NEET 2023 Question paper with answer key pdf F5 is available for download. NEET 2023 F5 question paper has been conducted by the NTA on May 7, 2023, in pen-paper mode. NEET 2023 question paper code F5 consists of 200 MCQs- 180 to be attempted in 200 minutes. Each of the 4 subjects (Zoology, Botany, Chemistry, Physics) in NEET F5 question paper 2023 have 50 MCQs (45 to be attempted). Check NEET Cutoff
You can download NEET 2023 question paper with answer key with solutions PDF for F5 using the links given below.
Latest: NEET Answer Key Available- Download Question Paper and PDFs
NEET 2023 Question Paper with Answer Key PDF F5 in English
| NEET 2023 F5 Question Paper with Answer Key | Check Solution |

Question 1:
A full wave rectifier circuit consists of two p-n junction diodes, a centre-tapped transformer, capacitor and a load resistance.
Which of these components remove the ac ripple from the rectified output?
View Solution
A capacitor acts as a filter in a rectifier circuit. It charges during the peaks of the output voltage and discharges during the troughs, thereby smoothing the pulsating DC output.
Quick Tip: Capacitors smooth out AC ripples in rectifiers by charging and discharging.
In a plane electromagnetic wave travelling in free space, the electric field component oscillates sinusoidally at a frequency of \(2.0 \times 10^{10}\) Hz and amplitude \(48~Vm^{-1}\). Then the amplitude of oscillating magnetic field is :
(Speed of light in free space \(= 3 \times 10^8~m/s\))
View Solution
A metal wire has mass \((0.4 \pm 0.002)~g\), radius \((0.3 \pm 0.001)~mm\) and length \((5 \pm 0.02)~cm\).
The maximum possible percentage error in the measurement of density will nearly be:
View Solution
In a series LCR circuit, the inductance \(L\) is 10 mH, capacitance \(C\) is 1 \(\mu\)F and resistance \(R\) is 100 \(\Omega\).
The frequency at which resonance occurs is:
View Solution
A football player is moving southward and suddenly turns eastward with the same speed to avoid an opponent.
The force that acts on the player while turning is:
View Solution
The errors in the measurement which arise due to unpredictable fluctuations in temperature and voltage supply are:
View Solution
The work functions of Caesium (Cs), Potassium (K) and Sodium (Na) are 2.14 eV, 2.30 eV and 2.75 eV respectively.
If incident electromagnetic radiation has an incident energy of 2.20 eV, which of these photosensitive surfaces may emit photoelectrons?
View Solution
A bullet is fired from a gun at the speed of 280 m/s in the direction 30° above the horizontal.
The maximum height attained by the bullet is (\(g = 9.8~m/s^2\), \(\sin 30^\circ = 0.5\)):
View Solution
A 12 V, 60 W lamp is connected to the secondary of a step down transformer, whose primary is connected to ac mains of 220 V.
Assuming the transformer to be ideal, what is the current in the primary winding?
View Solution
Resistance of a carbon resistor determined from colour codes is \((22000 \pm 5%)~\Omega\).
The colour of third band must be:
View Solution
In hydrogen spectrum, the shortest wavelength in the Balmer series is \(\lambda\).
The shortest wavelength in the Bracket series is:
View Solution
The equivalent capacitance of the system shown in the following circuit is:
View Solution
The half life of a radioactive substance is 20 minutes. In how much time, the activity of substance drops to \( \left( \frac{1}{16} \right) \) of its initial value?
View Solution
The angular acceleration of a body, moving along the circumference of a circle, is:
View Solution
Let a wire be suspended from the ceiling (rigid support) and stretched by a weight \( W \) attached at its free end. The longitudinal stress at any point of cross-sectional area \( A \) of the wire is:
View Solution
An electric dipole is placed at an angle of \( 30^\circ \) with an electric field of intensity \( 2 \times 10^5~N/C \). It experiences a torque equal to \( 4~Nm \). Calculate the magnitude of charge on the dipole, if the dipole length is 2 cm.
View Solution
The ratio of radius of gyration of a solid sphere of mass \( M \) and radius \( R \) about its own axis to the radius of gyration of the thin hollow sphere of same mass and radius about its axis is:
View Solution
The venturi-meter works on:
View Solution
The magnitude and direction of the current in the following circuit is:
View Solution
The temperature of a gas is –50° C. To what temperature the gas should be heated so that the rms speed is increased by 3 times?
View Solution
Given below are two statements:
Statement I: Photovoltaic devices can convert optical radiation into electricity.
Statement II: Zener diode is designed to operate under reverse bias in breakdown region.
View Solution
For Young’s double slit experiment, two statements are given:
Statement I: If screen is moved away from the plane of slits, angular separation of the fringes remains constant.
Statement II: If the monochromatic source is replaced by another of larger wavelength, angular separation decreases.
View Solution
The ratio of frequencies of fundamental harmonic produced by an open pipe to that of a closed pipe having same length is:
View Solution
Two coherent sources of intensities \( I \) and \( 4I \) are used in a Young’s double slit experiment. The fringe visibility is:
View Solution
Fringe visibility \( V = \frac{I_{max} - I_{min}}{I_{max} + I_{min}} \)
\[ \Rightarrow V = \frac{(\sqrt{I} + \sqrt{4I})^2 - (\sqrt{I} - \sqrt{4I})^2}{(\sqrt{I} + \sqrt{4I})^2 + (\sqrt{I} - \sqrt{4I})^2} = \frac{(1+2)^2 - (1-2)^2}{(1+2)^2 + (1-2)^2} = \frac{9 - 1}{9 + 1} = \frac{8}{10} = 0.8 \]
% Correction
This result is 0.8, not 0.6 — verify official answer key. Quick Tip: Use: \( V = \frac{2\sqrt{I_1 I_2}}{I_1 + I_2} \)
If maximum height of a projectile is increased by 4%, then its range increases approximately by:
View Solution
Height \( H \propto \sin^2\theta \), Range \( R \propto \sin 2\theta \). For small increases, % increase in R ≈ half that of H. Quick Tip: Small percentage increases in height reflect roughly half in range.
Two objects of same mass are moving with speeds 2 m/s and 3 m/s. The ratio of their kinetic energies is:
View Solution
Kinetic energy \( KE = \frac{1}{2}mv^2 \Rightarrow \frac{KE_1}{KE_2} = \frac{(2)^2}{(3)^2} = \frac{4}{9} \) Quick Tip: Kinetic energy is proportional to the square of velocity.
