JEE Main 2026 Jan 28 Shift 2 question paper is available here with answer key and solutions PDF. NTA conducted the second shift of the day on Jan 28, 2026, from 3 PM to 6 PM.
JEE Main 2026 28th Jan Shift 2 Question Paper with Solution PDF
- Download JEE Main 2026 Question Papers Pdf for all shfits
- Download JEE Main 2026 Session 1 Scorecard
- Predict your rank using collegedunia’s JEE Main Rank Predictor
Based on initial analysis, JEE Main Jan 28th Shift 2 was Moderate to Difficult. Students can access the JEE Main Jan 28 Shift 2 official questions with answer keys here.
Students can check the detailed paper analysis for JEE Main Jan 28th Shift 2 here.

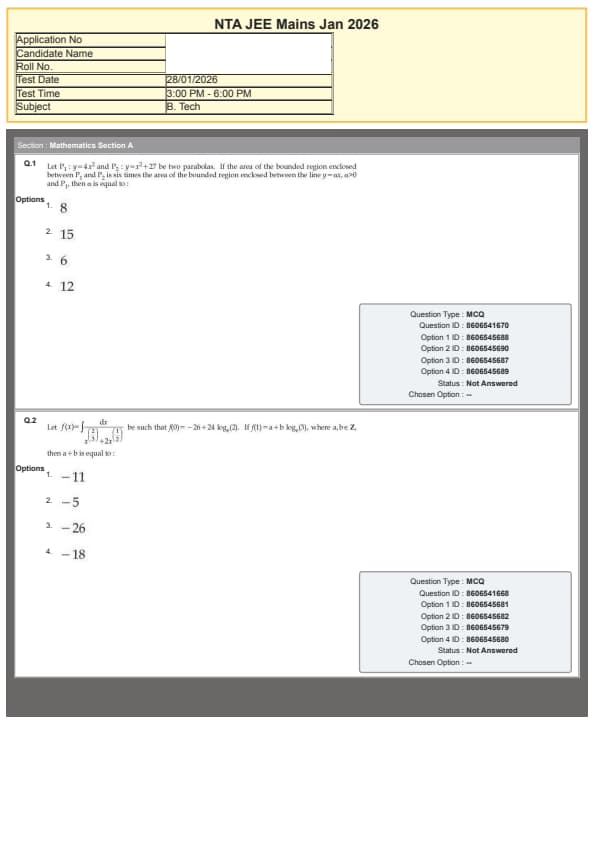
Let \(P_1 : y = 4x^2\) and \(P_2 : y = x^2 + 27\) be two parabolas.
If the area of the bounded region enclosed between \(P_1\) and \(P_2\) is six times the area of the bounded region enclosed between the line \(y = x\), the line \(x = 0\), and \(P_1\), then the required value is:
Let \[ f(x)=\int \frac{dx}{2\left(\frac{3}{2}\right)^x+2x\left(\frac12\right)^x} \]
such that \(f(0)=-26+24\log_e(2)\).
If \(f(1)=a+b\log_e(3)\), where \(a,b\in\mathbb{Z}\), then \(a+b\) is equal to:
Given below are two statements:
Statement I: \[ 25^{13}+20^{13}+31^{13} is divisible by 7 \]
Statement II:
The integral part of \(\left(7+4\sqrt3\right)^{25}\) is an odd number.
In the light of the above statements, choose the correct answer:
Let the ellipse \[ E:\ \frac{x^2}{144}+\frac{y^2}{169}=1 \]
and the hyperbola \[ H:\ \frac{x^2}{16}-\frac{y^2}{2^2}=1 \]
have the same foci.
If \(e\) and \(L\) respectively denote the eccentricity and the length of the latus rectum of \(H\),
then the value of \(24(e+L)\) is:
Let the arithmetic mean of \(\frac{1}{a}\) and \(\frac{1}{b}\) be \(\frac{5}{16}\), where \(a>2\).
If \(a,4,b\) are in A.P., then the equation \[ ax^2-ax+2(a-2b)=0 \]
has:
The sum of the coefficients of \(x^{499}\) and \(x^{500}\) in \[ (1+x)^{1000}+x(1+x)^{999}+x^2(1+x)^{998}+\cdots+x^{1000} \]
is:
Let \(y=y(x)\) be the solution of the differential equation \[ x\frac{dy}{dx}=y-x^2\cot x,\quad x\in(0,\pi) \]
If \(y\!\left(\frac{\pi}{2}\right)=\frac{\pi^2}{2}\), then \[ 6y\!\left(\frac{\pi}{6}\right)-8y\!\left(\frac{\pi}{4}\right) \]
is equal to:
An ellipse has its centre at \((1,-2)\), one focus at \((3,-2)\) and one vertex at \((5,-2)\).
Then the length of its latus rectum is:
Given below are two statements:
Statement I:
The function \(f:\mathbb{R}\to\mathbb{R}\) defined by \[ f(x)=\frac{x}{1+|x|} \]
is one-one.
Statement II:
The function \(f:\mathbb{R}\to\mathbb{R}\) defined by \[ f(x)=\frac{x^2+4x-30}{x^2-8x+18} \]
is many-one.
In the light of the above statements, choose the correct answer.
Let \[ f(x)=\lim_{\theta\to 0} \frac{\cos(\pi x-\theta)\,\sin(x-1)} {1+x^{\theta/2}(x-1)},\qquad x\in\mathbb{R}. \]
Consider the following statements:
[(I)] \(f(x)\) is continuous at \(x=1\).
[(II)] \(f(x)\) is continuous at \(x=-1\).
Then:
Let \(A\) be the focus of the parabola \(y^2=8x\).
Let the line \(y=mx+c\) intersect the parabola at two distinct points \(B\) and \(C\).
If the centroid of triangle \(ABC\) is \(\left(\frac{7}{3},\frac{4}{3}\right)\),
then \((BC)^2\) is equal to:
Let \([\,\cdot\,]\) denote the greatest integer function. Then \[ \int_{-\pi/2}^{\pi/2} \frac{12(3+[x])}{3+[\sin x]+[\cos x]}\,dx \]
is equal to:
Let \(P\) be a point in the plane of the vectors \[ \vec{AB}=3\hat{i}+\hat{j}-\hat{k} \quadand\quad \vec{AC}=\hat{i}-\hat{j}+3\hat{k} \]
such that \(P\) is equidistant from the lines \(AB\) and \(AC\).
If \(|\vec{AP}|=\frac{\sqrt5}{2}\), then the area of triangle \(ABP\) is:
Let \(Q(a,b,c)\) be the image of the point \(P(3,2,1)\) in the line \[ \frac{x-1}{1}=\frac{y-2}{2}=\frac{z-1}{1}. \]
The distance of \(Q\) from the line \[ \frac{x-9}{3}=\frac{y-9}{2}=\frac{z-5}{-2} \]
is:
The probability distribution of a random variable \(X\) is given below:
\[ \begin{array}{c|cccccccc} x & 4k & \frac{30k}{7} & \frac{32k}{7} & \frac{34k}{7} & \frac{36k}{7} & \frac{38k}{7} & \frac{40k}{7} & 6k
\hline P(X) & \frac{2}{15} & \frac{1}{15} & \frac{2}{15} & \frac{1}{5} & \frac{1}{15} & \frac{2}{15} & \frac{1}{5} & \frac{1}{15} \end{array} \]
If \(E(X)=\dfrac{263}{15}\), then \(P(X<20)\) is equal to:
Considering the principal values of inverse trigonometric functions, the value of \[ \tan\!\left(2\sin^{-1}\!\frac{2}{\sqrt{13}}-2\cos^{-1}\!\frac{3}{\sqrt{10}}\right) \]
is equal to:
Let the circle \(x^2+y^2=4\) intersect the \(x\)-axis at points \(A(a,0)\) and \(B(b,0)\).
Let \(P(2\cos\alpha,2\sin\alpha)\), \(0<\alpha<\frac{\pi}{2}\), and \(Q(2\cos\beta,2\sin\beta)\) be two points on the circle such that \((\alpha-\beta)=\frac{\pi}{2}\).
Then the point of intersection of lines \(AQ\) and \(BP\) lies on:
Let \[ A=\{z\in\mathbb{C}:|z-2|\le 4\} \quadand\quad B=\{z\in\mathbb{C}:|z-2|+|z+2|=5\}. \]
Then the maximum value of \(|z_1-z_2|\), where \(z_1\in A\) and \(z_2\in B\), is:
Evaluate: \[ \frac{6}{3^{26}}+\frac{10\cdot1}{3^{25}}+\frac{10\cdot2}{3^{24}}+\frac{10\cdot2^{2}}{3^{23}}+\cdots+\frac{10\cdot2^{24}}{3}. \]
The sum of all the elements in the range of \[ f(x)=\operatorname{sgn}(\sin x)+\operatorname{sgn}(\cos x) +\operatorname{sgn}(\tan x)+\operatorname{sgn}(\cot x), \]
where \[ x\neq \frac{n\pi}{2},\ n\in\mathbb{Z}, \qquad \operatorname{sgn}(t)= \begin{cases} 1,& t>0
-1,& t<0 \end{cases} \]
is:
If \[ \sum_{r=1}^{25}\left(\frac{r}{r^4+r^2+1}\right)=\frac{p}{q}, \]
where \(p\) and \(q\) are positive integers such that \(\gcd(p,q)=1\),
then \(p+q\) is equal to ___.
Three persons enter a lift at the ground floor. The lift will go up to the 10th floor.
The number of ways in which the three persons can exit the lift at three different floors,
if the lift does not stop at the 1st, 2nd and 3rd floors, is equal to ___.
If the distance of the point \(P(4\alpha,\alpha,\beta)\), \(\beta<0\), from the line \[ \vec r = 4\hat i-\hat k+\mu(2\hat i+3\hat k),\ \mu\in\mathbb{R}, \]
along a line with direction ratios \(3,-1,0\) is \(\dfrac{13}{\sqrt{10}}\),
then \(\alpha^2+\beta^2\) is equal to ___.
Let \(f\) be a differentiable function satisfying \[ f(x)=1-2x+\int_0^x (t-x)f(t)\,dt,\quad x\in\mathbb{R}, \]
and let \[ g(x)=\int_0^x \{f(t)+2\}^5(t-4)^6(t+12)^7\,dt. \]
If \(p\) and \(q\) are respectively the points of local minima and local maxima of \(g\),
then the value of \(|p+q|\) is ___.
Let \[ A=\begin{pmatrix}3 & -4
1 & -1\end{pmatrix} \]
and \(B\) be two matrices such that \[ A^{100}-100B+I=0. \]
Then the sum of all the elements of \(B^{100}\) is ___.
For a transparent prism, if the angle of minimum deviation is equal to its refracting angle,
the refractive index \(n\) of the prism satisfies:
Which one of the following is \emph{not} a measurable quantity?
Identify the correct statements:
[A.] Electrostatic field lines form closed loops.
[B.] The electric field lines point radially outward when charge is greater than zero.
[C.] The Gauss’s Law is valid only for inverse-square force.
[D.] The work done in moving a charged particle in a static electric field around a closed path is zero.
[E.] The motion of a particle under Coulomb’s force must take place in a plane.
Choose the correct answer from the options given below:
The time period of a simple harmonic oscillator is \[ T=2\pi\sqrt{\frac{m}{k}}. \]
Measured value of mass \(m\) has an accuracy of \(10%\) and time for 50 oscillations of
the spring is found to be \(60\,s\) using a watch of 2 s resolution.
Percentage error in determination of spring constant \(k\) is:
A Wheatstone bridge is initially at room temperature and all arms of the bridge have same value of resistances \[ (R_1=R_2=R_3=R_4). \]
When \(R_3\) resistance is heated, its resistance value increases by \(10%\).
The potential difference \((V_a-V_b)\) after \(R_3\) is heated is ___ V.

