JEE Main 2026 Jan 28 Shift 1 question paper is available here with answer key and solutions PDF. NTA has conducted the first shift of the day on Jan 28, 2026, from 9 AM to 12 PM.
JEE Main 2026 28th Jan Shift 1 Question Paper with Solution PDF
Based on initial analysis, JEE Main Jan 28th Shift 1 was Moderate to Difficult. Students can access the JEE Main Jan 28 Shift 1 official questions with answer keys here. Students can check the detailed paper analysis for JEE Main Jan 28th Shift 1 here.
Also Check:
- Download JEE Main 2026 Session 1 Question Paper for all Shifts
- Predict your rank using Collegedunia’s JEE Main Rank Predictor for free

If \( g(x) = 3x^2 + 2x - 3 \), \( f(0) = -3 \) and \( 4g(f(x)) = 3x^2 - 32x + 72 \), then \( f(g(2)) \) is equal to:
Let \( y = x \) be the equation of a chord of the circle \( C_1 \) (in the closed half-plane \( x \ge 0 \)) of diameter 10 passing through the origin. Let \( C_2 \) be another circle described on the given chord as diameter. If the equation of the chord of the circle \( C_2 \), which passes through the point \( (2, 3) \) and is farthest from the center of \( C_2 \), is \( x + ay + b = 0 \), then \( b \) is equal to:
Let \( S = \{x^3 + ax^2 + bx + c : a, b, c \in \mathbb{N} and a, b, c \le 20\} \) be a set of polynomials. Then the number of polynomials in \( S \), which are divisible by \( x^2 + 2 \), is:
The mean and variance of 10 observations are 9 and 34.2, respectively. If 8 of these observations are \( 2, 3, 5, 10, 11, 13, 15, 21 \), then the mean deviation about the median of all the 10 observations is:
Let \( y = y(x) \) be the solution of the differential equation \[ x\frac{dy}{dx} - \sin 2y = x^3(2 - x^3)\cos^2 y,\; x \ne 0. \]
If \( y(2) = 0 \), then \( \tan(y(1)) \) is equal to:
A bag contains 10 balls out of which \( k \) are red and \( (10-k) \) are black, where \( 0 \le k \le 10 \).
If three balls are drawn at random without replacement and all of them are found to be black, then the probability that the bag contains 1 red and 9 black balls is:
The common difference of the A.P.: \( a_1, a_2, \ldots, a_m \) is 13 more than the common difference of the A.P.: \( b_1, b_2, \ldots, b_n \).
If \( b_{31} = -277 \), \( b_{43} = -385 \) and \( a_{78} = 327 \), then \( a_1 \) is equal to:
The value of \[ \sum_{k=1}^{\infty} (-1)^{k+1}\left(\frac{k(k+1)}{k!}\right) \]
is:
If the distances of the point \( (1,2,a) \) from the line \[ \frac{x-1}{1}=\frac{y}{2}=\frac{z-1}{1} \]
along the lines \[ L_1:\ \frac{x-1}{3}=\frac{y-2}{4}=\frac{z-a}{b} \quad and \quad L_2:\ \frac{x-1}{1}=\frac{y-2}{4}=\frac{z-a}{c} \]
are equal, then \( a+b+c \) is equal to:
For three unit vectors \( \vec a, \vec b, \vec c \) satisfying \[ |\vec a-\vec b|^2 + |\vec b-\vec c|^2 + |\vec c-\vec a|^2 = 9 \]
and \[ |2\vec a + k\vec b + k\vec c| = 3, \]
the positive value of \( k \) is:
The value of \[ \lim_{x\to 0}\frac{\log_e\!\big(\sec(ex)\cdot \sec(e^2x)\cdots \sec(e^{10}x)\big)} {e^2-e^{2\cos x}} \]
is equal to:
Let \( z \) be a complex number such that \( |z-6|=5 \) and \( |z+2-6i|=5 \). Then the value of \( z^3+3z^2-15z+14 \) is equal to:
If \[ \frac{\tan(A-B)}{\tan A}+\frac{\sin^2 C}{\sin^2 A}=1, \quad A,B,C\in\left(0,\frac{\pi}{2}\right), \]
then:
The area of the region \[ R=\{(x,y): xy\le 8,\; 1\le y\le x^2,\; x\ge 0\} \]
is:
If \( \alpha,\beta \) where \( \alpha<\beta \), are the roots of the equation \[ \lambda x^2-(\lambda+3)x+3=0 \]
such that \[ \frac{1}{\alpha}-\frac{1}{\beta}=\frac{1}{3}, \]
then the sum of all possible values of \( \lambda \) is:
Let \( S=\{1,2,3,4,5,6,7,8,9\} \).
Let \( x \) be the number of 9-digit numbers formed using the digits of the set \( S \) such that only one digit is repeated and it is repeated exactly twice.
Let \( y \) be the number of 9-digit numbers formed using the digits of the set \( S \) such that only two digits are repeated and each of these is repeated exactly twice.
Then:
Let \( A, B, C \) be three \( 2\times2 \) matrices with real entries such that \[ B=(I+A)^{-1} \quad and \quad A+C=I. \]
If \[ BC=\begin{bmatrix}1 & -5
-1 & 2\end{bmatrix} \quad and \quad B\begin{bmatrix}x_1
x_2\end{bmatrix} =\begin{bmatrix}12
-6\end{bmatrix}, \]
then \( x_1+x_2 \) is:
Let \( ABC \) be an equilateral triangle with orthocenter at the origin and the side \( BC \) lying on the line \( x+2\sqrt{2}\,y=4 \). If the coordinates of the vertex \( A \) are \( (\alpha,\beta) \), then the greatest integer