The net magnetic flux through any closed surface is:
View Solution
A Carnot engine has an efficiency of 50% when its source is at a temperature 327\degree C. The temperature of the sink is:
View Solution
The minimum wavelength of X-rays produced by an electron accelerated through a potential difference of \( V \) volts is proportional to:
View Solution
An AC source is connected to a capacitor C. Due to decrease in its operating frequency:
View Solution
A vehicle travels half the distance with speed \( \vartheta \) and the remaining distance with speed \( 2\vartheta \). Its average speed is:
View Solution
The potential energy of a long spring when stretched by 2 cm is \( U \). If the spring is stretched by 8 cm, potential energy stored in it will be:
View Solution
The amount of energy required to form a soap bubble of radius 2 cm from a soap solution is nearly:
(surface tension of soap solution = 0.03 N m\(^{-1}\))
View Solution
Two bodies of mass \( m \) and \( 9m \) are placed at a distance \( R \). The gravitational potential on the line joining the bodies where the gravitational field equals zero, will be
(\( G \) = gravitational constant):
% Solution \textbf{Solution:}
Field is zero closer to smaller mass. Let position be \( x \) from \( m \):
\[ \frac{Gm}{x^2} = \frac{G(9m)}{(R-x)^2} \Rightarrow \frac{1}{x} = \frac{3}{R-x} \Rightarrow x = \frac{R}{4} \] Then potential \( V = -\frac{Gm}{x} - \frac{9Gm}{R - x} = -\frac{4Gm}{R} - \frac{36Gm}{3R} = -\frac{12Gm}{R} \)
View Solution
If \( \oint\limits_{S} \vec{E} \cdot d\vec{S} = 0 \) over a surface, then:
View Solution
The radius of inner most orbit of hydrogen atom is \( 5.3 \times 10^{-11} \, m \). What is the radius of third allowed orbit of hydrogen atom?
% Solution \textbf{Solution:}
Radius of \( n \)th orbit is given by \( r_n = n^2 \cdot r_1 \), where \( r_1 = 0.53 \, \text{\AA} \).
So, \( r_3 = 3^2 \cdot 0.53 = 9 \cdot 0.53 = 4.77 \, \text{\AA} \)
View Solution
A bullet from a gun is fired on a rectangular wooden block with velocity \( u \). When bullet travels 24 cm through the block along its length horizontally, velocity of bullet becomes \( \frac{u}{3} \). Then it further penetrates into the block in the same direction before coming to rest exactly at the other end of the block. The total length of the block is:
View Solution
10 resistors, each of resistance \( R \) are connected in series to a battery of emf \( E \) and negligible internal resistance. Then those are connected in parallel to the same battery, the current is increased \( n \) times. The value of \( n \) is:
View Solution
The net impedance of circuit (as shown in figure) will be:
% Solution \textbf{Solution:}
XL = \( 2\pi fL = 2\pi \cdot 50 \cdot \frac{50}{\pi} = 5000 \) mΩ = 5 Ω
XC = \( \frac{1}{2\pi fC} = \frac{1}{2\pi \cdot 50 \cdot \frac{10^{-3}}{\pi}} = 5 \, \Omega \)
Net reactance = \( X_L - X_C = 0 \), So Z = R = 10 Ω Oops! The calculation shows Z = 10 Ω, but correct answer is \( 10\sqrt{2} \, \Omega \). Let’s revisit: Z = \( \sqrt{(R^2 + (X_L - X_C)^2)} \) = \( \sqrt{(10^2 + 10^2)} = 10\sqrt{2} \)
View Solution
For the following logic circuit, the truth table is:
View Solution
The resistance of platinum wire at \( 0^{\circ}C \) is \( 2\Omega \) and \( 6.8\Omega \) at \( 80^{\circ}C \). The temperature coefficient of resistance of the wire is:
% Solution \textbf{Solution:}
\( R = R_0 (1 + \alpha T) \Rightarrow 6.8 = 2(1 + 80\alpha) \Rightarrow 3.4 = 1 + 80\alpha \Rightarrow \alpha = \frac{2.4}{80} = 0.03 = 3 \times 10^{-2} \)
View Solution
Two thin lenses are of same focal lengths \( f \), but one is convex and the other one is concave. When they are placed in contact with each other, the equivalent focal length of the combination will be:
View Solution
In the figure shown here, what is the equivalent focal length of the combination of lenses (Assume that all layers are thin)?
\( n_1 = 1.5, \, n_2 = 1.6, \, R_1 = R_2 = 20 \, cm \)
View Solution
The \( x \)-\( t \) graph of a particle performing simple harmonic motion is shown in the figure. The acceleration of the particle at \( t = 2 \, s \) is:
% Solution \textbf{Solution:}
From the graph, the motion is SHM. Displacement is given by \( x(t) = A \cos(\omega t) \). From the graph, amplitude \( A = 1 \) and time period \( T = 4 \, \text{s} \), so \( \omega = \frac{2\pi}{T} = \frac{\pi}{2} \). Acceleration is \( a = -\omega^2 x \). At \( t = 2 \, \text{s} \), \( x = \frac{1}{2} \Rightarrow a = -\left( \frac{\pi}{2} \right)^2 \cdot \frac{1}{2} = -\frac{\pi^2}{8} \cdot \frac{1}{2} = -\frac{\pi^2}{16} \)
View Solution
A student standing on a bridge throws a small ball vertically upwards with a velocity of \( 4 \, ms^{-1} \). The ball strikes the water surface after \( 4 \, s \). The height of the bridge above water surface is (Take \( g = 10 \, ms^{-2} \)):
View Solution
A very long conducting wire is bent in a semi-circular shape from A to B. The magnetic field at point P for steady current configuration is:
View Solution
A satellite is orbiting just above the surface of the Earth with period \( T \). If \( d \) is the density of Earth and \( G \) is the gravitational constant, the expression \( \frac{3\pi}{Gd} \) represents:
View Solution
A wire carrying a current \( I \) along the x-axis has length \( L \) and is in a magnetic field \( \vec{B} = 2\hat{i} + 3\hat{j} - 4\hat{k} \). The magnitude of the magnetic force on the wire is:
% Solution \textbf{Solution:}
Magnetic force \( \vec{F} = I \vec{L} \times \vec{B} \). Here, \( \vec{L} = L \hat{i} \). So,
\[ \vec{F} = IL \hat{i} \times (2\hat{i} + 3\hat{j} - 4\hat{k}) = IL (0 \hat{i} + 0 \hat{j} + 3 \hat{k} + 4 \hat{j}) = IL (3\hat{k} + 4\hat{j}) \] Magnitude = \( \sqrt{4^2 + 3^2} = 5 \), but component perpendicular to wire is only from \( \hat{j} \): \( 3IL \)
View Solution
An electric dipole is placed as shown. What is the potential at point \( P \) (in units of \( 10^2 \) V)? \( \epsilon_0 \) is the permittivity of free space, \( K = \frac{1}{4\pi \epsilon_0} \).
View Solution
Calculate the maximum acceleration of a moving car so that a body lying on the floor remains stationary. The coefficient of static friction between the body and floor is 0.15. \( g = 10 \, ms^{-2} \):
% Solution \textbf{Solution:}
Max acceleration \( a = \mu_s g = 0.15 \cdot 10 = 1.5 \, \text{ms}^{-2} \)
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: A reaction can have zero activation energy.