The speed of a longitudinal wave in a metallic bar is \(400\,m/s\).
If the density and Young’s modulus of the bar material increase by \(0.5%\) and \(1%\) respectively,
then the speed of the wave is changed approximately to ___ m/s.
Two p-n junction diodes \(D_1\) and \(D_2\) are connected as shown in the figure. \(A\) and \(B\) are input signals and \(C\) is the output.
The given circuit will function as a ___.

The mean free path of a molecule of diameter \(5\times10^{-10}\,m\)
at temperature \(41^\circC\) and pressure \(1.38\times10^5\,Pa\) is given as ___ m.
(Given \(k_B=1.38\times10^{-23}\,J/K\))
A nucleus has mass number \(\alpha\) and radius \(R_\alpha\).
Another nucleus has mass number \(\beta\) and radius \(R_\beta\).
If \(\beta=8\alpha\), then \(R_\alpha/R_\beta\) is:
A biconvex lens is formed by using two plano-convex lenses as shown in the figure.
The refractive index and radius of curvature of surfaces are also mentioned.
When an object is placed on the left side of the lens at a distance of \(30\,cm\),
the magnification of the image will be:

A small block of mass \(m\) slides down from the top of a frictionless inclined surface,
while the inclined plane is moving towards left with constant acceleration \(a_0\).
The angle between the inclined plane and ground is \(\theta\) and its base length is \(L\).
Assuming that initially the small block is at the top of the inclined plane,
the time it takes to reach the lowest point of the inclined plane is ___.