less than or equal to \( |\alpha+\sqrt{2}\beta| \) is:
If \[ \int \frac{1-5\cos^2 x}{\sin^5 x\cos^2 x}\,dx=f(x)+C, \]
where \( C \) is the constant of integration, then \[ f\!\left(\frac{\pi}{6}\right)-f\!\left(\frac{\pi}{4}\right) \]
is equal to:
Let \( f \) be a polynomial function such that \[ f(x^2+1)=x^4+5x^2+2,\quad for all x\in\mathbb{R}. \]
Then \[ \int_0^3 f(x)\,dx \]
is equal to:
In a G.P., if the product of the first three terms is \(27\) and the set of all possible values for the sum of its first three terms is \( \mathbb{R} - (a,b) \), then \( a^2+b^2 \) is equal to:
If \[ k=\tan\!\left(\frac{\pi}{4}+\frac{1}{2}\cos^{-1}\!\left(\frac{2}{3}\right)\right) +\tan\!\left(\frac{1}{2}\sin^{-1}\!\left(\frac{2}{3}\right)\right), \]
then the number of solutions of the equation \[ \sin^{-1}(kx-1)=\sin^{-1}x-\cos^{-1}x \]
is:
For some \( \theta\in\left(0,\frac{\pi}{2}\right) \), let the eccentricity and the length of the latus rectum
of the hyperbola \[ x^2-y^2\sec^2\theta=8 \]
be \( e_1 \) and \( l_1 \), respectively, and let the eccentricity and the length of the latus rectum of the ellipse \[ x^2\sec^2\theta+y^2=6 \]
be \( e_2 \) and \( l_2 \), respectively.
If \[ e_1^2=\frac{2}{e_2^2}\left(\sec^2\theta+1\right), \]
then \[ \left(\frac{l_1l_2}{e_1^2e_2^2}\right)\tan^2\theta \]
is equal to:
The value of \[ \sum_{r=1}^{20}\sqrt{\left|\pi\left(\int_0^r x|\sin \pi x|\,dx\right)\right|} \]
is:
Let \( PQR \) be a triangle such that \[ \vec{PQ}=-2\hat i-\hat j+2\hat k,\quad \vec{PR}=a\hat i+b\hat j-4\hat k,\ a,b\in\mathbb{Z}. \]
Let \( S \) be the point on \( QR \) which is equidistant from the lines \( PQ \) and \( PR \).
If \[ |\vec{PR}|=9 \quad and \quad \vec{PS}=\hat i-7\hat j+2\hat k, \]
then the value of \( 3a-4b \) is:
The electric current in the circuit is given as \[ i=i_0\left(\frac{t}{T}\right). \]
The r.m.s. current for the period \( t=0 \) to \( t=T \) is:
The magnitudes of power of a biconvex lens (refractive index \(1.5\)) and that of a plano-convex lens (refractive index \(1.7\)) are same.
If the curvature of the plano-convex lens exactly matches with the curvature of the back surface of the biconvex lens,
then the ratio of radii of curvature of the front and back surfaces of the biconvex lens is:
An atom \( ^8_3X \) is bombarded by a shower of fundamental particles and in 10 s this atom absorbed
10 electrons, 10 protons and 9 neutrons. The percentage growth in the surface area of the nucleus is recorded by:
Given below are two statements:
Statement I:
A plane wave after passing through a prism remains a plane wave, but passing through a small pin hole may become a spherical wave.
Statement II:
The curvature of a spherical wave emerging from a slit will increase for increasing slit width.
In the light of the above statements, choose the correct answer:
When both jaws of a vernier calipers touch each other, zero mark of the vernier scale is right to the zero mark of main scale. 4th mark on vernier scale coincides with a certain mark on the main scale. While measuring the length of a cylinder, observer observes 15 divisions on main scale and 5th division of vernier scale coincides with a main scale division. Measured length of cylinder is ____ mm.
(Least count of Vernier calliper = \(0.1\) mm)
In the potentiometer, when the cell in the secondary circuit is shunted with \(4\,\Omega\) resistance, the balance is obtained at a length \(120\) cm of wire. Now when the same cell is shunted with \(12\,\Omega\) resistance, the balance is shifted to a length of \(180\) cm. The internal resistance of the cell is ____ \( \Omega \).
Water drops fall from a tap on the floor, \(5\) m below, at regular intervals of time.
The first drop strikes the floor when the sixth drop begins to fall.
The height at which the fourth drop will be from the ground, at the instant when the first drop strikes the ground, is ____ m.
(\( g = 10 \, m s^{-2} \))
The electric field of an electromagnetic wave travelling through a medium is given by \[ \vec{E}(x,t)=25\sin(2\times10^{15}t-10^{7}x)\,\hat{n}. \]
Then the refractive index of the medium is ____.
(All given measurements are in SI units)
Three long straight wires carrying current are arranged mutually parallel as shown in the figure.
The force experienced by \(15\) cm length of wire \(Q\) is ____.
(\( \mu_0 = 4\pi \times 10^{-7}\,T m A^{-1} \))