Reason R: The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to threshold value is called activation energy.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
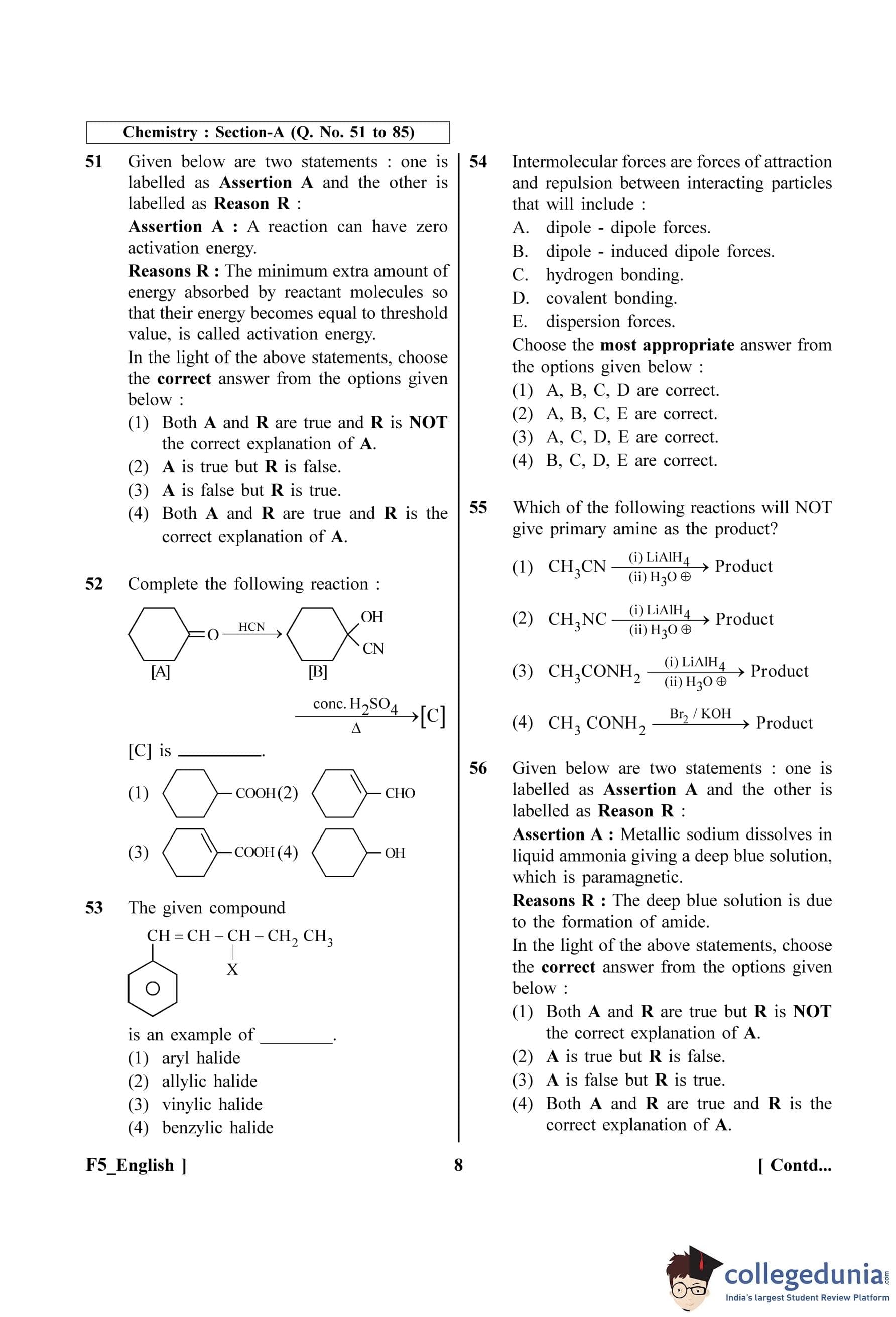
Complete the following reaction:
[C] is:
The given compound:
is an example of:
View Solution
Intermolecular forces are forces of attraction and repulsion between interacting particles. They include:
A. dipole-dipole forces
B. dipole-induced dipole forces
C. hydrogen bonding
D. covalent bonding
E. dispersion forces
Choose the most appropriate answer from the options below:
View Solution
Which of the following reactions will NOT give primary amine as the product?
View Solution
Given below are two statements:
Assertion A: Metallic sodium dissolves in liquid ammonia giving a deep blue solution, which is paramagnetic.
Reason R: The deep blue solution is due to the formation of amide.
Choose the correct answer from the options given below:
View Solution
For a certain reaction, the rate = k[A]\(^2\)[B], when the initial concentration of A is tripled keeping concentration of B constant, the initial rate would
View Solution
Taking stability as the factor, which one of the following represents correct relationship?
View Solution
Amongst the given options, which of the following molecules/ion acts as a Lewis acid?
View Solution
The number of \( \sigma \) bonds, \( \pi \) bonds and lone pair of electrons in pyridine, respectively are:
View Solution
The correct order of energies of molecular orbitals of N\(_2\) molecule, is:
View Solution
Consider the following reaction and identify the product (P).
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: In equation \( \Delta_rG = -nFE_cell \), value of \( \Delta_rG \) depends on \( n \).
Reason R: \( E_cell \) is an intensive property and \( \Delta_rG \) is an extensive property.
View Solution
Homoleptic complex from the following complexes is:
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: Helium is used in diving apparatus.
Reason R: Helium is less soluble in blood than nitrogen.