In an experiment, a set of readings are obtained as follows: \[ 1.24~mm,\ 1.25~mm,\ 1.23~mm,\ 1.21~mm. \]
The expected least count of the instrument used in recording these readings is ___ mm.
Number of photons of equal energy emitted per second by a \(6\,mW\) laser source
operating at wavelength \(663\,nm\) is ___.
(Given: \(h=6.63\times10^{-34}\,J·s\) and \(c=3\times10^8\,m/s\))
A particle starts moving from time \(t=0\) and its coordinate is given as \[ x(t)=4t^3-3t. \]
Consider the following statements:
[A.] The particle returns to its original position (origin) \(0.866\) units later.
[B.] The particle is \(1\) unit away from origin at its turning point.
[C.] Acceleration of the particle is non-negative.
[D.] The particle is \(0.5\) units away from origin at its turning point.
[E.] The particle never turns back as acceleration is non-negative.
Choose the correct answer from the options given below:
Match List–I with List–II.
\begin{tabular{l l
List–I & List–II
A. Coefficient of viscosity & I. \([ML^{-1}T^{-2}]\)
B. Surface tension & II. \([ML^{-2}T^{-2}]\)
C. Pressure & III. \([ML^{0}T^{-2}]\)
D. Surface energy & IV. \([ML^{-1}T^{-1}]\)
\end{tabular
Choose the correct answer from the options given below:
A plane electromagnetic wave is moving in free space with velocity \[ c=3\times10^{8}\ m/s \]
and its electric field is given as \[ \vec E = 54\sin(kz-\omega t)\,\hat{j}\ V/m, \]
where \(\hat{j}\) is the unit vector along the \(y\)-axis.
The magnetic field \(\vec B\) of the wave is:
A long cylindrical conductor with large cross section carries an electric current
distributed uniformly over its cross-section. Magnetic field due to this current is:
[A.] maximum at either end of the conductor
[B.] maximum at the axis of the conductor and minimum at the midpoint
[C.] minimum at the surface of the conductor
[D.] minimum at the axis of the conductor
[E.] same at all points in the cross-section of the conductor
Choose the correct answer from the options given below:
When the position vector \[ \vec r = x\hat{i}+y\hat{j}+z\hat{k} \]
changes sign as \(\vec r \rightarrow -\vec r\),
which one of the following vectors will \emph{not flip under sign change?
Identify the correct statements:
[A.] Effective capacitance of a series combination of capacitors is always smaller than the smallest capacitance of the combination.
[B.] When a dielectric medium is placed between charged plates of a capacitor, displacement of charges cannot occur due to insulation property of dielectric.
[C.] Increasing area of a capacitor plate or decreasing thickness of dielectric is an alternate method to increase the capacitance.
[D.] For a point charge, concentric spherical shells centered at the location of the charge are equipotential surfaces.
Choose the correct answer from the options given below:
As shown in the figure, a spring is kept in a stretched position with some extension by
holding the masses \(1\,kg\) and \(0.2\,kg\) with a separation more than spring natural length
and then released. Assuming the horizontal surface to be frictionless, the angular frequency
(in SI unit) of the system is ___.
(Given \(k=150\,N/m\))