Two wires \(A\) and \(B\) made of different materials have lengths \(6.0\) cm and \(5.4\) cm,
and areas of cross-sections \(3.0\times10^{-5}\,m^2\) and \(4.5\times10^{-5}\,m^2\), respectively.
They are stretched by the same magnitude under the same load.
If the ratio of Young’s modulus of \(A\) to that of \(B\) is \(x:3\), find the value of \(x\).
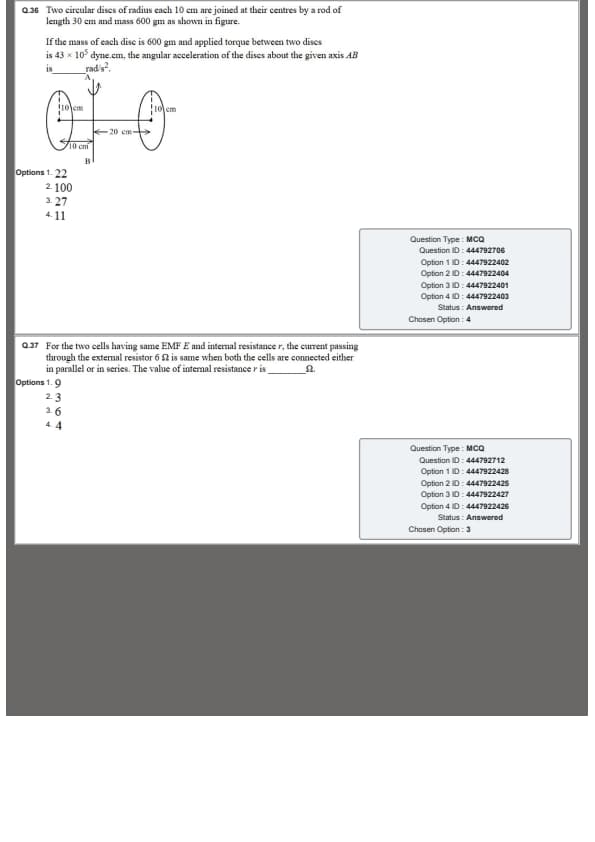
Two circular discs of radius \(10\) cm each are joined at their centres by a rod, as shown in the figure.
The length of the rod is \(30\) cm and its mass is \(600\) g.
The mass of each disc is also \(600\) g.
If the applied torque between the two discs is \(43\times10^{-7}\) dyne·cm,
then the angular acceleration of the system about the given axis \(AB\) is ____ rad s\(^{-2}\).