View Solution
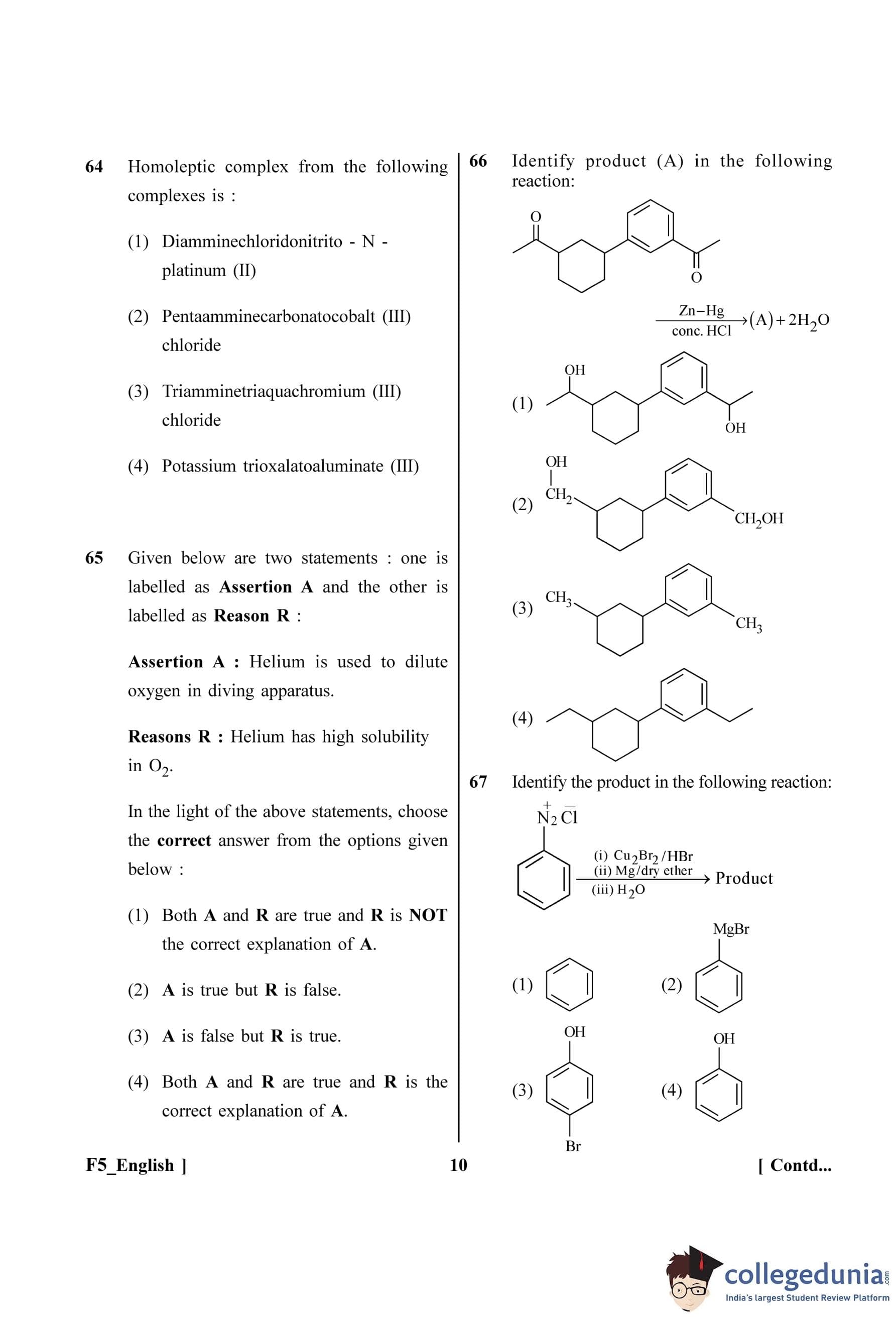
Identify the product A in the following reaction:
Identify the product in the following reaction:
Given below are two statements :
Statement I: A unit formed by the attachment of a base to 1' position of sugar is known as nucleoside
Statement II: When nucleoside is linked to phosphorous acid at 5'-position of sugar moiety, we get nucleotide.
View Solution
Which amongst the following molecules on polymerization produces neoprene?
View Solution
The relation between \(n_m\) (\(n_m\) = the number of permissible values of magnetic quantum number (\(m\))) for a given value of azimuthal quantum number (\(l\)), is
View Solution
The stability of \ce{Cu^{2+ is more than \ce{Cu^{+ salts in aqueous solution due to -
View Solution
Some tranquilizers are listed below. Which one from the following belongs to barbiturates?
View Solution
The element expected to form largest ion to achieve the nearest noble gas configuration is :
View Solution
Which of the following statements are NOT correct?
A. Hydrogen is used to reduce heavy metal oxides to metals.
B. Heavy water is used to study reaction mechanism.
C. Hydrogen is used to make saturated fats from oils.
D. The H-H bond dissociation enthalpy is lowest as compared to a single bond between two atoms of any element.
E. Hydrogen reduces oxides of metals that are more active than iron.
Choose the most appropriate answer from the options given below:
View Solution
A compound is formed by two elements A and B. The element B forms cubic close packed structure and atoms of A occupy 1/3 of tetrahedral voids. If the formula of the compound is \ce{A_xB_y, then the value of \(x + y\) is in option:
View Solution
Which one of the following statements is correct?
View Solution
Amongst the following, the total number of species NOT having eight electrons around the central atom in its outermost shell is,
\ce{NH3, \ce{AlCl3, \ce{BeCl2, \ce{CCl4, \ce{PCl5:
View Solution
Weight (g) of two moles of the organic compound, which is obtained by heating sodium ethanoate with sodium hydroxide in presence of calcium oxide is:
View Solution
Select the correct statements from the following:
A. Atoms of all elements are composed of two fundamental particles.
B. The mass of the electron is \(9.10939 \times 10^{-31}\) kg.
C. All the isotopes of a given element show same chemical properties.
D. Protons and electrons are collectively known as nucleons.
E. Dalton’s atomic theory regarded the atom as an ultimate particle of matter.
Choose the correct answer from the options given below:
View Solution
Which one is an example of heterogeneous catalysis?
