Hg)
From the following, how many compounds contain at least one secondary alcohol?
The wavelength of light absorbed for the following complexes are in the order
[Co(NH\(_3\))\(_6\)]\(^{3+}\) (I), [Co(H\(_2\)O)\(_6\)]\(^{3+}\) (II), [Co(CN)\(_6\)]\(^{3-}\) (III), [Co(NH\(_3\))\(_5\)(H\(_2\)O)]\(^{3+}\) (IV), [CoF\(_6\)]\(^{3-}\) (V)
Consider the following gaseous equilibrium in a closed container of volume \(V\) at temperature \(T\):
P\(_2\)(g) + Q\(_2\)(g) \(\rightleftharpoons\) 2PQ(g)
Initially, 2 moles each of P\(_2\)(g), Q\(_2\)(g) and PQ(g) are present at equilibrium. One mole each of P\(_2\) and Q\(_2\) are added. The number of moles of P\(_2\), Q\(_2\) and PQ at the new equilibrium respectively are
Given below are two statements:
Statement I: Cross aldol condensation between two different aldehydes will always produce four different products.
Statement II: When semicarbazide reacts with a mixture of benzaldehyde and acetophenone under optimum pH, it forms a condensation product with acetophenone only.
The wavelength of spectral line obtained in the spectrum of Li\(^{2+}\) ion, when the transition takes place between two levels whose sum is 4 and difference is 2, is
The heat of atomisation of methane and ethane are \(x\) kJ mol\(^{-1}\) and \(y\) kJ mol\(^{-1}\) respectively. The longest wavelength (\(\lambda\)) of light capable of breaking the C–C bond can be expressed in SI unit as:
Pair of species among the following having same bond order as well as paramagnetic character will be:
The unsaturated ether on acidic hydrolysis produces carbonyl compounds as shown below. Based on this, predict the solution/reagent that will help to distinguish "P" and "Q" obtained in the reaction.
Find out the statements which are not true.
A. Resonating structures with more covalent bonds and less charge separation are more stable.
B. In electromeric effect, an unsaturated system shows +E effect with nucleophile and –E effect with electrophile.
C. Inductive effect is responsible for high melting point, boiling point and dipole moment of polar compounds.
D. The greater the number of alkyl groups attached to the doubly bonded carbon atoms, higher is the heat of hydrogenation.
E. Stability of carbanion increases with increase in s-character of the carbon carrying negative charge.
The correct order of C, N, O and F in terms of second ionisation potential is
A student has planned to prepare acetanilide from aniline using acetic anhydride. The student has started from 9.3 g of aniline. However, the student has managed to obtain 11 g of dry acetanilide. The % yield of this reaction is
The half-life of \(^{65}\)Zn is 245 days. After \(x\) days, 75% of the original activity remained. The value of \(x\) in days is \underline{\hspace{1.5cm (Nearest integer).
(Given: \(\log 3 = 0.4771\) and \(\log 2 = 0.3010\))
Molar conductivity of a weak acid HQ of concentration 0.18 M was found to be \(\dfrac{1}{30}\) of the molar conductivity of another weak acid HZ with concentration 0.02 M. If \(\alpha_Q\) happened to be equal with \(\alpha_Z\), then the difference of the pK\(_a\) values of the two weak acids (pK\(_a\)(HQ) – pK\(_a\)(HZ)) is \underline{\hspace{1cm (Nearest integer).
(Given: degree of dissociation (\(\alpha \ll 1\) for both weak acids, \(\lambda^\circ\) : limiting molar conductivity of ions)
A chromium complex with formula CrCl\(_3\cdot\)6H\(_2\)O has a spin only magnetic moment value of 3.87 BM and its solution conductivity corresponds to 1:2 electrolyte. 2.75 g of the complex solution was initially passed through a cation exchanger. The solution obtained after the process was reacted with excess of AgNO\(_3\). The amount of AgCl formed in the above process is \hspace{1cm g (Nearest integer).
(Given: Molar mass in g mol\(^{-1\) Cr: 52; Cl: 35.5; Ag:108; O:16; H:1)
0.25 g of an organic compound “A” containing carbon, hydrogen and oxygen was analysed using combustion method. The increase in mass of CaCl\(_2\) tube and potash tube at the end of the experiment was found to be 0.15 g and 0.1837 g respectively. The percentage of oxygen in compound A is \underline{\hspace{1cm% (Nearest integer).
Grignard reagent RMgBr (P) reacts with water and forms a gas (Q). One gram of Q occupies 1.4 dm\(^3\) at STP. (P) on reaction with dry ice in dry ether followed by H\(_3\)O\(^+\) forms compound (Z). 0.1 mole of (Z) will weigh \underline{\hspace{1cm g (Nearest integer).






































Comments