For two identical cells each having emf \(E\) and internal resistance \(r\), the current through an external
resistor of \(6\,\Omega\) is the same when the cells are connected in series as well as in parallel.
The value of the internal resistance \(r\) is ____ \(\Omega\).
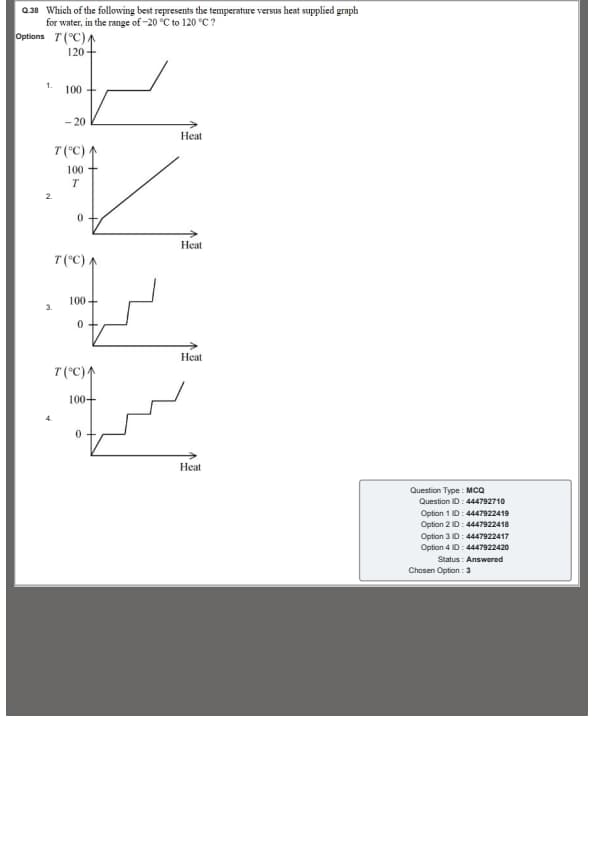
Which of the following best represents the temperature versus heat supplied graph for water,
in the range of \(-20^\circC\) to \(120^\circC\)?

Assuming in forward bias condition there is a voltage drop of \(0.7\) V across a silicon diode,
the current through diode \(D_1\) in the circuit shown is ____ mA.
(Assume all diodes in the given circuit are identical)