View Solution
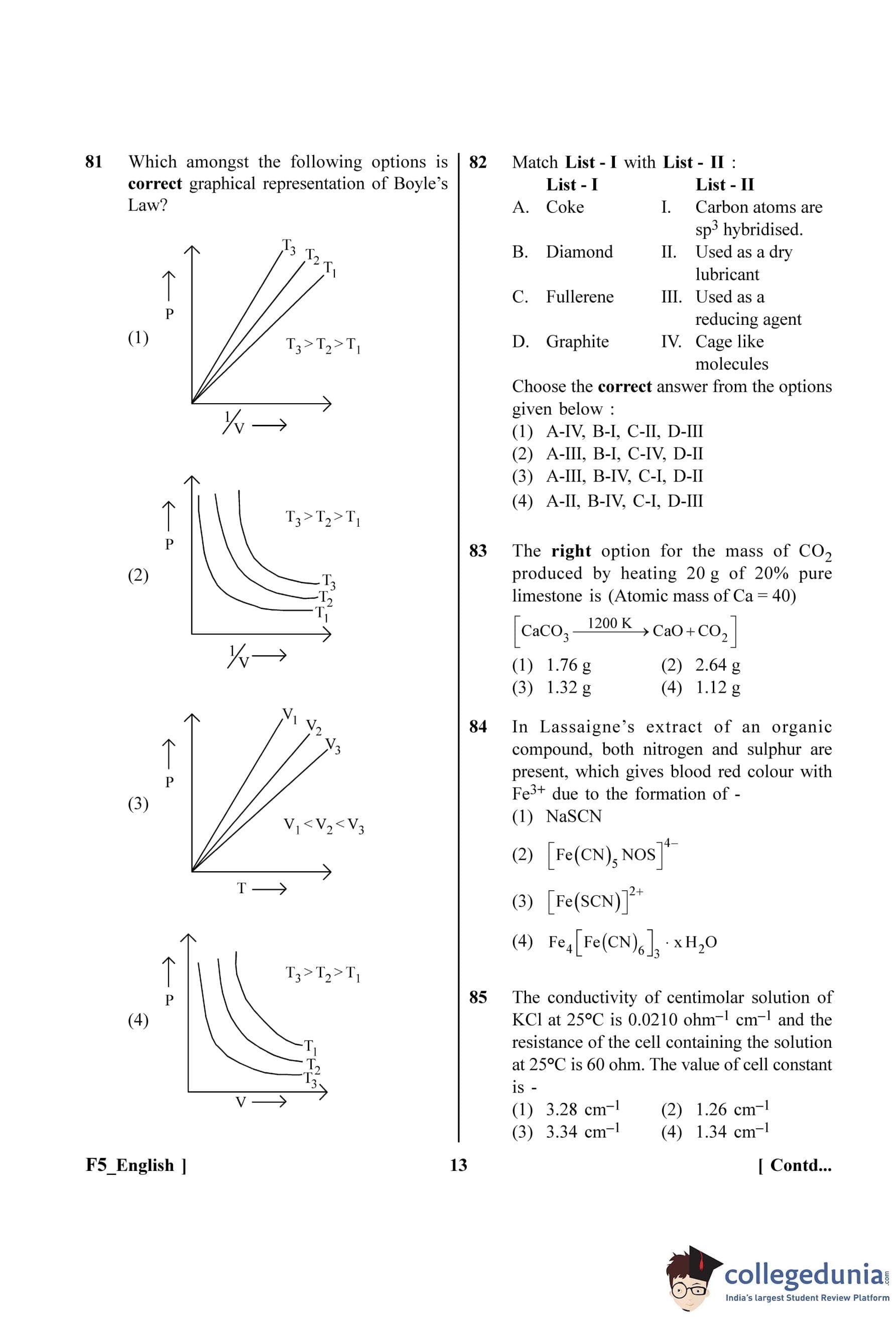
Which amongst the following options is correct graphical representation of Boyle’s Law?
Match List - I with List - II:
List - I List - II
A. Coke I. Carbon atoms are sp\textsuperscript{3 hybridised.
B. Diamond II. Used as a dry lubricant
C. Fullerene III. Used as a reducing agent
D. Graphite IV. Cage like molecules
Choose the correct answer from the options given below:
View Solution
The right option for the mass of \ce{CO2 produced by heating 20 g of 20% pure limestone is (Atomic mass of Ca = 40):
\[ \ce{CaCO3 ->[1200 K] CaO + CO2} \]
View Solution
In Lassaigne’s extract of an organic compound, both nitrogen and sulphur are present, which gives blood red colour with Fe\textsuperscript{3+ due to the formation of -
View Solution
The conductivity of centimolar solution of KCl at 25\textdegree C is 0.0210 ohm\textsuperscript{-1 cm\textsuperscript{-1 and the resistance of the cell containing the solution at 25\textdegree C is 60 ohm. The value of cell constant is -
View Solution
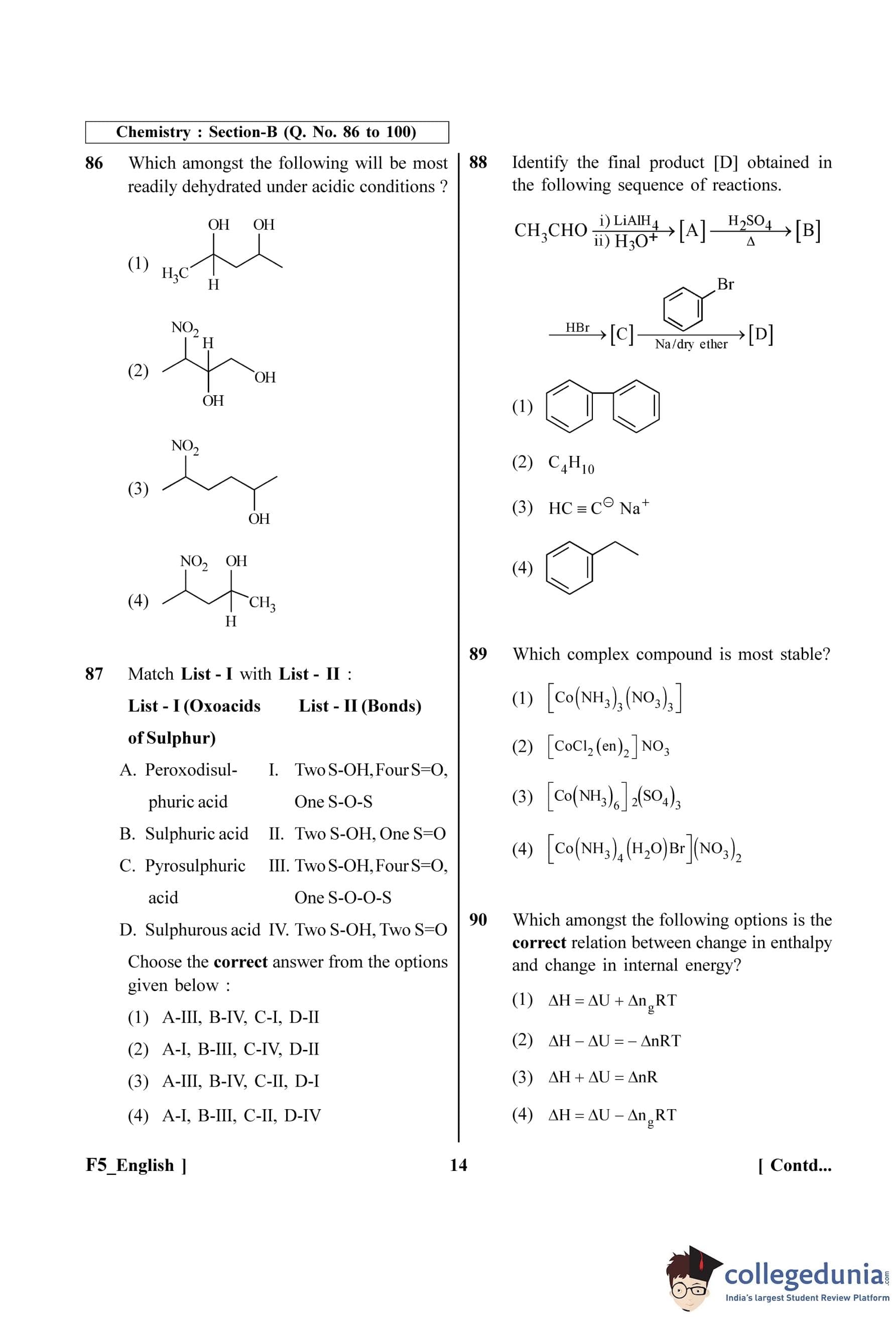
Which amongst the following will be most readily dehydrated under acidic conditions?