A flywheel having mass \(3\,kg\) and radius \(5\,m\) is free to rotate about a horizontal axis.
A string having negligible mass is wound around the wheel and the loose end of the string is connected
to a \(3\,kg\) mass. The mass is kept initially and released. Kinetic energy of the flywheel when the mass
descends by \(3\,m\) is ___ J. \((g=10\,m s^{-2})\)
% Given
Given: \[ M = 3\,kg, \quad R = 5\,m, \quad m = 3\,kg, \quad h = 3\,m, \quad g = 10\,m s^{-2} \]
Two tuning forks \(A\) and \(B\) are sounded together giving rise to 8 beats in 2 s.
When fork \(A\) is loaded with wax, the beat frequency is reduced to 4 beats in 2 s.
If the original frequency of tuning fork \(B\) is \(380\ Hz\), find the original frequency of tuning fork \(A\).
% Given
Given: \[ f_B = 380\ Hz \]
A beam of light consisting of wavelengths \(650\,nm\) and \(550\,nm\) illuminates
Young’s double slits with separation \(d=2\,mm\) such that the interference fringes are formed on
a screen placed at a distance \(D=1.2\,m\) from the slits. The least distance from the central maximum,
where the bright fringes due to both wavelengths coincide, is ____ \(\times10^{-5}\,m\).
% Given
Given: \[ \lambda_1=650\,nm,\quad \lambda_2=550\,nm,\quad d=2\times10^{-3}\,m,\quad D=1.2\,m \]
An inductor stores \(16\,J\) of magnetic field energy and dissipates \(32\,W\) of thermal
energy due to its resistance when an alternating current of \(2\,A\) (rms) and frequency \(50\,Hz\) flows through it. The ratio of inductive reactance to resistance is ___. \((\pi=3.14)\)
% Given
Given: \[ U=16\,J,\quad P=32\,W,\quad I=2\,A,\quad f=50\,Hz \]
A thermodynamic system is taken through the cyclic process \(ABC\) as shown in the \(P\!-\!V\)
diagram. The total work done by the system during the cycle \(ABC\) is ___ J.

% Given
Given:
The area enclosed by the cycle in the \(P\!-\!V\) diagram represents the work done.
Consider the elements N, P, O, S, Cl and F. The number of valence electrons present in the
elements with most and least metallic character from the above list is respectively.
The plot of \(\log_{10}K\) vs \(\frac{1}{T}\) gives a straight line.
The intercept and slope respectively are (where \(K\) is equilibrium constant).
The reactions which produce alcohol as the product are :
A student has been given 0.314 g of an organic compound and asked to estimate Sulphur. During the experiment, the student has obtained 0.4813 g of barium sulphate. The percentage of sulphur present in the compound is \hspace{2cm}. (Given Molar mass in g mol\(^{-1}\): S, 32; BaSO\(_4\), 233)
The cyclic cations having the same number of hyperconjugation are:

The correct order of acidic strength of the major products formed in the given reactions is:

Total number of alkali insoluble solid sulphonamides obtained by reaction of given amines with Hinsberg's reagent is:
Amines: Aniline, N-Methylaniline, Methanamine, N,N-Dimethylmethanamine, N-Methyl methanamine, Phenylmethanamine, N-propylaniline, N-phenylaniline, N,N-Dimethylaniline, Allyl amine, Isopropyl amine
Consider the following reactions:

The oxidation states of Cu in Z and Q, respectively are:
The wavelength of photon 'A' is 400 nm. The frequency of photon 'B' is \(10^{16}\,s^{-1}\). The wave number of photon 'C' is \(10^{5}\,cm^{-1}\). The correct order of energy of these photons is:
A student performed analysis of aliphatic organic compound 'X' which on analysis gave C = 61.01%, H = 15.25%, N = 23.74%.
This compound, on treatment with HNO\(_2\)/H\(_2\)O produced another compound 'Y' which did not contain any nitrogen atom. However, the compound 'Y' upon controlled oxidation produced another compound 'Z' that responded to iodoform test.
The structure of 'X' is:
Observe the following equilibrium in a 1 L flask: \[ \ce{A(g) <=> B(g)} \]
At \(T(K)\), the equilibrium concentrations of A and B are 0.5 M and 0.375 M respectively. 0.1 moles of A is added into the flask and heated to \(T(K)\) to establish the equilibrium again. The new equilibrium concentrations (in M) of A and B are respectively:
Given below are two statements:
Statement I: The increasing order of boiling point of hydrogen halides is HCl < HBr < HI < HF.
Statement II: The increasing order of melting point of hydrogen halides is HCl < HBr < HF < HI.
In the light of the above statements, choose the correct answer from the options given below:
Match List - I with List - II according to shape.
List - I
A. XeO\(_3\)
B. XeF\(_2\)
C. XeO\(_2\)F\(_2\)
D. XeOF\(_4\)
List - II
I. BrF\(_5\)
II. NH\(_3\)
III. [I\(_3\)]\(^{-}\)
IV. SF\(_4\)
Choose the correct answer from the options given below:
For the given reaction: \[ \ce{CaCO3 + 2HCl -> CaCl2 + H2O + CO2} \]
If 90 g \(\ce{CaCO3}\) is added to 300 mL of HCl which contains 38.55% HCl by mass and has density 1.13 g mL\(^{-1}\), then which of the following option is correct?
Given molar mass of H, Cl, Ca and O are 1, 35.5, 40 and 16 g mol\(^{-1}\) respectively.
Consider the following statements about manganate and permanganate ions. Identify the correct statements.
A. The geometry of both manganate and permanganate ions is tetrahedral.
B. The oxidation states of Mn in manganate and permanganate are +7 and +6, respectively.
C. Oxidation of Mn(II) salt by peroxodisulphate gives manganate ion as the final product.
D. Manganate ion is paramagnetic and permanganate ion is diamagnetic.
E. Acidified permanganate ion reduces oxalate, nitrite and iodide ions.
The correct increasing order of spin-only magnetic moment values of the complex ions \([\ce{MnBr4}]^{2-}\) (A), \([\ce{Cu(H2O)6}]^{2+}\) (B), \([\ce{Ni(CN)4}]^{2-}\) (C) and \([\ce{Ni(H2O)6}]^{2+}\) (D) is:
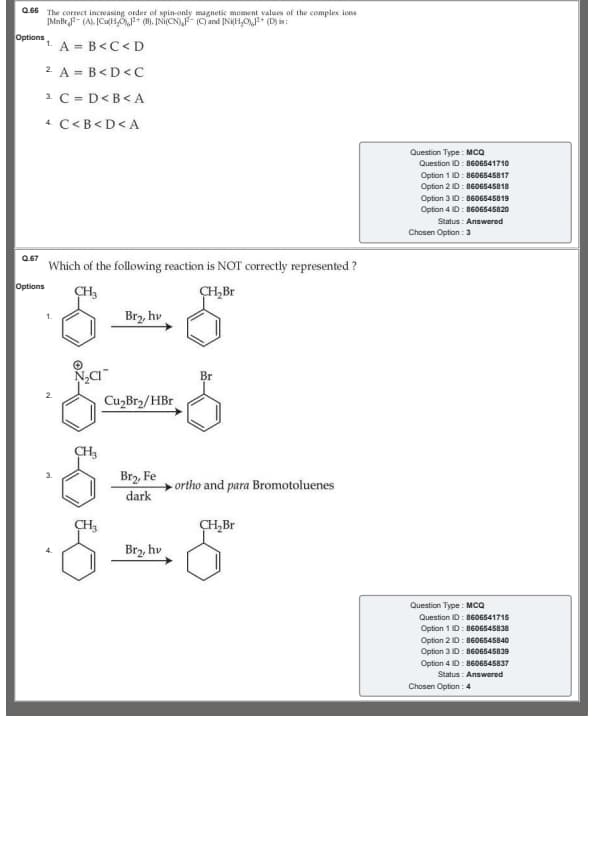
Which of the following reaction is NOT correctly represented?

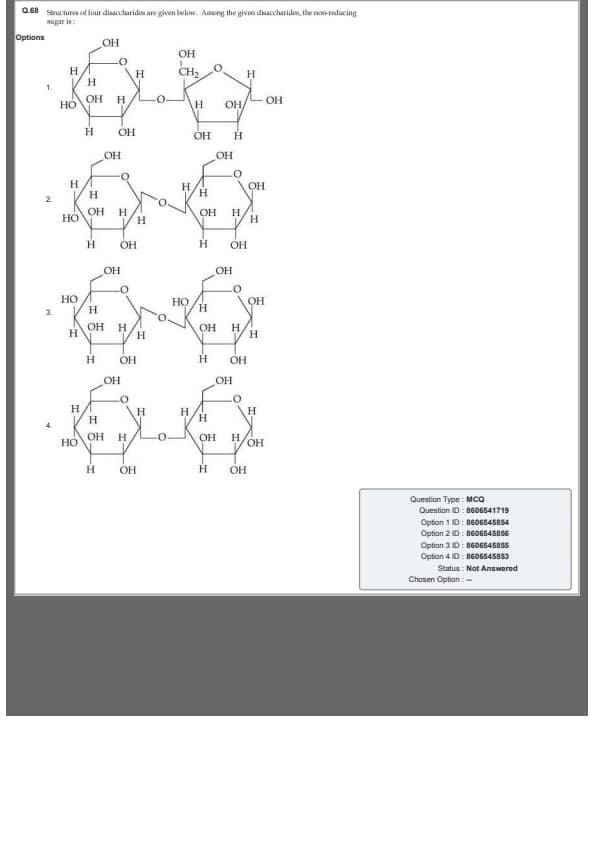
Structures of four disaccharides are given below. Among the given disaccharides, the non-reducing sugar is:

Identify the correct statements:
The presence of –NO\(_2\) group in benzene ring
A. activates the ring towards electrophilic substitutions.
B. deactivates the ring towards electrophilic substitutions.
C. activates the ring towards nucleophilic substitutions.
D. deactivates the ring towards nucleophilic substitutions.
Choose the correct answer from the options given below:
Consider the following aqueous solutions.
I. 2.2 g Glucose in 125 mL of solution.
II. 1.9 g Calcium chloride in 250 mL of solution.
III. 9.0 g Urea in 500 mL of solution.
IV. 20.5 g Aluminium sulphate in 750 mL of solution.
The correct increasing order of boiling point of these solutions will be:
[Given: Molar mass in g mol\(^{-1}\): H = 1, C = 12, N = 14, O = 16, Cl = 35.5, Ca = 40, Al = 27 and S = 32]
For strong electrolyte \(\Lambda_m\) increases slowly with dilution and can be represented by the equation: \[ \Lambda_m = \Lambda_m^0 - A\sqrt{c} \]
Molar conductivity values of the solutions of strong electrolyte AB at 18\(^\circ\)C are given below:
\[ \begin{array}{|c|c|c|c|c|} \hline c \,[mol L^{-1}] & 0.04 & 0.09 & 0.16 & 0.25
\hline \Lambda_m \,[S cm^2 mol^{-1}] & 96.1 & 95.7 & 95.3 & 94.9
\hline \end{array} \]
The value of constant \(A\) based on the above data [in \(S cm^2 mol^{-1}/(mol L^{-1})^{1/2}\)] unit is:
Consider the following two first-order reactions:
A \(\to\) B (first reaction)
C \(\to\) D (second reaction)
The rate constant for first reaction at 500 K is double of the same at 300 K. At 500 K, 50% of the reaction becomes complete in 2 hours. The activation energy of the second reaction is half of that of first reaction. If the rate constant at 500 K of the second reaction becomes double of the rate constant of first reaction at the same temperature; then rate constant for the second reaction at 300 K is ______ \(\times 10^{-3}\,hour^{-1}\) (nearest integer).
The number of isoelectronic species among \(\ce{S^{2-}}, \ce{C^{4-}}, \ce{Mn^{2+}}, \ce{Co^{3+}}\) and \(\ce{Fe^{3+}}\) is ‘n’. If ‘n’ moles of AgCl is formed during the reaction of complex with formula \(\ce{CoCl2(en)2NH3}\) with excess of AgNO\(_3\) solution, then the number of electrons present in the \(t_{2g}\) orbital of the complex is ________.
A volume of \(x\) mL of 5 M NaHCO\(_3\) solution was mixed with 10 mL of 2 M H\(_2\)CO\(_3\) solution to make an electrolytic buffer. If the same buffer was used in the following electrochemical cell to record a cell potential of 253.5 mV, then the value of \(x =\) ______ mL (nearest integer).
\[ \ce{Sn(s) | Sn(OH)2(s) | HSnO2^- (0.05 M) | OH^- (0.05 M) || Bi2O3(s) | Bi(s)} \]
Given: \[ E^\circ(\ce{HSnO2^- / Sn(OH)2}) = -0.90 \,V, \quad E^\circ(\ce{Bi2O3 / Bi}) = -0.44 \,V \] \[ pK_a(\ce{H2CO3}) = 6.11, \quad \frac{2.303RT}{F} = 0.059 \,V, \quad Antilog(1.29) = 19.5 \]
Two positively charged particles \(m_1\) and \(m_2\) have been accelerated across the same potential difference of 200 keV.
Given mass of \(m_1 = 1 \,amu\) and \(m_2 = 4 \,amu\).
The de Broglie wavelength of \(m_1\) will be \(x\) times that of \(m_2\). The value of \(x\) is ______ (nearest integer).












































Comments