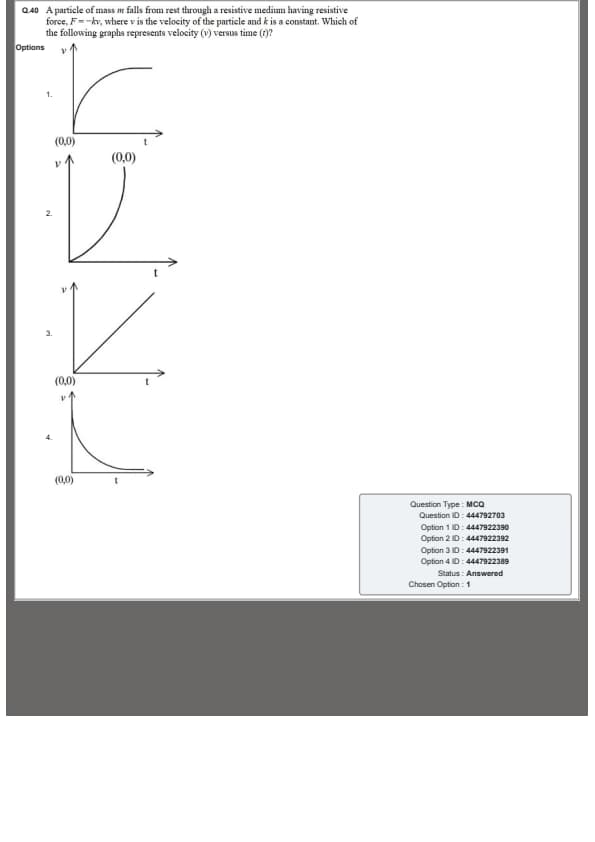
A particle of mass \(m\) falls from rest through a resistive medium having resistive force \(F=-kv\), where \(v\) is the velocity of the particle and \(k\) is a constant.
Which of the following graphs represents velocity \(v\) versus time \(t\)?

In the following \(p–V\) diagram, the equation of state along the curved path is given by \[ (V-2)^2 = 4ap, \]
where \(a\) is a constant. The total work done in the closed path is:

The magnetic field at the centre of a current carrying circular loop of radius \(R\) is \(16\,\muT\).
The magnetic field at a distance \(x=\sqrt{3}R\) on its axis from the centre is \(\,\_\_\_\_\_\ \muT\).
A block of mass \(5\) kg is moving on an inclined plane which makes an angle of \(30^\circ\)
with the horizontal. The coefficient of friction between the block and the inclined plane surface is \(\dfrac{\sqrt{3}}{2}\).
The force to be applied on the block so that the block moves \emph{down the plane without acceleration is ____ N.
(\( g = 10 \, m s^{-2} \))
10 kg of ice at \(-10^\circC\) is added to 100 kg of water to lower its temperature from \(25^\circC\).
Consider no heat exchange to surroundings.
The decrement in the temperature of water is ____ \( ^\circC \).
(Specific heat of ice \(=2100\,J kg^{-1}\!^\circC^{-1}\),
specific heat of water \(=4200\,J kg^{-1}\!^\circC^{-1}\),
latent heat of fusion of ice \(=3.36\times10^5\,J kg^{-1}\))
Two point charges of \(1\,nC\) and \(2\,nC\) are placed at two corners of an equilateral
triangle of side \(3\) cm. The work done in bringing a charge of \(3\,nC\) from infinity to the third
corner of the triangle is ____ \(\muJ\). \[ \left(\frac{1}{4\pi\varepsilon_0}=9\times10^9\,N m^2C^{-2}\right) \]
A convex lens of refractive index \(1.5\) and focal length \(f=18\) cm is immersed in water.
The difference in focal lengths of the given lens when it is in water and in air is \( \alpha \times f \).
Find the value of \( \alpha \).
(Given: refractive index of water \(=\dfrac{4}{3}\))
A solid sphere of radius \(10\) cm is rotating about an axis which is at a distance \(15\) cm from its centre. The radius of gyration about this axis is \( \sqrt{n} \) cm.
Find the value of \( n \).
The displacement of a particle executing simple harmonic motion with time period \(T\) is expressed as \[ x(t)=A\sin\omega t, \]
where \(A\) is the amplitude of oscillation.
If the maximum value of the potential energy of the oscillator is found at \[ t=\frac{T}{2\beta}, \]
then the value of \(\beta\) is ____.
The equivalent resistance between the points \(A\) and \(B\) in the given circuit is \[ \frac{x}{5}\,\Omega. \]
Find the value of \(x\).
The ratio of de Broglie wavelength of a deuteron with kinetic energy \(E\) to that of an alpha particle
with kinetic energy \(2E\) is \(n:1\).
(Assume mass of proton \(=\) mass of neutron.)
Find the value of \(n\).
Given below are two statements:
Statement I:
Griess–Ilosvay test is used for the detection of nitrite ion, which involves the use of sulphanilic acid and \(\alpha\)-naphthylamine reagent.
Statement II:
In the above test, sulphanilic acid is diazotized by the acidified nitrite ion, which on further coupling with \(\alpha\)-naphthylamine forms an azo-dye.
In the light of the above statements, choose the correct answer from the options given below.
The correct order of stability for the following carbanions is: \[ CH_2=CH^- ,\quad CH_3-CH_2^- ,\quad CH\equivC^- \]
Given below are two statements:
Statement I:
The number of pairs, from the following, in which \emph{both the ions are coloured are \([Sc^{3+},Ti^{3+}]\), \([Mn^{2+},Cr^{2+}]\), \([Cu^{2+},Zn^{2+}]\) and \([Ni^{2+},Ti^{4+}]\).
Statement II: \(Ti^{4+}\) is the strongest reducing agent among \(Th^{4+}, Ce^{4+}, Gd^{3+}\) and \(Eu^{2+}\).
In the light of the above statements, choose the correct answer.
Given below are two statements for the following reaction sequence:

Statement I:
Compound \(Z\) gives a yellow precipitate with NaOI.
Statement II:
Compound \(Q\) has two different types of hydrogen atoms (aromatic : aliphatic) in the ratio \(1:3\).
Choose the correct answer.
Given below are two statements:
Statement I:
The number of species among \(\mathrm{BF_4^-}\), \(\mathrm{SiF_4}\), \(\mathrm{XeF_4}\) and \(\mathrm{SF_4}\),
that have unequal E–F bond lengths is three. Here, E is the central atom.
Statement II:
Among \(\mathrm{O_2^-}\), \(\mathrm{O_2^{2-}}\), \(\mathrm{F_2}\) and \(\mathrm{O_2^+}\), \(\mathrm{O_2^+}\) has the highest bond order.
In the light of the above statements, choose the correct answer.
\(20.0\,dm^3\) of an ideal gas \(X\) at \(600\) K and \(0.5\) MPa undergoes isothermal reversible expansion
until the pressure of the gas becomes \(0.2\) MPa.
Which of the following option is correct?
(Given: \(\log 2 = 0.3010\), \(\log 5 = 0.6989\))
Consider a weak base \(B\) of \(pK_b = 5.699\). \(x\) mL of \(0.02\) M HCl and \(y\) mL of \(0.02\) M weak base \(B\) are mixed to make \(100\) mL of a buffer of pH \(=9\) at \(25^\circC\).
The values of \(x\) and \(y\) respectively are:
[Given: \(\log 2 = 0.3010,\ \log 3 = 0.4771,\ \log 5 = 0.699]
Regarding the hydrides of group 15 elements \(EH_3\) \((E = N, P, As, Sb)\),
select the correct statement(s):
[A.] The stability of hydrides decreases down the group.
[B.] The basicity of hydrides decreases down the group.
[C.] The reducing character increases down the group.
[D.] The boiling point increases down the group.
Choose the correct answer:
Method used for separation of mixture of products (B and C) obtained in the following reaction is:

In period 4 of the periodic table, the elements with highest and lowest atomic radii respectively are:
At temperature \(T\) K, \(2\) moles of liquid \(A\) and \(3\) moles of liquid \(B\) are mixed.
The vapour pressure of the ideal solution formed is \(320\) mm Hg.
At this stage, one mole of \(A\) and one mole of \(B\) are added to the solution.
The vapour pressure is now measured as \(328.6\) mm Hg.
The vapour pressures (in mm Hg) of pure \(A\) and pure \(B\) respectively are:
Consider the reaction: \[ Ph–CH=CH_2 \xrightarrow[peroxide]{HBr} Product \]
Which of the following statements are correct?
[A.] The reaction proceeds through a more stable radical intermediate.
[B.] The role of peroxide is to generate \(\mathrm{H^\bullet}\) radical.
[C.] During this reaction, benzene is formed as a byproduct.
[D.] \(1\)-Bromo-\(2\)-phenylethane is formed as a minor product.
[E.] The same reaction in absence of peroxide proceeds via a carbocation intermediate.
Choose the correct answer.
The wave numbers of three spectral lines of hydrogen atom are considered.
Identify the set of spectral lines belonging to the \emph{Balmer series}.
(\(R\) = Rydberg constant)
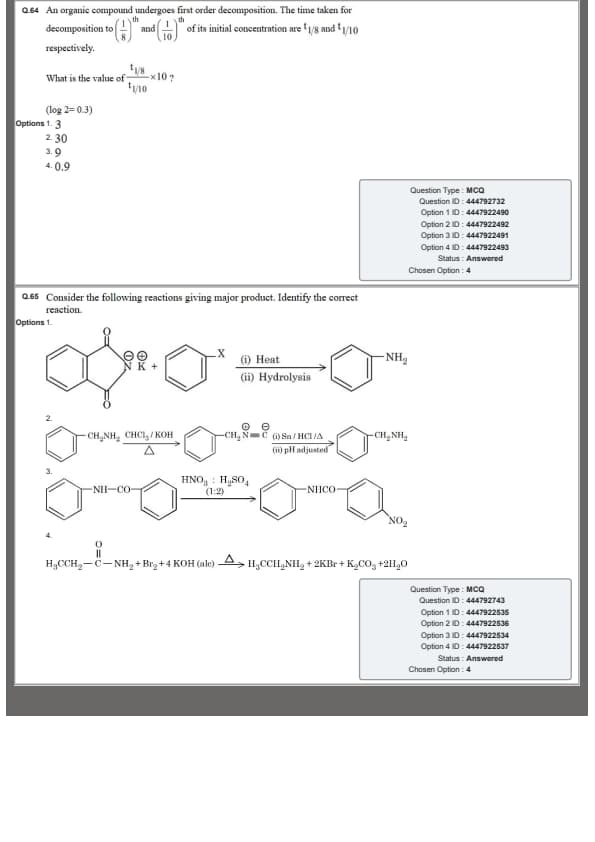
An organic compound undergoes first order decomposition.
The time taken for decomposition to \(\dfrac{1}{8}\) and \(\dfrac{1}{10}\) of its initial concentration
are \(t_{1/8}\) and \(t_{1/10}\) respectively.
Find the value of \[ \frac{t_{1/8}}{t_{1/10}}\times10 \]
(Given: \(\log 2 = 0.3\))
Consider the following reactions giving major product. Identify the correct reaction.

Consider the following reaction sequence:

Given:
Compound (x) has percentage composition \(76.6%\ C\), \(6.38%\ H\) and vapour density \(=47\).
Compound (y) develops a characteristic colour with neutral \(\mathrm{FeCl_3}\) solution.
Identify the \emph{INCORRECT} statement.
The figures below show:

Which of the following points in Figure 2 most accurately represents the nodal surface shown in Figure 1?
Given below are the four isomeric compounds \(P, Q, R, S\):