Match List - I with List - II :
List - I (Oxoacids of Sulphur) List - II (Bonds)
A. Peroxodisulphuric acid I. Two S-OH, Four S=O, One S-O-S
B. Sulphuric acid II. Two S-OH, One S=O
C. Pyrosulphuric acid III. Two S-OH, Four S=O, One S-O-O-S
D. Sulphurous acid IV. Two S-OH, Two S=O
View Solution
Identify the final product [D] obtained in the following sequence of reactions:
Which complex compound is most stable?
View Solution
Which amongst the following options is the correct relation between change in enthalpy and change in internal energy?
% Solution \textbf{Solution:}
The relation is derived from the First Law of Thermodynamics:
\(\Delta H = \Delta U + P\Delta V\).
For ideal gases, \(P\Delta V = \Delta n_gRT\), so:
\(\Delta H = \Delta U + \Delta n_gRT\)
View Solution
Consider the following reaction:
Identify products A and B.
Given below are two statements:
Statement I : The nutrient deficient water bodies lead to eutrophication.
Statement II : Eutrophication leads to decrease in the level of oxygen in the water bodies.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Consider the following compounds/species:
The number of compounds/species which obey Huckel’s rule is ______.
View Solution
Identify the major product obtained in the following reaction:
What fraction of one edge centred octahedral void lies in one unit cell of fcc?
View Solution
On balancing the given redox reaction,
\[ a Cr_2O_7^{2-} + b SO_3^{2-} (aq) + c H^+ (aq) \rightarrow 2a Cr^{3+} (aq) + b SO_4^{2-} (aq) + c \frac{1}{2} H_2O (l) \]
the coefficients \( a, b, c \) are found to be, respectively -
View Solution
To balance the given redox reaction, we first balance the Cr, SO₃²⁻ ions, and hydrogen ions. After balancing the half-reactions and ensuring the number of atoms and charges are equal on both sides, the coefficients \( a = 3 \), \( b = 8 \), and \( c = 1 \) are determined. Quick Tip: When balancing redox reactions, balance the atoms of elements involved in oxidation and reduction first, and then balance the charges using electrons.
The reaction that does NOT take place in a blast furnace between 900 K to 1500 K temperature range during extraction of iron is:
View Solution
Which of the following statements are INCORRECT?
A. All the transition metals except scandium form MO oxides which are ionic.
B. The highest oxidation number corresponding to the group number in transition metal oxides is attained in \( Sc_2O_3 \) to \( Mn_2O_7 \).
C. Basic character increases from \( V_2O_3 \) to \( V_2O_4 \) to \( V_2O_5 \).
D. \( V_2O_4 \) dissolves in acids to give \( VO_3^{2-} \) salts.
E. \( CrO_3 \) is basic but \( Cr_2O_3 \) is amphoteric.
Choose the correct answer from the options given below:
View Solution
The equilibrium concentrations of the species in the reaction \( A + B \rightleftharpoons C + D \) are 2, 3, 10 and 6 mol L\(^{-1}\), respectively at 300 K. \( \Delta G^\circ \) for the reaction is (R = 2 cal / mol K)
View Solution
Pumice stone is an example of -
View Solution
The historic Convention on Biological Diversity, ‘The Earth Summit’ was held in Rio de Janeiro in the year:
View Solution
The phenomenon of pleiotropism refers to
View Solution
Movement and accumulation of ions across a membrane against their concentration gradient can be explained by
View Solution
How many ATP and NADPH\textsubscript{2 are required for the synthesis of one molecule of Glucose during Calvin cycle?
View Solution
Which of the following stages of meiosis involves division of centromere?
View Solution
Given below are two statements:
Statement I: Endarch and exarch are the terms often used for describing the position of secondary xylem in the plant body.
Statement II: Exarch condition is the most common feature of the root system.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Frequency of recombination between gene pairs on same chromosome as a measure of the distance between genes to map their position on chromosome, was used for the first time by
View Solution
Which hormone promotes internode/petiole elongation in deep water rice?
View Solution
In tissue culture experiments, leaf mesophyll cells are put in a culture medium to form callus. This phenomenon may be called as:
View Solution
In angiosperm, the haploid, diploid and triploid structures of a fertilized embryo sac sequentially are:
View Solution
Which micronutrient is required for splitting of water molecule during photosynthesis?
View Solution
Axile placentation is observed in
View Solution
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: Late wood has fewer xylary elements with narrow vessels.
Reason R: Cambium is less active in winters.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Spraying of which of the following phytohormone on juvenile conifers helps in hastening the maturity period, that leads to early seed production?
View Solution
During the purification process for recombinant DNA technology, addition of chilled ethanol precipitates out
View Solution
Assertion A: The first stage of gametophyte in the life cycle of moss is protonema stage.
Reason R: Protonema develops directly from spores produced in capsule.
Choose the most appropriate answer:
View Solution
Among eukaryotes, replication of DNA takes place in -
View Solution
In the equation GPP – R = NPP, R refers to:
View Solution
In gene gun method used to introduce alien DNA into host cells, microparticles of ______ metal are used.
View Solution
Large, colourful, fragrant flowers with nectar are seen in:
View Solution
Family Fabaceae differs from Solanaceae and Liliaceae. With respect to the stamens, pick out the characteristics specific to family Fabaceae:
View Solution
Identify the pair of heterosporous pteridophytes among the following:
View Solution
Among ‘The Evil Quartet’, which one is considered the most important cause driving extinction of species?
View Solution
Upon exposure to UV radiation, DNA stained with ethidium bromide will show
View Solution
Expressed Sequence Tags (ESTs) refers to
View Solution
The reaction centre in PS II has an absorption maxima at
View Solution
Given below are two statements:
Statement I: The forces generated by transpiration can lift a xylem-sized column of water over 130 meters height.
Statement II: Transpiration cools leaf surfaces sometimes 10 to 15 degrees, by evaporative cooling.
In the light of the above statements, choose the most appropriate answer:
View Solution
What is the function of tassels in the corn cob?
View Solution
Cellulose does not form blue colour with Iodine because
View Solution
Assertion A: ATP is used at two steps in glycolysis.
Reason R: First ATP is used in converting glucose into glucose-6-phosphate and second ATP is used in conversion of fructose-6-phosphate into fructose-1,6-diphosphate.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
The thickness of ozone in a column of air in the atmosphere is measured in terms of:
View Solution
Identify the correct statements:
A. Detritivores perform fragmentation.
B. The humus is further degraded by some microbes during mineralization.
C. Water soluble inorganic nutrients go down into the soil and get precipitated by a process called leaching.
D. The detritus food chain begins with living organisms.
E. Earthworms break down detritus into smaller particles by a process called catabolism.
View Solution
What is the role of RNA polymerase III in the process of transcription in Eukaryotes?