\(P\): Aromatic compound containing an \(-\mathrm{OH}\) group
\(Q\): Aromatic compound containing an \(-\mathrm{CHO}\) group (aldehyde)
\(R\): Aromatic compound containing a ketone group
\(S\): Aromatic compound containing a ketone group
Identify the correct statements from below:
[A.] \(Q, R\) and \(S\) will give precipitate with \(2,4\)-DNP.
[B.] \(P\) and \(Q\) will give positive Baeyer’s test.
[C.] \(Q\) and \(R\) will give sooty flame.
[D.] \(R\) and \(S\) will give yellow precipitate with \(I_2/\mathrm{NaOH}\).
[E.] \(Q\) alone will deposit silver with Tollens’ reagent.
Choose the correct option.
The correct statement among the following is:
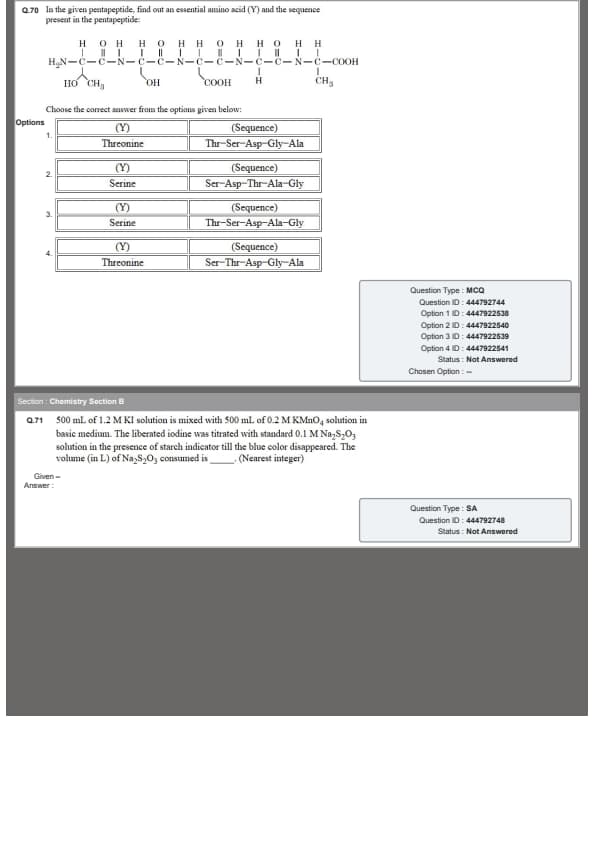
In the given pentapeptide, find out an essential amino acid (\(Y\)) and the sequence present in the pentapeptide.

Choose the correct answer from the options given below:
500 mL of \(1.2\) M KI solution is mixed with 500 mL of \(0.2\) M \( \mathrm{KMnO_4} \) solution in basic medium.
The liberated iodine is titrated with standard \(0.1\) M \( \mathrm{Na_2S_2O_3} \) solution in the presence of starch indicator
till the blue colour disappears.
The volume (in L) of \( \mathrm{Na_2S_2O_3} \) consumed is ____ (nearest integer).
Consider the dissociation equilibrium of the following weak acid: \[ \mathrm{HA \rightleftharpoons H^+(aq) + A^-(aq)} \]
If the \(pK_a\) of the acid is \(4\), then the pH of a \(10\ mM\) HA solution is ____ (Nearest integer).
(Given: The degree of dissociation can be neglected with respect to unity)
\(X\) is the number of geometrical isomers exhibited by \([\mathrm{Pt(NH_3)(H_2O)BrCl}]\).
\(Y\) is the number of optically inactive isomer(s) exhibited by \([\mathrm{CrCl_2(ox)_2}]^{3-}\).
\(Z\) is the number of geometrical isomers exhibited by \([\mathrm{Co(NH_3)_3(NO_2)_3}]\).
Find the value of \(X + Y + Z\).
\(0.53\ g\) of an organic compound \(X\) when heated with excess concentrated nitric acid and then with silver nitrate
gave \(0.75\ g\) of silver bromide precipitate. \(1.0\ g\) of \(X\) gave \(1.32\ g\) of \(\mathrm{CO_2}\) on combustion.
Find the percentage of hydrogen in compound \(X\). (Nearest integer)
[Given: Atomic masses (g mol\(^{-1}\)): H = 1, C = 12, Br = 80, Ag = 108, O = 16]
Consider the following redox reaction taking place in acidic medium: \[ \mathrm{BH_4^- (aq) + ClO_3^- (aq) \rightarrow H_2BO_3^- (aq) + Cl^- (aq)} \]
If the Nernst equation for the above balanced reaction is \[ E_{cell} = E^\circ_{cell} - \frac{RT}{nF}\ln Q, \]
then the value of \(n\) is ____ (Nearest integer).








































Comments