View Solution
The process of appearance of recombination nodules occurs at which sub stage of prophase I in meiosis?
View Solution
Unequivocal proof that DNA is the genetic material was first proposed by
View Solution
Which one of the following statements is NOT correct?
View Solution
Identify the correct statements:
A. Lenticels are the lens-shaped openings permitting the exchange of gases.
B. Bark formed early in the season is called hard bark.
C. Bark is a technical term that refers to all tissues exterior to vascular cambium.
D. Bark refers to periderm and secondary phloem.
E. Phellogen is single-layered in thickness.
View Solution
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: In gymnosperms the pollen grains are released from the microsporangium and carried by air currents.
Reason R: Air currents carry the pollen grains to the mouth of the archegonia where the male gametes are discharged and pollen tube is not formed.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II :
List I List II
A. M Phase I. Proteins are synthesized
B. G2 Phase II. Inactive phase
C. Quiescent stage III. Interval between mitosis and initiation of DNA replication
D. G1 Phase IV. Equational division
Choose the correct answer from the options given below:
View Solution
Which of the following statements are correct about Klinefelter’s Syndrome?
A. This disorder was first described by Langdon Down (1866).
B. Such an individual has overall masculine development. However, the feminine development is also expressed.
C. The affected individual is short-statured.
D. Physical, psychomotor and mental development is retarded.
E. Such individuals are sterile.
Choose the correct answer from the options given below:
View Solution
Main steps in the formation of Recombinant DNA are given below. Arrange these steps in a correct sequence.
A. Insertion of recombinant DNA into the host cell.
B. Cutting of DNA at specific location by restriction enzyme.
C. Isolation of desired DNA fragment.
D. Amplification of gene of interest using PCR.
Choose the correct answer from the options given below:
View Solution
How many different proteins does the ribosome consist of?
Choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: Gause’s “Competitive Exclusion Principle” states that two closely related species competing for the same resources cannot co-exist indefinitely and competitively inferior one will be eliminated eventually.
Statement II: In general, carnivores are more adversely affected by competition than herbivores.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Which of the following combinations is required for chemiosmosis?
Choose the correct answer from the options given below:
View Solution
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: A flower is defined as modified shoot wherein the shoot apical meristem changes to floral meristem.
Reason R: Internode of the shoot gets condensed to produce different floral appendages laterally at successive nodes instead of leaves.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II :
List I List II
A. Mutualism I. + (A), O(B)
B. Commensalism II. – (A), O(B)
C. Amensalism III. + (A), – (B)
D. Parasitism IV. + (A), + (B)
Choose the correct answer from the options given below:
View Solution
Match List I with List II :
List I List II
A. Cohesion I. More attraction in liquid phase
B. Adhesion II. Mutual attraction among water molecules
C. Surface tension III. Water loss in liquid phase
D. Guttation IV. Attraction towards polar surfaces
Choose the correct answer from the options given below:
View Solution
Melonate inhibits the growth of pathogenic bacteria by inhibiting the activity of
Choose the correct answer from the options given below:
View Solution
Match List I with List II :
List I List II
A. Iron I. Synthesis of auxin
B. Zinc II. Component of nitrate reductase
C. Boron III. Activator of catalase
D. Molybdenum IV. Cell elongation and differentiation
Choose the correct answer from the options given below:
View Solution
Match List I with List II :
List I List II
A. Oxidative decarboxylation I. Citrate synthase
B. Glycolysis II. Pyruvate dehydrogenase
C. Oxidative phosphorylation III. Electron transport system
D. Tricarboxylic acid cycle IV. EMP pathway
Choose the correct answer from the options given below:
View Solution
Which one of the following common sexually transmitted diseases is completely curable when detected early and treated properly?
View Solution
Match List I with List II :
List I List II
A. CCK I. Kidney
B. GIP II. Heart
C. ANF III. Gastric gland
D. ADH IV. Pancreas
Choose the correct answer from the options given below:
View Solution
Match List I with List II :
List I List II
A. Cartilaginous Joint I. Between flat skull bones
B. Ball and Socket Joint II. Between adjacent vertebrae in vertebral column
C. Fibrous Joint III. Between carpal and metacarpal of thumb
D. Saddle Joint IV. Between Humerus and Pectoral girdle
Choose the correct answer from the options given below:
View Solution
Which of the following statements are correct regarding female reproductive cycle?
A. In non-primate mammals, cyclical changes during reproduction are called oestrus cycle.
B. First menstrual cycle begins at puberty and is called menopause.
C. Lack of menstruation may be indicative of pregnancy.
D. Cyclic menstruation extends between menarche and menopause.
Choose the most appropriate answer from the options given below:
View Solution
Select the correct group/set of Australian Marsupials exhibiting adaptive radiation.
View Solution
Which of the following functions is carried out by cytoskeleton in a cell?
View Solution
Vital capacity of lung is ______.
View Solution
Match List I with List II :
List I List II
A. Gene ‘a’ I. β-galactosidase
B. Gene ‘y’ II. Transacetylase
C. Gene ‘i’ III. Permease
D. Gene ‘z’ IV. Repressor protein
Choose the correct answer from the options given below:
View Solution
Which one of the following techniques does not serve the purpose of early diagnosis of a disease for its early treatment?
View Solution
Broad palm with a single palm crease is visible in a person suffering from-
View Solution
Given below are two statements:
Statement I: Vas deferens receives a duct from seminal vesicle and opens into urethra as the ejaculatory duct.
Statement II: The cavity of the cervix is called cervical canal which along with vagina forms birth canal.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
In which blood corpuscles, the HIV undergoes replication and produces progeny viruses?
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Amniocentesis for sex determination is one of the strategies of Reproductive and Child Health Care Programme.
Reason R: Ban on amniocentesis checks increasing menace of female foeticide.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II with respect to human eye.
List I List II
A. Fovea I. Visible coloured portion of eye that regulates diameter of pupil.
B. Iris II. External layer of eye formed of dense connective tissue.
C. Blind spot III. Point of greatest visual acuity or resolution.
D. Sclera IV. Point where optic nerve leaves the eyeball and photoreceptor cells are absent.
Choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: Low temperature preserves the enzyme in a temporarily inactive state whereas high temperature destroys enzymatic activity because proteins are denatured by heat.
Statement II: When the inhibitor closely resembles the substrate in its molecular structure and inhibits the activity of the enzyme, it is known as competitive inhibitor.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Endometrium is necessary for implantation of blastocyst.
Reason R: In the absence of fertilization, the corpus luteum degenerates that causes disintegration of endometrium.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: A protein is imagined as a line, the left end represented by first amino acid (C-terminal) and the right end represented by last amino acid (N-terminal).
Statement II: Adult human haemoglobin consists of 4 subunits (two subunits of α type and two subunits of β type.)
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Once the undigested and unabsorbed substances enter the caecum, their backflow is prevented by-
View Solution
Given below are statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Nephrons are of two types: Cortical & Juxta medullary, based on their relative position in cortex and medulla.
Reason R: Juxta medullary nephrons have short loop of Henle whereas, cortical nephrons have longer loop of Henle.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: In prokaryotes, the positively charged DNA is held with some negatively charged proteins in a region called nucleoid.
Statement II: In eukaryotes, the negatively charged DNA is wrapped around the positively charged histone octamer to form nucleosome.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. P - wave I. Beginning of systole
B. Q - wave II. Repolarisation of ventricles
C. QRS complex III. Depolarisation of atria
D. T - wave IV. Depolarisation of ventricles
Choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. A Leopard and a Lion in a forest/grassland I. Competition
B. A Cuckoo laying egg in a Crow's nest II. Brood parasitism
C. Fungi and root of a higher plant in Mycorrhizae III. Mutualism
D. A cattle egret and a Cattle in a field IV. Commensalism
Choose the correct answer from the options given below:
View Solution
Which one of the following symbols represents mating between relatives in human pedigree analysis?
View Solution
Which of the following statements is correct?
View Solution
Which of the following is not a cloning vector?
View Solution
Match List I with List II:
List I List II
A. Vasectomy I. Oral method
B. Coitus II. Barrier method interruptus
C. Cervical caps III. Surgical method
D. Saheli IV. Natural method
Choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: RNA mutates at a faster rate.
Statement II: Viruses having RNA genome and shorter life span mutate and evolve faster.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. Peptic cells I. Mucus
B. Goblet cells II. Bile juice
C. Oxyntic cells III. Proenzyme pepsinogen
D. Hepatic cells IV. HCl and intrinsic factor for absorption of vitamin B12
Choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. Ringworm I. Haemophilus influenzae
B. Filariasis II. Trichophyton
C. Malaria III. Wuchereria bancrofti
D. Pneumonia IV. Plasmodium vivax
Choose the correct answer from the options given below:
View Solution
Which of the following are NOT considered as part of endomembrane system?
View Solution
Given below are two statements:
Statement I: Ligaments are dense irregular tissue.
Statement II: Cartilage is dense regular tissue.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. Taenia I. Nephridia
B. Paramoecium II. Contractile vacuole
C. Periplaneta III. Flame cells
D. Pheretima IV. Urecose gland
Choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. Heroin I. Effect on cardiovascular system
B. Marijuana II. Slow down body function
C. Cocaine III. Painkiller
D. Morphine IV. Interfere with transport of dopamine
Choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: Electrostatic precipitator is most widely used in thermal power plant.
Statement II: Electrostatic precipitator in thermal power plant removes ionising radiations.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Radial symmetry is NOT found in adults of phylum ______.
View Solution
Select the correct statements.
A. Tetrad formation is seen during Leptotene.
B. During Anaphase, the centromeres split and chromatids separate.
C. Terminalization takes place during Pachytene.
D. Nucleolus, Golgi complex and ER are reformed during Telophase.
E. Crossing over takes place between sister chromatids of homologous chromosome.
Choose the correct answer from the options given below:
View Solution
Which one of the following is NOT an advantage of inbreeding?
View Solution
The unique mammalian characteristics are:
View Solution
Given below are two statements:
Statement I: During G0 phase of cell cycle, the cell is metabolically inactive.
Statement II: The centrosome undergoes duplication during S phase of interphase.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Which of the following statements are correct regarding skeletal muscle?
A. Muscle bundles are held together by collagenous connective tissue layer called fascicle.
B. Sarcoplasmic reticulum of muscle fibre is a store house of calcium ions.
C. Striated appearance of skeletal muscle fibre is due to distribution pattern of actin and myosin proteins.
D. M line is considered as functional unit of contraction called sarcomere.
Choose the most appropriate answer from the options given below:
View Solution
Which of the following statements are correct?
A. Basophils are most abundant cells of the total WBCs.
B. Basophils secrete histamine, serotonin and heparin.
C. Basophils are involved in inflammatory response.
D. Basophils have kidney shaped nucleus.
E. Basophils are agranulocytes.
Choose the correct answer from the options given below:
View Solution
In cockroach, excretion is brought about by-
View Solution
Which of the following statements are correct?
A. An excessive loss of body fluid from the body switches off osmoreceptors.
B. ADH facilitates water reabsorption to prevent diuresis.
C. ANF causes vasodilation.
D. ADH causes increase in blood pressure.
E. ADH is responsible for decrease in GFR.
Choose the correct answer from the options given below:
View Solution
Select the correct statements with reference to chordates.
A. Presence of a mid-dorsal, solid and double nerve cord.
B. Presence of closed circulatory system.
C. Presence of paired pharyngeal gillslits.
D. Presence of dorsal heart.
E. Triploblastic pseudocoelomate animals.
Choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. Logistic growth I. Unlimited resource availability condition
B. Exponential growth II. Limited resource availability condition
C. Expanding age pyramid III. The percent individuals of pre-reproductive age is
largest followed by reproductive and post reproductive age groups
D. Stable age pyramid IV. The percent individuals of pre-reproductive and reproductive
age group are same
Choose the correct answer from the options given below:
View Solution
Match List I with List II:
List I List II
A. Mast cells I. Ciliated epithelium
B. Inner surface II. Areolar connective tissue
C. Blood III. Cuboidal epithelium
D. Tubular parts IV. Specialised connective tissue of nephron
Choose the correct answer from the options given below:
View Solution
The parts of human brain that helps in regulation of sexual behaviour, expression of excitement, pleasure, rage, fear etc. are:
(1) Corpora quadrigemina & hippocampus
(2) Brain stem & epithalamus
(3) Corpus callosum and thalamus
(4) Limbic system & hypothalamus
View Solution
Which of the following are NOT under the control of thyroid hormone?
A. Maintenance of water and electrolyte balance
B. Regulation of basal metabolic rate
C. Normal rhythm of sleep-wake cycle
D. Development of immune system
E. Support the process of R.B.Cs formation
Choose the correct answer from the options given below:
View Solution
Which of the following is characteristic feature of cockroach regarding sexual dimorphism?
View Solution
Which one of the following is the sequence on corresponding coding strand, if the sequence on mRNA formed is as follows 5' AUCGUCGACGCAUGCUGAUCG 3'?
View Solution



























NEET Previous Year Question Papers with Answer Keys
| NEET 2022 Question Papers | NEET 2021 Question Papers | NEET 2020 Question Papers |
| NEET 2019 Question Papers | NEET 2018 Question Papers | NEET 2017 Question Papers |








Comments